Trans. Nonferrous Met. Soc. China 23(2013) 2113-2117
SiC-MoSi2/ZrO2-MoSi2 coating to protect C/C composites against oxidation
Qian-gang FU, Jia-ping ZHANG, Zheng-zhong ZHANG, He-jun LI, Can SUN
C/C Composites Technology Research Center, state Key Laboratory of solidification processing Northwestern Polytechnical University, Xi’an 710072, China
Received 30 January 2013; accepted 6 May 2013
Abstract: To improve the oxidation resistance of carbon/carbon (C/C) composites in air at high temperatures, a SiC- MoSi2/ZrO2-MoSi2 coating was prepared on the surface of C/C composites by pack cementation and slurry method. The microstructures and phase compositions of the coated C/C composites were analyzed by scanning electron microscopy and X-ray diffraction, respectively. The result shows that the SiC-MoSi2/ZrO2-MoSi2 coating is dense and crack-free with a thickness of 250-300 μm. The preparation and the high temperature oxidation property of the coated composites were investigated. The as-received coating has excellent oxidation protection ability and can protect C/C composites from oxidation for 260 h at 1773 K in air. The excellent anti-oxidation performance of the coating is considered to come from the formation of ZrSiO4, which improves the stability of the coating at high temperatures.
Key words: carbon/carbon composites; SiC-MoSi2/ZrO2-MoSi2; coating; oxidation resistance
1 Introduction
Carbon/carbon (C/C) composites possess extraordinary and unique characteristics over traditional materials, such as low density, high specific strength, resistance to thermal shock and retention of mechanical properties at high temperatures, which makes them have great advantages used as thermal structural components in the aircraft and aerospace fields [1-3]. However, the oxidation of C/C composites begins to be significant above about 773 K, which limits their application [4]. As a result, oxidation is a major problem for C/C composites for the applications as high-temperature structural materials. Coating is an efficient method for protecting C/C composites used in high-temperature environment [5,6].
MoSi2 is an excellent material for oxidation resistant coating because of the self-sealing performance at high temperatures [7]. SiC also has outstanding anti-oxidation properties due to the self-sealing performance of SiO2 formed from the oxidation of SiC. Previous studies [8-10] indicated that the multi-phase coatings containing MoSi2 and SiC were proved to have good oxidation protective ability for C/C composites. However, the following two factors may cause serious failure of the coatings, one is that long time service at high temperatures reduces the thickness of the coating by the volatilization of SiO2; the other is that SiO2 possesses low viscosity at high temperatures, meaning that gaseous products can escape from the SiO2 film easily and produce holes in the coating [11].
ZrO2 possesses high melting point, and can react with SiO2 to produce ZrSiO4 [12-14]. Zircon (ZrSiO4) is an excellent refractory material. It exhibits several attractive properties for high temperature structural applications such as high melting point, excellent chemical stability and low thermal expansion coefficient [15]. Therefore, if the MoSi2 coating is doped with ZrO2, the stability of the glassy SiO2 at high temperatures would be improved, which is advantageous to the oxidation resistance of MoSi2 coating. Moreover, few literatures have concerned about introducing ZrO2 in MoSi2 coating. In this work, a SiC-MoSi2/ZrO2-MoSi2 coating was designed and prepared by a two-step method of pack cementation and slurry coating. The phase compositions and microstructures of the as-prepared SiC- MoSi2/ZrO2-MoSi2 multilayer coating were characterized.
Meanwhile, the anti-oxidation properties of the multi- layer coating were also investigated.
2 Experimental
Small specimens (10 mm×10 mm×10 mm) used as substrates were cut from bulk 2D C/C composites with a density of 1.70 g/cm3. These specimens were hand-polished using 100 and 400 grit SiC paper in turn, then cleaned ultrasonically with ethanol to remove any loose particles and dried at 373 K for 2 h.
The SiC-MoSi2/ZrO2-MoSi2 coating was prepared by a two-step technique of pack cementation and slurry coating. In the first step, Si, C and Mo were used as raw materials, the SiC-MoSi2 inner layer was prepared by pack cementation. The powder compositions were as follows: 50%-60% Si, 10%-25% graphite, 15%-25% Mo and 5%-15% Al2O3 (mass fraction). The first pack mixtures and C/C specimens were put in a graphite crucible, and then were heated to 2073-2273 K and held for 2-4 h in argon atmosphere to form a SiC-MoSi2 inner coating. In the second step, the ZrO2-MoSi2 outer layer was prepared by slurry coating. The powders for preparing the outer layer were composed of Si, Mo and ZrO2. Using PVA (polyvinyl alcohol) as dispersant, the above powders were mixed ultrasonically in a beaker. The as-obtained slurry was brushed directly on the surface of SiC-MoSi2 coated samples. The samples were heat treated at 2173-2473 K for 2 h in an argon protective atmosphere to form the ZrO2-MoSi2 outer coating. For comparison, SiC-MoSi2/MoSi2 coating was prepared by the same pack cementation and slurry coating.
To investigate the oxidation process of the as-coated samples, the oxidation test was carried out at 1773 K in air in an electrical furnace. The samples were weighed at room temperature by electronic balance with a sensitivity of ±0.1 mg. The anti-oxidation ability was evaluated by mass loss.
The phase composition, morphology and element distribution of the coating were analyzed by Rigaku D/max-3C X-ray diffraction (XRD) and JSM-6460 scanning electron microscopy (SEM) with energy dispersive spectroscopy (EDS).
3 Results and discussion
3.1 Structure and morphology of SiC-MoSi2/ ZrO2-MoSi2 coating
Figure 1 shows the SEM image and XRD pattern of the SiC-MoSi2 inner coating prepared by pack cementation. It can be seen that the inner layer obtained by the first step pack cementation is dense except the existence of several microcracks (see Fig. 1(a)). Although these cracks could be self-sealed partially during the high temperature oxidation test, C/C matrix would be oxidized by oxygen diffusing through the cracks at the temperature between the crack sealing temperature and the original oxidizing temperature of C/C composites. From Fig. 1(b), it can be seen that the inner coating is composed of MoSi2, SiC and a small quantity of Si.
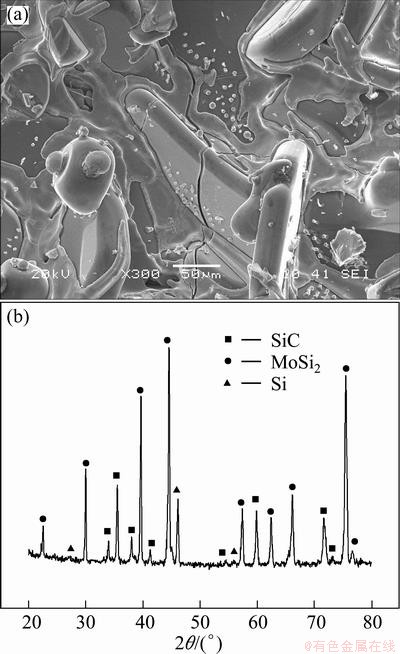
Fig. 1 SEM image (a) and XRD pattern (b) of SiC-MoSi2 inner coating prepared by pack cementation
Figure 2 shows the SEM image and XRD pattern of the ZrO2-MoSi2 outer coating prepared by slurry coating. As shown in Fig. 2(a), the coating has no big-size crack, while it is a little loose, owing to the volatilization of the dispersants that left holes during the process of sintering reaction at high temperatures. From Fig. 2(b), it can be seen that the coating is composed of ZrO2, MoSi2, SiC and Si. MoSi2 is formed by the reaction between Si and Mo. In addition, no Mo but a small quantity of Si is detected, meaning that Mo has been consumed completely during the process of heat treatment.
Figure 3 shows the cross-section of the SiC-MoSi2/ZrO2-MoSi2 coating prepared by the two-step technique of pack cementation and slurry coating. It can be observed that the thickness of the coating is about 250-300 μm, and the coating is dense and crack-free. The EDS analysis shows that the white phase can be distinguished as the mixture of ZrO2 and MoSi2.
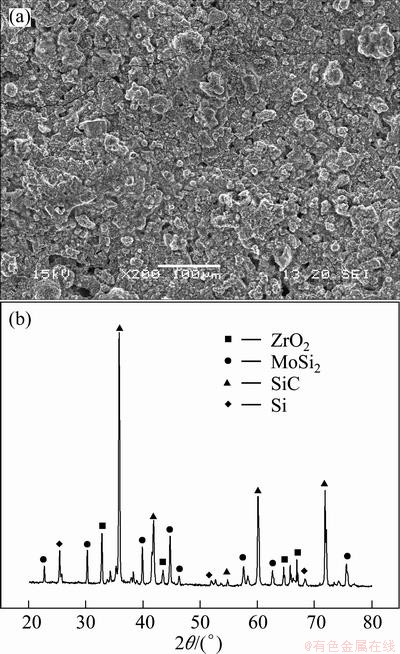
Fig. 2 SEM image (a) and XRD pattern (b) of ZrO2-MoSi2 outer coating prepared by slurry coating

Fig. 3 SEM image showing cross-section of SiC-MoSi2/ ZrO2-MoSi2 coating prepared by two-step technique of pack cementation and slurry coating
3.2. Oxidation process analyses of SiC-MoSi2/ZrO2- MoSi2 coating
Figure 4 shows the isothermal oxidation curves of the coated C/C samples at 1773 K in air. During the oxidation test, the coating mainly undergoes the following four chemical equations:
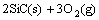 →
→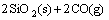 (1)
(1)
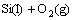 →
→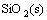 (2)
(2)
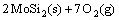 →
→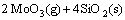 (3)
(3)
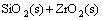

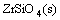 (4)
(4)
Figure 4 represents the isothermal oxidation curves of the C/C samples with SiC-MoSi2/MoSi2 coating and SiC-MoSi2/ZrO2-MoSi2 coating. As shown in Fig. 4(a), it can be seen that the oxidation process of the SiC-MoSi2/MoSi2 coated C/C composites could be divided into three stages. At the initial stage of oxidation (less than 15 h), the coated sample gains mass rapidly. After a short period of platform (between 15 and 20 h), the coated sample starts to exhibit the trend of mass loss. After oxidation at 1773 K in air for 160 h, the mass loss of the coated sample is up to 2.42%. As shown in Fig. 4(b), it can be seen that the SiC-MoSi2/ZrO2-MoSi2 coated sample loses only 1.31% mass after exposure to air at 1773 K for 260 h. That is to say, the introduction of ZrO2 could improve the oxidation resistance of SiC-MoSi2/MoSi2 coating. Compared with Fig. 4(a), it can also be found that the oxidation process of SiC-MoSi2/ZrO2-MoSi2 coated sample is similar to that of SiC-MoSi2/MoSi2 coated sample. The main difference is focused on the following two aspects, one is that the period of platform that the mass basically remains unchanged is much longer (between 20 and 116 h); the other is that the coated sample loses mass relatively slowly in the whole process, which suggests that the coating can protect the C/C substrate from oxidation at that temperature.
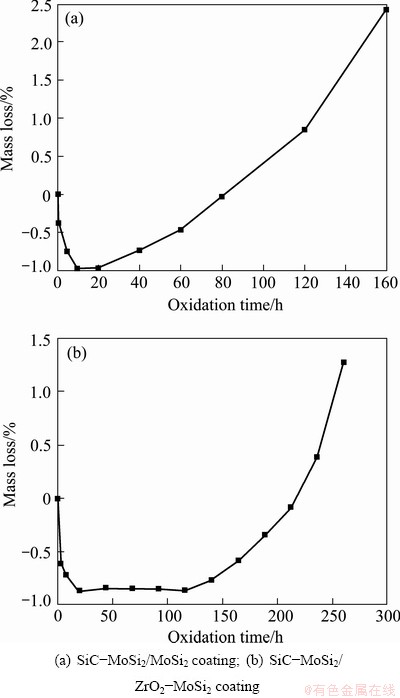
Fig. 4 Isothermal oxidation curves of coated C/C samples at 1773 K in air
Figure 5 shows the XRD pattern of SiC-MoSi2/ ZrO2-MoSi2 coating after oxidation at 1773 K. From Fig. 5, it can be seen that the coating is composed of SiO2, ZrSiO4, ZrO2 and SiC. ZrSiO4 phase forms due to the reaction between ZrO2 and SiO2. SiO2 might come from the oxidation of SiC, MoSi2 and Si (Eqs. (1)-(3)). The reason that only a small quantity of SiO2 is detected could be explained as follows. At about 1773 K, SiO2 transforms into amorphous glass state. During the oxidation test, the sample is taken out of the furnace directly into air within several seconds for weighing. The cooling rate from 1773 K to room temperature is very quick. Owing to this quick cooling, the majority of the glassy SiO2 is not able to crystallize before solidification. From Fig. 5, it can also be observed that a large quantity of ZrO2 is detected while the amount of ZrSiO4 is little. This phenomenon could be explained by Gibbs free energy in Eq. (4). DG of the reaction is calculated by thermodynamic equations [16,17] described as follows:
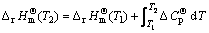 (5)
(5)
 (6)
(6)
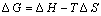 (7)
(7)
where 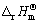 and
and  are the enthalpy of formation and standard molar entropy change, respectively; T1 is the room temperature and T2 is the processing temperature;
are the enthalpy of formation and standard molar entropy change, respectively; T1 is the room temperature and T2 is the processing temperature; 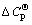 is the molar heat capacity at constant pressure which stands for the difference between the processing temperature and the room temperature. The value of DG is about -502.6 J/mol at 1773 K, which indicates that the reaction possesses weak spontaneity from the point of thermodynamics, so the amount of ZrSiO4 is little.
is the molar heat capacity at constant pressure which stands for the difference between the processing temperature and the room temperature. The value of DG is about -502.6 J/mol at 1773 K, which indicates that the reaction possesses weak spontaneity from the point of thermodynamics, so the amount of ZrSiO4 is little.
Figure 6 represents the SEM micrographs of the C/C samples with SiC-MoSi2/MoSi2 coating and SiC-MoSi2/ZrO2-MoSi2 coating. It can be observed that the surfaces of the above two coatings have changed into glass due to the formation of SiO2. Form Fig. 5, the peak of MoSi2 in SiC-MoSi2/ZrO2-MoSi2 coating disappears, indicating that MoSi2 is oxidized completely and produces SiO2 and MoO3 (Eq. (3)). However, the MoO3 phase is not detected due to the MoO3 (melting point of 1068 K) evaporation at this temperature, resulting in some holes on the coating, as shown in Fig. 6 [18]. At 1773 K, CO2 and CO generated from the oxidation of SiC and the C/C matrix would get out through the SiO2 film because of the low viscosity of the glass layer, which might also result in the formation of holes (see Fig. 6). These holes provide the entrance channels for oxygen to diffuse into the C/C matrix. Compared with the SiC-MoSi2/MoSi2 coated sample (see Fig. 6(a)), the size of the holes in SiC-MoSi2/ZrO2-MoSi2 coating obviously decreases (see Fig. 6(b)). From the above analysis, it can be concluded that by adding ZrO2 in MoSi2 coating, the stability of the glassy SiO2 film at high temperature can be reinforced, and the size of holes in the film is also reduced. The existence of ZrSiO4 phase can enhance the anti-oxidation performance, which might be attributed to the following aspects: 1) the thermal expansion coefficient of ZrSiO4 is close to that of SiC, which can eliminate the thermal expansion mismatch between glassy SiO2 and ZrSiO4; 2) the ZrSiO4 phase might have pining effect on the melting-point (2550 °C) in the case of the fact that the SiO2 glaze vaporizes fast [18]. This may be the reason that the oxidation protection for MoSi2-SiC/ZrO2-MoSi2 coating is superior to that for MoSi2-SiC/MoSi2 coating.
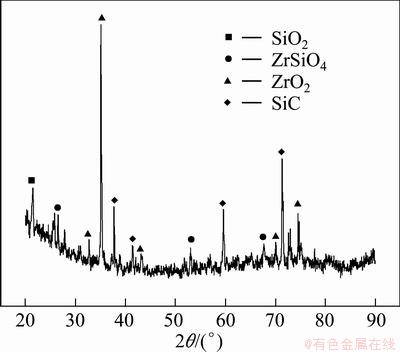
Fig. 5 XRD pattern of surface of SiC-MoSi2/ZrO2-MoSi2 coated C/C sample after oxidation at 1773 K in air for 260 h

Fig. 6 SEM images of coatings after oxidation at 1773 K in air for different time
4 Conclusions
1) A dense and crack-free MoSi2-SiC/ZrO2-MoSi2 coating for C/C composites is prepared by a two-step technique of pack cementation and slurry coating. The as-prepared coating has good oxidation protective ability and can efficiently protect C/C composites against oxidation at 1773 K for 260 h.
2) Owing to the generation of ZrSiO4 through the reaction between ZrO2 and SiO2, the stability of the glassy SiO2 film at high temperatures can be efficiently reinforced, which increases the oxidation resistance of the coating.
References
[1] SHEEHAN J E, BUESKING K W, SULLIVAN B J. Carbon-carbon composites [J]. Annual Review of Materials Science, 1994, 24: 19-44.
[2] PARK S J, SEO M K, LEE D R. Studies on the mechanical and mechanical interfacial properties of carbon-carbon composites impregnated with an oxidation inhibitor[J].Carbon, 2003, 41(15): 2991-3002.
[3] FITZER E. The future of carbon-carbon composites [J]. Carbon, 1987, 25(2): 163-190.
[4] JACOBSON N S, CURRY D M. Oxidation microstructure studies of reinforced carbon/carbon [J]. Carbon, 2006, 44(7): 1142-1150.
[5] HUANG J F, LI H J, XIONG X B, ZENG X R, LI K Z, FU Y W. Progress on the oxidation protective coating of carbon-carbon composites [J]. New Carbon Materials, 2005, 20(4): 373-379.
[6] YAN Zhi-qiao, XIONG Xiang, XIAO Peng, CHEN Feng, ZHANG Hong-bo, HUANG Bai-yun. Oxidation behavior of oxidation protective coatings for C/C-SiC composites at 1500 °C [J]. Transactions of Nonferrous Metals Society of China, 2009, 19(1): 61-64.
[7] LIU R J, ZHOU X G, ZHANG C R, ZHOU A C. The oxidation resistant MoSi2-SiC-Si coating for Cf/SiC ceramic matrix composites prepared by cementation [J]. Technique Aerospace Materials and Technology, 2000, 2(3): 45-48.
[8] JIAO G S, LI H J, LI K Z, WANG C, HOU D S. SiC-MoSi2-(Ti0.8Mo0.2)Si2 multi-composition coating for carbon/carbon composites [J]. Surface and Coatings Technology, 2006, 201(6): 3452-3456.
[9] FU Q G, LI H J, LI K Z, SHI X H, HU Z B, HUANG M. SiC whisker-toughened MoSi2-SiC-Si coating to protect carbon/carbon composites against oxidation [J]. Carbon, 2006, 44(9): 1866-1869.
[10] CHEN Y, WANG C G, ZHAO W, LU W B, CHEN A T, TAN T T. Fabrication of a SiC/Si/MoSi2 multi-coating on graphite materials by a two-step technique [J]. Ceramics International, 2012, 38(3): 2165-2170.
[11] FU Q G, LI H J, LI K Z, SHI X H, HUANG M. A MoSi2-SiC-Si/glass oxidation protective coating for carbon/carbon composites [J]. Carbon, 2006, 44(15): 3361-3364.
[12] GAO Y. Reaction of ZrO2-CaO-ZrSiO4 and ZrO2-Y2O3-ZrSiO4 detonation thermal sprayed coatings with manganese oxide at 1273 K [J]. Surface and Coatings Technology, 2005, 195(2-3): 320-324.
[13] HUANG J F, ZENG X R, LI H J, XIONG X B, SUN G L. ZrO2-SiO2 gradient multilayer oxidation protective coating for SiC coated carbon/carbon composites[J]. Surface and Coatings Technology, 2005, 190(2-3): 255-259.
[14] ARNO KAISER, MARKUS LOBERT, RAINER TELLE. Thermal stability of zircon (ZrSiO4) [J]. Journal of the European Ceramic Society, 2008, 28(11): 2199-2211.
[15] ZHANG Cheng-zhong, YOU Jing-lin, CHEN Hui, ZENG Hao, JIANG Guo-chang. Vibrational modes and in-situ high temperature raman spectra of zircon [J]. Journal of the Chinese Ceramic Society, 2006, 34(10): 1172-1176. (in Chinese)
[16] JARUNGTHAMMACHOTE S, DUTTA A. Equilibrium modeling of gasification: Gibbs free energy minimization approach and its application to spouted bed and spout-fluid bed gasifiers [J]. Energy Conversion and Management, 2008, 49(6): 1345-1356.
[17] VALERO A, VALERO A, VIEILLARD P. The thermodynamic properties of the upper continental crust: Exergy, Gibbs free energy and enthalpy [J]. Energy, 2012, 41(1): 121-127.
[18] ZHANG Wu-zhuang, ZENG Yi, GBOLOGAH L, XIONG Xiang, HUANG Bai-yun. Preparation and oxidation property of ZrB2-MoSi2/SiC coating on carbon/carbon composites [J]. Transactions of Nonferrous Metals Society of China, 2011, 21(7): 1538-1544.
C/C复合材料表面SiC-MoSi2/ZrO2-MoSi2抗氧化涂层
付前刚,张佳平,张正中,李贺军,孙 粲
西北工业大学 凝固技术国家重点实验室,C/C复合材料工程技术研究中心,西安 710072
摘 要:为了提高C/C复合材料在高温空气中的抗氧化性能,采用包埋法和涂刷法在C/C复合材料表面制备MoSi2-SiC/ZrO2-MoSi2涂层。借助扫描电镜和X射线衍射等测试手段对涂层的微观形貌和相组成进行分析,同时研究涂层的制备过程和高温静态空气中的抗氧化性能。结果表明:SiC-MoSi2/ZrO2-MoSi2涂层致密且无穿透性裂纹,厚度为250~300 μm,具有优异的抗氧化性能,在1773 K静态空气中的有效抗氧化时间为260 h,这主要是由于涂层中生成了ZrSiO4,有效提高了涂层的高温稳定性。
关键词:炭/炭复合材料;SiC-MoSi2/ZrO2-MoSi2;涂层;抗氧化性能
(Edited by Jing-hua FANG)
Foundation item: Projects (51221001, 51222207) supported by the National Natural Science Foundation of China; Project (090677) supported by the Program for New Century Excellent Talents in University of China; Project (B08040) supported by Program of Introducing Talents of Discipline to Universities, China
Corresponding author: Qian-gang FU; Tel: +86-29-88494197; Fax: +86-29-88495764; E-mail: fuqiangang@nwpu.edu.cn
DOI: 10.1016/S1003-6326(13)62704-1