Effect of NiO content on corrosion behaviour of Ni-xNiO-NiFe2O4 cermets in Na3AlF6-Al2O3 melts
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2004���6��
�������ߣ���� �λ��� ������ ������ ��ҵ��
����ҳ�룺1180 - 1186
Key words��NiFe2O4; cermet; corrosion behavior; inert anode; aluminum electrolysis
ժ Ҫ��1
Abstract: 5Ni-xNiO-NiFe2O4 cermets with different NiO contents were prepared and the corrosion behaviour in Na3AlF6-Al2O3 melts was investigated in laboratory electrolysis tests. The results indicate that adding NiO is un-favorable to the densification of NiFe2O4-xNiO ceramics, while small Ni doping can greatly improve the sintering property. The electrolysis tests show that excess NiO is beneficial to the reduction of Fe while has little effects on that of Ni in the bath; the steady-state concentrations of Ni, Fe are below the corresponding solubilities of NiFe2O4-xNiO, implying that corrosion mechanism changes while electrifying. Post-electrolysis examination of anodes shows that Ni metal leaches at the anode surface, yet the substrate ceramic prevents the penetration of bath and the further loss of metal phase.
������Ϣ��the National Basic Research Program of China
LI Jie(�� ��), DUAN Hua-nan(���), LAI Yan-qing(������),
TIAN Zhong-liang(������), LIU Ye-xiang(��ҵ��)
(School of Metallurgical Science and Engineering,Central South University, Changsha 410083, China)
Abstract: 5Ni-xNiO-NiFe2O4 cermets with different NiO contents were prepared and the corrosion behaviour in Na3AlF6-Al2O3 melts was investigated in laboratory electrolysis tests. The results indicate that adding NiO is un-favorable to the densification of NiFe2O4-xNiO ceramics, while small Ni doping can greatly improve the sintering property. The electrolysis tests show that excess NiO is beneficial to the reduction of Fe while has little effects on that of Ni in the bath; the steady-state concentrations of Ni, Fe are below the corresponding solubilities of NiFe2O4-xNiO, implying that corrosion mechanism changes while electrifying. Post-electrolysis examination of anodes shows that Ni metal leaches at the anode surface, yet the substrate ceramic prevents the penetration of bath and the further loss of metal phase.
Key words: NiFe2O4; cermet; corrosion behavior; inert anode; aluminum electrolysis CLC number: TF111.52; TG174
Document code: A
1 INTRODUCTION
It is well known that the current aluminum reduction cell with carbon consumable anode has many disadvantages. So the concept of inert anode was introduced, with which the cell reaction will be
Al2O3��2Al+3/2O2(1)
and the disadvantages can be avoided completely. However, the inert anode must meet some basic requirements: to exhibit a low corrosion rate in the high-temperature melts and environment of high oxidizability; not to contaminate the produced metal Al; to be economically feasible; to be a good electric conductor, etc. A lot of research work has been carried out, which can be divided into three classes, namely, metals[1, 2], ceramic oxides[3], and cermets[4, 5], but no material has been found to satisfy all these requirements. One of the most formidable challenges is the contradiction between the electronic conductivity and the corrosion property. Compared with metals and oxides, which can only meet one of these two and miss the other, the cermets seem, in this sense, more promising to become the bulk material for the inert anode.
Since 1980, Aluminum Company of America (Alcoa) conducted, with the support by US Department of Energy, a considerable work about the ferrites. One of the major successes was, as summarized in the final report[6], to determine the 17Cu(Cu-Ni)-18NiO-NiFe2O4 cermets to be researched further, because of its high electronic conductivity and good corrosion resistance during electrolysis. Thereafter, an intensive and extensive work was done, concentrating on such a material. However, both the report and the following work failed to exhibit the substantial proof to explain why the excess amount of NiO in the cermet was 18%.
In 1986, Young[7] pointed out that the solubility of Fe and Ni from NiFe2O4 are inversely related to each other as follows (for stoichiometric NiFe2O4):
![]()
where x(Fe2O3), x(NiO), ��(Fe2O3) and ��(NiO) are, respectively, the mole fraction and the activity coefficient of Fe2O3 and NiO in the melt which is saturated with NiFe2O4; k is a constant determined by temperature. The solubility of Fe2O3 is far greater than that of NiO, so a NiFe2O4-based anode should be composed of NiO-rich NiFe2O4. In 1996, Olsen et al[8] studied the corrosion behavior of different NiFe2O4-based cermets, in which the amounts of excess NiO were 0%, 17%, and 23%, respectively. However the results did not differentiate their corrosion properties effectively.
In the present work, the 5Ni-xNiO-NiFe2O4 cermets with varying bulk concentrations of NiO were synthesized, and tested in the Na3AlF6-Al2O3 melts with the respect of the corrosion behavior under conventional electrolysis condition. By analyzing the bath withdrawn during electrolysis, we expected to see how the NiO content affected the corrosion property and determined which composition behaved best.
2 EXPERIMENTAL
2.1 Preparation of cermets
The raw materials, nickel powder, NiO and Fe2O3 were all reagent grade.
NiFe2O4 based cermet samples were prepared by a conventional cold pressing-sintering pro- cess[9]. A proper amount of NiO and Fe2O3(the excess contents of NiO, compared with that of the stoichiometric NiFe2O4, in the ceramic phase are 0%, 10%, 20%, 30%, 40%, respectively ) were mixed by ball milling and then calcined to form the NiFe2O4-xNiO powder. The NiFe2O4-xNiO powder was mixed with 5% Ni by ball milling in the medium containing dispersant and adhesive. Finally, the mixed powders were cold pressed into cylindrical blocks (d20mm��40mm) at the pressure of (1.0-3.0)��108Pa and sintered at 1350�� for 2h in an atmosphere of efficaciously controlled oxygen partial pressure to get the desired cermet samples.
The relative density and porosity of the samples were tested according to ASTM C914-95(1999).
2.2 Electrolysis tests
The electrolyte was made up of reagent grade cryolite(Na3AlF6) and CaF2, technical grade Al2O3 and AlF3; the CR (NaF/AlF3 molar ratio) was kept to be 2.3, the concentrations of CaF2 and Al2O3 were both kept to be 5%(mass fraction). And the temperature was maintained at 965��, i.e. kept a superheat of 10��, monitored by a Pt-Pt10%Rh thermocouple.
The sketch of the experimental cells is presented in Fig.1. About 600g electrolyte was contained in the graphite crucible, which served as the counter electrode. The crucible was placed in a vertical laboratory furnace heated to the required temperature and kept for 1h before immersing the anode and electrifying 15min later. During electrolysis Al2O3 was added frequently based on the electrolytic consumption rate at 85% cathodic current efficiency. Bath samples were taken out and analyzed to determine the level of anode constituents in the melt during the tests. The anode-bath contacting area was controlled by the anode immerse depth, which was 1cm. The current and the cell voltage were supplied and monitored with a Multi-Purpose Potentiostat/Galvanostat (model 273A/10,Perkin-Elmer Instruments). The current was kept constant throughout the experiments, which normally lasted 8h.

Fig.1 Electrochemical cell
Some of the electrolyte samples taken during electrolysis were dissolved by HClO4 solution, and analyzed by atomic absorption spectrum (WFX-120, BRAIC), with the error of 5%; others from different places of the frozen bath were analyzed by X-ray fluorescence (Philips PW2424). Some of the anodes were sectioned, mounted, polished, and analyzed by XRMA (JSM-5600LV) using a quantitative energy dispersive spectrometer (EDS) connected to the SEM; others were washed in 30% AlCl3��6H2O solution at 80�� to remove the adhering bath, the volume losses(by immersion) of the anodes were determined[10, 11].
3 RESULTS AND DISCUSSION
3.1 Effect of NiO content on cermet density
The bulk density is of great importance to desirable inert anode material. Given low density, on one hand, there exist a lot of pores, which will lead to poor electric conductivity. The poor electric conductivity will further result in great anode ohmic voltage drop, high cell voltage, and high energy consumption. On the other hand, the low bulk density is detrimental to the corrosion resistance and antioxidation property. During electrolysis, the surface metal phase was corroded either due to electrochemical dissolution, or due to oxidation by the liberated oxygen and the following preferential chemical dissolution, leaving a lot of pores. If much electrolyte penetrated these pores, the decomposition and detachment of anode will be accelerated, thus resulting in ��catastrophic�� corrosion.
From Table 1, it can be found that in the range of 0-40%, the higher the NiO content was in the ceramic phase of NiFe2O4-xNiO, the lower the relative density and the higher the porosity. Obviously, NiO was unfavorable to improve the powder��s sintering property.
To eliminate the effect caused by different densities and highlight that brought by varying NiO contents, the present work, based on the previous research[12], doped 5% metal Ni and manufactured cermet anodes with different NiO contents to do electrolysis tests.
The density data for 5Ni-xNiO-NiFe2O4 cermets are shown in Table 2, from which it is obvious that the doped metal increased dramatically the sample densities. The relative density of the 5Ni-10NiO-NiFe2O4 cermet, in particular, increased from 92.96% to 98.11%, close to its theoretical density. Furthermore, though there still existed certain differences among these samples, the relative densities were all above 92%, thereby reducing the effect caused by density to some extent.
3.2 Effect of NiO content on the corrosion resistance
Young[7] pointed out that it took approximately 8h for stoichiometric NiFe2O4 in cryolite melts to reach steady-state concentrations, which were taken to be the solubilities. The work by LAI et al[13] showed that such a process would cost 4-6h. Therefore in present work all electrolysis experiments lasted 8h. Furthermore, because the alumina concentration was under saturation, the whole graphite crucible served as cathode, and the cathode area was far larger than the anode-bath contacting area. The produced Al at cathode probably either reacted with cathode carbon to form Al4C3 and dissolved into the bath again or dissolved into it directly, and the electrolysis duration was short, so after tests were finished we did not get any metal Al. Hence, only by analyzing the bath samples taken during electrolysis could we study the anode corrosion behavior. The analysis results are summarized in Tables 3 and 4.
Table 1 Densities and porosities of NiFe2O4-xNiO ceramics
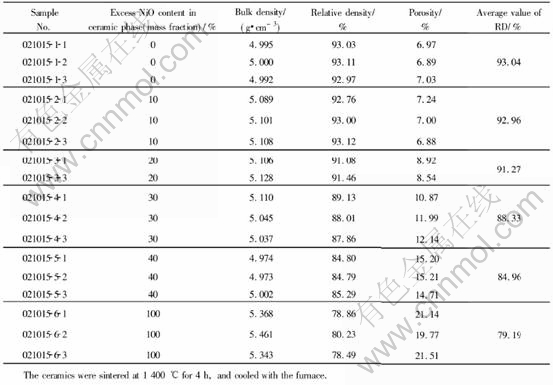
Table 2 Densities and porosities of 5Ni-xNiO-NiFe2O4 cermets
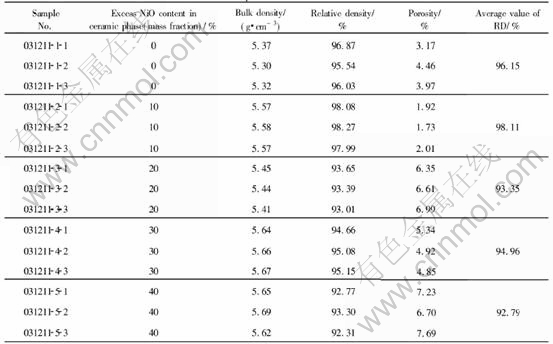
Table 3 Ni concentrations in bath during electrolysis (mass fraction, %)
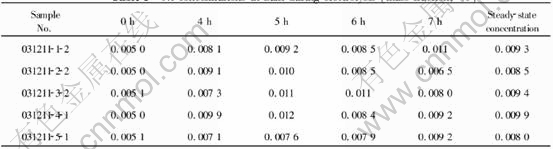
Table 4 Fe concentrations in bath during electrolysis (mass fraction, %)
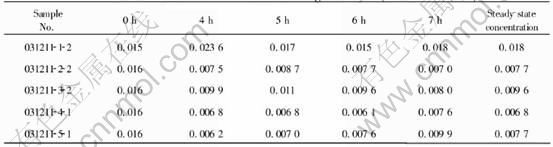
From Tables 3 and 4, it can be seen that the corrosion of cermets reaches the steady-state after about 4h, and such a steady-state value differs a lot from that of so called static corrosion, i.e. unpolarized corrosion. The solubility values of Fe and Ni from NiFe2O4 measured by Young[7] (bath ratio 1.1, melt with 6.5% Al2O3, 1000��) were 0.058% and 0.009%. The values of Fe and Ni from NiFe2O4-xNiO by LAI[13] (bath ratio 1.15, melt with 5% Al2O3, 960��) varied in 0.054%-0.072% and 0.0079%-0.0085%. While in the current work, the steady-state value for Fe was in the range of 0.0068%-0.018%, far below the above mentioned solubility; the value for Ni varied in 0.0080%-0.0099%, close to its solubility.
As shown in these two Tables, the mass ratios Fe/Ni neither ranged from 3 to 8, as Young[7] mentioned, nor were near 9, as LAI[13] reported, but were close to 1. This might be due to several reasons. First, the addition of Ni and NiO restrained the dissolution of Fe from anodes, and decreased the mass ratio. Second, the polarized corrosion mechanism of ceramic phase in the cermets, affected by chemical dissolution, electrochemical corrosion and so on, was probably different from the static corrosion mechanism (unpolarized corrosion mechanism), which was only controlled by chemical dissolution. However, the exact mechanism requires further studies.
Again from Tables 3 and 4, it could be found that the increasing excess amount of NiO in the ceramic phase reduced the content of Fe in the electrolyte from 0.018% to 0.0068%-0.0096%, while had little influence on that of Ni. And for Fe particularly, when the content of NiO varied in 10%-40%, its steady-state concentrations differed little. This is interesting because it implies that the excess addition of NiO is favorable to improving the material corrosion resistance, however, the amount of addition has a limited effect on such an improvement. Olsen and Thonstad[8], in their work about Cu-NiO-NiFe2O4 cermets containing 0%, 17%, and 23% excess NiO, found as well that the steady-state values were quite similar for all the anode compositions with respect to concentrations of Ni, Fe, and Cu.
So, considering the previous work about electric conductivity of the NiFe2O4-NiO ceramics[14], and the effect of NiO content on corrosion resistance in the current work, we safely concluded that the cermet with 10% excess NiO behaved best and was worthy of further studies.
3.3 Preliminary study of corrosion mechanism
To obtain more information about the corrosion mechanism of the cermets, anode 031211-5-1, after electrolysis experiment, was sectioned, mounted and polished. The backscattered electron picture and corresponding X-ray images for Al, Ca, Fe, and Ni were shown in Fig.2.
From Fig. 2 (a), it was obvious that the metal phase leached preferentially, on the right there was a lot of holes and pores left. The X-ray mapping images near the surface (b) and (c) show that all the aim elements were not distributed evenly; there was a clear gradient across the surface. Al and Ca scattered much more in the adhering electrolyte than in the bulk anode material and pores, which implied that though metal phase exhibited a preferential corrosion, the remained ceramic phase could effectively hold back the bath penetration and the loss of the metal phase in the underlying cermet because of high relative density. Fe and Ni were fairly rare in electrolyte, indicating the reaction between bath and anode material was limited.
As discussed by Ray[4], the anode material may be corroded via several mechanisms such as oxidation, chemical dissolution (fluorination), electrochemical dissolution, reduction by dissolved metal Al, electrolyte penetration, and grain boundary attack. Under conventional electrolysis conditions, chemical dissolution and electrolyte penetration serve as two major corrosion mechanisms.
For the former one, there are several possible reactions competing with reaction (1) during electrolysis:
2AlF3 (s)+3FeO(s)=3FeF2(s)+ 3/2O2(g)+2Al(l)(3)
2AlF3(s)+Fe2O3(s)=2FeF3(s)+ 3/2O2(g)+2Al(l)(4)
2AlF3(s)+3NiO(s)=3NiF2(s)+ 3/2O2(g)+2Al(l)(5)
In current melts (CR 2.3, at 965��), assuming the activity of AlF3 is 1.5��10-3[15], the remained are all unit activity, the decomposition voltages for reactions (3), (4) and (5) are 2.59V, 2.25V, and 2.07V, respectively. While under the same condition, the decomposition voltage of reaction (1) with unit activity of alumina at 965�� is 2.18V. During electrolysis, the cell reactions (3-5) compete with reaction (1), and will be more favored at high aluminum fluoride activity, at lower temperatures, and with low alumina level.
About the electrolyte penetration, it may result from two reasons. The first one is due to pores left by the electrochemical dissolution of metal phase when anodes are polarized, or the metal phase oxidation and the following preferential chemical dissolution, bath penetrates into these pores by capillary effects. The second one is the selective dissolution of Fe, since in the ceramic phase of the NiFe2O4-based anode, Fe has a fairly high solubility in cryolite, compared with Ni. But in present work, due to high anode bulk density, short electrolysis period, low metal content in cermets, and addition of excess NiO, the above-mentioned factors are effectively restrained or eliminated, so the bath penetration is not severe.
4 CONCLUSIONS
1) Increasing the contents of NiO in NiFe2O4-xNiO ceramic is detrimental to the sintering property, decreasing the density; but the addition of 5% Ni can obviously improve the sintering property. With the same ceramic composition, the densities of 5Ni-xNiO-NiFe2O4 cermets are 3.11%-7.83% higher than those of corresponding NiFe2O4-xNiO ceramics. The relative density of 5Ni-10NiO-NiFe2O4 cermet, particularly, reaches 98.11%, close to its theoretical density.

Fig.2 SEM backscattered pictures and corresponding X-ray mapping images for Al, Ca, Fe, and Ni from an area near anode surface of anode 031211-5-1 after electrolysis
2) The addition of NiO in 5Ni-xNiO-NiFe2O4 cermets can improve the corrosion resistance, but the adding amount affects the property to a limited extent. Considering properties including density, electric conductivity, and corrosion resistance, 5Ni-10NiO-NiFe2O4 cermet behaves best and should be further studied. During electrolysis, the steady-state concentrations of Ni and Fe in the melts are lower than their solubilities without polarization, and their ratio is not stoichiometric, which implies the corrosion mechanism must be different from that in unpolarized condition, and the exact corrosion mechanism remains to be further studied.
3) From the post-examination with SEM/EDS of the anodes, the metal Ni is found to be corroded preferentially and produce many pores, but the remained ceramic phase can effectively hold back the electrolyte penetration because of high density. How to reduce the preferential dissolution of metal should be an emphasis for future research work.
REFERENCES
[1]Windisch C F Jr, Marschman S C. Electrochemical polarisation studies on Cu and Cu-containing cermet anodes for the aluminium industry [A]. Zabreznik R D. Light Metals [C]. Warrendale, PA, USA: TMS, 1987. 351-355.
[2]Hryn J N, Sadoway D R. Cell testing of metal anodes for the aluminium electrolysis [A]. Das S K. Light Metals [C]. Warrendale, PA, USA: TMS, 1993. 475-483.
[3]YANG J, LIU Y, WANG H. The behaviour and improvement of SnO2-based inert anodes in aluminium electrolysis [A]. Das S K. Light Metals [C]. Warrendale, PA, USA: TMS, 1993. 493-495.
[4]Ray S R. Inert anodes for hall cells [A]. Miller R E. Light Metals [C]. Warrendale, PA, USA: TMS, 1986. 287-298.
[5]Tarcy G R. Corrosion and passivation of cermet inert anodes in cryolite-type electrolytes [A]. Miller R E. Light Metals [C]. Warrendale, PA, USA: TMS, 1986. 309-320.
[6]Weyand J D, De Young D H, Ray S P, et al. Inert Anodes for Aluminium Smelting [R]. PA 15069, Washington DC: Aluminum Company of America, February, 1986.
[7]De Young D H. Solubilities of oxides for inert anodes in cryolite -based melts [A]. Miller R E. Light Metals [C]. Warrendale, PA, USA: TMS, 1986. 299-307.
[8]Olsen E, Thonstad J. The behaviour of nickel ferrite cermet materials as inert anodes [A]. Hale W. Light Metals [C]. Warrendale, PA, USA: TMS, 1996. 249-257.
[9]ZHANG Gang, LAI Yan-qing, TIAN Zhong-liang, et al. Preparation of nickel ferrite based cermets for aluminum electrolysis [J]. Journal of Material Science and Engineering, 2003, 21 (4): 44-47. (in Chinese)
[10]Wang H, Thonstad J. The behaviour of inert anodes as a function of some operating parameters [A]. Campbell P G. Light Metals [C]. Warrendale, PA, USA: TMS, 1989. 283-290.
[11]Xiao Hai-ming, Hovland R, Rolseth S, et al. On the corrosion and the behavior of inert anodes in aluminum electrolysis [A]. Cutshall E R. Light Metals [C]. Warrendale, PA, USA: TMS, 1992. 389-399.
[12]TIAN Zhong-liang, LAI Yan-qing, ZHANG Gang, et al. Preparation of NiFe2O4-Cu based cermet inert anodes in aluminium electrolysis [J]. The Chinese Journal of Nonferrous Metals, 2003, 13(6): 1540-1545. (in Chinese)
[13]LAI Yan-qing, TIAN Zhong-liang, QIN Qing-wei, et al. Solubility of composite oxide ceramics in Na3AlF6-Al2O3 melts [J]. Journal of Central South University of Technology, 2003, 34 (3): 245-248. (in Chinese)
[14]QIN Qing-wei, LAI Yan-qing, ZHANG Gang, et al. Preliminary testing of NiFe2O4-NiO as ceramic matrix of cermet inert anode in aluminum electrolysis [J]. Trans Nonferrous Met Soc China, 2003, 12 (5): 1208-1212.
[15]Ray S P. Effect of Cell Operating Parameters on Performance of Inert Anodes in Hall-Heroult Cells [A]. Zabreznik R D. Light Metals [C]. Warrendale, PA, USA: TMS, 1987. 367-380.
Foundation item: Project (G1999064903) supported by the National Basic Research Program of China; Project (50204014) supported by the National Natural Science Foundation of China; Project (2001AA335013) supported by the Hi-tech Research and Development Program of China
Received date: 2004-06-07; Accepted date: 2004-09-03
Correspondence: DUAN Hua-nan; Tel: +86-731-8830474; E-mail: micahel_dhn@yahoo.com.cn

