Carburization of ferrochromium metals in chromium ore fines containing coal during voluminal reduction by microwave heating
来源期刊:中南大学学报(英文版)2009年第1期
论文作者:陈津 赵晶 张猛 晏泓 周剑雄
文章页码:43 - 48
Key words:chromium ore fines containing coal (COFCC); ferrochromium metal; microwave heating; voluminal reduction; carburization
Abstract: Chromium ore fines containing coal (COFCC) can be rapidly heated by microwave to conduct the voluminal reduction, which lays a foundation of getting sponge ferrochromium powders with a lower content of C. Under the conditions of COFCC with n(O):n(C) (molar ratio) as 1.00:0.84 and n(SiO2):n(CaO) as 1.00:0.39, the samples were heated by 10 kW microwave power to reach the given temperatures and held for different times respectively. The results show that the low-C-Cr ferrochromium metal phase in the reduced materials forms before the high-C-Cr ferrochromium metal phase does. With increasing temperature the C content of ferrochromium metals is in a positive correlation with the content of Cr. The C content of ferrochromium metal in reduced materials is 0-10.07% with an average value of 4.68%. With the increase of holding time the Cr content in ferrochromium metals is in a negative correlation with the content of C, while the content of Fe changes in the contrary way. In the microwave field the kinetic conditions of carburization are closely related with the temperature of microwave heating, holding time and carbon fitting ratio.
基金信息:the National Natural Science Foundation of China
J. Cent. South Univ. Technol. (2009) 16: 0043-0048
DOI: 10.1007/s11771-009-0007-5![]()
CHEN Jin(陈 津)1, ZHAO Jing(赵 晶)1, ZHANG Meng(张 猛)1,
YAN Hong(晏 泓)1, ZHOU Jian-xiong(周剑雄)2
(1. College of Materials Science and Engineering, Taiyuan University of Technology, Taiyuan 030024, China;
2. Institute of Mineral Resource, Chinese Academy of Geological Science, Beijing 100037, China)
Abstract: Chromium ore fines containing coal (COFCC) can be rapidly heated by microwave to conduct the voluminal reduction, which lays a foundation of getting sponge ferrochromium powders with a lower content of C. Under the conditions of COFCC with n(O)?n(C) (molar ratio) as 1.00?0.84 and n(SiO2)?n(CaO) as 1.00?0.39, the samples were heated by 10 kW microwave power to reach the given temperatures and held for different times respectively. The results show that the low-C-Cr ferrochromium metal phase in the reduced materials forms before the high-C-Cr ferrochromium metal phase does. With increasing temperature the C content of ferrochromium metals is in a positive correlation with the content of Cr. The C content of ferrochromium metal in reduced materials is 0-10.07% with an average value of 4.68%. With the increase of holding time the Cr content in ferrochromium metals is in a negative correlation with the content of C, while the content of Fe changes in the contrary way. In the microwave field the kinetic conditions of carburization are closely related with the temperature of microwave heating, holding time and carbon fitting ratio.
Key words: chromium ore fines containing coal (COFCC); ferrochromium metal; microwave heating; voluminal reduction; carburization
1 Introduction
Chromium is an element of easily-forming carbides [1-2]. Cr-Fe double carbides ([CrxFey]Cz) are mainly formed because of the limitless mutual solubility of Cr in α-Fe [3]. In the production of liquid-state ferrochromium, the ferrochromium contains 6%-9% C. The higher the temperature, the more the carbon in ferrochromiums [4]. At present, the production process of low or extra-low-C ferrochromium is relatively complicated by the means of agglomeration technology (such as Perrin process, solid-state vacuum decarburization process), which restricts the applications for the low or extra-low-C ferrochromium [5-8]. In order to use the high-C ferrochromium, in the well-sized steelworks the argon oxygen decarburization (AOD) or vacuum oxygen decarburization (VOD) process is used by increasing temperature and vacuum to acquire the carbon elimination and chromium conservation to the greatest degree.
Chromium ore fines containing coal (COFCC) can be volumetrically heated by microwave rapidly [9]. Medium or low carbon sponge ferrochromium powder can be obtained by controlling the kinetic conditions of carburization because of the rapid reaction rate of voluminal reduction and the relatively low reaction temperature in solid phase compared with those in liquid phase. Carbon-bearing chromium ore pellet (briquette) has a characteristic of self-reduction by the conventional heating [10-11], and the content of C in ferrochromium metals increases with the increase of carbon fitting ratio [12]. When powdery materials are heated by microwave, the carburization of the ferrochromium metals is still unclear. In this work, the carburization of the reduced COFCC at low carbon fitting ratio was studied, and the carburization control conditions during the voluminal reduction by microwave heating were discussed through the experiment of bulk-mass powdery materials.
2 Experimental
2.1 Materials
All the raw materials in the experiments were picked up from the iron and steel works. 1 000 g of COFCC was composed of 800 g of Indian chromium ore fines (Table 1), 150 g of anthracite fines (Table 2) and 50 g of lime fines (Table 3). n(O)?n(C) (molar ratio) was 1.00?0.84, and n(SiO2)?n(CaO) was 1.00?0.39 in COFCC.
Table 1 Chemical compositions of India chromium ore fines (mass fraction, %)

Table 2 Chemical compositions of anthracite fines (mass fraction, % )
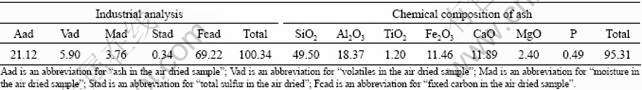
Table 3 Chemical compositions of lime fines (mass fraction, %)
![]()
2.2 Procedure
All materials were separately ground to 74 μm (more than 95% in mass fraction) and then mixed together in the mass proportion to prepare COFCC. The crucible used in experiments was made of light fire bricks with inner diameter of 65 mm and height of 260 mm. 1 kg of COFCC was put into the crucible and set in the microwave metallurgical oven (under atmospheric pressure without protective gas). The frequency of microwave heating was 2.450 GHz. The samples were heated by 10 kW microwave power up to the given temperatures of 1 273, 1 373, 1 473, and 1 575 K, and kept for different times, respectively. The temperature of the stuff was measured by Pt-Pt/10%Rh thermocouple.
The instrument used for analyzing the physical phase of samples was super probe made up of the electron probe microanalyzer (EPMA, JXA8800R, Japan) and X-ray energy dispersive spectrometer (EDS, ISIS300, Oxford Company). The working conditions of the instrument were as follows: accelerating voltage 20 kV and current 2×10-2 μA. The quantitative analysis was used to make sure the metal contents of samples while the semiquantitative analysis was applied to confirming the content of C and O. The electron images were semiquantitative analysis images.
3 Results and discussion
Figs.1 and 2 show the relationship between temperature and contents of Cr, Fe, C, S and P in ferrochromium metals reduced from COFCC by microwave heating. In the ferrochromium metals, contents of Cr and Fe are in a negative correlation, corresponding to the experimental results obtained in a conventional furnace in Ref.[13]. Furthermore, the content of Cr increases with increasing content of C, and P decreases with increasing the content of S, while the content of Fe changes just to the contrary. Under the condition of low molar ratio of C to O, Cr is much easier to be carburized than Fe, and easier to integrate with P instead of S.
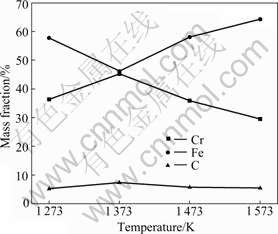
Fig.1 Relationship between temperature and contents of Cr, Fe and C in ferrochromium metals
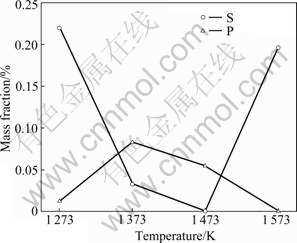
Fig.2 Relationship between temperature and contents of S and P in ferrochromium metals
Figs.3 and 4 show the relationship between holding time and the contents of Cr, Fe, C, S and P in the ferrochromium metals. The contents of Cr and Fe are also in a negative correlation, the content of Cr is in a negative correlation with the content of C, but in a positive correlation with the contents of P and S. The content of Fe changes just to the contrary.
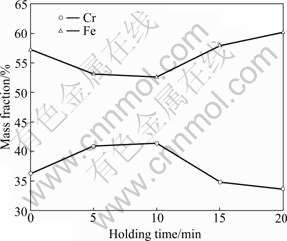
Fig.3 Relationship between holding time and contents of Cr and Fe in ferrochromium metals at 1 273 K
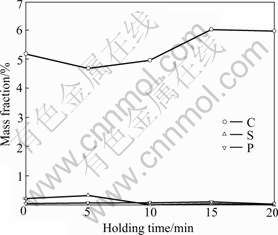
Fig.4 Relationship between holding time and contents of C, S, and P in ferrochromium metals at 1 273 K
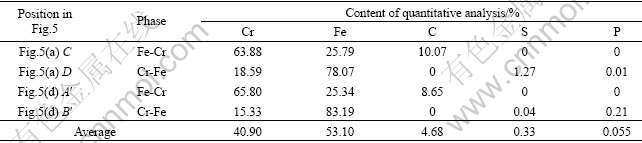
It is worth noting that ferrochromium metal can be classified into two kinds. One is high-C-Cr ferrochromium metal phase (Cr 63.88%-65.80%, Fe 25.34%-25.79%, C8.65%-10.07%), the other is low-C-Cr ferrochromium metal phase (Cr 15.33%-18.59%, Fe 78.07%-83.19%, without C) (Fig.5 and Table 4). According to the microscopic structure of boundary between the two phases (Fig.5(c)), the low-C-Cr ferrochromium metal phase is obviously nibbled by the high-C-Cr ferrochromium metal phase, indicating that the former forms before the latter. Moreover, the content of C in ferrochromium metals increases with the increase of high-C-Cr ferrochromium phase. Table 5 shows the average contents of Cr, Fe, C, S, and P in ferrochromium metal phase at 1 273 K. The content of C is lower than 10.07%, with an average value of 4.68%.

Fig.5 Electron back scatter diffraction (EBSD) images of ferrochromium metal using voluminal reduction of COFCC by microwave heating for 5 min at 1 273 K: (a) Dot-shape structure of ferrochromium metals in reduced materials; (b) Sharp-nibbling structure of ferrochromium metals in slag phase; (c) Structure of high-C-Cr phase nibbling low-C-Cr phase in ferrochromium metals; (d) Structure of residual slag in low-C-Cr phase
Table 4 Chemical compositions of reduced materials and ferrochromium metal obtained by super probe (1 273 K, 5 min of holding time)
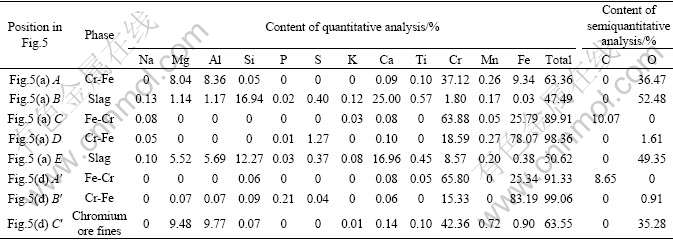
Table 5 Average values of mainly chemical compositions in ferrochromium metal obtained at 1 273 K for 5 min
4 Carburization of ferrochromium metals in microwave field
Cr2O3 of chromium ores mainly exists in the form of spinel, [Fe, Mg]O?[Cr, Al, Fe]2O3, with different contents of Al2O3, FeO, Fe2O3, and MgO. Indian chromium ore used in the experiment belongs to the low-Mg, Al-bearing chromium ore. It is proven that the carbon reduction reaction for chromium ore fines takes place as follows [14]:
CrxOy+yC=xCr+yCO (Initial reaction)
CrxOy+yCO=xCr+yCO2 (Leading reaction)
yCO2+yC=2yCO (Leading reaction)
The voluminal reduction reaction of COFCC by the microwave heating is in an accord with reactive mechanism. Here, the initial reaction temperature of Boudouard reaction is 974 K:
CO2+Cgraphite=2CO, ![]() =166 550-171 T(Tinitial=974 K) (1)
=166 550-171 T(Tinitial=974 K) (1)
When Cr2O3 exists in the form of the ferrichromspinel [FeCr2O4], the initial reaction temperature to form the metal iron is 1 184 K [15]:
FeO・Cr2O3+C=Fe+Cr2O3+CO,![]() =163 830-138.43 T (Tinitial=1 184 K) (2)
=163 830-138.43 T (Tinitial=1 184 K) (2)
When the reaction temperature to form metal chromium is 1 448 K:
FeO・Cr2O3+4C=Fe+2Cr+4CO,![]() =983 772-679.6 T (Tinitial=1 448 K) (3)
=983 772-679.6 T (Tinitial=1 448 K) (3)
During the carbon reduction of chromium ore, once the metal phase occurs, it can be carburized at once [16]. In other words, metal iron is firstly reduced from FeO [17], and then carburized to form Fe3C at the same time:
3α-Fe+C=Fe3C, ![]() =29 037-28.033 T,(298-1 273 K, Tinitial=1 035 K) (4)
=29 037-28.033 T,(298-1 273 K, Tinitial=1 035 K) (4)
3γ-Fe+C=Fe3C, ![]() =11 234-11.004 T,(1 000-1 683 K, Tinitial=1 021 K) (5)
=11 234-11.004 T,(1 000-1 683 K, Tinitial=1 021 K) (5)
The formation of metal iron, especially the formation of iron carbide, it accelerates the decomposition and reduction of Cr2O3 in chromium ore fines, and promotes the formation of metal chromium. Furthermore, the metal chromium can not only form the solid solution with α-Fe, but also be combined with carbon to form various complex carbides rapidly at different temperatures. The carburization process of ferrochromium metal phase can be carried out in the following ways.
(1) Cr2O3 is reduced through carbon diffusion, and then various chromium carbides are formed at different temperatures:
2/3Cr2O3+26/9C=4/9Cr3C2+2CO, ![]() =114 410-83.05 T (Tinitial=1 378 K) (6)
=114 410-83.05 T (Tinitial=1 378 K) (6)
2/3Cr2O3+18/7C=4/21Cr7C3+2CO,![]() =115 380-82.09 T (Tinitial=1 406 K) (7)
=115 380-82.09 T (Tinitial=1 406 K) (7)
2/3Cr2O3+53/23C=4/69Cr23C6+2CO,![]() =118 270-81.75 T (Tinitial=1 447 K) (8)
=118 270-81.75 T (Tinitial=1 447 K) (8)
The chromite (FeO・Cr2O3) is initially reduced to metal iron above 1 184 K (reaction 2), but the formation temperature of Fe3C is only 1 035 K (reaction 4), while the metal chromium is reduced above 1 448 K (reaction 3) and Cr3C2 is formed only at 1 378 K (reaction 6). Therefore, metal iron is reduced initially at a low temperature, and then carburized to form Fe3C. With increasing temperature, metal chromium is obtained by reducing Cr2O3 with carbon and simultaneously carburized to form chromium carbide or complex carbide.
(2) Chromium carbide is formed from the reaction of CO with metal chromium [18].
From Eqn.(3), metal chromium can be reduced at 1 448 K, and then reacts with CO as follows:
2CO=C+CO2, 3Cr+2C=Cr3C2,![]() =-790 78-17.657 T (9)
=-790 78-17.657 T (9)
Because CO can be easily absorbed on the surface of metal chromium, the chromium carbide can be formed under a certain carbon potential. Actually, the partial pressure of CO is very high during the voluminal reduction of COFCC by microwave heating owing to the Boudouard reaction. The chromium ore fines can be reduced at a high carbon partial pressure to provide the thermodynamics and kinetics conditions for the carburization of metal chromium. This experiment was done at a low molar ratio of C to O (0.84), but the carburization of ferrochromium metal phase occurred in different degrees, which indicates that metal chromium is much easier to be carburized than metal iron.
(3) Low-C chromium carbide is formed from the reduction reaction of high-C chromium with Cr2O3.
The oxygen of Cr2O3 in chromium ore fines can not only be combined with CO or C, but combined with C in the chromium carbides.
5Cr2O3+27Cr3C2=13Cr7C3+15CO (10)
Cr2O3+3Cr7C3=Cr23C6+3CO (11)
2Cr2O3+Cr23C6=27Cr+6CO (12)
Under the atmospheric pressure, reactions (10), (11) and (12) were carried out at 1 240, 1 600, and 2 000 K, respectively. Under a certain carbon potential (pCO), the higher the temperature, the easier the formation of low-C carbide. In the experiment, it is impossible to form the pure metal chromium owing to reactions (10) and (11).
Carburization rate is generally determined by the carburization concentration, the temperature and the thickness of carburized layer [19-20]. In the solid-solid-phase voluminal reduction process of COFCC by microwave heating, the carburization of CO dominates in view of the leading reaction of Boudouard reaction:
2CO=[C]+CO2 (13)
Generally, the carburization process of CO takes place according to the following three stages: (1) the enhancement of carbon potential―increasing CO concentration on the material surface and decreasing CO2 concentration at a certain temperature; (2) the diffusion of carbon―diffusing into the inner of ferrochromium metal phase to reach the carbon potential (pCO) for carburization; (3) the equilibrium stage of carburization―the carbon concentration inside the ferrochromium metal phase equilibrizing with CO concentration outside. If CO concentration reaches the critical carburization concentration, it will be absorbed and decomposed on the surface of ferrochromium metal phase, producing the high active carbon atoms to carburize:
FeO?CrxOy+zCO=FeO?CrxOy?zCO (14)
(Chemical absorption of CO)
FeO?CrxOy?zCO=Fe?xCr?C?(z-1)CO2 (15)
(Interfacial chemical reaction)
Fe?xCr?C?(z-1)CO2=Fe?xCr?C+(z-1)CO2 (16)
(Physical desorption of CO2)
But in the voluminal reduction process of COFCC by microwave heating, the carburization of ferrochromium metal phase mainly includes two stages: firstly, carbon atoms transfer from gas phase to the surface of chromium ore fines for reduction; and secondly, carbon atoms diffuse from the surface of ferrochromium metal to the inside for reducing carburization. So it is not a simple carburizing process for ferrochromium metal in the voluminal reduction process of COFCC by microwave heating, but a complex carburizing process in the course of reduction. At the certain temperature and carbon potential, the main influence factor of carburization rate is the transfer coefficient of carbon, which will be mainly depended on chemical compositions of carburization. The experiment shows that transfer coefficient of carbon increases with increasing contents of CO and CO2 when their total content is lower than 50% in carburizing ambience[21]. The transfer coefficient of carbon is so small that the velocity of carburization is too slow when the total content of CO and CO2 is lower than 15%.
According to the analysis of carburization in reduction process, Fe3C primitively forms on the outside of chromium ore fines. On account of instability for Fe3C, it will change to (Fe, Cr)3C2 step by step with the reduction process [17]. The carbon in complex carbides will simultaneously diffuse inside the chromium ore fines and capture the oxygen to form CO. Then the complex carbides change to low-C carbide such as (Fe,Cr)7C3 (Fig.6). In theory, the content of C inside the reduced ferrochromium metal should be lower than that outside.

Fig.6 Sketch map of reducing carburization for chromium ore fines
Furthermore, CO is easy to be absorbed on the surface of chromium ore fines to carry out the chemical absorption due to the molecule polarization of CO in the microwave field and the energy accumulation[18], which accelerates the carburization reaction. But compared with the traditional methods of ferrochromium production, the dynamic conditions of carburization process are weaker than those for liquid-phase reaction because of the rapid heating rate and short reaction time for COFCC in microwave field.
5 Conclusions
(1) During the voluminal reduction of COFCC by microwave heating with n(O)?n(C) (molar ratio) as 1.00? 0.84 and n(SiO2)?n(CaO) as 1.00?0.39, the high-C-Cr ferrochromium metal phases and the low-C-Cr ferrochromium metal phases are main. The low-C-Cr ferrochromium metal phases form before the high-C-Cr ferrochromium metal phases. In the ferrochromium metal phases, contents of Cr and Fe are in a negative correlation.
(2) With increasing temperature the contents of C and P are in a positive correlation with the content of Cr, while the content of S is in a negative correlation. At the low molar ratio of C and O, metal chromium is easier to be carburized than metal iron. The content of C in ferrochromium metals is lower than 10.07% with an average of 4.68% at 1 273 K.
(3) With increasing holding time the content of Cr is in a negative correlation with content of C, while a positive correlation with contents of P and S, but Fe changes just to the contrary.
(4) The kinetic conditions of reducing carburization are related to the microwave heating temperature, holding time, and carbon fitting ratio of reduced materials.
References
[1] LE?KO A, NAVARA E. Microstructural characterization of high-carbon ferrochromium [J]. Materials Characterization, 1996, 36(4): 349-356.
[2] GEMINIANI A, GRANDE M A. Chrome carbide investigated [J]. Metal Powder Report, 2003, 58(3): 23-24.
[3] DETROYE M, RENIERS F, BUESS-HERMAN C, VEREECKEN J. AES-XPS study of chromium carbides and chromium iron carbides [J]. Applied Surface Science, 1999, 144(4): 78-82.
[4] AKY?ZL? M, ERIC R H. Slag-metal equilibrium in the smelting of high-carbon ferrochromium [J]. Journal of the South African Institute of Mining and Metallurgy, 1992, 92(4): 101-110.
[5] ZHU De-qing, LI Jian, PAN Jian, HE Ao-ping. Sintering behaviours of chromite fines and the consolidation mechanism [J]. International Journal of Mineral Processing, 2008, 86(1): 58-67.
[6] HONKANIEMI M, KROGERUS H, DAAVITTILA J, OIKARINEN P. The importance of chromite pre-treatment in the production of ferrochromium [C]// Sixth International Ferro Alloys Congress. Cape Town, 1992, 1: 79-86.
[7] BASSON J, CURR T R, GERICKE W A. South Africa’s ferroalloys industry-present status and future outlook [C]// The Eleventh International Ferro Alloys Congress. New Delhi, 2007, 1: 3-24.
[8] LEOT’EV L I, ZHUCHKOV V I, SMIRNOV L A, DASHEVSKII V Y. Global and Russian ferroalloy production [J]. Steel in Translation, 2007, 37(3): 283-287.
[9] XUE Zheng-liang, HU Hui-jun, ZHANG You-ping, TING Xiu-mei, CUI Hui-jun, LIU Jin-ying. High temperature reduction properties of synthetic chromite by carbon [J]. Journal of Wuhan University of Science and Technology: Natural Science Edition, 2004, 27(1): 1-3. (in Chinese)
[10] HIROSHI G. The reduction behavior of synthetic chromites by carbon [J]. Transactions of the Iron and Steel Institute of Japan, 1980, 20: 154-162.
[11] LIN Q, LIU R, CHEN N. Kinetics of direct reduction of chrome iron ore [J]. Journal of Thermal Analysis and Calorimetry, 1999, 58(2): 317-322.
[12] XUE Zheng-liang, HU Hui-jun, ZHANG You-ping, LI Zheng-bang, ZHOU Yu-sheng. Condense behaviour of metal phase in synthetic chromite briquette by carbon during high temperature reduction [J]. Ferro-Alloys, 2004(5): 1-5. (in Chinese)
[13] SOYKAN O, ERIC R H, KING R P. The reduction mechanism o f a natural chromite at 1 416 ℃ [J]. Metallurgical and Materials Transactions B, 1991, 22(1): 53-63.
[14] NIAYESH M J, DIPPENAR R J. The solid state reduction of chromite [C]// Sixth International Ferro Alloys Congress. Cape Town, 1992, 1: 57-63.
[15] ZHANG You-ping, LI Zheng-bang, XUE Zheng-liang. Application technology of chromite ore fines for producing stainless steel master alloy [J]. Special Steel, 2003, 24(1): 29-32. (in Chinese)
[16] JIANG Guo-chang, XU Jiang-lun, XU Kuang-di. A basic study on reduction process of chrome ore agglomerate [J]. Ferro-Alloys, 1989(5): 23-30. (in Chinese)
[17] XU Rong-jun, NI Rui-ming, ZHANG Sheng-bi, MA Zhong-ting. Research on reduction thermodynamics of carbon-bearing chromite pellets [J]. Sintering and Pelletizing, 1996, 21(3): 1-4. (in Chinese)
[18] CHAKRABORTY D, RANGANATHAN S, SINHA S N. Investigations on the carbothermic reduction of chromite ores [J]. Metallurgical and Materials Transaction B, 2005, 36(4): 437-444.
[19] GOLDSTEIN J I, MOREN A E. Diffusion modeling of the carburization process [J]. Metallurgical and Materials Transaction A, 1978, 9(11): 1515-1525.
[20] TANG Dian, QIU Yu-lang, CHEN Zai-liang. Carburizing and nitriding of ferrous metals in ancient China [J]. Teat Treatment of Metals, 2002, 27(8): 50-53. (in Chinese)
[21] ZHANG Yao-li, WU Yan. A study on the carburizing velocity [J]. Industrial Heating, 2000(3): 40-42. (in Chinese)
Foundation item: Project(50474083) supported by the National Natural Science Foundation of China; Project supported by the Baoshan Iron & Steel Co. Ltd. of China
Received date: 2008-03-06; Accepted date: 2008-05-15
Corresponding author: CHEN Jin, Professor, PhD; Tel: +86-351-5683869; E-mail: chenjinty@sohu.com
(Edited by YANG Hua)

