J. Cent. South Univ. Technol. (2007)01-0028-04
DOI: 10.1007/s11771-007-0006-3
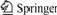
Influencing factors of pyrite leaching in germ-free system
OU Le-ming(欧乐明), HE Rong-quan(何荣权), FENG Qi-ming(冯其明)
(School of Resources Processing and Bioengineering, Central South University, Changsha 410083, China)
Abstract: The effect of mineral particle size, pulp potential and category of oxidant on pyrite leaching was studied. The results show that a smaller mineral particle size leads to a higher leaching rate of pyrite, and the optimum result with pyrite leaching rate of 2.92% is obtained when mineral particle size is less than 0.037 mm. The pulp potential reflects the leaching process. The increase of pulp potential can improve pyrite leaching. The leaching rate and velocity of pyrite can be enhanced rapidly by adding strong oxidant. The kind and the method of adding oxidant have important effect on the pyrite leaching. Appropriate concentration of Fe3+ can enhance pyrite leaching but the precipitation generated by high concentration of ferric ion covers the surface of pyrites and prevents the leaching process. The leaching rate increases with the constant addition of H2O2.
Key words: pyrite; leaching rate; mineral particle size; pulp potential; oxidant
1 Introduction
Pyrite (FeS2) is of reducibility, which can be oxidized when exposed in air or in aqueous solution. The oxidation of pyrite has both advantage and disadvantage for some practical applications. For example, the pyrite oxidation should be inhibited to reduce acid waste water in waste water treatment process, but when it comes to desulphurization from coal, the pyrite is expected to be fully oxidated so that sulphur can be removed completely[1]. During bioleaching and acid leaching processes, pyrite is expected to be oxidized as fast as possible to increase the leaching velocity[2]. In flotation process, the oxidation of pyrite should be encouraged in some cases, but sometimes it should be inhibited[3]. In a word, whether the pyrite should be oxidized or not is determined by practical applications. At present, researchers have paid more attention to the influencing factors of oxidation of pyrite, such as temperature, leaching time, pulp concentration, mineral particle size, pH value and so on[4-6]. Reports on influencing factors in bioleaching of pyrite are available[7-8], but there are few reports on germ-free acid leaching. Therefore, in this study, the influencing factors of pyrite leaching in the germ-free system were investigated.
2 Experimental
2.1 Preparation of samples
All pyrites used in the experiments were bought
from Changsha mineral market. The crystals of pyrite were intact and contained few impurities. The ores were
crashed and ground by porcelain mill and then sieved into four different sizes: <0.037 mm, 0.037-0.050 mm, 0.050-0.074 mm and 0.074-0.154 mm. These samples were vacuum dried and finally saved in sealed glass bottles for preparation. The chemical analysis indicates that the pyrite contains 49.02% S and 40.11% Fe.
2.2 Experimental method
The experiments were carried out in shake flasks, which were put in an oscillator. The ratio of liquid to solid was 10?1. The leaching solution was sulfuric acid prepared before the experiments. Analytically pure sulfuric acid was used to adjust the original pH value of the leaching solution. The rotational speed of the oscillator was set to be 160 r/min, and the temperature was set at 30 ℃. All the water used in experiments was distilled water.
During the experiments, the contents of Fe2+ and total iron ions(TFe) in the leaching solution and the pulp potential were tested. The concentration of Fe2+ was tested by titration with potassium dichromate. The content of TFe was tested by atomic absorption spectrometry. The pulp potential was monitored with platinum reference electrode. The value of pulp potential, Em, was converted to standard hydrogen electrode potential, E, by the equation of E=Em+244.5 mV.
3 Results and discussion
3.1 Effect of mineral size on leaching of pyrite
Mineral particle size is one of the important influencing factors during the leaching of pyrite, which should be paid much attention especially during bioleaching[9]. Therefore, mineral particle size was investigated at first in the experiments. The results are shown in Fig.1.
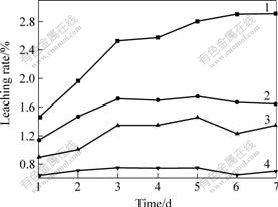
Fig.1 Relationship between leaching rate of pyrite and leaching time at original pH value of 2.0
Mineral particle size/mm: 1―<0.037; 2―0.037-0.050; 3―0.050-0.074; 4―0.074-0.154
From Fig.1 it can be seen that the leaching rates of pyrite with various sizes increase with lengthening leaching time. Especially when the particle size is less than 0.037 mm, the leaching rate increases in a bigger margin than others, and a leaching rate of 2.92% is obtained after leaching for 7 d. It is also shown that the smaller the particle size, the higher the leaching rate. After leaching for 7 d, the leaching rate of pyrite with size less than 0.037 mm is 2.22% which is higher than that of the mineral with size of 0.074-0.154 mm. The smaller mineral particle size means larger specific surface and more surface activity site, which make the leaching easier. This result is identical with the effect of mineral particle size on bioleaching investigated by YUAN et al[9].
3.2 Effect of pulp potential on leaching of pyrite
It has been proved that the pyrite leaching is an electrochemical process by thermodynamics analysis and previous experiments. Therefore, pulp potential is a key influencing factor for leaching of pyrite. LAN et al[10] suggested that the releasing rate of SO42- was very slow when the pulp potential was lower than 0.35 V, and so was the oxidation processing of pyrite. When pulp potential enhanced, the oxidation would be accelerated, and the pH value would drop rapidly. Based on some correlative reports, the following conclusion can be drawn that the leaching rate of pyrite is related to the pulp potential, when the pulp potential is higher than 0.60 V, the oxidation of pyrite occurs fiercely, however, when it is lower than 0.60 V, the reaction is very weak.
It is discovered that the pulp potential ties with the leaching system through the measure of pulp potential. Fig.2 shows that the average pulp potential decreases with increasing the mineral particle size. Based on the analysis of Fig.1, the conclusions that mineral particle size influences the pulp potential, and thus influences the leaching efficiency are obtained. The finer the particle, the higher the potential and the leaching rate.

Fig.2 Relationship between pulp potential and leaching time of pyrite with different sizes
Particle size/mm: 1―<0.037; 2―0.037-0.050; 3―0.050-0.074; 4―0.074-0.154
Fig.3 shows the effect of oxidant on relationship between leaching time and pulp potential, indicating that when there is no oxidant in the leaching solution, the pulp potential increases firstly with the time prolonging and then keeps stable; when Fe3+ is added, the pulp potential decreases with increasing the time and then keeps stable; when hydrogen peroxide is added, the potential decreases in the first three hours, and then increases gradually to a stable value. The different changing trend indicates different mechanism of reactions. Therefore, by controlling the pulp potential the leaching of pyrite can be controlled.
3.3 Effect of oxidant on leaching of pyrite
Oxidation plays an important role in the leaching of pyrite. The effects contain two aspects. The first is the kind of oxidant such as Fe3+, dissolved oxygen and H2O2. MOSES[11] suggested that the effect of Fe3+ on pyrite oxidation was greater than that of dissolved oxygen. During the leaching, the quantity of Fe2+ created by the effect of Fe3+ is 15 times larger than that of dissolution oxygen. The second is the effect of oxidant concentration on pyrite leaching, generally, the higher the oxidant concentration, the higher the leaching rate.
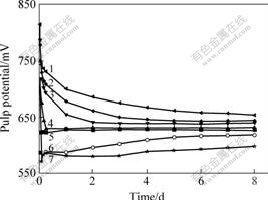
Fig.3 Effect of oxidant on relationship between leaching time and pulp potential
1―0.16 mol/L Fe3+; 2―0.08 mol/L Fe3+; 3―0.04 mol/LFe3+; 4―0.01 mol/L Fe3+; 5―No oxidant; 6―1 mL/d H2O2; 7―1 mL H2O2
3.3.1 Effect of Fe3+ on leaching of pyrite
Based on Ref.[10] and the analysis of thermo- dynamics, Fe3+ can improve the leaching of pyrite. Therefore, the experiments using ferric sulfate as oxidant were carried out. The results are shown in Figs.4 and 5.
Fig.4 shows that the concentration of Fe2+ increases with prolonging leaching time, but in the meantime the increasing extent slows down, and with increasing concentration of Fe3+, the concentration of Fe2+ change remarkably. Fig.5 shows the content of total iron, TFe, almost keep unchangeable. When the concentration of Fe3+ is low, the content of total iron presents an increasing trend, and when the concentration of Fe3+ is high, the content of total iron presents a decreasing trend. With increasing concentration of Fe3+, the hydrolysis of Fe3+ results in precipitation. From Figs.4 and 5, the addition of Fe3+ can increase the content of Fe2+ rapidly. Fe2+ is generated by the reduction of Fe3+ and the oxidation of pyrite.
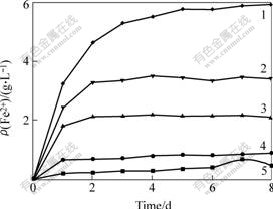
Fig.4 Relationship between mass concentration of
Fe2+(ρ(Fe2+)) and leaching time at different concentrations of Fe3+(c(Fe3+))
c(Fe3+)/(mol?L-1): 1―0.16; 2―0.08; 3―0.04; 4―0.01; 5―0
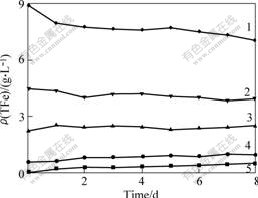
Fig.5 Relationship between ρ(TFe) and leaching time at different c(Fe3+)
c(Fe3+)/(mol?L-1): 1―0.16; 2― 0.08; 3―0.04; 4―0.01; 5―0
Figs.3-5 show that the pulp potential and the concentration of Fe2+ correlate each other when c(Fe3+)=0.16 mol/L. With prolonging leaching time, the pulp potential decreases and the concentration of Fe2+ increases. When the pulp potential keeps stable, the concentration of Fe2+ presents a stable trend. The relationship between pulp potential and mass concentration of ρ(Fe2+) indicates that pulp potential changes with the process of leaching. Therefore, the conclusion that an oxidation process occurs during the leaching of pyrite can be made. At the same time, it can be seen that the content of Fe2+ keeps stable after leaching for 5 d, but the content of total iron presents a decreasing trend. The reason may be the precipitation of Fe(OH)3. The precipitation covers the surface of pyrite and prevents the leaching.
3.3.2 Effect of H2O2 on leaching of pyrite
H2O2 is also an effective oxidant which can improve the leaching of pyrite besides Fe3+. The experiments using H2O2 as oxidant were investigated. The amounts of H2O2 were 0, 1 mL and 1 mL/d, respectively. The results are shown in Fig.6, indicating that the addition of H2O2 enhances the leaching velocity obviously. However, the leaching rate with H2O2 is almost close to that without H2O2 after leaching for 8 d, and the difference between the two is only 0.03%. From Fig.6 (curve 2), the leaching rate comes to a stable value after leaching 4 d with the addition of H2O2, because S0 or some dense sulfur layers are generated on the surface of pyrite, which hinder the leaching. Comparing curve 2 with curve 3, the gap between the two is very small, and the leaching rate keeps increasing when H2O2 is added constantly. The leaching rate keeps stable if the H2O2 is added at one time, this is because the constant addition of H2O2 provides dissolved oxygen for the reaction, which results in the disappearance of S0 on the surface of pyrite, and the effect of inhibition is eliminated.
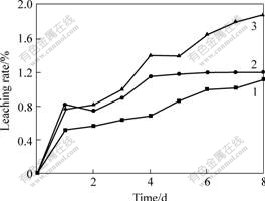
Fig.6 Effect of H2O2 on leaching rate of pyrite
Amount of H2O2: 1―0; 2―1 mL; 3―1 mL/d
4 Conclusions
1) Particle size is an important factor influencing the leaching of pyrite. Finer particle size will improve the leaching. When particle size is less than 0.037 mm, the leaching rate of pyrite is 2.92%.
2) Pulp potential is also a key influencing factor for the leaching of pyrite. The pulp potential is related to particle size, concentration of oxidant and categories of oxidant. There is a certain functional relation between pulp potential and leaching rate, and also the leaching velocity of pyrite.
3) The oxidant such as Fe3+ and H2O2 can enhance the pulp potential effectively, thus enhance the leaching rate. H2O2 is better than Fe3+ for the leaching. After leaching for a certain time, the leaching process will be hindered by the precipitation of excessive Fe3+. The leaching rate comes to a stable value after leaching for 4 d with the addition of H2O2, but constant addition of H2O2 can provide dissolved oxygen for the reaction and can eliminate the inhibition effect of S0 on the surface of pyrite.
References
[1] YI Ping-gui, YU Qing-sen, ZONG Han-xing. Thermodynamic analysis for chemical desulfurization of pyrite in coal[J]. Coal Conversion, 1999, 22 (1): 47-52.( in Chinese)
[2] YANG Hong-ying, YANG Li, ZHAO Yu-shan. Active alignment of sulphide minerals biooxidized by Thiobacillus ferrooxidans[J]. Nonferrous Metals, 2002, 54(2): 42-45.(in Chinese)
[3] FENG Qi-min, CHEN Jin. Flotation Electrochemistry of Sulphide Ore[M]. Changsha: Center South University of Technology Press, 1992.(in Chinese)
[4] MCKIBEEN M A, BARNES H L. Oxidation of pyrite in temperature acidic solution: rate laws and surface textures[J]. Geochim Cosmochim Acta, 1986, 50: 1509-1520.
[5] WILLIAMSON M A, RIMSTIDT J D. The kinetics and electrochemical rate-determining step of aqueous pyrite oxidation[J]. Geochim Cosmochim Acta, 1994, 58(24): 5443-5454.
[6] HOLMES P A, CRUNDWELL F K. The kinetics of the oxidation of pyrite by ferric ions and dissolved oxygen: an electrochemical study[J]. Geochim Cosmochim Acta, 2000,64(2): 263-274.
[7] ROJAS-CHAPANA J A, GIERSIG M, TRIBUTSCH H. The path of sulfur during bio-oxidation of pyrite by Thiobacillus ferrooxidans[J]. J Fuel, 1996, 75(8): 923-930.
[8] YAHYA A, JOHNSON D B. Bioleaching of pyrite at low pH and low redox potentials by novel mesophilic gram-positive bacteria[J]. Hydrometallurgy, 2002, 63(2): 181-188.
[9] YUAN Xin, YUAN Chu-xiong. The microbe process technology study on non-metallic mineral (Ⅲ): the pyrite microbiological oxidation study[J]. China Nonmetallic Mining Industry Herald, 2000, 17(5): 22-39. (in Chinese)
[10] LAN Ye-qing, ZHOU Gang, LIU Zheng-hua,et al. Pyrite oxidation under different conditions[J]. Journal of Nanjing Agricultural University, 2000, 23(1): 81-84.(in Chinese)
[11] MOSES C O, NORDSTROM D K, HERMAN J S, et al. Aqueous pyrite oxidation by dissolved oxygen and by ferric iron[J]. Geochim Cosmochim Acta, 1987, 51(6): 1561-1571.
(Edited by CHEN Wei-ping)
Foundation item: Project(2004CB619204) supported by the National Key Fundamental Research and Development Program of China; Projects(50321402, 50574101) supported by the National Natural Science Foundation of China
Received date: 2006-04-28; Accepted date: 2006-06-02
Corresponding author: OU Le-ming, PhD; Tel: +86-731-8830913; E-mail: ou-leming@163.com