网络首发时间: 2017-09-30 11:28
稀有金属 2018,42(05),546-554 DOI:10.13373/j.cnki.cjrm.xy17040020
基于AB5合金复合贮氢材料的研究进展
罗龙 吴文远 边雪 李培玉 齐健博
东北大学冶金学院
内蒙古科技大学分析测试中心
摘 要:
制备复合贮氢材料是改善贮氢合金性能, 克服单一合金缺点的一条有效途径。基于AB5贮氢合金的特点, 对利用AB5合金制备复合贮氢材料等方面的研究进行了总结。AB5贮氢合金一般具有CaCu5晶体结构, 具有相对较高的吸氢容量、良好的循环稳定性、低平衡压、吸氢动力学快和良好的抗杂质性等优点, 尤其与其他贮氢材料进行复合作为燃料电池用贮氢罐的候选材料, 具有很好的开发应用前景。主要涉及了AB5/AB2, AB5/Mg (Mg基) 、AB5/V-Ti, AB5/碳材料等二元复合贮氢材料的研究进展。阐述了各种复合材料的相组成、微观结构、贮氢性能以及作用机制等, 并对今后复合贮氢材料的研究方向进行了展望, 如利用材料计算方法对复合材料热力学、动力学等方面进行理论计算、新制备工艺开发、AB5合金与多元体系贮氢材料进行复合。
关键词:
AB5合金;复合贮氢材料;贮氢性能;晶体结构;
中图分类号: TG139.7
作者简介:罗龙 (1983-) , 男, 内蒙古乌兰察布人, 博士研究生, 讲师, 研究方向:贮氢合金;E-mail:ljd0928@aliyun.com;;吴文远, 教授;电话:024-83680527;E-mail:wuwy@smm.neu.edu.cn;
收稿日期:2017-04-11
基金:国家自然科学基金项目 (51274060);内蒙古自然科学基金项目 (2014MS0529);基本科研业务费项目 (N150204019);内蒙古科技大学创新基金项目 (2014QDL048) 资助;
Research Progress of Composite Hydrogen Storage Materials Based on AB5 Alloy
Luo Long Wu Wenyuan Bian Xuan Li Peiyu Qi Jianbo
School of Metallurgy, Northeastern University
Analytical and Testing Center, Inner Mongolia of Science and Technology
Abstract:
Preparation of composite hydrogen storage materials is an effective way to improve the properties of hydrogen storage alloys and overcome the shortcomings of single alloys. Based on the characteristics of AB5 hydrogen storage alloys, the preparation of composite hydrogen storage materials by AB5 alloy was summarized. AB5 hydrogen storage alloys with a Ca Cu5 crystal structure had relatively high hydrogen absorption capacity, good cycle stability, low equilibrium pressure, hydrogen absorption kinetics and good anti impurity.In particular, composite hydrogen storage materials based on AB5 alloy can be used as a candidate material for hydrogen storage tanks for fuel cells, and have a good prospect of development and application. This research progress of AB5/AB2, AB5/Mg ( Mg-based) , AB5/V-Ti, AB5/carbon materials and other two component composite hydrogen storage material was summarized. The phase composition, microstructure, hydrogen storage properties and mechanism of the composites were discussed. The future research directions of composite hydrogen storage materials were prospected, such as the materials computation methods used to calculate the thermodynamics and dynamics of composite materials, new process development and multi system composite hydrogen storage materials based on AB5 alloy.
Keyword:
AB5 alloy; composite hydrogen storage materials; hydrogen storage properties; crystal structure;
Received: 2017-04-11
氢是清洁无污染, 可再生的能源[1], 能量密度在120~142 m J・kg-1之间, 是石油的三倍[2], 被认为是解决逐渐枯竭的化石燃料和保护地球环境很好的替代能源, 从使用角度来说, 氢的存贮是“氢经济”的重点, 所以, 储氢材料在用于设计未来的清洁能源系统方面吸引了越来越多的兴趣[3]。目前, 一般市售的吸氢金属间化合物是AB5型合金, 也是目前研究最为系统的合金[4], 原型是La Ni5, 一般具有Ca Cu5晶体结构 (图1 (a) [5]) , 吸氢后形成La Ni5H6, 晶体结构如图1 (b) [6], 体积贮氢容量为0.115 g・cm-3[7], 质量贮氢容量为1.4%[8]。AB5合金具有相对较高的吸氢容量、良好的循环稳定性、低平衡压、吸氢动力学快和良好的抗杂质性[9]等优点;但是, 由于Ca Cu5型六方晶体结构的限制, AB5型合金容量较少 (<1.5% (质量分数) ) 并且材料相对较贵[10], 达不到美国能源部 (DOE) 设定的2017年目标质量贮氢容量为5.5%, 体积贮氢容量为40 kg・m-3[11]的要求, 成为制约AB5合金应用的瓶颈。
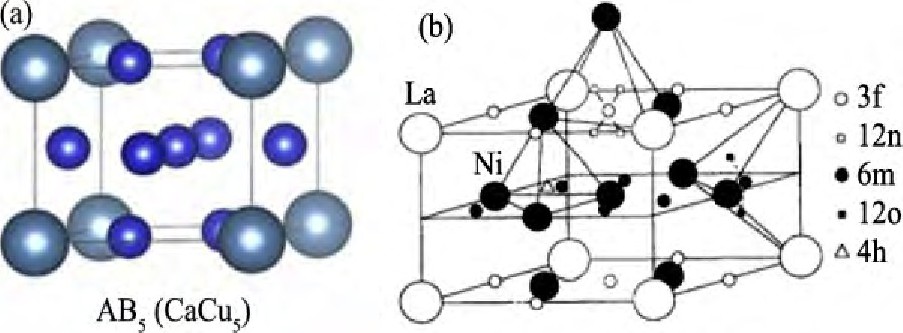
图1 La Ni5结构, La Ni5H6结构Fig.1 Structures of La Ni5[5] (a) and La Ni5H6[6] (b)
制备复合材料是改善贮氢材料性能的一条有效途径[12], 一些研究发现复合后的贮氢合金的容量远远大于单相合金[13]。本文对国内外近几年基于AB5合金的复合贮氢材料的研究动态及发展现状进行了概述, 并对今后发展方向进行了展望。
1 AB5与AB2复合
AB2型Ti基或Zr基Laves相合金, 由于其在室温下具有较大贮氢容量 (2.0%) [9], 也是目前Ni-MH电池负极的一种候选材料。但是由于其表面容易氧化以及合金表面电化学催化活性差, 致使该合金活化相对困难。为了改善这一状况, 孙昊等[14]将混合合金球磨40 h后, 制备了复合合金, 最大放点容量达到669.7 m Ah・g-1, 包覆于合金表面的镍不但对合金性能起到催化作用, 提高了合金的表面活性, 还有效地提高了合金的综合储氢性能;韩树民等[15]机械球磨Zr0.9Ti0.1 (Mn0.35Ni0.50V0.15) 2与10%AB5, 制备了复合合金, 生成了许多新鲜表面, 另外, 一些AB5颗粒被粘附在AB2颗粒表面。AB2-AB5复合合金的活化周期从14次减少到7次, 最大放电容量从330 m Ah・g-1增加到347 m Ah・g-1, 增加了5.2%。Chu等[16]将AB2合金Ti0.9Zr0.2Mn1.5Cr0.3V0.3与10%La Ni3.8Mn0.3Al0.4Co0.5机械球磨, 经过2 h球磨后, 通过扫描电镜 (TEM) 观察发现, AB5细颗粒分散在主体颗粒上;最大放电容量达到310.4 m Ah・g-1, 相比原始Ti0.9Zr0.2Mn1.5Cr0.3V0.3电极放电容量48.6 m Ah・g-1, 有了极大的提高;阳极极化测试发现提高了合金中氢原子的扩散。除了采用机械球磨制备AB2-AB5复合合金以外, Han等[17]采用烧结La Ni5和Zr0.9Ti0.1 (Mn0.35Ni0.55V0.15) 2混合粉末, 制备了La Ni5-x%AB2复合合金, 合金最大放电容量从188 m Ah・g-1 (x=0) 提高到305 m Ah・g-1 (x=10) , 同时复合合金相对于La Ni5, 具有更好的低温电化学性能。之后, 该研究者又通过对AB2型Zr0.9Ti0.9Ni0.1Mn0.6V0.3和La Ni5熔炼处理进一步对AB2-x%La Ni5复合贮氢合金进行了研究[18], 证实了La Ni5的添加提高了AB2合金的活化性能。综上研究分析认为, AB5型合金不仅本身具有贮氢性能, 而且也对氢化和氢化物分解过程具有催化作用, 所以有利于改善AB2合金的活化性能。
2 AB5与Mg (Mg基) 复合
镁基合金在过去的30年中作为贮氢材料被广泛研究[19], 主要基于以下原因: (1) 具有良好的吸氢性能; (2) 是轻质固态贮氢材料[20]; (3) 贮氢容量达到7.6%[21,22]。更重要的是Mg价格便宜, 地壳中含量丰富[23]。镁基合金具有良好的耐热性, 容易回收利用, 能够在平衡状态下与其它元素形成固溶体和化合物[24]。
然而, 因为Mg-H之间的强键合, 镁基合金热力学及动力学性能很差[25], 限制了实际应用。研究人员基于优势互补原则将其与AB5进行复合, 取得了很大效果。
2.1 AB5/Mg复合材料
Raman等[26]采用射频感应熔炼制备了Mg-x%CFMm Ni5复合贮氢材料, 其中合金 (x=30) 具有比无铈Mm Ni5更好的吸放氢动力学性能, 贮氢量达到5.6%。Davidson等[27]采用高能球磨制备了Mgx%Mm Ni4.6Fe0.6复合贮氢合金, 合金 (x=30) 在350℃吸氢5.0%, 吸放氢速度达到80 cm3・min-1。Zhu等[28]采用高能球磨制备了纳米复合材料Mm Ni5-x Mx-Mg, 球磨20 h后, 复合材料由Mm Ni5-xMx, Mg, Mm2Mg173个相组成, Mm Ni5-x Mx-10%Mg复合材料比Mm Ni5-xMx吸氢快十倍, Mm Ni5-xMx-30%Mg复合材料吸氢量达到2.3%, 远远高于Mm Ni5-xMx的1.25%;之后又通过研究[29]证明了形成纳米复合材料是提高吸氢性能的有效途径。Liang等[30]采用机械球磨Mg-50%La Ni5制备了纳米复合材料, 在低温下具有良好的吸氢动力学, 在302 K, 1.5 MPa氢压下500 s吸氢2.5%, 温度在523 K以上, 吸氢4.1%, 这与Delchev等[31]的研究结果基本一致。Vijay等[32]同样采用机械球磨制备了纳米尺度的Mg-x%Mm Ni5混合物, 氢气气氛下球磨12 h后, Mg-10%Mm Ni5在100℃吸氢5.1%, 研究表明纳米结构的Mg与分散在Mg晶粒表面和晶界的Mm Ni5加强了吸氢性能, Mm Ni5可能起到氢扩散到Mg晶粒的通道作用。Gu等[33]采用氢化燃烧合成Mg-30%La Ni5, 随后机械球磨, 制备了复合材料, 373 K吸氢3.8%, 而采用单一机械球磨或氢化燃烧合成制备的复合材料都低于1.5%;脱氢开始温度为470 K, 低于单一氢化燃烧合成的放氢开始温度;放氢速度比单一球磨或氢化燃烧合成制备的复合材料都快。Spassov等[34]研究了不同球磨条件下制备的Mg-10% (原子分数) La Ni5复合纳米材料的吸氢动力学性能, 研究发现Mg H2+La Ni5和Mg+La Ni5干磨制备的材料比Mg+La Ni5在庚烷下球磨制备的材料吸放氢动力学要快, 达到0.035%・s-1~0.4%・s-1 (质量分数) , 原因是干磨得到的复合材料粒度更细。伟伟等[35]在商业化AB5中加入少量Mg (≤7%) 进行机械合金化, 发现合金的最大容量、放电性能以及循环稳定性都变得更好。
2.2 AB5/Mg2Ni复合材料
除了以上纯Mg与AB5形成复合材料外, 许多研究人员将Mg基合金Mg2Ni与AB5复合制备了不同的贮氢材料, 取得了许多有益的效果。Zhu等[36]采用机械球磨制备了Mg2Ni/Mm Ni5-x (Co Al Mn) x纳米相复合材料, 放电容量大于单相合金Mg2Ni和Mm Ni5-x (Co Al Mn) x的简单叠加, 存在纳米复合增强效应, 晶粒尺度小于100 nm时, 增强效应最强, 放电容量最大。Pu等[37]采用氢化燃烧合成了Mg2Ni H4, 然后与x%Mm Ni3.8Co0.75Mn0.4Al0.2机械球磨5 h制备了复合贮氢材料, 随着AB5含量的增加, 复合材料的容量先减小后增大, 最高达到509 m Ah・g-1 (x=40) ;复合材料的动力学性能明显提高, 分析认为AB5合金与原位形成的Ni颗粒之间的协同催化效应起到了积极作用。王琳瑛等[38]同样选择Ml Ni3.8Co0.75Mn0.4Al0.2和Mg2Ni合金球磨复合, 其中添加30%AB5制备的复合材料电化学反应催化活性明显提高, 电化学性能改善效果最佳。除以上AB5来改善Mg2Ni的复合贮氢材料外, 另外的一些研究人员用Mg2Ni反过来改善AB5, 制备复合材料。Liu等[39,40]采用二步重熔法制备了Mm0.3Ml0.7Ni3.55Co0.75Mn0.4Al0.3/x%Mg2Ni新型复合合金, 合金的活化性能及最大容量随Mg2Ni的含量先增加后减小, 复合材料中Mg2Ni含量为5%时, 室温下活化性能 (Cn=6) 与最大容量 (274.4 m Ah・g-1) 最好。田晓等[41,42]在Liu[37,38]研究的基础上, 分别研究了快淬速度及退火温度对Mm0.3Ml0.7Ni3.55Co0.75Mn0.4Al0.3/5%Mg2Ni复合合金结构和电化学性能的影响, 研究表明快淬及退火处理后, 复合合金都由 (La, Mg) Ni3新相和La Ni5相组成, 当快淬速度为20 m・s-1时合金最大放电容量达到344 m Ah・g-1;退火态合金的最大放电容量随退火温度的升高先增强后减弱, 其中退火温度为1023 K时, 合金容量303 m Ah・g-1。
3 AB5与V-Ti基复合
V-Ti基固溶体合金具有BCC结构, 具有较高的体积贮氢容量 (VH2为0.16 g・cm-3, 大约为液体氢的2.25倍[43], La Ni5H6为0.115 g・cm-3[7]) 和质量贮氢容量 (理论上为3.8%[8,44], La Ni5为1.4%[7]) , 氢化反应条件温和[45], 抗粉化性能好, 动力学性能优越[46]等优点。
尽管V基贮氢合金具有许多优点, 但是还存在如活化性能差、吸放氢平台倾斜、有效贮氢容量低、吸放氢平台滞后效应明显、循环稳定性差等问题, 达不到实际应用要求[47]。为了改善这些问题, 研究人员采用了多种手段来改善其吸放氢性能, 其中就包括与AB5形成新型复合材料。
王艳芝等在V基合金与AB5复合的研究中做了大量的工作[48,49,50,51,52,53,54], 其中采用两步电弧熔炼法制备的Ti0.10Zr0.15V0.35Cr0.10Ni0.30+x%AB5复合贮氢合金, 合金主相在添加AB5后并没有改变, 仍然是体心立方结构的钒基固溶体相和六方结构的C14Laves相, 复合过程中生成了第二相, 第二相的含量随添加AB5的增加而增加;复合过程存在明显的协同效应, 与基体合金相比, 复合材料电化学性能明显提高, 分析认为与第二相的形成有关。Yu等[55]采用机械球磨Ti-30V-15Mn-15Cr+20%La (Ni Mn Co Al) 5, 球磨时间30 min, 复合材料电极表现出更高的放电容量和更长的循环寿命, 这可能是由于La (Ni Mn Co Al) 5合金颗粒均匀分散在复合材料表面, 这些颗粒不仅起到电催化和微电流收集的作用, 而且阻止充放电循环过程中V溶解到碱性溶液中。这与其他研究人员[56,57]将V基合金与AB5球磨, 得到的改善效果基本一致。同样, 在AB5中加入少量纯钒形成的复合贮氢合金的性能同样优于单一AB5。Kumar等[58]将La Ni5+20%V粉末在高纯氩气下高能球磨4 h, 制备的复合材料吸氢容量及动力学性能比La Ni5要高, 其中动力学性能的提高主要与晶格膨胀、费米能级降低及纯钒催化性能有关。
AB5与其他合金形成复合合金的制备主要是通过电弧熔炼、感应熔炼或者机械球磨方法, 制备方法不同得到的合金性能差别较大, 详见表1[59,60], 通过研究分析发现, 恰当的制备工艺可以改变复合合金表面或内部的组织, 从而改善复合合金性能;增加适当的热处理可能会进一步改善复合合金性能。同时要考虑到, 热处理会造成某种元素一定程度的挥发损耗, 一般来讲, 时间越长, 温度越高, 损耗越大, 对合金性能影响越大。
4 AB5与碳材料复合
最近, 因为碳材料 (如活性炭、碳纳米管和石墨纳米纤维) 高循环稳定性及快速吸氢动力学的特点, 其吸氢性能引起了研究人员的极大兴趣。碳材料吸氢是基于H2与材料表面之间较弱的范德华力, 由于氢分子与吸附表面之间的弱结合, 常温常压下存储的容量很低 (<1%) [61]。一些研究结果表明, 碳材料与吸氢合金结合, 是制备常温常压下具有良好吸氢性能复合材料的一条可行途径。Li等[62]首先在简单球磨条件下用金属镍粉对石墨进行改性, 然后与90%AB5合金混合成复合材料, 电化学测试结果显示, 复合材料与AB5相比充放电极化更低, 3C倍率放电容量提高22%, 氢扩散更快。之后, Li等[61]又在不同条件下用金属镍粉对活性炭进行改性, 将改性的活性炭与90%AB5合金混合成复合材料, 电化学测试结果显示, 利用金属镍对活性炭表面的电化学还原/氧化反应具有良好的催化作用, 水热法改性的活性炭与AB5形成的复合材料放电容量在1C和3C倍率条件下放电, 分别提高16%和59%。Ren等[63]采用浸渍还原法将10%MNi4.8Sn0.2 (M=La, Nd) 纳米颗粒负载于碳纳米管上, 制备出复合贮氢材料, 由于AB5颗粒与预处理碳纳米管之间的协同效应, 复合材料贮氢容量可以达到2.96%和2.88%, 是MNi4.8Sn0.2的3倍, 而且具有很好的循环稳定性。Cui等[64]为了改善商业化AB5合金的高倍率放电能力, 将粒度50μm的AB5合金粉与石墨烯纳米片进行球磨30 min, 得到复合贮氢材料电极在放电电流密度3000 m A・g-1下, 容量保持率为68.3%, 是单一合金电极的3.2倍。如此优越的性能是由于: (1) 合金较小的粒度减小了氢原子扩散的距离; (2) 石墨烯纳米片的高电导率加速了电荷转移; (3) 石墨烯纳米片与合金之间的互相联系降低了内阻。海山等[65]将AB5合金与10%多壁碳纳米管 (CNTs) 均匀混合后机械球磨制备了复合贮氢材料, 球磨后复合合金的最大放电容量等电化学性能均得到明显改善。分析认为, 球磨过程中细长的CNTs不断断裂, 缺陷增多, 比表面积增大, 有利于氢原子的进出、吸附和储存;同时, CNTs的氢催化作用和毛细管作用产生大量氢, 并形成较高的氢压, 使氢更容易进入合金颗粒内部, 即提高了电极表面的电荷迁移速率, 从而有利于储氢量的提高, 这与Tsai等[66]的研究结论基本一致。
表1 合金制备方法及特征Table 1 Preparation methods and characteristics[59,60] 下载原图
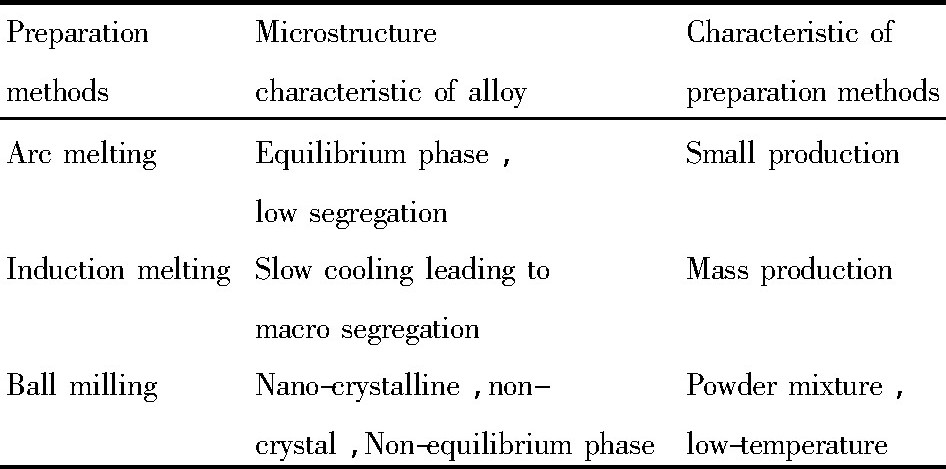
表1 合金制备方法及特征Table 1 Preparation methods and characteristics[59,60]
5 AB5与其他复合
除以上复合体系外, 研究人员出于不同的研究目的还将AB5合金与其他材料进行复合, 得到一些有益的结论。Lim等[67]为了提高La-Mg-Ni基合金的循环稳定性, 将商业AB5合金粉末与LaMg-Ni基合金粉混合成复合材料, 结果表明添加AB5的复合合金相比La-Mg-Ni基合金循环稳定性提高, 复合材料循环寿命的改善很可能归因于AB5和La-Mg-Ni基合金之间的负载分担。同样, Huang等[68]将商业AB5合金粉末与La-Mg-Ni基合金粉在氩气气氛下机械球磨1 h, 制备的复合材料循环稳定性也明显提高, 与参考文献[67]的结论一致。Zhang等[69]采用烧结AB5和La Mg3制备复合材料, 其中烧结温度为1123 K, AB5-5%La Mg3的复合材料放电容量为358 m Ah・g-1, 容量比商业化AB5高7.2%。为了进一步改善AB5合金的性能, 研究人员将有机物[70,71], Pd[72,73,74,75], Co[76], Cu[77], Ni[78]等与AB5采用不同的制备方法得到复合贮氢材料, 电化学性能均有所改善。
6 结语
AB5合金存在诸如良好的循环稳定性、低平衡压、吸氢动力学快和良好的抗杂质性等优点;但是, 合金容量较少、材料相对较贵成为其大规模应用难以突破的瓶颈。要想取得进一步发展, 制备复合材料是简单有效的方法。近十多年来, AB5合金在复合贮氢材料中的应用研究取得了丰富成果, 但是离大规模实际应用还有一段距离, 需要从以下几个方面做进一步研究: (1) 更多的利用材料计算方法对复合材料热力学、动力学等方面进行理论计算, 为实验提供理论指导。 (2) 探索新的复合材料制备方法, 适当增加合理的热处理。 (3) 改进已有制备工艺, 同时考虑提高贮氢性能及降低材料成本。 (4) 将AB5合金与多元体系贮氢材料进行复合。
参考文献
[1] Lin H C, Lin K M, Wu K C, Hsiung H H, Tsai H K.Cyclic hydrogen absorption-desorption characteristics of Ti Cr V and Ti0.8Cr1.2V alloys[J].International Journal of Hydrogen Energy, 2007, 32 (18) :4966.
[2] Ouyang L Z, Huang J M, Wang H, Wen Y J, Zhang Q A, Sun D L, Zhu M.Excellent hydrolysis performances of Mg3RE hydrides[J].International Journal of Hydrogen Energy, 2013, 38 (7) :2973.
[3] Lin J, Lu C, Sun L S, Liang F, Cao Z Y, Wang L M.Ti-V-Ni with graphene-mixing icosahedral quasicrystalline composites:preparation, structure and its application in Ni-MH rechargeable batteries[J].International Journal of Hydrogen Energy, 2016, 41 (2) :1098.
[4] Godula-Jopek A, Jehle W, Wellnitz J.Hydrogen storage technologies[J].Wiley-VCH, 2012, 90 (5) :3.
[5] Liu W, Webb C J, Gray E, Mac A.Review of hydrogen storage in AB3alloys targeting stationary fuel cell applications[J].International Journal of Hydrogen Energy, 2016, 41 (5) :3485.
[6] Yasuda K, Brodowsky H.Fermi energy effects on absorption of hydrogen in La Ni5and La Co5alloys[J].Journal of Alloys and Compounds, 1995, 231:454.
[7] Chen P, Zhu M.Recent progress in hydrogen storage[J].Mater Today, 2008, 11 (12) :36.
[8] Pei P, Zhang P L, Zhang B, Song X P.V based hydrogen storage alloys and alloying research[J].Materials Review, 2006, 20 (10) :123. (裴沛, 张沛龙, 张蓓, 宋西平.V系储氢合金及其合金化[J].材料导报, 2006, 20 (10) :123.)
[9] Rusmana N A A, Daharib M.A review on the current progress of metal hydrides material for solid-state hydrogen storage applications[J].International Journal of Hydrogen Energy, 2016, 41 (28) :12108.
[10] Pan H, Liu Y, Gao M, Zhu Y, Lei Y.The structural and electrochemical properties of La0.7Mg0.3 (Ni0.85Co0.15) x (x=3.0~5.0) hydrogen storage alloys[J].International Journal of Hydrogen Energy, 2003, 28 (11) :1219.
[11] Cao Z J, Ouyang L Z, Wang H, Liu J W, Sun D L, Zhang Q A, Zhu M.Advanced high-pressure metal hydride fabricated via Ti-Cr-Mn alloys for hybrid tank[J].International Journal of Hydrogen Energy, 2015, 40 (6) :2717.
[12] Xu X Q, Cui J D, Zhang Y, Yang Z M.Preparation and hydrogen sorption performance of porous titanium getter[J].Chinese Journal of Rare Metals, 2017, 41 (10) :1125. (徐晓强, 崔建东, 张艳, 杨志民.多孔Ti吸气材料的制备及吸氢性能研究[J].稀有金属, 2017, 41 (10) :1125.)
[13] Wang Z M, Peng C H, Ouyang L Z, Li Z X, Zhu M.Enhancement of electrochemical characteristics of Mg2Ni/Mm Ni5-x (Co Al Mn) xby forming nano-phase[J].Acta Metallurgica Sinica, 2002, 38 (2) :189. (王仲民, 彭成红, 欧阳柳章, 李祖鑫, 朱敏.纳米化对Mg2Ni/Mm Ni5-x (Co Al Mn) x复合储氢合金电极特性的影响[J].金属学报, 2002, 38 (2) :189.)
[14] Sun H, Feng D C, Ren H P, Zhang Y H.Hydrogen storage properties of Mg22Y2Ni10Cu2+xwt.%Ni composite prepared by ball milling[J].Chinese Journal of Rare Metals, 2016, 40 (6) :284. (孙昊, 冯佃臣, 任慧平, 张羊换.球磨Mg22Y2Ni10Cu2+xwt.%Ni复合材料储氢性能研究[J].稀有金属, 2016, 40 (6) :284.)
[15] Han S M, Zhao M S, Wu Y M, Zheng Y Z.Electrode characteristics and microstructure of AB2-AB5composite hydrogen storage alloy formed by ball-milling process[J].Chinese Journal of Inorganic Chemistry, 2003, 19 (3) :262. (韩树民, 赵敏寿, 吴耀明, 郑炀曾.利用球磨制备AB2-AB5复合贮氢合金及其电极性能和微观结构研究[J].无机化学学报, 2003, 19 (3) :262.)
[16] Chu H L, Zhang Y, Sun L X, Qiu S J, Qi Y N, Xu F, Yuan H T.Structure and electrochemical properties of composite electrodes synthesized by mechanical milling Ni-free Ti Mn2-based alloy with La-based alloys[J].Journal of Alloys and Compounds, 2007, 446-447:614.
[17] Han S M, Zhao M S, Zhang Z, Zheng Y Z, Jing T F.Effect of AB2alloy addition on the phase structures and electrochemical characteristics of La Ni5hydride electrode[J].Journal of Alloys and Compounds, 2005, 392:268.
[18] Han S M, Zhang Z, Zhao S M, Zheng Y Z.Electrochemical characteristies and microstructure of Zr0.9Ti0.9Ni0.1Mn0.6V0.3-La Ni5composite hydrogen storage alloys[J].International Journal of Hydrogen Energy, 2006, 31 (5) :563.
[19] Andreasen A.Hydrogenation properties of Mg-Al alloys[J].International Journal of Hydrogen Energy, 2008, 33 (24) :7489.
[20] Aburto A, Orgaz E.Ab initio structural and electronic investigation of magnetic RNi Sn (R=La, Ce, Pr, Nd) intermetallics and their hydrides[J].Physical Review B, 2007, 75 (4) :045130.
[21] de Castro J F R, Santos S F, Costa A L M, Yavari A R, Botta F W J, Ishikawa T T.Structural characterization and dehydrogenation behavior of Mg-5at.%Nb nanocomposite processed by reactive milling[J].Journal of Alloys and Compounds, 2004, 376 (1-2) :251.
[22] Dornheim M, Doppiu S, Barkhordarian G, Boesenberg U, Klassen T, Gutfleisch O R Bormanna.Hydrogen storage in magnesium-based hydrides and hydride composites[J].Scripta Materialia, 2007, 56 (10) :841.
[23] Shao H Y, Liu T, Wang Y T, Xu H R, Li X G.Preparation of Mg-based hydrogen storage materials from metal nanoparticles[J].Journal of Alloys and Compounds, 2008, 465 (1-2) :527.
[24] Liang G.Synthesis and hydrogen storage properties of Mg-based alloys[J].Journal of Alloys and Compounds, 2004, 370 (1-2) :123.
[25] Ponthieu M, Fernàndez J F, Cuevas F, Bodega J, Ares J R, Adeva P, Sánchez C.Thermodynamics and reaction pathways of hydrogen sorption in Mg6 (Pd, TM) (TM=Ag, Cu and Ni) pseudo-binary compounds[J].International Journal of Hydrogen Energy, 2014, 39 (32) :18291.
[26] Sai Raman S S, Srivastava O N.Hydrogenation behaviour of the new composite storage materials Mg-xwt.%CFMm Ni5[J].Journal of Alloys and Compounds, 1996, 241 (1-2) :167.
[27] Davidson D J, Sai Raman S S, Srivastava O N.Investigation on the synthesis, characterization and hydrogenation behaviour of new Mg-based composite materials Mgxwt.%Mm Ni4.6Fe0.4prepared through mechanical alloying[J].Journal of Alloys and Compounds, 1999, 292 (1) :194.
[28] Zhu M, Zhu W H, Chung C Y, Che Z X, Li Z X.Microstructure and hydrogen absorption properties of nanophase composite prepared by mechanical alloying of Mm Ni5-x (Co Al Mn) xand Mg[J].Journal of Alloys and Compounds, 1999, 293-295:531.
[29] Zhu M, Zhu W H, Gao Y, Che X Z, Ahn J H.The effect of Mg content on microstructure and hydrogen absorption properties of mechanical alloyed Mm Ni3.5 (Co Al Mn) 1.5-Mg[J].Materials Science and Engineering A, 2000, 286 (1) :130.
[30] Liang G, Boily S, Huot J, Van Neste A, Schulz R.Hydrogen absorption properties of a mechanically milledMg-50wt.%La Ni5composite[J].Journal of Alloys and Compounds, 1998, 268 (1-2) :302.
[31] Delchev P, Sarafska T, Spassov T.Microstructure and hydriding properties of ball-milled Mg-10at.%Mm Ni5 (Mm=La, Ce-rich mischmetal) composites[J].Journal of Alloys and Compounds, 2006, 417 (1-2) :85.
[32] Vijay R, Sundaresan R, Maiya M P, Murthy S S.Application of Mg-xwt%Mm Ni5 (x=10~70) nanostructured composites in a hydrogen storage device[J].International Journal of Hydrogen Energy, 2007, 32 (32) :2390.
[33] Gu H, Zhu Y, Li L.Hydrogen storage properties of Mg-30wt.%La Ni5composite prepared by hydriding combustion synthesis followed by mechanical milling (HCS+MM) [J].International Journal of Hydrogen Energy, 2009, 34 (3) :1405.
[34] Spassov T, Delchev P, Madjarov P.Hydrogen storage in Mg-10at.%La Ni5nanocomposites, synthesized by ball milling at different conditions[J].Journal of Alloys and Compounds, 2010, 495 (1) :149.
[35] Wei W, Tian X, Tegus O, Guo Q.Phase structure and electrochemical properties of Ml Ni3.55Co0.75Mn0.4Al0.3/Mg composite alloys prepared by mechanical alloying[J].Chinese Rare Earths, 2015, 36 (1) :69. (伟伟, 田晓, 特古斯, 国庆.机械合金化Ml Ni3.55Co0.75Mn0.4Al0.3/Mg复合储氢合金的相结构和电化学性能[J].稀土, 2015, 36 (1) :69.)
[36] Zhu M, Wang Z M, Peng C H, Zeng M Q, Gao Y.The effect of grain refining on the discharge capacity of Mg2Ni/Mm Ni5-x (Co Al Mn) xcomposite prepared by mechanical alloying[J].Journal of Alloys and Compounds, 2003, 349 (1-2) :284.
[37] Pu Z G, Zhu Y F, Zhu J Y, Yuan J G, Zhang J G, Chen W, Fang J J, Li L Q.Kinetics and electrochemical characteristics of Mg2Ni H4-xwt.%Mm Ni3.8Co0.75Mn0.4Al0.2 (x=5, 10, 20, 40) composites for Ni-MH battery[J].International Journal of Hydrogen Energy, 2014, 39 (8) :3887.
[38] Wang L Y, Ding Y, Ruan A J, Ma L Q.Research on the electrochemical properties of Mg2Ni-x MLNi3.8Co0.75Mn0.4Al0.2hydrogen storage alloy prepared by mechanical milling[J].Light Alloy Fabrication Technology, 2010, 38 (4) :39. (王琳瑛, 丁毅, 阮爱杰, 马立群.球磨法制备Mg2Ni-x MLNi3.8Co0.75Mn0.4Al0.2储氢合金的电化学性能研究[J].轻合金加工技术, 2010, 38 (4) :39.)
[39] Liu X D, Huang L H, Tian X, Feng H W, Chi B.Activation characteristics and microstructure of Mg2Ni/Mm0.3Ml0.7Ni3.55Co0.75Mn0.4Al0.3composite hydrogen storage alloys prepared by two-step re-melting[J].International Journal of Hydrogen Energy, 2007, 32 (18) :4939.
[40] Huang L H, Liu X D, Yan S F, Che G D.Phase structure and cycle stabilities of Mg2Ni/Mm0.3Ml0.7Ni3.55Co0.75Mn0.4Al0.3composite hydrogen storage alloys prepared by two-step re-melting[J].Journal of Rare Earths, 2007, 25 (s1) :139.
[41] Tian X, Tegus O, Hai S, Yao Z Q.Effects of rapid quenching on electrochemical properties of Ml Ni3.55Co0.75Mn0.4Al0.3/5wt%Mg2Ni composite hydrogen storage alloy[J].Journal of Inorganic Materials, 2012, 27 (11) :26. (田晓, 特古斯, 海山, 姚占全.快淬对Ml Ni3.55Co0.75Mn0.4Al0.3/5wt%Mg2Ni复合储氢合金电化学性能的影响[J].无机材料学报, 2012, 27 (11) :26.)
[42] Tian X, Yun G H, Shang T, Wang H Y, Hai S.Effects of annealing temperature on structure and electrochemical properties of Mm (Ni Co Mn AI) 5/5wt%Mg2Ni hydrogen storage alloys[J].Journal of Functional Materials, 2013, 19 (44) :2859. (田晓, 云国宏, 尚涛, 王鸿钰, 海山.退火温度对Mm (Ni Co Mn AI) 5/5wt%Mg2Ni储氢合金结构和电化学性能的影响[J].功能材料, 2013, 19 (44) :2859.)
[43] Lototsky M V, Yartys V A, Zavaliy I Y.Vanadiumbased BCC alloys:phase-structural characteristics and hydrogen sorption properties[J].Journal of Alloys and Compounds, 2005, 404:421.
[44] Luo L, Wu W Y, Bian X, Qi J B.Research progress of vanadium-based solid solution hydrogen storage alloys[J].Chinese Journal of Rare Metals, 2017, 41 (11) :1265. (罗龙, 吴文远, 边雪, 齐健博.钒基固溶体贮氢合金的研究进展[J].稀有金属, 2017, 41 (11) :1265.)
[45] Song X P, Pei P, Zhang P L, Chen G L.The influence of alloy elements on the hydrogen storage properties in vanadium-based solid solution alloys[J].Journal of Alloys and Compounds, 2008, 455 (1-2) :392.
[46] Yan Y G, Chen Y H, Wu C L, Tao M D, Liang H.A low-cost BCC alloy prepared from a Fe V80 alloy with a high hydrogen storage capacity[J].Journal of Power Sources, 2007, 164 (2) :799.
[47] Pukazhselvan D, Kumar V, Singh S K.High capacity hydrogen storage:basic aspects, new developments and milestones[J].Nano Energy, 2012, 1 (4) :566.
[48] Wang Y Z, Zhao M S, Li S C, Wang L M.Structure and electrochemical characteristics of melted composite Ti0.10Zr0.15V0.35Cr0.10Ni0.30-La Ni5hydrogen storage alloys[J].Electrochimica Acta, 2008, 53 (27) :7831.
[49] Wang Y Z, Zhao M S, Wang L M.Effect of La-Mgbased alloy addition on structure and electrochemical characteristics of Ti0.10Zr0.15V0.35Cr0.10Ni0.30hydrogen storage alloy[J].International Journal of Hydrogen Energy, 2009, 34 (6) :2646.
[50] Wang Y Z, Zhao M S, Wang L M.Effect of AB5alloy on Ti0.10Zr0.15V0.35Cr0.10Ni0.30hydrogen storage alloy[J].Journal of Applied Electrochemistry, 2009, 39 (9) :1565.
[51] Wang Y Z, Zhao M S.Structure and electrochemical characteristics of Ti0.10Zr0.15V0.35Cr0.10Ni0.30-La Ni4Al0.4Mn0.3Co0.3composite hydrogen storage alloy[J].International Journal of Hydrogen Energy, 2010, 35 (15) :8268.
[52] Wang Y Z, Zhao M S.Distinct synergistic effect in Ti0.10Zr0.15V0.35Cr0.10Ni0.30+1.0wt.%La Ni5hydrogen storage composite electrode[J].International Journal of Hydrogen Energy, 2012, 37 (4) :3276.
[53] Wang Y Z, Zhao M S.Electrochemical characteristics and synergetic effect of Ti0.10Zr0.15V0.35Cr0.10Ni0.30-1.0wt.%La Ni5hydrogen storage composite electrode[J].Journal of Rare Earths, 2012, 30 (2) :146.
[54] Wang Y Z, Zhao M S.Structure and electrochemical characteristics of Ti-V-based solid solution/AB5-type LaMg-based alloy composite hydrogen storage material[J].Journal of Inorganic Materials, 2012, 27 (5) :463. (王艳芝, 赵敏寿.Ti-V基固溶体/AB5型镧镁基合金复合储氢材料的结构与电化学性能[J].无机材料学报, 2012, 27 (5) :463.)
[55] Yu X B, Li F, Wu Z, Xia B J, Xu N X.Enhanced electrochemical properties of ball-milled Ti-30V-15Mn-15Cr+20wt%La (Ni Mn Co Al) 5alloy electrodes[J].Physics Letters A, 2004, 320 (4-5) :312.
[56] Park H S, Chourashiya M, Yang D C, Park C N, Park C J.Hydrogen storage and electrochemical characteristics of Ti0.32Cr0.43-xV0.25Fex (x=0~0.08) alloys and its composites with Lm Ni4.1Al0.25Mn0.3Co0.65alloy[J].Journal of Alloys and Compounds, 2012, 513 (3) :566.
[57] Lee H G, Chourashiya M G, Park C N, Park C J.Hydrogen storage and electrochemical properties of the Ti0.32Cr0.43-x-yV0.25FexMny (x=0~0.055, y=0~0.080) alloys and their composites with Mm Ni3.99Al0.29Mn0.3Co0.6alloy[J].Journal of Alloys and Compounds, 2013, 566 (4) :37.
[58] Kumar S, Tirpude A, Taxak M, Krishnamurthy N.Hydrogen absorption kinetics in powdered V+80wt.%La Ni5composite[J].Journal of Alloys and Compounds, 2013, 580 (12) :S179.
[59] Hu Z L.Hydrogen Storage Materials[M].Beijing:Chemical Industry Press, 2000.186. (胡子龙.贮氢材料[M].北京:化学工业出版社, 2000.186.)
[60] Li D, Lou Y W, Du J L, Pu C H, Huang T S, Li Z L, Wu Z, Li C H.Research progress of vanadium-based hydrogen storage alloy[J].Materials Review, 2015, 29 (12) :92. (李朵, 娄豫皖, 杜俊霖, 蒲朝辉, 黄铁生, 李志林, 吴铸, 李重河.钒基储氢合金的研究进展[J].材料导报, 2015, 29 (12) :92.)
[61] Li X F, Xia T C, Dong H C, Shang Q, Song Y H.Preparation of nickel modified activated carbon/AB5alloy composite and its electrochemical hydrogen absorbing properties[J].International Journal of Hydrogen Energy, 2013, 38 (21) :8903.
[62] Li X F, Wang L Z, Dong H C, Song Y H, Shang H D.Electrochemical hydrogen absorbing properties of graphite/AB5alloy composite electrode[J].Journal of Alloys and Compounds, 2012, 510 (1) :114.
[63] Ren J W, Liao S J, Liu J M.MNi4.8Sn0.2 (M=La, Nd) -supported multi-walled carbon nanotube composites as hydrogen storage materials[J].Chinese Science Bulletin, 2007, 52 (12) :1616.
[64] Cui R C, Yang C C, Li M M, Jin B, Ding X D, Jiang Q.Enhanced high-rate performance of ball-milled Mm Ni3.55Co0.75Mn0.4Al0.3hydrogen storage alloys with graphene nanoplatelets[J].Journal of Alloys and Compounds, 2017, 693:126.
[65] Hai S, Tian X, Tegus O, Temurjorig.Effect of millingtime on the electrochemical properties of AB5/10wt.%CNTs composite hydrogen storage alloys[J].Journal of Inner Mongolia University (Natural Science Edition) , 2013, 44 (2) :188. (海山, 田晓, 特古斯, 特木日朝日格.球磨时间对AB5/10wt.%CNTs复合储氢合金电化学性能的影响[J].内蒙古大学学报, 2013, 44 (2) :188.)
[66] Tsai P J, Chiu T C, Tsai P H, Lin K L, Lin K S, Chan S L I.Carbon nanotube buckypaper/Mm Ni5composite film as anode for Ni/MH batteries[J].International Journal of Hydrogen Energy, 2012, 37 (4) :3491.
[67] Lim K L, Liu Y N, Zhang Q A, Lin K S, Chan S L I.Cycle stability improvement of La-Mg-Ni based alloys via composite method[J].Journal of Alloys and Compounds, 2016, 661:274.
[68] Huang H X, Huang K L.Effect of AB5alloy on electrochemical properties of Mm0.80Mg0.20Ni2.56Co0.50Mn0.14Al0.12hydrogen storage alloy[J].Powder Technology, 2012, 221 (5) :365.
[69] Zhang Z, Han S M, Li Y, Jing T F, Wang X T.The effects of sintering temperature on the phase structure and electrochemical properties of AB5-5mass%La Mg3composite alloy[J].Journal of Alloys and Compounds, 2006, 421 (1-2) :289.
[70] Kim J H, Yamamoto K, Yonezawa S, Takashima M.Effects of Ni-PTFE composite plating on AB5-type hydrogen storage alloy[J].Materials Letters, 2012, 82:217.
[71] Shen W Z, Han S M, Li Y, Song J Z, Tong Q.Study on surface modification of AB5-type alloy electrode with polyaniline by electroless deposition[J].Electrochimica Acta, 2010, 56 (2) :959.
[72] Rivera M A, Pal U, Wang X Y, Gonzalez-Rodriguez J G, Gamboa S A.Rapid activation of Mm Ni5-xMx based MH alloy through Pd nanoparticle impregnation[J].Journal of Power Sources, 2006, 155 (2) :470.
[73] Williams M, Nechaev A N, Lototsky M V, Yartys V A, Solberg J K, Denys R V, Pineda C, Li Q, Linkov V M.Influence of aminosilane surface functionalization of rare earth hydride-forming alloys on palladium treatment by electroless deposition and hydrogen sorption kinetics of composite materials[J].Materials Chemistry and Physics, 2009, 115 (1) :136.
[74] Modibane K D, Williams M, Lototskyy M, Davids M W, Klochko Ye, Pollet B G.Poisoning-tolerant metal hydride materials and their application for hydrogen separation from CO2/CO containing gas mixtures[J].International Journal of Hydrogen Energy, 2013, 38 (23) :9800.
[75] Lototsky M V, Williams M, Yartys V A, Klochko Ye V, Linkov V M.Surface-modified advanced hydrogen storage alloys for hydrogen separation and purification[J].Journal of Alloys and Compounds, 2011, 509 (2) :S555.
[76] Zhao X Y, Yao Y, Ma L Q, Yang M, Ding Y, Shen X D.Synergistic effects in an AB5-Co material as an anode for a secondary alkaline battery[J].International Journal of Hydrogen Energy, 2010, 35 (9) :4342.
[77] Raju M, Ananth M V, Vijayaraghavan L.Influence of electroless coatings of Cu, Ni-P and Co-P on Mm Ni3.25Al0.35Mn0.25Co0.66alloy used as anodes in Ni-MH batteries[J].Journal of Alloys and Compounds, 2009, 475 (1-2) :664.
[78] Feng H W, Liu X D, Tian X, Chi B, Huang L H, Xu J.Electrochemical properties of the Mm0.3Ml0.7Ni3.55Co0.75Mn0.4Al0.3alloy mixed with Ni powder[J].International Journal of Hydrogen Energy, 2009, 34 (4) :1886.


