J. Cent. South Univ. Technol. (2007)03-0285-06
DOI: 10.1007/s11771-007-0056-6

Devitrification behaviour of rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy prepared by melt spinning method
XIAO Yu-de(肖于德)1, 2, LI Min(李 敏)1, ZHONG Jue(钟 掘)2, LI Wen-xian(黎文献)1, MA Zheng-qing(马正青)1
(1. School of Materials Science and Engineering, Central South University, Changsha 410083, China;
2. School of Mechanical and Electrical Engineering, Central South University, Changsha 410083, China)
Abstract:Rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy was prepared by using melt spinning. Its calorimetric behavior was characterized by using differential scanning calorimeter in a continuous or isothermal heating mode. phase transformation was investigated, with a special interest in primary crystallization, by using an in-situ examination of X-ray diffractometry (XRD) and high resolution transmission electron microscopy (HRTEM). The results show that, the whole devitrification of rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy involves two main processes of primary crystallization and secondary crystallization that consist mainly of two reactions. For primary crystallization, the apparent activation energies, EIso and EKis and growth activation energies Eg are about 153, 166 and 288 kJ/mol, respectively. The interdiffusion of Al atoms is a rate-controlled step of formation of the α(Al) particles, but slow diffusion of Ni and Nd atoms plays a significant role in retarding growth of the a (Al) particles. For secondary crystallization, EIso, EKis and Eg of the first reaction are about 291, 208 and 290 kJ/mol, and those of the second reaction are about 367, 269 and 372 kJ/mol. The two reactions of secondary crystallization are controlled mainly in an interface-controlled three-dimensional mode, depending mainly on slow diffusion of Ni and Nd atoms.
Key words: rapid solidification; Al-rich amorphous alloy; crystallization; nucleation and growth
1 Introduction
Rapidly solidified Al-based amorphous alloys have been paid more and more attention in the novel material research field since they were discovered independently by HE et al[1] and INOUE et al[2] in late of 1980s, not only due to their high strength, hardness and low density, but also because a new-type Al-based amorphous/ nanocrystalline composites can be obtained by partial crystallization of the amorphous alloys[3-7].
The Al-based amorphous/nanocrystalline alloys containing transition metal (TM=Fe, Co, Ni, Cu, etc) and rare earth (RE=La, Y, Ce, Nd, etc) elements can be obtained by a suitable processing combination of rapid solidification and heat treatment[8-11]. Many of these Al-based amorphous alloys exhibit high tensile strength above 950-1 150 MPa and the amorphous/nano- crystalline composites produced via partial crystallization of the amorphous alloys have a significantly improved tensile strength up to 1 560 MPa. Many investigations[4-14] indicate that partial crystallization of Al-rich amorphous alloy may be an effective way to obtain novel light-mass structural materials with high strength and moderate ductility.
The amorphous/nanocrystalline composites consist of novel ultra-fine microstructures with large quantity of a(Al) nanoparticles embedded dispersively in an Al-rich amorphous matrix[8-14]. The annealing treatment based on primary crystallization of the Al-rich amorphous alloy is an important well-controlled method to this kind of ideal nanoscale structure with optimized combination of mechanical properties. However, many careful studies ought to be done firstly to understand the overall devitrification process of Al-based fully amorphous alloys, in particular primary crystallization, in order to seek for a suitable fabrication process and/or optimized annealing parameters.
Therefore, our main objective is to understand carefully the overall devitrification process of rapidly solidified Al-Ni-Cu-Nd amorphous alloy under continuous and isothermal heating regime in this work. Exothermic behavior during devitrification of rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy was analyzed by using differential scanning calorimetry (DSC) in a continuous or isothermal heating mode, and phase transformation was investigated, with a special interest in primary crystallization, by using an in-situ examination of X-ray diffractometry (XRD) and high resolution transmission electron microscopy (HRTEM).
2 Experimental
The rapidly solidified Al-based amorphous ribbons were prepared in a single copper roll melt-spinner after being melted with high pure Al, Ni, Cu, and Nd in vacuum. The ribbons are typically less than 3 mm in width and 30 mm in thickness, with chemical composition of Al87Ni7Cu3Nd3 (molar fraction, %).
Differential scanning calorimetric curves were obtained in an Ar atmosphere by using a Perkin-Elmer DSC-7 in continuous heating regimes at heating rate of 5, 10, 20, 40, 60, 80, 160 and 240 ℃/min, and in isothermal heating regimes at temperatures of 80, 100, 120, 130, 160, 260, 280, 290, 300 and 320 ℃, heated/ cooled at 160 ℃/min. The XRD experiments were carried out in a SIEMENS D500 X-ray diffractometer with a monochromatic Cu Ka (mean wave length of 0.154 18 nm) radiation, scanning at a step of 0.03? over a 2θ angle range of 30?-50?. The HRTEM foils, prepared by ion milling in a liquid nitrogen (N2) bath to avoid crystallization during thinning, were examined using a CM30 high-resolution transmission electron microscope at 300 keV.
3 Results and analysis
3.1 Exothermic reactions of rapidly solidified
Al87Ni7Cu3Nd3 amorphous alloy in continuous heating regime
Fig.1 shows a typical DSC curve of Al87Ni7Cu3Nd3 amorphous alloy in continuous heating regime at a heating rate of 60 ℃/min. Two main exothermic peaks are observed on the curves. The two peaks correspond to main devitrifying stages, namely, primary crystallization and secondary crystallization. Several typical X-ray diffraction patterns of rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy heated continuously to various temperatures at 60 ℃/min are demonstrated partially in Fig.2. It indicates that, the first peak, appeared in the temperature range from 110 to 320℃ with peak temperature of 155 ℃, is broad and asymmetric, resulting from primary crystallization of the amorphous phase to precipitate α(Al) crystal particles, and the second between 320 and 370 ℃ is sharp and symmetric, corresponding to further transformation of the residual amorphous phase into a multi-phase mixture of α(Al) crystal and some intermetallic compounds, mainly Al3Ni, Al8Cu4Nd and Al11Nd3.
It is worth noting that, the second DSC peak seems to consist mainly of two overlapped sub-peaks on the continuous heating DSC curve at 60 ℃/min. The two peaks overlapped each other can be separated by fitting this part of DSC curve using a Lorentzian model for multi-peaks in a Microcal Origin 6.0 software, seen in the inset of Fig.1, and their appearing temperatures are 346 and 351 ℃, respectively. This indicates that, two reactions occur in the residual amorphous phase during secondary crystallization, and the two reactions are not completely separated in the Al87Ni7Cu3Nd3 amorphous ribbons.
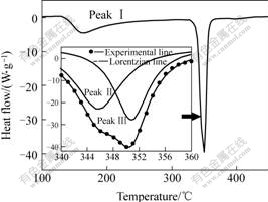
Fig.1 DSC curve of rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy in continuous heating regime at 60 ℃/min

Fig.2 XRD patterns of rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy heated continuously to
various temperatures at 60 ℃/min
3.2 Effect of heating temperature on exothermic
reaction of rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy in isothermal heating regime
Fig.3 shows several typical DSC curves of rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy in isothermal heating regime at various heating temperatures. Only Peak Ⅰ corresponding to primary crystallization is detected on each of the isothermal DSC curves below 160 ℃, shown in Fig.3(a), while Peaks Ⅱ and Ⅲcorresponding to secondary crystallization can be discovered at 260-320 ℃, shown in the Fig.3(b). Peak Ⅰ can hardly appear on the isothermal DSC curves at the higher temperatures perhaps due to primary crystallization occurring during heating at 160 ℃/min. Fig.4 shows XRD patterns of rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy in isothermal heating regime at 300 ℃. It indicates that Al3Ni, Al8Cu4Nd and Al11Nd3 phases precipitate successively from the residual amorphous matrix after primary crystallization. With the heating temperature improving, all of the exothermic peaks shift towards left. Namely, both the onset and peak time of the DSC peaks is shortened with improving the isothermal temperature.
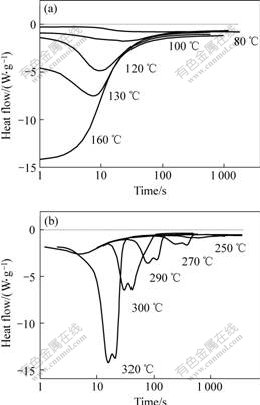
Fig.3 DSC curves of rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy in isothermal heating regime at
various heating temperatures
(a) PeakⅠ, (b) Peaks Ⅱ and Ⅲ
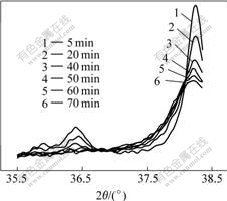
Fig.4 XRD patterns of rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy in isothermal heating regime at 300 ℃
The Johnson-Mehl-Avrami-Kolmogorov(JMAK) equation is the most widely used approach for the description of isothermal reaction[15-17] :
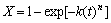 (1)
(1)
where t is the time of the reaction without incubation time τ, X is the relative reaction degree as a function of time t at the given temperature T, n is the Avrami exponent, depending on rate of nucleation and morphology of growth during the reaction, and k is the Avrami reaction rate constant, depending on thermal history of the reaction system.
k is associated with the reaction activation energy, E(in J/mol), and the temperature, T(in K), through the Arrhenius temperature dependence:
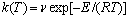 (2)
(2)
where R is the ideal gas constant, 8.314 (J/(mol?K), and v is the frequency factor.
At a certain time (tp), the reaction rate, dX/dt, reaches its maximum, i.e.,
 (3)
(3)
By taking the second derivation of Eqn.(1), and combining with Eqn.(2), tp-T equation relating to the apparent activation energy E and isothermal temperature T, can be finally derived:
 (4)
(4)
tp-T equation can be deduced into the following equation if 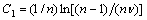 :
:
 (5)
(5)
where EIso is the apparent activition energy in isothermal heating regime. In addition, here n=2.5 for primary crystallization, and n=4 for secondary crystallization, according to Refs.[18-19]. The apparent activation energy EIso for the crystallization process is usually determined with Eqn.(5). Fig.5 reveals the plots of ln tp versus 1/T for various DSC peaks, from which EIso can be figured out. According to Eqn.(5), all of the plots would be linear and activation energy, EIso, can be easily determined from the slope of the plot, given in Fig.5. The apparent activation energies EIso of the crystallization processes corresponding to PeaksⅠ, Ⅱ, and Ⅲ are about 153, 291, and 367 kJ/mol, respectively.
3.3 Effect of heating rate on exothermic reaction of
rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy in continuous heating regime
The main exothermic peaks are isolated from several typical DSC curves of rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy in continuous heating regime at various heating rates, in order to distinguish clearly from each other. The PeakⅠcorresponding to primary crystallization and the Peaks Ⅱ and Ⅲ to secondary crystallization are shown in Figs.6(a) and (b). With heating rate improving, the exothermic peaks shift towards higher temperature, and both the onset temperature and the peak temperature of the DSC peaks are elevated with improving the heating rate.
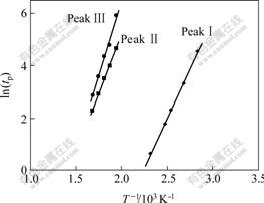
Fig.5 Plots of lntp versus 1/T for determining apparent activation energy of rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy
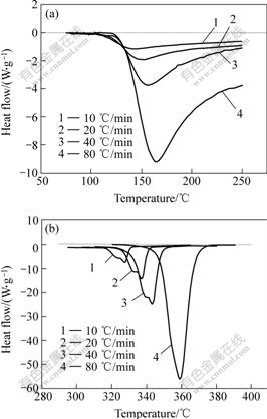
Fig.6 DSC curves of rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy in continuous heating regime at
various heating rates
(a) PeakⅠ, (b) PeaksⅡ and Ⅲ
To apply JMAK Equation to describe the non-isothermal reaction, it is necessary to take account of the dependence of k on time, t. Thus[20-23],
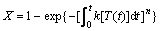 (6)
(6)
where k is a function of T through the Arrhenius temperature dependence, as shown in Eqn.(2), and T is a function of time, dT=βdt, β is the heating rate in K/min.
Starting from Eqn.(6) and Eqn.(2), and making d2X/dt2=0 at a certain temperature (Tp), the following equation can be obtained:
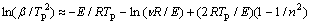 (7)
(7)
and then the so-called Kissinger Equation relating to temperature, Tp, and heating rate, β, can be finally derived[20-23]:
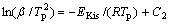 (8)
(8)
where EKis is the apparent activation energy determined by using the Kissinger method, Tp is the characteristic temperature, R is the gas constant, and C2 is a constant. The apparent activation energy for the crystallization process is usually determined with the Kissinger equation without any appreciable error[20]. Fig.7 reveals plots of 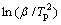 versus 1/Tp for various DSC peaks, from which EKis can be figured out. According to Eqn.(8), all of the plots would be linear and the activation energy, EKis, can be easily determined from the slope of the plot, given in Fig.7. The apparent activation energies EKis of the crystallization processes corresponding to PeaksⅠ, Ⅱand Ⅲ are about 166, 208 and 269 kJ/mol, respectively.
versus 1/Tp for various DSC peaks, from which EKis can be figured out. According to Eqn.(8), all of the plots would be linear and the activation energy, EKis, can be easily determined from the slope of the plot, given in Fig.7. The apparent activation energies EKis of the crystallization processes corresponding to PeaksⅠ, Ⅱand Ⅲ are about 166, 208 and 269 kJ/mol, respectively.
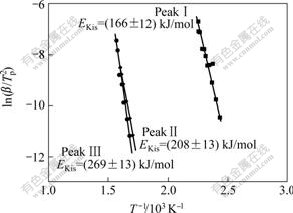
Fig.7 Plots of 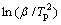 versus 1/T for determining apparent
versus 1/T for determining apparent
activation energy of rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy
4 Discussion
The Kissinger plots exhibit straight lines, with little scattering, suggesting that within the actual range of heating rates there is no obvious change in the reaction mechanism for the corresponding crystallization. The activation energy of primary crystallization is close to that (119-142 kJ/mol) of interdiffusion in pure aluminum, which indicates that the interdiffusion of Al atoms is a rate-controlled step for the primary crystallization, leading to α(Al) crystal particle precipitation. It is a very slow reaction due to extremely slow diffusion rate of Al, Ni, Nd and Cu atoms at low temperature. Obviously, the secondary crystallization resulting in the formation of the intermetallic compounds of Al3Ni, Al8Cu4Nd and Al11Nd3 is controlled by diffusion of Ni, Nd and Cu atoms with large diffusion activation energies. The intermetallic compounds form at a large rate and higher temperature. However, it must be noted that, the facts that the activation energy is larger than that of Al interdiffusion and the first DSC exothermic peak is broadened and asymmetric also indicate that the neodymium, nickel, and copper atoms with low diffusion coefficients in the aluminum have a remarkable (or non-negligible) retarding effect on nucleation and growth of α(Al) crystal particles, especially in the later stage of primary crystallization.
During primary crystallization, α(Al) crystal particles precipitate, multiply and grow in the amorphous matrix, as shown in Fig.8, accompanied by exclusion of solute atoms with low diffusing rate into the surrounding amorphous phase of α(Al) crystal particles. The rejected solute atoms build up in the front of advancing interface, and form a solute-atom-enriched concentration diffusion field around the Al crystal particles. This will lead to soft impingement effect on migration of phase interface, caused by dragging of solute-atoms build-up and mutual interaction of diffusion field[24]. With development of primary crystallization, the soft impingement effect becomes more and more violent, and it is more and more difficult for α(Al) crystal particles to grow. It implies that slow diffusion of Ni and Nd atoms plays a more and more dominant role in retarding growth of α(Al) particles during primary crystallization of rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy, with the temperature improving.

Fig.8 Bright field HRTEM micrograph of ribbon heated continuously up to about 320 ℃
The activation energy for crystallization can be expressed as the sum of nucleation and growth activation energies[18], E=aEn+bEg, in which a+b=1. In general, En is much smaller than Eg for primary crystallization of the Al-rich amorphous alloy, and even smaller than E. However, Eg can be suggested to be calculated by a modified Kissinger method expressed as follows [18]:
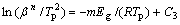 (12)
(12)
where Tp is the characteristic temperature, β is the heating rate, m is a numerical factor related to the growth mechanism, R is the gas constant, and C3 is a constant. In this work, m=n-1, and the Avrami exponent n corresponding to Tp is about 2.5 for the primary crystallization in a diffusion-controlled growth mode, and 4 for the secondary devitrification in an interface- controlled three-dimensional mode. Hence, Eg can be calculated to be about 288, 290, 372 kJ/mol, by slopes of 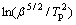 and
and 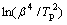 vs 1/Tp, seen in Fig.9, via the modified Kissinger method. Therefore, primary crystallization is mainly controlled in a diffusion-controlled growth mode instead of the interface-controlled nucleation mode in continuous heating regime.
vs 1/Tp, seen in Fig.9, via the modified Kissinger method. Therefore, primary crystallization is mainly controlled in a diffusion-controlled growth mode instead of the interface-controlled nucleation mode in continuous heating regime.

Fig.9 Plots of 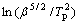 or
or 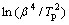 versus 1/T for determining values of E by modified Kissinger method
versus 1/T for determining values of E by modified Kissinger method
5 Conclusions
1) The whole devitrification of rapidly solidified Al87Ni7Cu3Nd3 amorphous alloy involves two main processes: primary crystallization resulting in two-phase mixture of α(Al) plus residual amorphous phase, and secondary crystallization corresponding to some inter-metallic phases appearing.
2) For primary crystallization, the apparent activation energies, EIso and EKis of primary crystallization are about 153 and 166 kJ/mol, and the growth activation energy Eg is about 288 kJ/mol. The interdiffusion of Al atoms is a rate-controlled step of formation of the α(Al) particles, but slow diffusion of Ni and Nd atoms plays a significant role in retarding growth of the α(Al) particles.
3) For secondary crystallization with two reactions, the apparent activation energies, EIso and EKis of the first reaction are about 291 and 208 kJ/mol and the growth activation energy Eg is about 290 kJ/mol, and those of the second reaction are about 367, 269 and 372 kJ/mol. The two reactions of secondary crystallization are controlled mainly in an interface-controlled three- dimensional mode, depending mainly on slow diffusion of Ni and Nd atoms.
References
[1] HE Y, POON S J, SHIFET G J. Synthesis and properties of metallic glasses that contain aluminum[J]. Science, 1988, 241: 1640-1642.
[2] INOUE A, OHTERA K, KITA K, et al. New amorphous alloys with good ductility in Al-Ce-M (M=Nb, Fe, Co, Ni or Cu) systems[J]. Japanese Journal of Applied Physics, 1988, 27(8): 1796-1799.
[3] SHIFLET G J, HE Y, POON S J. Mechanical properties of a new class of metallic glasses based on aluminum[J]. Journal of Applied Physics, 1988, 64(12): 6863-6865.
[4] KIM Y H, INOUE A, MASUMOTO T. Increase in mechanical strength of Al-Y-Ni amorphous alloy by dispersion of nanoscale fcc-Al particle[J]. Materials Transaction JIM, 1991, 32(4): 381-338.
[5] GREER A L. Metallic glasses[J]. Science, 1995, 267: 1947-1953.
[6] INOUE A, KIMURA H M, SASAMORI K, et al. Synthesis and high mechanical strength of Al-based alloys consisting mainly of nanogranular amorphous particles[J]. Materials Science and Engineering, 1996, A217(3): 401-406.
[7] INOUE A, KIMURA H. Fabrications and mechanical properties of bulk amorphous, nanocrystalline, nanoquasicrystalline alloys in aluminum-based system[J]. Journal of Light Metals, 2001, 10(1): 31-41.
[8] DAS S K, PEREPEZKO J H, WU R I, et al. Undercooling and glass formation in Al-based alloys[J]. Materials Science and Engineering, 2001, A304/306(1): 159-168.
[9] GICH M, GLORIANT T, SURI?ACH S, et al. Glass forming ability and crystallization processes within Al-Ni-Sm system[J]. Journal of Non-Crystalline Solids, 2001, 289(3): 214-220.
[10] WU R I , WILDE G, PEREPEZKO J H. Glass formation and primary nanocrystallization in Al-base metallic glasses[J]. Materials Science and Engineering, 2001, A301(1): 12-23.
[11] INOUE A. Amorphous, nanoquasicrystalline and nanocrystalline alloys in Al-based systems[J]. Progress in Materials Science, 1998, 43(1): 365-415.
[12] TSAI A A, KAMIYAMA K, KAWAMURA Y, et al. Formation and precipitation mechanism of nanoscale Al particles in Al-Ni based amorphous alloy[J]. Acta Mater, 1997, 45(6): 1477-1487.
[13] GREER A L. Crystallization of amorphous alloys[J]. Metallurgical and Material Transaction, 1996, 27(3): 549-556.
[14] ALLEN D R, FOLEY J C, PEREPEZKO J H. Nano-crystal development during primary crystallization of amorphous alloys[J]. Acta Metallurgica et Materialia, 1998, 46(2): 431-440.
[15] KEMPEN A T W, SOMMER F, MITTEMEIJER E J. Determination and interpretation of isothermal and non-isothermal transformation kinetics: The effective activation energied in terms of nucleation and growth[J]. Journal of Materials Science, 2002, 37(5): 1321-1332.
[16] CHENG Kang-guo. Evaluation of crystallization kinetics of glasses by non-isothermal analysis[J]. Journal of Materials Science, 2001, 36 (4): 1043- 1048.
[17] NATARAJ D, PRABAKAR K, NARAYANDASS K, et al. Determination of kinetic parameters of Bi2Se3 thin films by computation[J]. Crystal Research and Technology, 2000, 35(9): 1087-1094.
[18] YE F, LU K. Crystallization kinetics of Al-La-Ni amorphous alloy[J]. Journal of Non-Crystalline Solids, 2000, 289(3): 228-235.
[19] ABDEL-FATTAH G, NASSER A, AFAF G, et al. Decomposition and Precipitation Mechanisms in Supersaturated Al-Mg alloys[J]. High Temperatures-High Pressures, 1999, 31(3): 613-625
[20] ZHANG Hao-yue, MITCHELL B S. A method for determining crystallization kinetic parameters from one nonisothermal calorimetric experiment[J]. Journal of Materials Research, 2000, 15(4): 1000-1007.
[21] SOIFER L, KORIN E. Effect of heating rate on crystallization kinetics of amorphous Al91La5Ni4 by DSC[J]. Journal of Thermal Analysis and Calorimetry, 1999, 56(3): 437-446.
[22] GOGEBAKAN M, WARREN P J, CANTOR B. Crystallization behaviour of amorphous Al85Y11Ni4 alloy[J]. Materials Science and Engineering, 1997, A226-228(1): 168-172.
[23] XIAO Yu-de, LI Wen-xian. Studies on primary crystallization of rapidly quenched Al-Ni-Cu-Nd metallic glass by using high resolution transmission electron microscopy[J]. Acta Metallurgical Sinica, 2003, 16(5): 407-415.
[24] XIAO Yu-de, LI Wen-xian, MA Zheng-qing. Investigation on crystallization process in rapidly solidified Al-Nd-Ni amorphous alloy prepared by melt spinning[J]. Trans Nonferrous Met Soc China, 2004, 14(4): 665-669.
(Edited by YANG Bing)
Foundation item: Project(2005-5) supported by the Postdoctoral Foundation of Central South University, China; Project(2005038560) supported by the Postdoctoral Science Foundation of China
Received date: 2006-08-24; Accepted date: 2006-09-27
Corresponding author: XIAO Yu-de, PhD; Professor; Tel: +86-731-8830261; E-mail: xiaoyude@mail.csu.edu.cn