XPS characteristics of sulfur of bio-oxidized arsenic-bearing gold concentrate and changes of surface nature of bio-oxidation residue
YANG Hong-ying(杨洪英), GONG En-pu(巩恩普), YANG Li(杨 立),
CHEN Gang(陈 刚), FAN You-jing(范有静), ZHAO Yu-shan(张玉山), Lǚ Jiu-ji(吕久吉)
(School of Materials and Metallurgy, Northeastern University, Shenyang 110004, China)
Abstract: During bio-oxidation of sulfides, the chemical state change of sulfur is a complex and key factor. It is not only an indicator of the extent and intensity of the bio-oxidation, but also controls the property of bio-leaching medium and the period of oxidation. The chemical state of sulfur in sulfides oxidized by leaching bacteria was studied with XPS. Sulfide minerals in the arsenic-bearing gold concentrate consist of pyrite, arsenopyrite, chalcopyrite, galena, sphalerite and so on. In order to probe the pattern of the chemical state change of sulfur in the bio-oxidation residue of arsenic-bearing gold concentrate, the structure of the grains, and the surface nature of the residue, XPS test was carried out through different sputtering duration. The study of XPS clearly shows that: sulfides is progressively oxidized from the surface of minerals to the core by leaching bacteria; the chemical valence of sulfur changes from S2- or [S2]2- to [SO4]2-; sulfur in the core is in a reduction state, S2- or [S2]2-, but exists in an oxidation state S6+ on the surface; due to the chemical state change of sulfur, mineral phase of the bio-oxidation residue is also changed(sulfides inside, while sulfates outside); the layered structure is found in the grains of the bio-oxidation residue.
Key words: bio-oxidation; arsenic-bearing gold concentrate; XPS; chemical state; sulfur; surface nature CLC number: TH111.31
Document code: A
1 INTRODUCTION
The bacterial leaching mechanism of sulfides has been studied for years[1-8]. Bio-chemical reactions on the interface between bio-leaching solution and sulfides are interrelated to the crystal structure and surface nature of sulfides[9]. The property of sulfide mineral and its surface nature directly control the direction and duration of the process of bacteria oxidation. The nature of a mineral appears to play an important role during bio-oxidation process. A lot of membrane protein and many kinds of active polar groups, such as ─OH, ─COOH, ─SH, ─NH2 are found on the mucous layer of leaching bacteria. These active polar groups establish new equilibrium with chemical bonds attaching on surfaces of minerals. After the sulfide minerals are oxidized with leaching bacteria, the surface nature of sulfides is changed completely. Oxidized mineral materials and bio-metabolic products are found on the surface. These bio-metabolic products will definitely influence the cyanidation process later on. The change of crystal structure leads to the change of mineral phase in the gold concentrate. The most direct evidences are the change in valence of various elements with multiple valences, the change in the nature of sulfide components, the decomposition of sulfide crystals, and the final form of new mineral phase. XPS is an important means to investigate the valences of elements on the surface of a mineral. Sulfur is a significant element showing the extent of bio-oxidation of arsenic-bearing gold concentrate. The study of XPS of sulfur of the bio-oxidized grains has revealed the chemical behaviors of sulfur during bio-oxidation process and its bio-oxidation characteristics in bio-oxidized sulfides.
2 SAMPLE
Samples used in this study are arsenic-bearing gold concentrate collected from Guangxi, China. The main chemical compositions of different grain grades are shown in Table 1. The mineralogical study shows that the metal minerals mainly consist of arsenopyrite and pyrite, and a little bit of chalcopyrite, galena, and sphalerite; stibnite is very little. The gangue minerals mainly are quartz, feldspar and calcite. Ultra-submicro grains of gold are occluded in arsenopyrite or pyrite crys-
Table 1 Major elements of arsenic-bearing gold concentrate(mass fraction, %)
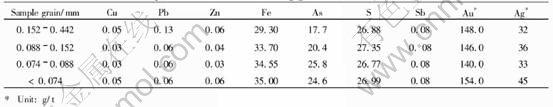
tals. Test samples of bio-oxidation are arsenic-bearing gold concentrate. The size of grains is below 0.074mm. After the gold concentrate is oxidized with leaching bacteria, the metal luster on sulfide minerals disappears completely. It is shown by the X-ray diffraction that some sulphates form in the oxidizing slags through bio-oxidation. The surface of some sulfide grains is deeply corroded, having rough surface and earth-luster. The corrosive extent of different minerals in the arsenic-bearing gold concentrate is varied, which completely depends on their mineralogical nature.
3 XPS CHARACTERISTICS OF BIO-OXIDATION RESIDUE
3.1 XPS test
XPS is the most efficient technology to investigate the chemical characteristics of the surface of minerals. It can provide some key information, such as element distribution on the surface of minerals, valences of elements, chemical states[10, 11]. The instrument used in this study was MICROLAB.MKⅡ-X made by VG Company, Britain. The testing condition was Mg as excitation source.
3.2 XPS characteristics of sulfur of bio-oxidation residue
In order to discuss oxidation extent of the bio-oxidation residue and the change of the valence of sulfur, a special technique, sputtering, was utilized. XPS test was conducted through different durations of sputtering.
Through the study, XPS characteristics of sulfur at the surface of samples and at different depths in different samples were discovered. Changes of chemical state of sulfur during bio-oxidation process have been well studied. Two bio-oxidation residue samples, 8-4-2 and 8-1-2, were tested. The results are listed in Table 2.
Fig.1 shows XPS characteristic spectrum lines of sulfur in sample 8-4-2. It shows XPS characteristics of sulfur after different periods of sputtering. The XPS characteristics reflect the change of the valence of sulfur on the surface or at different depths of sample after bio-oxidation. Line 1 is the XPS characteristic line of sulfur on the surface of the sample, which has two peaks. The main peak
Table 2 XPS characteristics of bio-oxidation residue
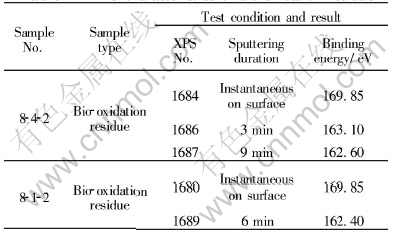

Fig.1 XPS characteristic spectrum lines of sulfur of sample 8-4-2
of binding energy is 169.85eV. Line 2 is the XPS characteristic line of sulfur of the same sample after 3min of sputtering. It also shows two peaks, however, the main peak is obviously displaced at 163.10eV. Line 3 is the XPS characteristic line of sulfur after 9min of sputtering. It has only one peak with binding energy of 162.60eV. Beyond of peaks, the lines show dental feature. Fig.2 shows XPS characteristic spectrum lines of sulfur of sample 8-1-2. Line 1 is the XPS characteristic line of sulfur on the surface of the sample. The main peak is clear and its binding energy is 169.85eV. Line 2 is the XPS characteristic spectrum line of the same sample sputtered for 6min; the binding energy displaces from 169.85eV to 162.40eV. The obvious displacement of the binding energy indicates a big change of the valence of sulfur.
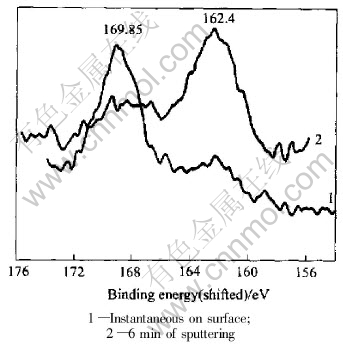
Fig.2 XPS characteristic spectrum lines of sulfur of sample 8-1-2
3.3 XPS characteristics of sulfur of chemically treated bio-oxidized residue
Samples 8-4-1 and 8-1-1 were collected from the arsenic-bearing gold concentrate that was bio-oxidized by leaching bacteria and then treated through a chemical medicament of acidification after bio-oxidation. Testing was conducted after the oxidation layers and bio-metabolic products of samples had been washed away. The XPS characteristics of sulfur of samples 8-4-1 and 8-1-1 are listed in Table 3.
Table 3 XPS Characteristics of treated bio-oxidized residue
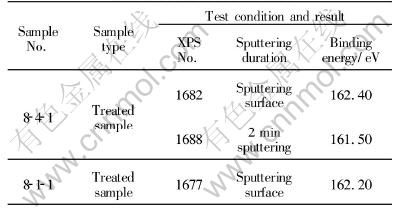
Fig.3 shows the XPS characteristics of sulfur in sample 8-4-1, a sample of chemically treated acidification of bio-oxidation residue. Line 1 shows the XPS characteristic of the surface of the sample. The curve is smooth and the peak is clear with binding energy of 162.40eV. Line 2 is the XPS characteristic spectrum line of the same sample after it was sputtered for 2min. The peak binding energy is 161.50eV.

Fig.3 XPS characteristic spectrum lines of sulfur of sample 8-4-1
Fig.4 shows the XPS characteristic of sulfur on the surface of sample 8-1-1. The sample was also treated with the same chemical method. The binding energy is 162.20eV.
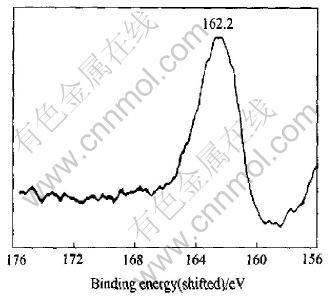
Fig.4 XPS characteristic spectrum line of sulfur of sample 8-1-1
4 ANALYSIS OF XPS CHARACTERISTICS
4.1 Geo-chemical characteristics of element sulfur
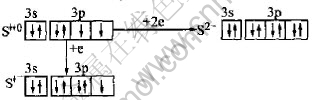
The electron configuration of sulfur is 3s23p4, which leads to sulfur having geo-chemical characteristics of multiple valences under different circumstances. Sulfur atom can be excited to S2- state or S1- state when it acts with other elements under reduction condition[12]:
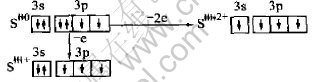
Sulfur can be excited to S1+ or S2+ state under some condition[12]:
Sulfur have different valences and can show different chemical behaviors under different reduction-oxidation conditions. With oxygen fugacity increasing, the chemical state of sulfur will change as follow:
S2-→S1-→S0→S4+→S6+
H2S― FeS2― S ―SO2 ―SO2-4
→Oxidation strengthening
Sulfur atom in the structure of sulfide minerals can be presented as argon form and/or ultra-argon form. There are six types of charge, S2+, S1+, S0, S1-, S2-, S3-[12]. In fact, Geo-chemical characteristic of element sulfur in the structure of sulfide minerals in nature is quite complicated. Therefore, the oxidation of sulfide minerals has become a growing interest in bio-hydrometallurgy[13-16]. At present, study on bio-oxidation process of arsenic-bearing gold concentrate is just beginning, but becomes more and more important. During biological oxidation, sulfides are oxidized, their crystal structures are broken, and then gold grains occluded in the crystals of sulfides, such as arsenopyrite and pyrite, are exposed through electrons being transferred from bacteria to sulfides. Later, gold will be extracted from the bio-oxidized arsenic-bearing gold concentrate with cyaniding processing.
4.2 Chemical state of bio-oxidized residue
The main mineral compositions of arsenic-bearing gold concentrate are sulfides. The chemical state of the bio-oxidation residue is changed dramatically after bacterial oxidation. Because the duration of sputtering is different, which means that bacteria reach into different depths of the sample, the chemical states of sulfur are different at different depths. For instance, Fig.1 shows XPS characteristic lines of sulfur of samples sputtered for different durations(instantaneous sputtering on the surface, sputtering for 3min, or sputtering for 9min). With the increase of sputtering duration and sputtering depth, the main peak of binding energy decreases(169.85eV, 163 .10eV, 162.60eV) and the shapes of peaks of spectrum lines are evidently different. Fig.2 shows XPS characteristic lines of sulfur of another sample after instantaneously sputtering on the surface or sputtering for 6min. The main peaks of binding energy are 169.85eV and 162.40eV, respectively. As on Fig.1, two lines are quite different. The peak at 169.85eV of XPS characteristic line of sulfide with instantaneously sputtering is the binding energy of S2p3/2, which reveals the electron lamella of atom as S2p. The chemical state of sulfur changes to [SO4]2-, the valence of sulfur is S6+, the mineral phase is sulfates, such as Fe2(SO4)3. The binding energy has an obvious displacement and becomes 163eV after sputtering for 3min. It is the binding energy of S2p1/2 and indicates that the electron lamella of atom is S2p, sulfur is in reduced state, the valence of sulfur is S2- or [S2]2-, the phase of mineral belongs to sulfide minerals. By sputtering for 9min, the binding energy is 162.6eV, which is the binding energy of S2p3/2, sulfur exists as S2- or [S2]2- , the mineral phase also belongs to sulfide minerals[17, 18]. The XPS characteristic lines of sulfur of sample 8-2-1 clearly show a displacement of binding energy and changes of chemical state of sulfur. Although the mineral compositions are so complex, including sulfides such as pyrite, arsenopyrite, sphalerite, galena, chalcopyrite, and sulfates, that they can produce some interference, the general trend shows that the chemical state of sulfur is changed dramatically from the surface to different depths of grains of the bio-oxidized gold concentrate. The study shows that a change of the main peak of binding energy of sulfur of the bio-oxidation residue has occurred through bio-oxidation process.
Sample 8-4-2
Sputtering duration:
Instantaneous→3min→9min
Binding energy:
69.85eV→163 .10eV→162.60eV
Chemical valence:
[SO4]2-→ S2-or [S2]2-→ S2-or [S2]2
Chemical state:
Oxidized state→ Reduced state→More reduced state
Sample 8-1-2
Sputtering period: Instantaneous→6min
Binding energy: 169.85eV→162.40eV
Chemical valence: [SO4]2-→S2- or [S2]2-
Chemical state: Oxidized state→Reduced state
The data above demonstrate that bacterial oxidation starts from surface to core of sulfides, which results in changes of the chemical states of sulfur and its mineral phase, sulfates on the surface to sulfides in the core. The study of XPS characteristics of sulfur of the bio-oxidation residue also reveals that grains of the bio-oxidation residue possess a layered structure.
4.3 Surface modification of bio-oxidation residue
Fig.3 and Fig.4 show XPS characteristics of sulfur of samples 8-4-1 and 8-1-1, from which the oxidation film and bio-metabolic products were washed away. Comparing Fig.3 and Fig.4 with Fig.1 and Fig.2, it can be clearly seen that the XPS characteristics of sulfur are very different from those of samples with bio-metabolic products and oxidation films. XPS characteristic spectrum lines of sulfur on the surface of chemically treated samples have smooth single peaks. There is a big displacement of binding energy. The main peak is 162.4eV. The peak of 169.85eV disappears because of no S6+ in sample 8-4-1 or sample 8-1-1. It also shows that the chemical state of sulfur of the samples treaded chemically are S2- and [S2]2-. Their mineral phases are sulfides. The fresh surface of sulfides with S2-, [S2]2- is exposed again; in turn, the surface energy will become higher. Therefore, the bacterial oxidation will be further advanced.
5 CONCLUSIONS
1) The chemical state of sulfur is changed during bio-oxidation processing from the surface to the core of bio-oxidation residue, which leads to changes of mineral phase of sulfur. Sulfur on the surface of the residue is presented as S6+; the main mineral phase is sulfates. While, sulfur in the core is presented as S2-, and so the main mineral phase is sulfides.
2) A layered structure is observed in the grains of bio-oxidation residue. The outer sulfur is in oxidation state, while the inner sulfur is in reduction state.
3) In order to accelerate bio-oxidation, it is necessary to chemically treat the bio-oxidation residue; therefore, the chemical valence of sulfur on the surface of grains will change from S6+ to S2- or [S2]2-. Through the chemical treatment, grains can obtain higher surface energy and then the bio-oxidation can be advanced further.
Acknowledgements
The authors thank Dr. YU Guang-hua, University of Science and Technology Beijing, China, for the XPS test.
REFERENCES
[1]Tributsch H. Direct versus indirect bioleaching [J]. Hydrometallurgy, 2001, 59: 177-185.
[2]Sand W, Gehrke T, Jozsa P G, et al. (Bio) chemistry of bacterial leaching―direct vs. indirect bioleaching [J]. Hydrometallurgy, 2001, 59: 159-175.
[3]Hansford G S, Vargas T. Chemical and electrochemical basis of bioleaching processes [J]. Hydrometallurgy, 2001, 59 : 135-145.
[4]Dopson M, Lindstrom E B. Potential role of Thiobacillus caldus in arsenopyrite leaching [J]. Appl Environ Microbiol, 1999, 65: 36- 40.
[5]Boon M, Brasser H J, Hansford G S, et al. Comparison of the oxidation kinetics of different pyrites in the presence of Thiobacillus ferrooxidans or Leptospirillum ferrooxidans [J]. Hydrometallurgy, 1999, 53: 57-72.
[6]Brierley J A, Brierley C L. Present and future commercial applications in biohydrometallurgy [J]. Hydrometallurgy, 2001, 59: 233-240.
[7]Yahya A, Johnson D B. Bioleaching of pyrite at low pH and low redox potentials by novel mesophilic Gram-positive bacteria [J]. Hydrometallurgy, 2002, 63: 181- 188.
[8]Rojas-Chapana J A, Tributsch H. Biochemistry of sulfur extraction in bio-corrosion of pyrite by Thiobacillus ferrooxidans [J]. Hydrometallurgy, 2001, 59: 291-300.
[9]YANG Hong-ying, YANG Li, ZHAO Yu-shan. Submicro-battery effect and selective bio-oxidation model of gold-bearing arsenopyrite by Thiobacillus ferrooxidans [J]. Trans Nonferrous Met Soc China, 2002, 12(6): 1199-1202.
[10]WANG Dian-fen. Applications of X-ray Photoelectron Spectroscopy in Research of Non-Metallic Materials[M]. Wuhan: Wuhan University of Technology Press, 1994.(in Chinese)
[11]WANG Jian-qi, WU Wen-hui, FENG Da-ming. Electronic Energy Spectrumlogy(XPS/XAES/UPS) Quotation [M]. Beijing: National Defence Industry Press,1992.(in Chinese)
[12]WANG Pu, PAN Zhao-lu, WENG Ling-bao. Systematic Mineralogy [M]. Beijing: Geological Publishing House, 1992.(in Chinese)
[13]Hyland M M, Jean G, Bancroft G M. XPS and AES studies of Hg(Ⅱ) sorption and desorption reaction on sulfide minerals [J]. Geochim Cosmochim Acta, 1990, 54: 1957-1967.
[14]LIU Ying-jun, CAO Li-ming, LI Zhao-lin,et al. Element Geochemistry [M]. Beijing: Science Press,1984.(in Chinese)
[15]Mycroft J R, et al. Detection of sulfur and polysulphides on electrochemically oxidized pyrite surfaces by X-ray photoelectron spectroscopy and Raman spectroscopy [J]. J Elecrtoanal Chem, 1990, 290: 139-152.
[16]Hyland M M, Bancroft G M. An XPS study of gold deposition at low temperature on sulfide minerals: Reducing agents [J]. Geochim Cosmochim Acta, 1989, 53: 367-372.
[17]LI Ying-tang, TIAN Shu-yan. Application Mineralogy [M]. Beijing: Science Press, 1995.(in Chinese)
[18]JIA Jian-ye, XIE Xian-de, WU Da-qing, et al. An XPS study on surfaces of common sulfide minerals [J]. Geological Journal of China Universities, 2000, 6: 255-259.(in Chinese)
(Edited by YUAN Sai-qian)
Foundation item: Projects(50074015; 50274024) supported by the National Natural Science Foundation of China; project (20022022) supported by the Natural Science Foundation of Liaoning Province
Received date:2004 -05-25; Accepted date: 2004-08-06
Correspondence: YANG Hong-ying, Professor, PhD; Tel: +86-24-83679555; E-mail: hyyang@ mail.nue.edu.cn