Isolation and characterization of YNTC-1,a novel Alicyclobacillus sendaiensis strain
来源期刊:中南大学学报(英文版)2008年第4期
论文作者:丁建南 何环 张成桂 于一尊 邱冠周
文章页码:508 - 514
Key words:bioleaching; sulphide minerals; isolation; strain YNTC-1; Alicyclobacillus sendaiensis; Sulfobacillus thermosulfidooxi- dans
Abstract:
A heterotrophic acidothermophilic bacterial strain, YNTC-1, was isolated from an acidic hot spring in Tengchong, Yunan, China. YNTC-1 grows at pH value of 1.5-8.0 and temperature of 40-70 ℃, with optimal pH and temperature at 3.0 and 55 ℃, respectively. The cells of the strain are in shape of short rod, with 1.0-1.2 μm in length and 0.7-0.8 μm in diameter, and with distinct spores at both poles of each cell. The predominant fatty acids in cellular membrane of the strain are C18:1 ω7c. 16s rRNA gene analysis reveals that this strain is closely related to Alicyclobacillus sendaiensis, with over 99% sequence similarity. Based on phenotypic and genotypic analyses, YNTC-1 is identified as a member of A. sendaiensis. Considering some important morphological and biochemical differences between strain YNTC-1 and A. sendaiensis ATCC 27009T, YNTC-1 may be proposed to be a novel subspecies of A. sendaiensis. However, this viewpoint has to be confirmed by further studies. Co-bioleaching of pyrite and chalcopyrite with strain YN22, Sulfobacillus thermosulfidooxidans, shows that strain YNTC-1 has no evident influence on bioleaching rates of these two sulphide minerals.
基金信息:the Chinese National Natural Science Foundation for Innovative Research Groups
the Major State Basic Research and Development Program of China
J. Cent. South Univ. Technol. (2008) 15: 508-514
DOI: 10.1007/s11771-008-0096-6![]()
DING Jian-nan(丁建南)1, 2, HE Huan(何 环)1, ZHANG Cheng-gui(张成桂)1,
YU Yi-zun(于一尊)3, QIU Guan-zhou(邱冠周)1
(1. School of Resources Processing and Bioengineering, Central South University, Changsha 410083, China;
2. Biological Resources Institute, Jiangxi Academy of Sciences, Nanchang 330029, China;
3. Institute of Subtropical Agriculture, the Chinese Academy of Sciences, Changsha 410125, China)
Abstract: A heterotrophic acidothermophilic bacterial strain, YNTC-1, was isolated from an acidic hot spring in Tengchong, Yunan, China. YNTC-1 grows at pH value of 1.5-8.0 and temperature of 40-70 ℃, with optimal pH and temperature at 3.0 and 55 ℃, respectively. The cells of the strain are in shape of short rod, with 1.0-1.2 ?m in length and 0.7-0.8 ?m in diameter, and with distinct spores at both poles of each cell. The predominant fatty acids in cellular membrane of the strain are C18:1 ω7c. 16s rRNA gene analysis reveals that this strain is closely related to Alicyclobacillus sendaiensis, with over 99% sequence similarity. Based on phenotypic and genotypic analyses, YNTC-1 is identified as a member of A. sendaiensis. Considering some important morphological and biochemical differences between strain YNTC-1 and A. sendaiensis ATCC 27009T, YNTC-1 may be proposed to be a novel subspecies of A. sendaiensis. However, this viewpoint has to be confirmed by further studies. Co-bioleaching of pyrite and chalcopyrite with strain YN22, Sulfobacillus thermosulfidooxidans, shows that strain YNTC-1 has no evident influence on bioleaching rates of these two sulphide minerals.
Key words: bioleaching; sulphide minerals; isolation; strain YNTC-1; Alicyclobacillus sendaiensis; Sulfobacillus thermosulfidooxi- dans
1 Introduction
Microbial hydrometallurgy is often considered more environmentally friendly and economical than conventional pyrometallurgy[1-3]. Bacterial leaching of metal sulfides has developed rapidly in the last decades. Recovering metals from sulphide minerals by acidophiles has developed into a successful and expanding area of biotechnology[4]. Based on the great commercial importance of acidophilic organisms in hydrometallurgy, this research focused on screening acidophiles from acidic water samples such as acidic mine drainages and acidic hot springs. A number of autotrophic or mixtrophic acidophilic microorganisms capable of oxidizing ferrous iron and sulfur were isolated and identified, including several acidothermophiles. However, microbes in the samples were considerably diverse and some heterotrophic acidophilic bacteria were found coexisting with the isolated autotrophic or mixtrophic acidophiles. This suggests a special interaction between the two groups of inhabitants, which may result in both partners to gain benefits[5]. It was reported that Acidithiobacillus ferrooxidans and Leptospirillum ferrooxidans, the most widely used leaching microbes in the processing of metal sulfide ores, were both sensitive to organic acids and other small molecular organic compounds, which inhibited the growth of these microbes. Heterotrophic bacteria could remove this inhibition by metabolising the organic materials, enhance the growth of the autotrophs and as a result improve leaching rate of metal sulfides[6-7].
With view of the above discussion, a study was undertaken to identify a newly isolated heterotrophic acidothermophilic strain YNTC-1 coexisting in an acidic hot spring with the ferrous iron and sulfur oxidizing acidothermophile, strain YN22 of Sulfobacillus thermosulfidooxidans, and experiments for co- bioleaching of pyrite and chalcopyrite by mixing the two bacteria were carried out to determine whether the heterotroph has any influence on the leaching rates of the two sulfide minerals, although it could not oxidize ferrous iron or sulfur.
2 Experimental
2.1 Strain isolation
A water sample was collected from an acidic hot spring with a temperature of 80 ℃ and pH 3.5, located at Tengchong, Yunnan of southwestern China. 10 mL of the sample was inoculated for the enrichment of ferrous iron oxidizing microorganisms in 100 mL liquid medium containing (g/L) (NH4)2SO4 (0.4), MgSO4・7H2O (0.5), K2HPO4 (0.2), KCl (0.1), FeSO4・7H2O (20) and yeast extract (0.1). The medium was acidified to pH 1.8 with H2SO4 and autoclaved before filter-sterilized ferrous iron was added. The hydrothermal sample was continuously enriched at 60 ℃ and 180 r/min three times. A ferrous iron and sulfur oxidizer, strain YN22 of Sulfobacillus thermosulfidooxidans, was successfully isolated by serial dilution of the finally enriched culture. The enriched culture was then re-enriched for heterotrophic acidothermophiles by adding 10 mL of the culture in 100 mL of another medium consisting of (g/L) (NH4)2SO4 (2.0), KCl (0.1), MgSO4?7H2O (0.5), K2HPO4 ( 0.5), glucose (1.0) and yeast extract (0.1), with pH adjusted to 2.5 by KOH or H2SO4 solution. The re-enrichment cultivation was conducted in a 250-mL flask on a rotary shaker at 170 r/min and 60 ℃. In the course of incubation, the turbidity and the odor of the liquid medium were monitored. After an obvious color change and a volatile aroma of the medium were observed, 10 mL of the culture broth was transferred to a new 250-mL flask containing 100 mL fresh medium. These operations were repeated three times and the final enriched culture was serially diluted with sterilized liquid medium. Aliquot of 0.1 mL, respectively from 105, 106, 107 and 108 times diluted microbial suspensions was spread onto the solid medium (pH 3.0) prepared by gelling the liquid medium for heterotrophic microbes with 1.5% (ratio of mass to volume, g/mL) agar. Three dishes were inoculated for each dilution and incubated at 60 ℃. A beaker of sterilized water was necessary in the incubator to prevent the solid medium from being dried. One of the colonies, YNTC-1, appearing on the solid agar medium after incubation for 2 d, was picked up and inoculated into the corresponding liquid medium. The isolate was plated three times to ensure its purity. Purity of strain YNTC-1 was confirmed by microscopic examination until all cells were similar in shape and size.
2.2 Strain identification
Unless otherwise stated, all cultures of strain YNTC-1 for identification study were carried out in the liquid medium for heterotroph as described above.
2.2.1 Morphological and physiological study
To estimate the influence of temperature and initial pH on bacterial growth, strain YNTC-1 was cultured in the liquid medium (pH 2.5) and bacteria at stationary phase were collected by centrifugation and washed twice with dilute sulfuric acid (pH 2.5) for inoculation. The collected bacteria (2.9×106 mL-1) were re-suspended in one group of 250-mL flasks containing 100 mL medium inoculated at the same pH value of 2.5 and different temperatures from 30 to 80 ℃ for optimal temperature determination, and in the other group of 250-mL flasks containing the same medium inoculated at the same temperature of 60 ℃ and different pH values from 2.0 to 8.0 for optimal pH value determination. All cultures were repeated in triplicate. Liquid samples (100 ?L) were withdrawn from the flasks and diluted with sterilized water if necessary at a 2 h interval to measure bacterial population density in culture supernatants by direct cell counts with a Petroff-Hauser chamber on an Olympus CX31 microscope.
Following optimal temperature and pH value determination, the strain was triply inoculated at the optimal temperature and pH value on a rotary shaker (170 r/min) using the same method as mentioned above and the population density of the strain was monitored by direct counting under microscope every 2 h as described above. The data of this experiment were used to construct a growth curve of the strain.
The cells at exponential phase were harvested by centrifugation (10 000 r/min for 5min) for morphological study. Morphological characteristics of the harvested cells were observed by optical microscope (Olympus CX31) and their substructures were examined by scanning electron microscope (SEM, JEOL JSM-6360 LV).
2.2.2 Polymerase chain reaction(PCR) amplification of 16S rRNA gene
To extract genomic DNA of strain YNTC-1 for 16S rRNA gene amplification, stationary phase cells of the strain were collected, washed with 1.0 mol/L H2SO4 three times and then re-suspended in 200 ?L Tris-EDTA (TE) buffer (pH 8.0). Genomic DNA was extracted using an EZ-10 Spin Column Genomic DNA Minipreps Kit (Bio Basic Inc.) according to the introduction of the kit. Primers 63f (5′-CAGGCCTAACACATGCAAGTC-3′) and 1387r (5′-GGGCGGWGTGTACAAGGC-3′) were provided by Sunbiotech Co., Ltd. Beijing, to amplify 16S rRNA gene of the strain. Amplification was performed in a 25 ?L mixture containing 2.5 ?L 10×PCR buffer, 2.5 ?L (25 mmol) MgCl2, 1 μL (10 mmol) dNTP mix (Fermentas), 1 ?L of each primer (62.5 μmol), 1 ?L (5 units) Taq polymerase (Fermentas), 1 ?L DNA template and 15 ?L sterilized double distilled water. Negative- DNA control was made in the same volume including identical components except the template DNA, which was replaced by 1 ?L of sterilized double distilled water. The DNA amplifications were carried out in a Biometra DNA thermal cycler with a temperature program consisting of the initial denaturation at 94 ℃ for 5 min, followed by 33 cycles of denaturation at 94 ℃ for 45 s, annealing at 55 ℃ for 45 s, polymerization at 72 ℃ for 90 s, and final elongation at 72 ℃ for 10 min. The product of PCR was purified using an E.Z.N.A? gel extraction kit (Omega Bio-Tek, Inc.) and cloned with pBS-T PCR products clone kit (Tiangen Biotech Co. Ltd., Beijing) according to the method described by XIA et al[8].
Based on the similarities in 16S rDNA sequences between strain YNTC-1 and the related species, a phylogenetic tree demonstrating the relationship among the involved strains was constructed with softwares Clustal X (1.8) and MEGA3.
2.2.3 Fatty acid analysis
Strain YNTC-1 was cultured in the medium described by GOTO et al[9]. Cells at exponential phase were centrifugally harvested, washed with sterilized water three times and freeze-dried. Fatty acids were analyzed using gas chromatograph (HP6890, HP Corp, USA) and data analysis was performed with Sherlock microbial identification system (Sherlock Version 4.5, MIDI Corp. USA)[10]. All the tests for fatty acid analysis were completed in the Institute for Microbiology and Epidemiology of Academy of Military Medical Sciences of Beijing, China.
2.3 Bioleaching of pyrite and chalcopyrite
The two co-isolated strains, YN22 and YNTC-1, were used in leaching pyrite (FeS2) and chalcopyrite (CuFeS2) to elucidate the influence of strain YNTC-1 on the bioleaching rates of the two sulfide minerals. Strain YN22 was cultivated in YE (yeast extract)-supplemented 9K medium with the composition (g/L) of (NH4)2SO4 (3.0), Ca(NO3)2 (0.01), KCl (0.1), MgSO4.7H2O (0.5), K2HPO4 (0.5), YE (0.2) and FeSO4・7H2O (30), and strain YNTC-1 was grown in the medium described in the enrichment of this strain. After two subcultures, cells of the two strains were respectively obtained by centrifugation and washed twice using H2SO4 solution (pH 2.0) as the inoculums for the bioleaching tests of pyrite and chalcopyrite. The pyrite and chalcopyrite powders used in this experiment had a size distribution of 95% smaller than 74 ?m, and their chemical compositions (Table 1) were analyzed by atomic absorption spectrometry (AAS). The oxidative dissolutions of pyrite and chalcopyrite by pure culture of strain YN22 and mixed culture of YN22 and YNTC-1 were respectively studied in 250-mL shake flasks containing the same medium as that used for strain YN22 with 3% (ratio of mass to volume, g/mL) mineral powder. Pure cultures were inoculated with 1.0×107 /mL viable cells of strain YN22 and mixed cultures were co-inoculated by strain YN22 and strain YNTC-1, each with a viable bacterial mass equivalent to 1.0×107 /mL cells. The cultures (in triplicate) for bioleaching tests were run at 54 ℃, pH 2.0 and 170 r/min, and sampled at regular intervals to determine concentrations of soluble iron and copper using AAS. Parallel cultures without bacteria (also in triplicate) were performed as controls under the identical conditions.
Table 1 Chemical compositions of pyrite and chalcopyrite used in leaching tests (mass fraction, %)

3 Results and discussion
3.1 Identification of strain YNTC-1
3.1.1 Colony and cell morphology
When grown on agar-solidified medium, strain YNTC-1 produces white, round, creamy and convex- shaped colonies with incomplete edges, 0.5-2.0 mm in diameter (Fig.1). The cells of strain YNTC-1 are oval to short rods, 0.75-0.81 ?m in diameter and 1.0-1.2 ?m in length, with round ends (Fig.2). Distinct spores at both poles of each cell can be observed in some individuals of this strain, as shown with white arrows in Fig.2.

Fig.1 Colony morphology of strain YNTC-1
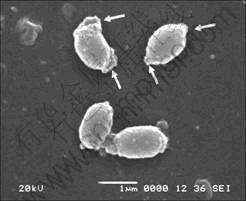
Fig.2 SEM image of strain YNTC-1
3.1.2 Temperature and pH ranges
The growth status of strain YNTC-1 at different temperatures is shown in Fig.3. It is indicated that the strain is thermophilic and able to survive in a quite wide temperature range from 30 to 80 ℃, with an optimum at 55 ℃. No growth is detected at 85 ℃ or at temperature lower than 25 ℃ after inoculation for 7 d. Strain YNTC-1 grows in a pH range from 1.5 to 8.0, with optimal growth at pH 3.0 (Fig.4), but no growth is detected at pH lower than 1.0 or higher than 8.5.

Fig.3 Effect of temperature on growth of strain YNTC-1 (ρ is cell density)
Strain YNTC-1 grew in the liquid medium as mentioned in the enrichment of this strain and viable bacterial density was monitored by direct counting under optical microscope at regular intervals during 48 h of inoculation at pH 3.0, 55 ℃ and 170 r/min. The data indicate that the strain can be activated in a short time and grows fast, requiring only 14 h to reach a bacterial density of 2.41×108 /mL from the initial 1.00×106 /mL (Fig.5).
3.1.3 Phylogeny
The nearly complete sequence consisting of 1 424 bases of the amplified 16S rDNA of strain YNTC-1 was determined. 16S rDNA sequence analysis indicates that the closest relative of strain YNTC-1 is Alicyclobacillus sendaiensis, with a 16S rDNA sequence similarity over 99%. Phylogenetic tree based on 16S rDNA sequences of YNTC-1 and the species within genus Alicyclobacillus was constructed using softwares Clustal X1.8 and MEGA3.01 (Fig.6). The results also clearly show thatstrain YNTC-1 is the most closely related to Alicyclobacillus sendaiensis.

Fig.4 Effect of pH value on growth of strain YNTC-1 (ρ is cell density)

Fig.5 Growth curve of strain YNTC-1 (ρ is cell density)

Fig.6 Phylogenetic tree based on 16 rRNA gene sequences of strain YNTC-1 and species within Alicyclobacillus (GenBank accession numbers are shown in parentheses; numbers depict bootstrap values obtained for bootstrap sampling of 1 000)
3.1.4 Fatty acid composition
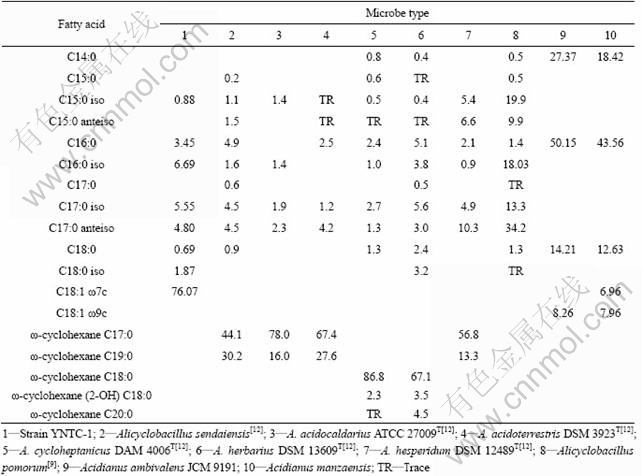
There are 8 groups of fatty acids found in strain YNTC-1 by gas chromatograph. The predominant one is the unsaturated C18:1 ω7c, accounting for 76.07% of the total fatty acids detected in this strain, as demonstrated in column 1 of Table 2. The other 7 groups C16:0 iso, C17:0 iso, C17:0 anteiso, C16:0, C18:0 iso, C15:0 iso and C18:0, which account for 6.69%, 5.55%, 4.80%, 3.45%, 1.87%, 0.88% and 0.69%, respectively (Table 2).
According to the characteristics of genus Alicyclobacillus, ω-alicyclic acids are the predominant membrane fatty acids in the genus[11-12] and the main factors for the identification of this genus. These fatty acids contain six- or seven-carbon rings, and play an important role in the growth of Alicyclobacillus bacteria in thermal and acidic environments. However, Alicyclobacillus pomorum has optimal temperatures of 45-50 ℃ for growth and is identified as a species in the genus, although it is a microbe without ω-alicyclic fatty acids (Table 2). Thus the presence of ω-alicyclic acids does not necessarily relate to the physiological property of being acidothermophilic or to species identification of genus Alicyclobacillus. The fact that ω-alicyclic acids are not absolutely necessary for acidothermophilic property is reinforced by Acidianus ambivalens and A. manzaensis. The two acidothermophilic archaea possess no ω-alicyclic acids (Table 2), but have optimal growth at temperatures from 60 to 70 ℃ and pH values between 1.5 and 2.0.
Alicyclobacillus, once assigned to genus Bacillus, consists of 8 species: Alicyclobacillus acidiphilus, A. acidocauldarius, A. acidoterrstris, A. cycloheptanicus, A. herbarius, A. hepsperidum, A. pomorum and A. sendaiensis[13]. These species are grouped into genus Alicyclobacillus according to their distinct 16S rRNA gene sequences, morphological and physiological characteristics, and unique membrane component of ω-alicyclic fatty acid lipids including ω-cyclohexane, ω-cycloheptane, ω-alicyclic, or straight- and branched-chain fatty acid lipids[12, 14].
Alicyclobacillus species are acidophilic and thermophilic bacteria, growing well in acidic and thermal environments. Most of existing Alicyclobacillus strains were isolated from sulfide-rich mine drainages and acidic hot springs except some from acidic beverages, for example, fruit juice[15]. CHEN et al[16] surveyed Alicyclobacillus bacteria in some hot springs of south China and found that the genus had extensive distribution. Strain YNTC-1 was isolated from a sample collected from an acidic hot spring in Tengchong, Yunnan, southwestern China. Although this strain does not have ω-alicyclic fatty acids, the phylogenic analysis of 16S rDNA and physiological characteristics have clearly indicated that it should be classified into genus Alicyclobacillus, being identified as a member of A. sendaiensis. Considering the difference in fatty acid composition between strain YNTC-1 and A. sendaiensis, the strain may be proposed to be a novel subspecies of A. sendaiensis. However, this viewpoint has to be confirmed by further studies.
Table 2 Membrane fatty acid compositions of strainYNTC-1, related Alicyclobacillus species and two archaea (data in each column are percentages of membrane fatty acids in a species, %)
3.2 Influence of strain YNTC-1 on bioleaching rates of pyrite and chalcopyrite
The changes of iron and copper leaching rates with time during bioleaching of chalcopyrite are shown in Fig.7. The leaching rates of iron and copper with pure culture of strain YN22 are nearly the same as those with the mixed culture of YN22 and strain YNTC-1, which suggests that the heterotrophic strain YNTC-1 has no effective influence on the bioleaching rate of chalcopyrite by strain YN22. From the leaching curve, it is obvious that the dissolution rate of iron (from slope of the curves) is higher than that of copper both in the pure culture of YN22 and the mixed culture of YN22 and YNTC-1 in the first 21 d. The dissolution of iron slightly levels off after that time, whereas the copper leaching continues, and there are clear precipitates at the bottom of flasks. According to the description of chalcopyrite leaching mechanism and the precipitation of jarosite, the leaching rate of copper is often inhibited by the passivation[17]. Because ferric ion is a very important oxidant in bioleaching and responsible for chalcopyrite dissolution[18-19], the precipitation of ferric ions will consume the oxidant, in other words, the leaching rate of copper will slow down with the decrease of ferric ions. That is the reason why the curves of copper dissolution level off after 28 d (Fig.7). At the same time, although the leaching rate of copper by mixed culture is slightly higher than that by pure culture, both leaching trends of copper have no distinct difference. The final leaching rates of iron and copper with pure culture and mixed culture are 20.35% (iron), 27.76% (copper) and 20.61% (iron), 29.02% (copper), respectively.
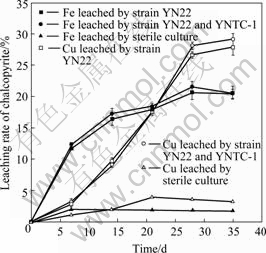
Fig.7 Effect of strain YNTC-1 on iron and copper ions leaching rates of chalcopyrite
The iron leaching rates of pyrite with pure strain YN22 and mixed culture of YNTC-1 and YN22 are shown in Fig.8. The iron dissolution goes up continuously with time for both pure and mixed cultures, and the final leaching rates in both cultures are 52.3% and 56.9% respectively. There is no remarkable difference between these two cultures either. This result is in disaccord with that by NAOKO and JOHNSON[20]. They reported that a mixed culture of Leptospirillum MT6 and the obligate heterotroph Alicyclobacillus Y004 oxidized pyrite more rapidly and more completely than a pure culture of Leptospirillum MT6. And they suggested that mutualistic interactions between physiologically distinct moderately thermophilic acidophiles, involving transfer of organic and inorganic carbon and transformations of iron and sulfur, maybe have critical role in optimizing pyrite dissolution.
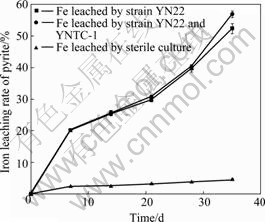
Fig.8 Effect of strain YNTC-1 on iron leaching rate of pyrite
The copper and iron dissolutions are almost negligible for the sterile controls in both chalcopyrite and pyrite leaching compared with inoculated cultures (Figs.7 and 8).
4 Conclusions
1) YNTC-1, a heterotrophic acidothermophilic bacterial strain, is co-isolated with a mixtrophic strain (YN22) of Sulfobacillus thermosulfidooxidans from an acidic hot spring in Tengchong, Yunnan, southwestern China. The optimum temperature and pH value for strain YNTC-1 growth are 55 ℃ and 3.0, respectively. When grown on agar-solidified medium, the strain produces white, round, creamy and convex-shaped colonies. The cells of this strain are oval to short rods, with 0.75-0.81 ?m in diameter and 1.0-1.2 ?m in length, and with round ends. Distinct spores at both poles of each cell can be observed in some individuals of this strain. 16S rDNA sequence analysis indicates that the closest relative of strain YNTC-1 is Alicyclobacillus sendaiensis, with a 16S rDNA sequence similarity over 99%. The predominant fatty acids are the unsaturated C18:1 ω7c, accounting for 76.07% of the total fatty acids detected in this strain. On the basis of phenotypic, genotypic and phylogenetic analyses, YNTC-1 is identified as a member of A. sendaiensis.
2) Strain YNTC-1 may be proposed to be a novel subspecies of A. sendaiensis due to some important morphological and biochemical differences observed between the two bacteria. However, further studies are needed for this view.
3) The leaching rates of chalcopyrite and pyrite with pure culture of strain YN22 of S. thermosulfidooxidans and mixed culture of strain YN22 and strain YNTC-1 have no distinct difference, indicating that strain YNTC-1 has no evident influence on the bioleaching rates of these two sulphide minerals mediated by strain YN22, S. thermosulfidooxidans.
References
[1] WITNE J Y, PHILLIPS C V. Bioleaching of Ok Tedi copper concentrate in oxygen- and carbon dioxide-enriched air [J]. Minerals Engineering, 2001, 14(1): 25-48.
[2] KREBS W, BROMBACHER C, BOSSHARD P P, BACHOFEN R, BRANDL H. Microbial recovery of metals from solids [J]. FEMS Microbiol Rev, 1997, 20(3/4): 605-617.
[3] BOSECKER K. Bioleaching: Metal solubilization by microorganisms [J]. FEMS Microbiol Rev, 1997, 20(3/4): 591-604.
[4] ROHWERDER T, GEHRKE T, KINZLER K, SAND W. Bioleaching review (part A): Progress in bioleaching: Fundamentals and mechanisms of bacterial metal sulfide oxidation [J]. Appl Microbiol Biotechnol, 2003, 63(3): 239-248.
[5] JOHNSON D B. Biodiversity and ecology of acidophilic microorganisms [J]. FEMS Microbiol Ecol, 1998, 27(4): 307-317.
[6] JOHNSON D B. Importance of microbial ecology in the development of new mineral technologies [J]. Hydrometallurgy, 2001, 59(2/3): 147-157.
[7] RODGERS L E, HOLDEN P J, FOSTER L J R. Culture of Acidiphilium cryptum BV1 with halotolerant Alicyclobacillus-like spp.: Effects on cell growth and iron oxidation [J]. Biotechnology Letters, 2002, 24(18): 1519-1524.
[8] XIA Jin-lan, PENG An-an, HE Huan. A new strain Acidithiobacillus albertensis BY-05 for bioleaching of metal sulfides ores [J]. Trans Nonferrous Met Soc China, 2007, 17(1): 168-175.
[9] GOTO K, MOCHIDA K, ASAHARA M, SUZUKI M, KASAI H, YOKOTA A. Alicyclobacillus pomorum sp. nov.: A novel thermo- acidophilic, endospore-forming bacterium that does not possess ω-alicyclic fatty acids, and emended description of the genus Alicyclobacillus [J]. International Journal of Systematic and Evolutionary Microbiology, 2003, 53(5): 1537-1544.
[10] WU Yu-ping, XU Jian-ming, WANG Hai-zhen. Application of Sherlockmis in identification of soil bacteria [J]. Acta Pedologica Sinica, 2006, 43(4): 642-647. (in Chinese)
[11] GOTO K, MATSUBARA H, MOCHIDA K, MATSUMURA T, HARA Y, NMA M, YAMASATO K. Alicyclobacillus herbarius sp. nov.: A novel bacterium containing ω-cycloheptane fatty acids, isolated from herbal tea [J]. International Journal of Systematic and Evolutionary Microbiology, 2002, 52(1): 109-113.
[12] NAOKI T, YURI I, OSAMU S, HISASHI H, TORU N, TOKUZO N. Alicyclobacillus sendaiensis sp. nov., a novel acidophilic, slightly thermophilic species isolated from soil in Sendai, Japan [J]. International Journal of Systematic and Evolutionary Microbiology, 2003, 53(4): 1081-1084.
[13] CHEN Shi-qiong, CHEN Wen-feng, HU Xiao-song, ZHAO Guang-hua. Rapid identification of Alicyclobacillus isolated from concentrated apple juice processing by 16S rDNA PCR-RFLP [J]. Journal of Chinese Institute of Food Science and Technology, 2006, 6(2): 99-102. (in Chinese)
[14] SIMBAHAN J, DRIJBER R, BLUM P. Alicyclobacillus vulcanalis sp. nov., a thermophilic, acidophilic bacterium isolated from Coso Hot Springs, California, USA [J]. International Journal of Systematic and Evolutionary Microbiology, 2004, 54(5): 1703-1707.
[15] CHEN Shi-qiong, TANG Qing-yan, ZHANG Xiao-dong, ZHAO Gung-hua, HU Xiao-song, LIAO Xiao-jun, CHEN Fang, WU Ji-hong, XIANG Hua. Isolation and characterization of hermo- acidophilic endospore-forming bacteria from the concentrated apple juice-processing environment [J]. Food Microbiology, 2006, 23(5): 439-445.
[16] CHEN Zhi-wei, JIANG Cheng-ying, LIU Shuang-jiang. Survey on and phylogeny of Alicyclobacillus species in hot springs of southern China’s Guangdong and Yunnan Provinces [J]. Microbiology, 2004, 31(3): 50-54. (in Chinese)
[17] SHU Rong-bo, RUAN Ren-man, WEN Jian-kan. Review on passivation of chalcopyrite during bioleaching process [J]. Chinese Journal of Rare Metal, 2006, 30(3): 395-400. (in Chinese)
[18] YASUHIRO K, MASAHIKO T, SATORU A. Copper recovery from chalcopyrite concentrate by acidophilic thermophile Acidianus brierleyi in batch and continuous-flow stirred tank reactors [J]. Hydrometallurgy, 2001, 59(2/3): 271-282.
[19] FALCO L, POGLIANI C, CURUTCHET G. A comparison of bioleaching of covellite using pure cultures of Acidithiobacillus ferrooxidans and acidithiobacillus thiooxidans or a mixed culture of Leptospirillum ferrooxidans and Acidithiobacillus thiooxidans [J]. Hydrometallurgy, 2003, 71(1/2): 31-36.
[20] NAOKO O, JOHNSON D B. Biooxidation of pyrite by defined mixed cultures of moderately thermophilic acidophiles in pH-controlled bioreactors: Significance of microbial interactions [J]. Biotechnology and Bioengineering, 2004, 87(5): 574-583.
Foundation item: Project(50621063) supported by the Chinese National Natural Science Foundation for Innovative Research Groups; Project (2004CB619201) supported by the Major State Basic Research Development Program of China
Received date: 2007-12-22; Accepted date: 2008-01-30
Corresponding author: QIU Guan-zhou, Professor; Tel: +86-731-8879212; E-mail: qgzfblw@yahoo.com.cn
(Edited by CHEN Wei-ping)

