Trans. Nonferrous Met. Soc. China 27(2017) 932-938
Electrochemical response to biomass of bacterial cells of Achromobacter sp. CH-1 growth
Yun-yan WANG1,2, Li-yuan CHAI1,2, Qing-wei WANG1,2,3, Zhi-hui YANG1,2, Rong DENG1
1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. Chinese National Engineering Research Centre for Control and Treatment of Heavy Metal Pollution, Changsha 410083, China;
3. Changsha Science Environmental Technology Co., Ltd., Changsha 410000, China
Received 30 May 2016; accepted 6 January 2017
Abstract: A simple and effective method has been developed to reflect the growth of bacteria CH-1 via the potential of the solution. The results indicate that during the bacterial cultivation, the biomass increases and the potential of the solution decreases over time. The relationship between biomass and potential of the solution could be expressed by the equation with constants of a and b which are all related to species, batch, number, and growth environment of bacterial. When the initial pH value is 10 and the initial biomass is 6.55×107 cell/mL, the correlated equation of the biomass and the potential of the solution could be divided into two segments. The growth of bacteria CH-1 is different under various experimental conditions, but the biomass is directly related to the potential of the solution regardless of the conditions of different initial pH values and bacteria number.
Key words: electrochemical response; Cr(VI); bacteria CH-1; biomass; potential of solution
1 Introduction
Chromium is one of the major trace heavy metal pollutants in the environment. Chromium in environmental waters typically comes from industrial pollution sources, including tanning factories, steel works, wood preservation and artificial fertilizers [1-4]. It is widely known that the toxicity and biological activity of the element depend on not only the total amount, but also its chemical form. Chromium species exist mainly in two different oxidation states in environmental water: Cr(VI) and Cr(III), which have contrasting physiological effects. Cr(III) is considered as an essential trace element for the maintenance of an effective glucose, lipid, and protein metabolism in mammals. On the other hand, Cr(VI) can be toxic for biological systems and cancerogenic in human beings.
The conventional methods for removal of Cr(VI) from wastewater include chemical precipitation, adsorption, and ion exchange, and so on [5,6]. In our previous works [7-22], the novel technologies for detoxification of chromium-containing slag and for removal of chromium-containing wastewater by microorganism Achromobacter sp. CH-1 were put forward, and then the correlated researches have been done.
During the process of bacterial cultivation, it is of great importance to control the growth of bacterial no matter what the relationship between its growth and product composition is. Therefore, the convenient, effective and quick means for the biomass measurement is fundamental to promptly determine the changing tendency of the bacterial growth and to adjust the cell metabolizing in the process of cell cultivation.
Nowadays, the conventional methods used in laboratory to measure the total amounts of bacteria are microscope direct notation, flat colony notation, photoelectric turbidimetry, smear straining method, minim calorimetry, photoluminescence method, electronic autocounter, and centrifugal volumetric method [23]. Moreover, there are other methods to characterize the bacteria amount by using some substances. For instance, the biomass for some special microorganisms could be estimated according to their specific lipid component contained in the cells. Within the scope of the environmental and ecological investigations, phosphatide is considered as the sign of the microorganism biomass in the sea and the bayou sediment. Electronic impedance methods based on the electrochemistry technology can be used to confirm the growth and breed situation of some bacteria in specific culture medium via testing the electronic impedance continuously. However, in spite of many approaches to measure the growth of cells, there are still many problems in the testing process because of the complexity of the physiological and biochemical characteristics of the cells and levity of the environment. Furthermore, biomass testing methods are still not practically used because their quality and precision are very poor, the in situ testing methods during the manufacture process are not mature yet and the relationship between the cell growth and the other parameters of the physiological and biochemical characteristics and so on is not clear.
Cr(VI)-reducing bacteria are widespread and Cr(VI) reduction occurs under both aerobic and anaerobic conditions [24,25]. During the numerous environmental parameters, the redox potential of the solution plays a very important role in the Cr(VI) reduction process by bacteria, and different bacteria can only reduce the Cr(VI) at different redox potentials in the aqueous solution [26]. During the industrial process, the parameters that can be obtained on-site are scanty and many physiological and biochemical characteristics parameters are still required to be tested. If the correlated relationship between the cell growth and some easily-testing parameters can be found, it is very useful to optimize the operation conditions, to magnify and to design the biochemical process via the changing tendency of the cell concentration reflected from some easily on-site testing parameters.
In this study, in order to characterize the growth situation of bacteria CH-1 via detecting the potential of the solution simply, the relationship between the subtotals of bacteria CH-1 (N) and potential (E) of the solution was established through testing N and E at different time.
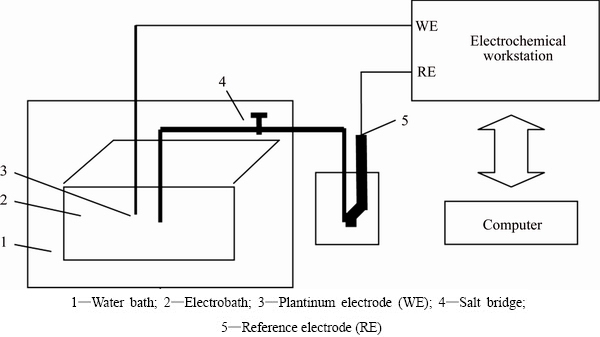
Fig. 1 Set-up of potential measurement system
2 Experimental
2.1 Bacterial strains
The strain studied in this work was Achromobacter sp. by gene sequencing of 16S rRNA and nominated as CH-1 separated from nature, which could reduce soluble and toxic chromate with high concentration to the insoluble and less toxic Cr(III) presented as Cr(OH)3 precipitate in aerobic cultures, no Cr(VI) decreasing was observed in anaerobic cultures. In contrast to other chromium(VI) reducing microorganisms, CH-1 showed higher chromium(VI) reduction rate at high pH value. Reduction performed under alkaline conditions at pH value of 7 to 11, and optimum pH value was 10. The cultivation method was described in Refs. [8-11].
2.2 Medium and hexavalent chromium ions
Medium is composed of 4 g/L carbon source, 4 g/L nitrogen source, and 2 g/L NaCl. Cr(VI) solution with different concentrations of Cr(VI) using K2Cr2O7 dissolved in water.
2.3 Analysis methods
Achromobacter sp. CH-1 was cultivated in a horizontal shaker with a speed of 120 r/min at 30 °C. Then, the cell number and solution potentials were determined after being cultivated for 2, 4, 6, 8, 10, and 12 h, respectively.
Cell number was monitored by microscope. The pH value was measured with LP115 pH meter. The set-up of potential measurement system is described in Fig. 1, which consisted of a polymethyl methacrylate electrobath with the sizes of 100 cm × 50 cm × 50 cm, a saturated calomel electrode (SCE) as a reference electrode, a platinum electrode as a working electrode, a water bath, an electrochemical workstation and a computer system. SCE and platinum electrode were connected with the electrochemical workstation (CHI660A) monitored by the computer. The solution potential was represented by the open circuit potential between SCE and the platinum electrode. All potentials in this work were expressed versus the potential of SCE. All experiments had three replicates.
3 Results and discussion
3.1 Relationship between ln N and E
The results of our previous research indicated that the optimal ability of reducing Cr(VI) in alkaline media by bacteria CH-1 could be obtained under the condition of initial pH 10, temperature 30 °C and the initial bacteria inoculation amount 20% (volume fraction). The bacteria CH-1 was cultivated under the optimal condition in this study. During the experimental process, the cell number (N) of the samples and the potential (E) of the solution were determined at regular intervals. The experimental results are listed in Table 1.
Table 1 Bacteria biomass and changes of solution potential
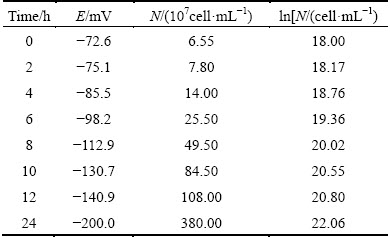
As shown in Table 1, during the process of cell cultivation, the cell concentration increases and potential of the solution decreases over time. The relationship between the potential of the solution and logarithm of cells number (ln N) could be fitted in three ways, as shown in Fig. 2, Fig. 3 and Fig. 4, respectively. And the fitted equations are as follows:
-E=-467.56948+29.52356ln N (R=0.97487) (1)
-E=1645.19916-183.75817ln N+5.35867(ln N)2 (R=0.99901) (2)
-E=-3751.41836+628.07733ln N-35.23523(ln N)2+0.6747(ln N)3 (R=0.99975) (3)
Obviously, according to the fitted line and the correlated coefficients, the liner correlation of the simple fitted equation is not satisfactory (Fig. 2), the quadratic fitted equation is good enough (Fig. 3), and the cubic fitted equation is optimal (Fig. 4). However, the cubic fitted equation is too complex to explain the physical significance of the constant and the coefficients.
Combining the characteristics of microorganism growth process with the physical significance of slope and intercept of the fitted line, the following fitted way (Fig. 5) is more preferable.
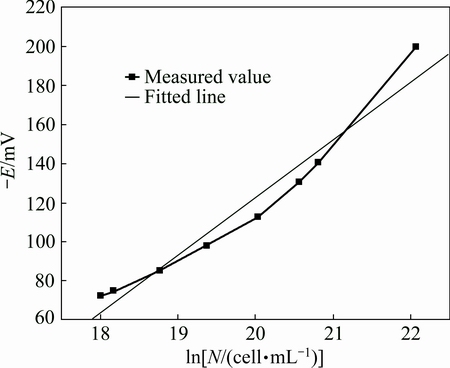
Fig. 2 Simple fitted curve of ln N-E

Fig. 3 Quadratic fitted curve of ln N-E
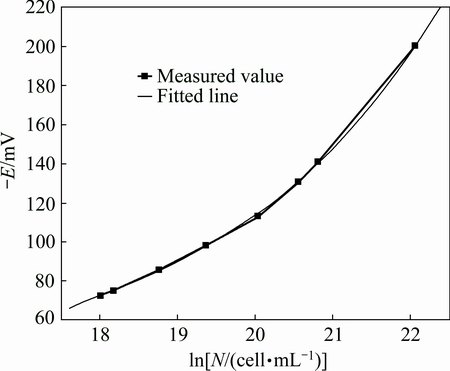
Fig. 4 Cubic fitted curve of ln N-E

Fig. 5 Relationship between ln N and E
It can be seen from Fig. 5 that with the growth of bacteria, the potential of the solution decreases slowly at the beginning and then speeds up later, which is similar to the growth curve of bacteria. The bacteria grow slowly in the lag phase while the potential of the solution decreases. However, in the logarithm phase, the bacteria grow rapidly and correspondingly the potential of the solution decreases rapidly. It can be analyzed from the two segments of Fig. 5 that the potential of the solution and the logarithm of cells are linear correlativity. The linear equation of former segment can be expressed as follows:
-E =-267.04+18.84×ln N (R=0.9983) (4)
The linear equation of latter segment is
-E=-761.03+43.49×ln N (R=0.9972) (5)
3.2 Validation of relationship between ln N and E
The bacteria CH-1 cultivation experiment under the optimal conditions of pH 10 and bacteria inoculation amount 20% was repeated to validate the relationship between the potential of the solution and the logarithm of cells. The results are shown in Table 2 and Fig. 6.
Table 2 Bacteria growth and changes of solution potential at optimized condition

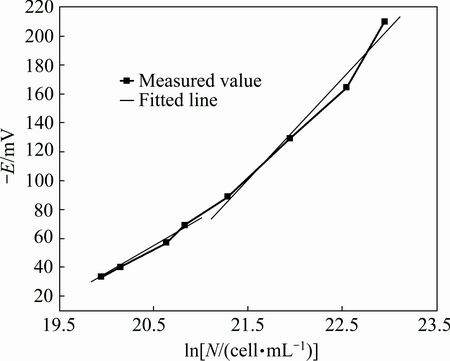
Fig. 6 Validation of relationship between ln N and E at optimized condition
It can be concluded from the two segments of Fig. 6 that the potential of the solution and the logarithm of cells are also linear correlativity. The linear equation of former segment is
-E =-742.76+38.89×ln N (R=0.9940) (6)
The linear equation of latter segment is
-E=-1404.26+70.00×ln N (R=0.9898) (7)
Obviously, Eqs. (4)-(7) are of great difference under the same experimental conditions. The reason is that the activity of the bacteria is different in different batches. However, the linear correlativity between the potential of the solution and the biomass is similar, which can be described as
-E=a+bln N (8)
where a is the electric charge quantity of unit cell concentration, b is the effect degree of cell concentration on electric charge quantity.
It can be seen from Eq. (8) that with the value of a becoming more negative, the electric charge quantity of unit cell concentration increases and the activity and reduction ability of cell becomes stronger; with the value of b increasing, the effect degree of cell concentration on electric charge quantity increases and the activity of cell becomes better. Both a and b are related to the species of bacteria, batch, amount and the growth environment of bacteria.
Although every experimental conditions and bacteria growth situation are different, the relationship between the potential of the solution and the biomass is similar. Therefore, we can gain the bacteria growth situation in site in the industrial process by measuring the potential of the solution and the biomass in every batch experiment and then calculating the constants a and b in the correlation equation.
3.3 Effect of initial bacteria inoculation amounts on relationship between ln N and E
Cultivating the bacteria CH-1 under the optimal conditions, the experiments were conducted under the conditions of same bacteria batch and initial pH value but different initial bacteria inoculation amounts.
Figures 7 and 8 show the relationship between the potential of the solution and the logarithm of cells with the initial bacteria inoculation amounts of 2.03×108 and 6.6×108 cell/mL. It can be deduced that the linear equations of the latter segments can be respectively expressed as follows:
-E=-1505.51+78.67×ln N (R=0.9932) (9)
-E =-1787.08+89.76×ln N (R=0.9928) (10)
Equations (9) and (10) illustrate that the initial bacteria inoculation amount has a little effect on the relationship between the potential of the solution and the logarithm of cells under the condition of same bacteria batch, pH value and other experimental conditions.

Fig. 7 Relationship between ln N and E with initial biomass of 2.03×108 cell/mL

Fig. 8 Relationship between ln N and E with initial biomass of 6.6×108 cell/mL
3.4 Effect of initial pH values on relationship between ln N and E
The effects of the initial pH value on the relationship between the potential of the solution and the logarithm of cells are shown in Figs. 9 and 10.

Fig. 9 Relationship between ln N and E at pH of 9

Fig. 10 Relationship between ln N and E at pH of 11
Similarly, the linear equations of the latter segments are
-E=-1511.81+77.72×ln N (R=0.9974) (11)
-E=-1690.28+89.04×ln N (R=0.9988) (12)
Equations (11) and (12) show that the initial pH value has a little effect on the relationship between the potential of the solution and the logarithm of cells under the conditions of same bacteria batch, initial bacteria inoculation amount and other experimental conditions. The values of a and b in Eqs. (11) and (12) reveal that the bacteria activity is better when the initial pH value is 11 than that of 9.
4 Conclusions
1) During the process of cell cultivation, the cell concentration increases and the potential of the solution decreases with time course. There is a linear correlativity between the potential of the solution and the logarithm of cells when cultivating the bacteria CH-1 at the initial pH of 10 and bacteria inoculation amount of 6.55×107 cell/mL. The linear equation of the former segment is -E=-267.04+18.84×ln N with R of 0.9983, the latter segment is -E=-761.03+43.49×ln N with R of 0.9972.
2) The linear correlativity of the potential of the solution and the biomass is -E=a+bln N. Both a and b are related to the species of bacteria, batch, amount and the growth environment of bacteria, which are the key science and technology question because of their profound physical meaning.
3) Initial bacteria amount and initial pH value have a little effect on the relationship between the potential of the solution and the logarithm of cells. The bacteria activity is better when the initial pH value is 11 than that of 9.
4) Determination of the potential of the solution could quickly and simply response bacteria activity and growth of the system, and then the optimal controlling and adjusting means could be adopted to the detoxification process.
References
[1] YANG Y, LIU H, XIANG X H, et al. Outline of occupational chromium poisoning in China [J]. Bulletin of Environmental Contamination & Toxicology, 2013, 90(6): 742-749.
[2] DAI J, REN F L, TAO C Y. Adsorption of Cr(VI) and speciation of Cr(VI) and Cr(III) in aqueous solutions using chemically modified chitosan [J]. International Journal of Environmental Research & Public Health, 2012, 9(5): 1757-1770.
[3] HE Zhi-guo, LI Shu-zhen, WANG Li-sha, ZHONG Hui. Characterization of five chromium-removing bacteria isolated from chromium-contaminated soil [J]. Water Air & Soil Pollution, 2014, 225(3): 1-10.
[4] LI Ming-ming, ZHU Jian-yu, GAN Min, WANG Qian-fen, JIE Shi-qi, CHAI Li-yuan. Characteristics of chromium coprecipitation mediated by acidithiobacillus ferrooxidans DC [J]. Water Air Soil Pollution, 2014, 225: 2071.
[5] CHEN R, CHAI L, LI Q, SHI Y, WANG Y, et al. Preparation and characterization of magnetic Fe3O4/CNT nanoparticles by RPO method to enhance the efficient removal of Cr(VI) [J]. Environmental Science & Pollution Research International, 2013, 20(10): 7175-7185.
[6] XU Wen-bin, LI Xiao-bin, ZHOU Qiu-sheng, PENG Zhi-hong, LIU Gui-hua, QI Tian-gui. Remediation of chromite ore processing residue by hydrothermal process with starch [J]. Process Safety and Environmental Protection, 2011, 89: 179-185.
[7] CHAI Li-yuan, HE De-wen, YU Xia, LIU Hui, MIN Xiao-bo, CHEN Wei-liang. Technological progress on detoxification and comprehensive utilization of chromium-containing slag [J]. Transactions of Nonferrous Metals Society of China, 2002, 12(3): 514-518.
[8] CHAI Li-yuan, YANG Zhi-hui, WANG Yun-yan, DENG Rong, ZHU Wen-jie, HUANG Shun-hong, Potential-pH diagram for “Leucobacter sp Ch-1-Cr-H2O” system [J]. Journal of Hazardous Materials, 2008, 157(2-3): 518-524.
[9] MA Ze-min, ZHU Wen-jie, LONG Huai-zhong, CHAI Li-yuan, WANG Qing-wei. Chromate reduction by resting cells of Achromobacter sp. Ch-1 under aerobic conditions [J]. Process Biochemistry, 2007 (42): 1028-1032.
[10] ZHU Wen-jie, CHAI Li-yuan, MA Ze-min, WANG Yun-yan, XIAO Hai-juan, ZHAO Kun. Anaerobic reduction of hexavalent chromium by bacterial cells of Achromobacter sp. Ch1 [J]. Microbiological Research, 2008, 163(6): 616-623.
[11] ZHU Wen-jie, YANG Zhi-hui, MA Ze-min, CHAI Li-yuan. Reduction of high concentrations of chromate by Achromobacter sp. CH-1 isolated from Changsha, China [J]. World Journal of Microbiology and Biotechnology, 2008, 24(7): 991-996.
[12] ZHU Wen-jie, CHEN Ling-li, YANG Zhi-hui, CHAI Li-yuan. Automatic detection of Staphylococcus aureus and Shigella dysenteriae with separated electrodes series piezoelectric sensing technique [J]. World Journal of Microbiology and Biotechnology, 2008, 24(7): 1073-1079.
[13] WANG Yun-yan, YANG Zhi-hui, CHAI Li-yuan, ZHAO Kun. Diffusion of hexavalent chromium in chromium-containing slag as affected by microbial detoxification [J]. Journal of Hazardous Materials, 2009, 169(1-3): 1173-1178.
[14] YANG Jing, CHAI Li-yuan, WANG Yun-yan, HE Xu-wen. Transportation and distribution of chromium in the anaerobic sludge treating the chromium-containing wastewater [J]. International Journal of Environment and Pollution, 2009, 38(3): 256-266.
[15] CHAI Li-yuan, WANG Yun-yan, YANG Zhi-hui, WANG Qing-wei, WANG Hai-ying, Detoxification of chromium-containing slag by Achromobacter sp. CH-1 and selective recovery of chromium [J]. Transactions of Nonferrous Metals Society of China, 2010, 20(8): 1500-1504.
[16] HUANG Shun-hong, PENG Bing, YANG Zhi-hui, CHAI Li-yuan, ZHOU Li-cheng, Chromium accumulation, microorganism population and enzyme activities in soils around chromium- containing slag heap of steel alloy factory [J]. Transactions of Nonferrous Metals Society of China, 2009, 19(1): 241-248.
[17] HUANG Shun-hong, PENG Bing, YANG Zhi-hui, CHAI Li-yuan, XU You-ze, SU Chang-qing, Spatial distribution of chromium in soils contaminated by chromium-containing slag [J]. Transactions of Nonferrous Metals Society of China, 2009, 19(3): 756-764.
[18] WANG Yang-yang, YANG Zhi-hui, PENG Bing, CHAI Li-yuan, WU Bao-lin, WU Rui-ping. Biotreatment of chromite ore processing residue by Pannonibacter phragmitetus BB [J]. Environ Sci Pollut Res, 2013, 20: 5593-5602.
[19] CHAI L Y, HUANG S H, YANG Z H, PENG B, HUANG Y, CHEN Y H. Hexavalent chromium reduction by Pannonibacter phragmitetus BB isolated from soils under chromium-containing slag heap [J]. Journal of Environmental Science and Health, Part A, 2009, 44 (6): 615-622.
[20] LIAO Ying-ping, MIN Xiao-bo, YANG Zhi-hui, CHAI Li-yuan, ZHANG Shu-juan, WANG Yang-yang. Physicochemical and biological quality of soil in hexavalent chromium-contaminated soils as affected by chemical and microbial remediation [J]. Environ Sci Pollut Res, 2014, 21: 379-388.
[21] WANG Y, PENG B, YANG Z, TANG C, CHEN Y, et al. Treatment of Cr(VI) contaminated water with Pannonibacter phragmitetus BB [J]. Environmental Earth Sciences, 2014, 71(10): 4333-4339.
[22] WANG Yang-yang, CHAI Li-yuan, LIAO Qi, TANG Chong-jian, LIAO Ying-ping, PENG Bing, YANG Zhi-hui. Structural and genetic diversity of hexavalent chromium-resistant bacteria in contaminated soil [J]. Geomicrobiology Journal, 2016, 33(3-4): 222-229.
[23] HAO Dong-xia, LIU Ben-fa, WU Zhao-liang. Research situation and development on testing method of cell growth [J]. Microbiology Aviso, 2001, 28(6): 82-85. (in Chinese)
[24] GVOZDYAK P L, MOGILAVICH N F, RYLSKII A F, GRISHCHENKO N I. Reduction of hexavalent chromium by collection strains of bacteria [J]. Mikrobiologiyal, 1986, 55(6): 962-965.
[25] DELEO P C,EHRLICH H L. Reduction of hexavalent chromium by Pseudomonas fluorescensLB300 in batch and continuous cultures [J]. Applied Microbiology and Biotechnology, 1994,40(5): 756-759.
[26] LLOVERA S,BONET R,SIMON-PUJOL MD,CONGREGADO F. Francisco Congregado. Chromate reduction by resting cells of Argobacterium radiobacter EPS-916 [J]. Applied Environmental Microbiology, 1993, 59(10): 3516-3518.
Cr(VI)还原菌Achromobacter sp. CH-1生长量的电化学响应
王云燕1,2,柴立元1,2,王庆伟1,2,3,杨志辉1,2,邓 荣1
1. 中南大学 冶金与环境学院,长沙 410083;
2. 中南大学 国家重金属污染防治工程技术研究中心,长沙 410083;
3. 长沙赛恩斯环保科技有限公司,长沙 410000
摘 要:溶液电势可简单有效地表征碱性介质中Cr(VI) 还原菌Achromobacter sp. CH-1的生长情况。Achromobacter sp. CH-1培养过程中,溶液电势随着细菌数的增加而降低,溶液电势与细菌数对数呈线性相关,线性方程中常数a、b与细菌的种类、批量、数量和细菌的生长环境有关。初始pH为10、初始细菌数为6.55×107 cell/mL时培养CH-1菌,溶液电势与细菌数的对数值的相关曲线可分两段表征。不同初始pH与不同细菌数条件下CH-1菌的生长情况不同,但细菌的生长量与溶液电势直接相关。
关键词:电化学响应;Cr(VI);还原菌Achromobacter sp. CH-1;生物量;溶液电势
(Edited by Xiang-qun LI)
Foundation item: Projects (51304251, 51504299) supported by the National Natural Science Foundation of China; Project (201509050) supported by Special Program on Environmental Protection for Public Welfare, China
Corresponding author: Qing-wei WANG; Tel: +86-731-88830577; Fax: +86-731-88710171; E-mail: qw_wang@csu.edu.cn
DOI: 10.1016/S1003-6326(17)60109-2