Application of ionic liquids in hydrometallurgy of nonferrous metals
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2010���3��
�������ߣ������ ��� ��һ��
����ҳ�룺513 - 520
Key words��ionic liquids; hydrometallurgy; nonferrous metals; metal oxides; mineral processing��extraction and separation of metal ions
Abstract: Ionic liquids as green solvents have shown important application in the extraction and separation of nonferrous metals. The new application perspective, the important fundamental and the applied studies of the extraction and separation of nonferrous metals in ionic liquids, including the dissolution and corrosion of metal and metal oxide, hydrometallurgy of chalcopyrite and metallic oxidized ore, and extraction and separation of metal ions, are introduced.
������Ϣ��the National Natural Science Foundation of China
the Natural Science Foundation of Yunnan Province, China
the Science Foundation of the Education Department of Yunnan Province, China
the Science Foundation of Kunming University of Science and Technology, China

TIAN Guo-cai(�����), LI Jian(�� ��), HUA Yi-xin(��һ��)
Faculty of Metallurgical and Energy Engineering, Kunming University of Science and Technology,
Kunming 650093, China
Received 6 July 2009; accepted 13 January 2010
Abstract: Ionic liquids as green solvents have shown important application in the extraction and separation of nonferrous metals. The new application perspective, the important fundamental and the applied studies of the extraction and separation of nonferrous metals in ionic liquids, including the dissolution and corrosion of metal and metal oxide, hydrometallurgy of chalcopyrite and metallic oxidized ore, and extraction and separation of metal ions, are introduced.
Key words: ionic liquids; hydrometallurgy; nonferrous metals; metal oxides; mineral processing��extraction and separation of metal ions
1 Introduction
Non-ferrous metals are important strategic resources and have a wide range of applications in industries as diverse as industrial equipment, health, transport, energy, construction, automotive and aircraft, electronic equipment, and packaging. Most nonferrous metals are obtained by hydrometallurgical processes, for example, aqueous acids or alkalis are predominantly used to dissolve the metal oxides, sulfides, or silicates. Electrowinning and solvent extraction are frequently used to recover and concentrate the metals. A limited number of high-temperature molten salts have also been used extensively for the recovery of refractory metals, such as titanium and aluminum, from their ores[1]. Nonferrous metal industry has a rapid development and has achieved remarkable progress in recent years. However, the production of non-ferrous metals from natural mineral ores is of in general high energy consumption, large acid consumption, high environmental pollution, and serious corrosion, furthermore, the ores need to be sourced from less rich, low grade and/or geologically complex sites as high quality ore bodies are progressively mined out. Therefore, the development of highly effective, low-temperature and environmentally benign technologies for metals processing to reduce energy consumption and to lower investment costs and the greenhouse gas emissions is urgent[2]. Ionic liquids are considered to be the most promising candidates due to their low toxicity and little environmental impact in recent years. The application of ionic liquids as solvents to metallurgical ore beneficiation may offer potential as environmentally sensitive media and alternatives to hydrometallurgical processes.
Ionic liquids (ILs), also called room temperature ionic liquids (RILS) and ambient temperature molten salts, which are liquids at ambient temperature[3], are completely composed of organic cations and inorganic/organic anions. They have many fascinating physical properties which make them attract fundamental interest to many chemists, since both the thermodynamics and kinetics of reactions carried out in ionic liquids are different from those in conventional molecular solvents, then the chemistry is different and unpredictable at our current state of knowledge. And they have been widely and successfully used in synthesis and preparation of materials, catalysts, electro-deposition of metals, and fuel cell[4-6]. Common characteristics of ILs for solvent and electrochemical application include[4-6] the following items: 1) Non-flammability and very low or negligible vapor pressure. The non-flammability of ILs is particularly valuable for solvents used for exothermic reactions. Negligible vapor pressure means that solvent evaporation is eliminated, reducing the need for respiratory protection and exhaust systems, which can be used in high vacuum systems and in distillation or sublimation of products and by-products, which are not possible with traditional low-boiling organic solvents. 2) ILs can dissolve a wide range of inorganic and organic compounds. It is important for dissolving disparate combinations of reagents into the same phase. 3) Wide liquid range and thermal stability, which allows access to wider temperature ranges and allows more tremendous kinetic control of chemical or electrochemical processes than that attained by using conventional molecular solvent/electrolyte systems. And this characteristic is also useful for temperature- dependent separation techniques, such as extraction, precipitation or crystallization. 4) Lower melting points, air and water stability, which considerably increases the scope of the electrochemical reactions. 5) Wide electrochemical window, high electrochemical stability and ion conductivity, which allows investigation of electrochemical processes that are previously beyond solvent limits, and can be used in place of traditional solvent-based electrolytes. It is prone to volatility, flammability, leakage, and corrosion. It has been widely and successfully used in electrodeposition of metals (epically reactive metals) and in many electrochemical devices including fuel cells, solar cells, batteries and capacity. 6) ILs are designer solvents. Different cations and anions can form different kinds of ionic liquids and their properties can be adjusted to suit the requirements of a particular process by changing the cations and anions. Properties such as density, melting point, viscosity can be easily varied by simply changing the structure of the ions. Changes with structure is the miscibility of water in these ionic liquids, which can be of substantial benefit when carrying out solvent extractions or product separations, as the relative solubility of the ionic and extraction phase can be adjusted to make the separation as easy as possible.
Preliminary studies have shown that ionic liquids have potential as solvents and electrolytes for metal recovery, and the feasibility of these solvents has been demonstrated for the extraction of gold and silver from a mineral matrix[7], the recovery of uranium and plutonium from spent nuclear fuel[8], electrodeposition and electrowinning of metals (especially Li, Na, Al, Mg and Ti) from ionic liquids[9-11]. Research works have shown that the electrodeposition of Al from ionic liquids can save 30%-50% energy consumption compared with the traditional process, and this technology has been strongly supported as an industrial technologies project by the partners of University of Alabama, Albany Research Center, Century Aluminum Company, Secat Inc. and University of Kentucky. Ionic liquids as green solvents and electrolytes have shown important and potential application in the extraction and separation of nonferrous metal. In this work, the new application perspective and the important fundamental and applied studies of the extraction and separation of nonferrous metal in ionic liquids including metal oxide processing, hydrometallurgy of chalcopyrite and metallic oxidized ore, and extraction and separation of metal ions are introduced.
2 Processing metal oxides with ionic liquids
Metal oxide processing is essential for metal extraction, waste recycling and catalyst preparation[12]. Metal oxides are insoluble in most molecular solvents and generally strong aqueous mineral acids are used for their dissolution, especially high temperature molten salts are also used extensively for the recovery of refractory metals, such as titanium and aluminum, from their ores[1, 12-13]. However, there are many disadvantages in these processes such as large acid consumption, recycle of acid waste, environmental pollution, high energy consumption, and serious corrosion. Therefore, there has been a worldwide interest in seeking for new and green solvents for processing metal oxides under ambient atmosphere and low temperature with low consumption of energy, acid and oxygen as well as free of any pollutants release. Ionic liquids that have abilities to dissolve a wide range of inorganic and organic compounds provide a potential way to process metal oxides greenly at low temperature. In recent years, many research works have been focused and much progress has been achieved in this filed.
In 1997, DAI and co-workers[14] determined the solubility of UO3 in imidalozium-based chloroaluminate melts to be 24.58 mmol at 65 ��. The dissolution of V2O5 in (Emim) chloroaluminate ionic liquids was studied by BELL et al[15]. The oxide was found to be very soluble in the basic melt, 0.15 g in 1 g, but also soluble in the neutral melt. The authors reported that V2O5 reacts with the acidic chloroaluminate melt, forming a volatile VOCl3 compound.
High solubility of inorganic salts such as LiCl and AgCl, aromatic acids, amino acids, and metal oxides CuO in choline chloride eutectics [HOC2H4N(CH3)3]Cl(ChCl)/urea mixtures was reported by ABBOTT et al in 2003[16]. One year later, they reported the solubility of ZnO, CuO, and Fe3O4 in three deep eutectic solvents (DES) formed between choline chloride and carboxylic acids at 50 ��[17]. They found that Fe3O4 is most soluble in the oxalic acid/ChCl mixture, whereas it is 20 times less soluble in the phenylpropionic acid mixture, but CuO shows the opposite behavior. It is suggested that solvents can be designed to preferentially extract one metal oxide with respect to another. The majority of the more ionic transition metal oxides were found to be soluble in a DES although the more covalent metal oxides such as aluminates and silicates are insoluble in all DES studied to date, which suggests that metal ions could be extracted from an aluminosilicate matrix without dissolving the matrix. Metals such as copper can be recovered electrochemically from the DES with high current efficiencies using bulk electrolysis, which could have potential applications to mineral extraction or metal oxide processing.
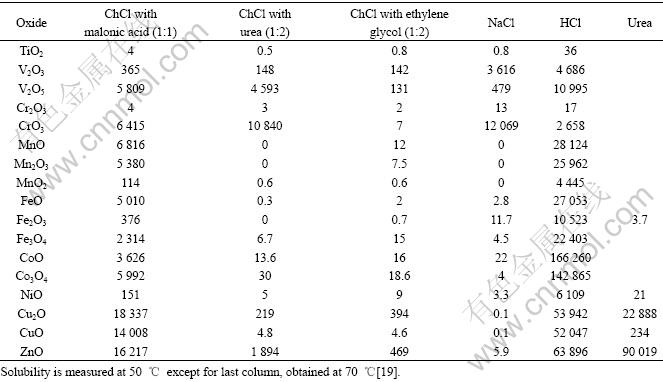
In 2006, ionic liquid choline chloride (ChCl) and urea were applied to the processing of electric arc furnace dust by ABBOTT et al[18]. It was found that the solubility of ZnO, Cu2O and PbO2 is large, and zinc and lead can be selectively removed and subsequently electrowon from the liquid, whereas the insoluble iron and aluminosilicates can be recycled in the electric arc furnace. The solubility of 17 common metal oxides have been determined in three ionic liquids based on choline chloride with urea, malonic acid, and ethylene glycol and compared with aqueous solutions of HCl and NaCl[19]. Table 1 gives the details. It is found that the order of solubility is: HCl��malonic acid��urea��NaCl��ethylene glycol. Higher solubility was observed with the more ionic oxides such as ZnO in an analogous manner than those expected for aqueous acidic solutions, whereas more covalent oxides such as TiO2 exhibit negligible solubility.
Table 1 Solubility of metal oxides in eutectic mixtures of ChCl with malonic acid (1:1), urea (1:2), and ethylene glycol (1:2), compared with that in aqueous 0.181 mol/L NaCl and 3.14 mol/L HCl after 2 d (10-6)
The task-specific ionic liquid protonated betaine bis(trifluoromethyl sulfonyl)imide([Hbet]Tf2N) was used for selective solubilization of metal oxides and metal salts by ABBOTT et al[20]. They found that the metal oxides such as Sc2O3, Y2O3, La2O3, Pr6O11, Nd2O3, Sm2O3, Eu2O3, Gd2O3, Tb4O7, Dy2O3, Ho2O3, Er2O3, Tm2O3, Yb2O3, Lu2O3, UO3, PbO, ZnO, CdO, HgO, CuO, Ag2O, NiO, PdO, and MnO can be soluble in the ionic liquid [Hbet][Tf2N] and insoluble or very poorly soluble for iron and cobalt oxides, as well as aluminum oxide and silicon oxide. Very recently, it has been reported the solubility of metal oxides in various task-specific ionic liquids, such as betainium bis(trifluoromethylsulfonyl) imide ([Hbet]Tf2N), N-butyl-N-dimethylbetainium bis(tri fluoromethyl-sulfonyl)-imide ([C4Hbet]Tf2N), N-hexyl-N-dimethylbetainium bis(trifluoromethylsulfonyl)imide([C6Hbet]Tf2N), N-carboxymethyl-N-methylpyrro- lidinium bis(trifluoromethylsulfonyl)imide([HbetmPyr]-Tf2N), N-carboxy-methyl-N-methylpiperidinium bis-(trifluoromethylsulfonyl)imide([HbetmPip]Tf2N), N-carboxymethyl-N-methylmorpholinium bis-(trifluoromethylsulfonyl)imide([HbetmMor]Tf2N),N- carboxymethyl-N-methyl morpholiniumethylester bis- (trifluoromethylulfonyl)imide ([EtHbetmMor]Tf2N), N-carboxymethyl-pyridiniumbis(trifluoromethylsulfonyl)imide ([HbetPy]Tf2N), 1-carboxymethyl-3-methyl- imidazolium bis(trifluoromethylsulfonyl)imide ([HbetmIm]Tf2N )[21]. It is found that the oxides Sc2O3, Y2O3, La2O3, Pr6O11, Nd2O3, Sm2O3, Eu2O3, Gd2O3, Tb4O7, Dy2O3, Ho2O3, Er2O3, Tm2O3, Yb2O3, Lu2O3, UO3, PbO, ZnO, CdO, HgO, CuO, Ag2O, NiO, and PdO as well as the hydroxides Pb(OH)2, Zn(OH)2, Cd(OH)2, Cu(OH)2, Ni(OH)2, Fe(OH)2, Fe(OH)3, Co(OH)2, Cr(OH)3, Mn(OH)2,LiOH, NaOH, KOH, RbOH, CsOH, Mg(OH)2, Ca(OH)2, Sr(OH)2, and Ba(OH)2 can be soluble in the ionic liquids, whereas Co3O4, CoO, Co2O3, Cr2O3, FeO, and Fe2O3 are found to be insoluble in the ionic liquids under the experimental conditions used to dissolve the other oxides. However, these oxides could be solubilized in the ionic liquids (including [Hbet]Tf2N) by using a digestion bomb at higher temperature.
ZHANG and co-workers[22] reported recently that alumina (Al2O3) can be soluble in the 1-ethyl-3-methyl-imidazolium hydrogen sulfate ([Emim]HSO4) ionic liquid and the solubility was 3.81 g/L at 20 ��, which suggests that this system might satisfy the electrolysis of alumina, and alumina could be reduced to aluminum at a platinum electrode. With normal deposition at -0.54 V and under potential deposition (UPD) at -0.26 V, the deposition is a diffusion controlled process[22].
The research works mentioned above show that most of the metal oxides can be selectively solved in ionic liquids, which provides a new method to obtain the specific metal ion in ionic liquids to further extraction and separation. Especially, it may be used as a potential ��green�� leaching agent for low-grade ore and refractory oxide ore. Although much progress has been achieved, there are challenging issues remaining. The low solubility of metal salts is a serious drawback for possible applications of ionic liquids that require high concentrations of dissolved metal salts, e.g., the electro-deposition of metals or solvents for the synthesis of nano-particles[23]. Unfortunately, quantitative data about the solubility of metal salts in ionic liquids are still very scarce. The dissolution mechanism of metal oxides in ionic liquids is an key issue that needs to be further investigated because it has recently been reported that the coordination environments of the solvates in ionic liquids might be quite unique[24-25].
3 Ionic liquids in mineral processing
The most important process in hydrometallurgy is efficient separation of metals from their ores followed by metals recovery from concentrate. In recent decades, increasing importance has been attached to hydrometallurgical processing of sulfide ores, such as chalcopyrite, as more attentions have been paid to the green and sustainable development of metallurgical industry[7, 11, 26-27]. Some leaching processes, including chloride leaching, bioleaching and pressure sulfate leaching, have been developed to recover copper from chalcopyrite concentrates to date. However, due to the highly toxic nature of cyanide and environmental consequences, the process is very controversial and provokes examination of new leaching species. Therefore, there has been a worldwide interest in seeking for new and green hydrometallurgical processes for the production of copper operated under ambient atmosphere and low temperature with low consumption of energy, acid and oxygen as well as free of pollutants release. Ionic liquids were examined as solvents (either as a neat liquid or as aqueous mixtures) for the leaching of gold, silver, copper and base metals from sulphidic ores[7, 26-27]. At present, cyanides are mainly used for the commercial hydrometallurgical leaching of gold and silver from ores and concentrates.
The possibility of application of ionic liquids in processing of copper sulfide ores and base metal sulfides supported by preliminary findings in the use of ionic liquids in the electrorefining of chalcopyrite (CuFeS2) was also indicated by McCLUSCEY et al[11]. 1-butyl-3-methyl-imidazolium tetrafluroborate ([Bmim] BF4) ionic liquid with Fe(BF4)3 was used as the leaching agent of chalcopyrite, after 8 h with ferric tetrafluroborate in water: [Bmim]BF4 of 1?1 and an extraction of 90% of available copper was achieved at 100 ��.
WHITEHEAD et al[7] reported the recovery of gold and silver from ore in 1-butyl-3-methyl-imidazolium hydrogen sulfate ionic liquid ([Bmim]HSO4) with iron(��) sulfate oxidant and thiourea added. The extraction of gold was achieved to be larger than 85% from synthetic oxidic ore as well as natural sulphidic ore at 20-50 �� using ionic liquid as a solvent. Gold extraction was close to results achieved for aqueous system H2SO4/tiourea/Fe2(SO4)3, whereas recovery of silver from the natural sulphidic ore was significantly higher (��60%) for the neat IL compared with an aqueous acid solution (��10%). Moreover, high selectivity for the extraction of gold and silver was reported, with minimal selectivity of other metals (Cu, Zn, Pb and Fe).
The [Bmim]HSO4 has been applied as a solvent medium to leach gold, silver, copper and base metals from sulphidic ores using thiourea (mainly) in the presence of iron(��) as oxidant by WHITEHEAD et al[26](Fig.1). They found that copper extraction from chalcopyrite was much more efficient than iron extraction in the ionic liquid medium at 70 ��, increasing from 55% to 87% as the ionic liquid composition increased from 10% (mass fraction) in water to 100%. Gold and silver recoveries were larger than 85% and larger than 60%, respectively, in the presence of iron(��)/thiourea at 20-50 �� in this ionic liquid [7, 26]. Extension of n-alkyl chain length in imidazolium cation resulted in decreasing extraction efficiency of gold and silver, which might result from the increasing viscosity of ILs. The analysis of leaching results obtained for ILs with varied chain length and different types of anions showed that [Bmim]HSO4 was the most effective medium when taking into account of the relatively low price of this compound[7, 27].
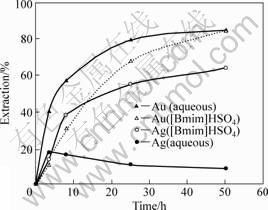
Fig.1 Comparative leaching of gold and silver from ore using aqueous H2SO4 or [Bmim]HSO4 in presence of Fe2(SO4)3 and tiourea[7, 27]
Leaching experiments with [Bmim]HSO4 ILs were carried with pure ionic liquid and their aqueous solutions in stationary flask to leach the chalcopyrite concentrate in a temperature range of 50-90 �� with pulp densities of 10 g/L and 100 g/L under air atmosphere in our group[28]. Copper extraction increased from 51.8% to 87.8% as the ionic liquid concentration in leaching solution increased from 10% (volume fraction) to 100%. Copper extraction was very low at temperature below 70 ��, whereas increased markedly at moderately elevated temperature from 70 to 90 ��, suggesting that a moderate temperature (��70 ��) was needed to break bonds in chalcopyrite crystal lattice to accelerate the chemical reaction.
Preliminary researches performed in last decade show that ionic liquids have good selectivity for gold and silver. It is indicated that ILs may be used as a potential solvent to replace traditional cyanides in the commercial hydrometallurgical leaching of gold and silver from ores and concentrates, and to develop efficient processing technology for the recovery of gold and silver from ores.
4 Ionic liquids in solvent extraction of metal ions
Solvent extraction is a process that allows the separation of two or more components due to their unequal solubilities in two immiscible liquid phases. It is an important method in hydrometallurgy in separation of metal ions from their solution. The unique properties, such as good stability, nonvolatility, wide liquid range, low flammability and adjustable miscibility and polarity, being hydrophilic and hydrophobic depending on the structures of cations and anions, make ILs attractive as alternative media in liquid-liquid extraction of metal ions from aqueous solutions.
DAI et al[29] observed large distribution coefficient value for extraction of Sr(NO3)2 from aqueous solutions by using dicyclo-hexyl-18-crown-6 (DCH18C6) as extractant and imidazolium hexaflurophosphates and bis[(trifluoromethane)-sulfonyl]amides as solvents. CHUN et al [30] observed the low extraction efficiency of competitive alkali metal salt from aqueous in CnmimPF6 (n=4-9). The high extraction efficiency appears when extractant DCH18C6 is added; and the selectivity is K+��Rb+��Sr2+ Cs+��Na+��Li+. ROGERS et al[31] reported the extraction of sodium, cesium and strontium nitrates from aqueous solution into 18-crown-6 (18C6), DCH 18C6 and 4, 4��(5��)-di-(tert- butylcyclohexano)-18-crown-6 (Dtb18C6) in CnmimPF6 (n=4, 6, 8). It was found that the best extraction effects is Dtb18C6 and the selectivity is Sr2+��Cs+��Na+.
Since the partitioning of metal ions from aqueous solutions into ionic liquids is inefficient as a result of the tendency of the metal cations to remain hydrated in the aqueous phase, additional extractants, in types of crown ethers[30], calixarenes[31], ditizone[32] and others[33-60] were used. These species significantly enhance the partitioning of metal ions by forming complexes. Many research works have been focused on the extraction and separation of alkali metals[30-31, 38-39], alkali metals extracting heavy metals[29, 37-39, 41-42], heavy and radioactive metals[32-33, 36-37, 42-49] , rare earth metals[43-60], and part works of this field have been reviewed by ZHAO et al[35] and CHEN et al[57]. The progress in IL extractions of metal ions, including alkali, alkaline earth, heavy and radioactive metals, and rare earth in recent years are given in Table 2.
Table 2 Extractions of metal ions in ionic liquids
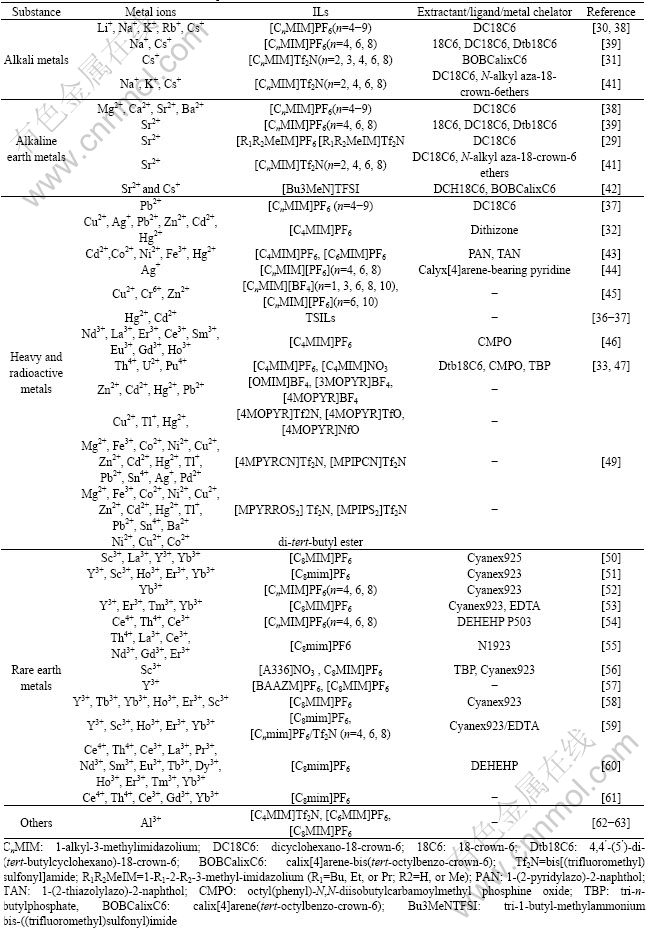
To improve the selection and the solubility of metal ion, the commonly used methods include a metal ion-ligating functional group in structure of one of the ions named Task Specific ionic liquids, which plays dual roles of both hydrophobic solvents and extractants. VISSER et al[36-37] presented new thiourea, urea and thioether derivative of ILs designed to extract heavy metal ions (e.g. Hg2+ and Cd2+), and the same methods have been used to the extraction and separation of rare earth metals by CHEN et al[56-57].
Research results described above indicate that the application of ILs as an alternative solvent to replace traditional organic solvents in liquid-liquid extraction of metal ions is very promising and may offer potential for development of efficient processing of nuclear materials.
5 Conclusions
Ionic liquids as a new-type of ��green�� media have shown important and potential application in the extraction and separation of nonferrous metal. It is indicated that ionic liquids process can be taken at or near room temperature and can significantly lower the energy consumption, operation costs, and pollutant emissions. In recent years, many efforts have been preformed and various important progress has been obtained on the extraction and separation of nonferrous metal in ionic liquids, such as metal oxide processing, extraction and separation of metal ions, mineral processing, electrodeposition and electrorefining of metals. In summary, ionic liquids are unique and promising solvents for extractions because of their non-volatility, adjustable hydrophobicity and polarity, and dissolution ability.
However, there are still some problems to be solved, since information about ionic liquids is not well known up to now and most of works are only performed in laboratory. How about the relationships between the properties and the structure of ionic liquids? Variations in cations and anions can produce a large number (1018) of ionic liquids and properties of ionic liquids depend on the structure of ions. How to do and how we start from for a specific research? What��s the mechanism for metal oxide and mineral processing, electrodeposition and extraction of metal ions? How do the ions such as metal ions, cations and anions of ionic liquids behave?
References
[1] FRAY D J, CHEN G Z. Reduction of titanium and other metal oxides using electrode oxidation [J]. Mater Sci Technol, 2004, 20: 295-300.
[2] CHERGINETS V. Handbook of solvents [M]. WYPYCH G. Toronto: Chem Tec Publishing, 2001: 1484-1496.
[3] WELTON T. Room-temperature ionic liquids: solvents for synthesis and catalysis [J]. Chem Rev, 1999, 99(8): 2071-2083.
[4] LI Ru-xiong. Green solvents-synthesis and application of ionic liquids [M]. Beijing: Chemical Industry Engineering Press, 2004: 10-16. (in Chinese)
[5] ZHANG Suo-jiang, L? Xing-mei. Ionic liquids from fundamental study to industrial application [M]. Beijing: Science Press, 2006: 12-16. (in Chinese)
[6] DENG You-quan. Ionic liquids��properties, preparation and application [M]. Beijing��China SINO-PEC Press, 2006: 334-344. (in Chinese)
[7] WHITEHEAD J A, LAWRENCE G A, MCCLUSKEY A. Green leaching: recyclable and selective leaching of gold-bearing ore in an ionic liquid [J]. Green Chem, 2004, 6: 313-315.
[8] THIED R C, SEDDON K R, PITNER W R, ROONEY D W. Nuclear fuel reprocessing, WO 99 41752 [P]. 1999.
[9] ZHANG M, KAMAVARAM V, REDDY R G. Aluminum electrowinning in ionic liquids at low temperature [J]. Light Metals, 2005: 583-588.
[10] WU B, REDDY R G, ROGERS R D. Production, refining and recycling of lightweight and reactive metals in ionic liquids, US 2002070122 [P]. 2002.
[11] McCLUSKEY A, LAWRANCE G A, LEITCH S K, OWEN M P, HAMILTON I C. Ionic liquids industrial applications for green chemistry [M]. ROGERS R D, SEDDON K R. Washington D C: American Chemical Society, 2002, 818: 199-212.
[12] FRAY D J. Emerging molten salt technologies for metals production [J]. JOM, 2001, 53: 26-31.
[13] ENDRES F, MACFARLANE D, ABBOTT A. Electrodeposition from ionic liquids [M]. Weinheim: WILEY-VCH Verlag, 2008: 83-120.
[14] DAI S, SHIN Y S, TOTH L M, BARNES C E. Comparative uv-vis studies of uranyl chloride complex in two basic ambient-temperature melt systems [J]. Inorg Chem, 1997, 36: 4900-4902.
[15] BELL R C, CASTLEMAN A W, THORN D L. Vanadium oxide complexes in room-temperature chloroaluminate molten salts [J]. Inorg Chem, 1999, 38: 5709-5715.
[16] ABBOTT A P, CAPPER G, DAVIES D L, RASHEED R K, TAMBYRAJAH V. Novel solvent properties of choline chloride/urea mixtures [J]. Chem Commun, 2003(11): 70-77.
[17] ABBOTT A, BOOTHBY D, CAPPER G, DAVIES D L, RASHEED R K. Deep eutectic solvents formed between choline chloride and carboxylic acids: versatile alternatives to ionic liquids[J]. J Am Chem Soc, 2004, 126: 9142-9147.
[18] ABBOTT A P, CAPPER G, DAVIES D L, SHIKOTRA P. Processing metal oxides using ionic liquids[J]. Trans Inst Min Metall C, 2006, 115: 115-119.
[19] ABBOTT A P, CAPPER G, DAVIES D L, MCKENZIE S O. Solubility of metal oxides in deep eutectic solvents based on choline chloride [J]. J Chem Eng, 2006, 51: 1280-1282.
[20] NOCKEMANN P, THIJS B, PITTOIS S, THOEN J, GLORIEUX C, VAN HECKE K, VAN MEERVELT L, KIRCHNER B, BINNEMANS K. Task-specific ionic liquid for solubilizing metal oxides [J]. J Phys Chem B, 2006, 110: 20978-20992.
[21] NOCKEMANN P, THIJS B, TATJANA N. Carboxyl-functionalized task-specific ionic liquids for solubilizing metal oxides[J]. Inorg Chem, 2008, 47(21): 9987-9999.
[22] MA Jiang-hua, LI Yu-ping, LI Hui-quan, ZHANG Yi. Synthesis of 1-ethyl-3-methylimidazolium hydrogen sulfate and its application in the electrolysis of aluminum [J]. The Chinese Journal of Process Engineering, 2007, 7: 1083-1088. (in Chinese)
[23] ABBOTT A P, McKENZIE K J. Application of ionic liquids to the electrodeposition of metals [J]. Phys Chem Chem Phys, 2006, 8: 4265-4279.
[24] HINES C C, COCALIA V A, ROGERS R D. Using ionic liquids to trap unique coordination environments: Polymorphic solvates of ErCl3 (OH2)4?2 ([C2mim]Cl) [J]. Chem Commun, 2008(8): 226-228.
[25] HINES C C, CORDES D B, Griffin S T, WATTS S I, COCALIA V A, ROGERS R D. Flexible coordination environments of lanthanide complexes grown from chloride-based ionic liquids [J]. New J Chem, 2008, 32: 872�C877.
[26] WHITEHEAD J A, ZHANG J, PEREIRA N, McCLUSKEY A, LAWRANCE G A. Application of 1-alkyl-3-methyl- imidazolium ionic liquids in the oxidative leaching of sulphidic copper, gold and silver ores [J]. Hydrometallurgy, 2007, 88: 109-120.
[27] LUCZAK J, JOSKOWSKA M, HUPKA J. Imidazolium ionic liquids in mineral processing [J]. Physicochemical Problems of Mineral Processing, 2008, 42: 223-236.
[28] DONG Tie-guang, HUA Yi-xin, ZHANG Qi-bo, ZHOU Dan-gui. Leaching of chalcopyrite with Br?nsted acidic ionic liquid at ambient pressure and low temperature [J]. Hydrometallurgy, 2009, 99: 33-38.
[29] DAI S, JU Y H, BBARNES C E. Solvent extraction of strontium nitrate by a crown ether using room-temperature ionic liquids [J]. J Chem Soc Dalton Trans, 1999(8): 1201-1202.
[30] CHUN S, DZYUBA S V, BARTSCH R A. Influence of structural variation in room-temperature ionic liquids on the selectivity and efficiency of competitive alkali metal salt extraction by a crown ether [J]. Anal Chem, 2001, 73: 3737-3741.
[31] LUO H, DAI S, BONNESEN P V, BUCHANAN A C, HOLBREY J D, BRIDGES N, ROGERS R D. Extraction of cesium ions from aqueous solutions using calix[4]arene-bis(tertoctylbenzo-crown-6) in ionic liquids[J]. Anal Chem, 2004, 76: 3078-3083.
[32] WEI G T, YANG Z, CHEN C J. Room temperature ionic liquid as a novel medium for liquid/liquid extraction of metal ions [J]. Anal Chim Acta, 2003, 488: 183-192.
[33] VISSER A E, ROGERS R D. Room-temperature ionic liquids: New solvents for f-element separations and associated solution chemistry [J]. J Solid State Chem, 2003, 171: 109-113.
[34] KOZONOI N, IKEDA Y. Extraction mechanism of metal ion from aqueous solution to the hydrophobic ionic liquid, 1-butyl-3-methylimidazolium nonafluorobutanesulfonate [J]. Monatsh Chem, 2007, 138: 1145-1151.
[35] ZHAO Hua, XIA Shu-qian , MA Pei-sheng. Use of ionic liquids as ��green�� solvents for extractions [J]. J Chem Technol Biotechnol, 2005, 80: 1089-1096.
[36] VISSER A, SWATLOWSKI R P, REICHERT R M, MAYTON R, SHEFF S, WIERZBICKI A, DAVIS J H, ROGERS R D. Task-specific ionic liquids for the extraction of metal ions from aqueous solutions [J]. Chem Commun, 2001: 135-136.
[37] VISSER A, SWATLOWSKI R P, ROGERS R D. Task-specific ionic liquids incorporating novel cations for the coordination and extraction of Hg2+ and Cd2+: Synthesis, characterization, and extraction studies [J]. Environ Sci Technol, 2002, 36: 2523-2529.
[38] BARTSCH RA, CHUN S, DZYUBA S V. Ionic liquids as novel diluents for solvent extraction of metal salts by crown ethers, ionic liquids: Industrial applications for green chemistry [M]. ROGERS R D, SEDDON K R. Washington D C: American Chemical Society, 2002: 58-68.
[39] VISSER A E, SWATLOSKI R P, REICHERT W M, GRIFFIN S T, ROGERS R D. Traditional extractants in nontraditional solvents: groups 1 and 2 extraction by crown ethers in room temperature ionic liquids [J]. Industrial & Engineering Chemistry Research, 2000, 39: 3596-3604.
[40] DAI S, JU Y H, BARNES C E. Solvent extraction of strontium nitrate by a crown ether using room-temperature ionic liquids [J]. Journal of the Chemical Society, Dalton Transactions, 1999, 8: 1201-1202.
[41] LUO H, DAI S, BONNESEN P V. Solvent extraction of Sr2+ and Cs+ based on room-temperature ionic liquids containing monoaza-substituted crown ethers [J]. Analytical Chemistry, 2004, 76: 2773-2779.
[42] CHEN P Y. The assessment of removing strontium and cesium cations from aqueous solutions based on the combined methods of ionic liquid extraction and electrodeposition [J]. Electrochimica Acta, 2007, 52(17): 5484-5492.
[43] VISSER A E, SWATLOSKI R P, GRIFFIN S T, HARTMAN D H, ROGERS R D. Liquid/liquid extraction of metal ions in room temperature ionic liquids [J]. Separation Science and Technology, 2001, 36: 785-804.
[44] SHIMOJO K, GOTO M. Solvent extraction and stripping of silver ions in room-temperature ionic liquids containing calixarenes [J]. Analytical Chemistry, 2004, 76: 5039-5044.
[45] VIDAL S, NEIVA CORREIA M J, MARQUES M M, ISMAEL M R, ANGELINO REIS M T. Studies on the use of ionic liquids as potential extractants of phenolic compounds and metal ions [J]. Separation Science and Technology, 2004, 39: 2155-2169.
[46] NAKASHIMA K, KUBOTA F, MARUYAMA T, GOTO M, Ionic liquids as a novel solvent for lanthanide extraction [J]. Analytical Sciences, 2003, 19: 1097-1098.
[47] BASTON G M N, BRADLEY A E, GORMAN T, HAMBLETT I, HARDACRE C, HATTER J E, HEALY M J F, HODGSON B, LEWIN R, LOVELL K V, SEDDON K R. Ionic liquids for the nuclear industry: A radiochemical, structural, and electrochemical investigation [M]//In Ionic Liquids Industrial Applications for Green Chemistry. ROGERS R D, SEDDON K R. Washington D C: American Chemical Society, 2002: 162-177.
[48] PAPAICONOMOU N, LEE J M, SALMINEN J, STOSCH M, PRAUSNITZ J M. Selective extraction of copper, mercury, silver, and palladium ions from water using hydrophobic ionic liquids [J]. Ind Eng Chem Res, 2008, 47: 5080-5086.
[49] JITENDRA R, HARJANI A, SINGER R D. Removal of metal ions from aqueous solutions using chelating task-specific ionic liquids [J]. Dalton Trans, 2008(34): 4595-4601.
[50] SUN Xiao-qi, WU Dong-bei, CHEN Ji, LI De-qian. Separation of scandium(��) from lanthanides(��) with room temperature ionic liquid based extraction containing Cyanex 925 [J]. J Chem Technol Biotechnol, 2007, 82: 267-272.
[51] SUN Xiao-qi, PENG Bo, JI Yang, CHEN Ji, LI De-qian. The solid�Cliquid extraction of yttrium from rare earths by solvent (ionic liquid) impreganated resin coupled with complexing method [J]. Separation and Purification Technology, 2008, 63: 61-68.
[52] PENG Bo, SUN Xiao-qi,, CHEN Ji. Extraction of yttrium (��) into [C8mim]PF6 containing cyanex 923 [J]. Journal of Rare Earth, 2007, 25(Z1): 153-156.
[53] SUN Xiao-qi, PENG Bo, JI Yang, CHEN Ji, LI De-qian. An effective method for enhancing metal-ions selectivity of ionic liquid-based extraction system: adding water-soluble complexing agent [J]. Talanta, 2008, 74(4): 1071-1074.
[54] ZUO Yong, CHEN Ji, LI De-qian. Extraction and separation of thorium (��) from lanthanides (��) with room temperature ionic liquids containing primary amine N1923 [C]//International Solvent and Extraction Conference 2008. Tuscon, America, 2008: 320-325.
[55] ZUO Yong, CHEN Ji, LI De-qian. Reversed micellar solubilization extraction and separation of thorium (��) from rare earth (��) by primary amine N1923 in ionic liquid [J]. Separation and Purification Technology, 2008, 63: 684-690.
[56] SUN Xiao-qi, PENG Bo, JI Yang, CHEN Ji, LI De-qian. Application of rtil-based system cyanex923(925)/[Csmim][PF6] and tbp/[A336][NO3] in scandium(��) extraction [J]. Journal of the Chinese Rare Earth Society, 2007, 25(4): 417-421. (in Chinese)
[57] CHEN Ji, LI De-qian, Application of ionic liquids on the rare earth separation [J]. Acta Chemical Process, 2008(Z1): 54-59. (in Chinese)
[58] LIU Ying-hui, SUN Xiao-qi, LUO Fang, CHEN Ji. Ionic liquids and cyanex 923-doped organic-inorganic hybrid materials for Y(��) and Lanthauides (��) separation [J]. Chinese Journal of Rare Metals, 2007, 31(3): 395-398.
[59] SUN Xiao-qi, PENG Bo, JI Yang, Chen Ji, LI De-qian, LUO Fang. Ionic liquids based ��all-in-one�� synthesis and photoluminescence properties of lanthanide fluorides [J]. J Phys Chem C, 2008, 112: 10083-10088.
[60] ZUO Yong, LIU Ying-hui, CHEN Ji, LI De-qian. Extraction and recovery of cerium(��) along with fluorine(��) from bastnasite leaching liquor by DEHEHP in [C8mim]PF6 [J]. J Chem Technol Biotechnol, 2009, 84: 949-956.
[61] ZUO Yong, LIU Ying-hui, CHEN Ji, LI De-qian. The separation of cerium(��) from nitric acid solutions containing thorium(��) and lanthanides(��) using pure [C8mim]PF6 as extracting phase [J]. Ind Eng Chem Res, 2008, 47 (7): 2349-2355.
[62] ZHANG M, KAMAVARAM V, REDDY R G. Application of fluorinated ionic liquids in the extraction of aluminum [J]. Light Metals, 2004: 315-319.
[63] WU B, REDDY R G, ROGERS R D. Production, refining and recycling of lightweight and reactive metals in ionic liquids. US6881321 [P]. 2008.
Foundation item: Project(50904031) supported by the National Natural Science Foundation of China; Project(2008E0049M) supported by the Natural Science Foundation of Yunnan Province, China; Project(07Z40082) supported by the Science Foundation of the Education Department of Yunnan Province, China; Project(2007-16) supported by the Science Foundation of Kunming University of Science and Technology, China
Corresponding author: TIAN Guo-cai; Tel: +86-871-5162008; E-mail: tiangc@iccas.ac.cn; tiangc01@gmail.com
DOI: 10.1016/S1003-6326(09)60171-0

