
Boron removal from metallurgical grade silicon by oxidizing refining
WU Ji-jun(伍继君)1, MA Wen-hui(马文会)1, YANG Bin(杨 斌)1, DAI Yong-nian(戴永年)1, K. MORITA2
1. National Engineering Laboratory for Vacuum Metallurgy, Faculty of Materials and Metallurgical Engineering, Kunming University of Science and Technology, Kunming 650093, China;
2. Institute of Industrial Science, The University of Tokyo, Bunkyo Tokyo 113-8656, Japan
Received 14 April 2008; accepted 29 September 2008
Abstract: A purification process was developed to remove impurity element boron from the metallurgical grade silicon by the electric arc furnace refining. The thermodynamic equilibria calculation and experiment to remove boron in the oxidizing atmosphere were performed and analyzed. Boron is removed as the gaseous species BxOy and BxHzOy in O2 and H2O-O2 atmosphere respectively. The equilibrium pressure of BxHzOy is 105-1010 times that of BxOy. Boron is removed and its content in silicon is reduced from 18×10-6 to 2×10-6 in the Ar-H2O-O2 atmosphere in the electric arc furnace.
Key words: thermodynamics; metallurgical grade silicon; purification; oxidizing refining; electric arc furnace
1 Introduction
Since 2000, the booming of photovoltaic (PV) industry has being lasted for about 8 years. The worldwide production of photovoltaic modules was 1 787 MW in 2005 and it is projected to approach 18 GW annually by 2020. In 2005, more than 90% of the solar cells were made using crystalline silicon. The growth of the PV industry is limited by the availability of silicon feedstock, and is directly dependent on the electronic industry processes. In other words, the photovoltaic industry will face a shortage of silicon feedstock in the near future[1-3]. At present, the supply of solar grade silicon is short of demand and the new technique of production and purification with large scale and low price must be explored. The purification of metallurgical grade silicon is being paid close attention by investigators.
Impurity elements Fe, Al, Ca, Ti, P, B, etc, in silicon can be removed by the metallurgical method. Among them, the metallic elements with very small segregation coefficient can be separated from silicon by the directional solidification[4]. B and P contents in silicon materials will affect the conversion efficiency of solar grade cells. Phosphorus can be volatilized from silicon in form of high volatile P2[5]. But the boron removal is still a challenge for its large segregation coefficient to silicon (k0=0.8) and low vapor pressure (pB=10-6 Pa at 1 773 K)[6-7]. The common practice of boron removal is by pyrometallurgical refining step with the calcium silicate slag of CaO-SiO2, CaO-Al2O3-SiO2 or NaO0.5-CaO-SiO2 to obtain solar grade silicon[8-11]. Plasma refining is restricted for the apparatus despite of its availability in boron removal[12-13]. Even though, boron can be also vaporized from silicon in form of gaseous oxides or hydrates[14].
In this work, the oxidizing refining method of metallurgical grade silicon was used for the boron removal and the process and feasibility were presented. Based upon the thermodynamic equilibria analysis of molten silicon system in O2 and H2O-O2 atmosphere, it is demonstrated that the boron content in metallurgical grade silicon can be effectively reduced. And the experimental setup and results were presented.
2 Thermodynamic equilibrium analysis
The thermodynamic equilibrium analysis aims at the form and limit of boron removal by calculating the Gibbs free energy and vapor pressure of gaseous species at different temperature, and determining system pressure and gas composition, therefore, to provide a guideline or reference for oxidizing refining application of metallurgical grade silicon. The melting and boiling points of Si and B are 1 685, 2 540 and 2 573, 2 820 K, respectively. Silicon is in molten state at the determinate temperature range of 1 685-2 500 K. Boron oxides BO, B2O, BO2, B2O2, B2O3 and hydrates BHO, BH2, BHO2, BH2O2, BH3O3, B2H4O4, B3H3O3, B3H3O6 are volatile according to the thermodynamic data. So, the impurity element boron in metallurgical grade silicon can be changed into gaseous species through oxidization in O2 and H2O-O2 atmosphere and then be removed. The effects of other impurities elements such as Fe, Al, Ca, Ti, P, C, Cu, V, etc, on Si and B are ignored here.
2.1 Oxidizing refining of O2 atmosphere
The impurity element boron dissolved in molten silicon and expressed as [B] is oxidized into gaseous boron oxide species (BxOy) by O2 in the temperature range of 1 685-6 000 K. The potential reactions in [B]-O2 and Si-O2 are listed in Table 1.
Table 1 Reactions for Si-B-O system

Based on the handbook of NIST-JANAF Thermochemical Tables[15], the relationship between the Gibbs free energy change ( ) of chemical oxides (BxOy, SiO2, SiO) and temperature (T/K) was calculated and shown in Fig.1, where the preferential order of oxidizing products is B2O3>B2O2>BO>BO2>B2O. The
) of chemical oxides (BxOy, SiO2, SiO) and temperature (T/K) was calculated and shown in Fig.1, where the preferential order of oxidizing products is B2O3>B2O2>BO>BO2>B2O. The  values of SiO2 and SiO are much negative in the temperature range of 1 685-2 100 K and 2 100- 2 500 K, respectively. The silicon is the principal part in the molten system and the reactions (6) and (7) firstly occur at 1 685-2 100 K and 2 100-2 500 K, respectively. According to Fig.1, we conclude that the impurity element B in metallurgical grade silicon may not be oxidized into gaseous species at 1 600-2 500 K and at this temperature region only Si oxidation takes place to form SiO2 and SiO.
values of SiO2 and SiO are much negative in the temperature range of 1 685-2 100 K and 2 100- 2 500 K, respectively. The silicon is the principal part in the molten system and the reactions (6) and (7) firstly occur at 1 685-2 100 K and 2 100-2 500 K, respectively. According to Fig.1, we conclude that the impurity element B in metallurgical grade silicon may not be oxidized into gaseous species at 1 600-2 500 K and at this temperature region only Si oxidation takes place to form SiO2 and SiO.
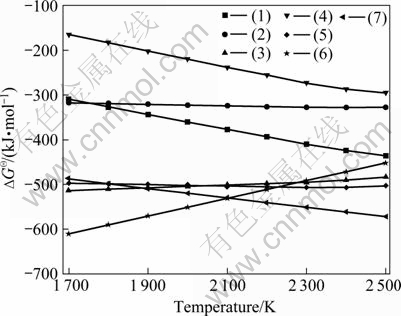
Fig.1 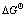 of oxides vs temperature for Si-B-O system (reacting gas: O2)
of oxides vs temperature for Si-B-O system (reacting gas: O2)
The  ―T curve of gaseous boron oxides (BxOy) is declined downwards and opposite to that of SiO2. The reactions (8)-(12), given in Table 2, between SiO2 and [B] will occur at the determinate temperature and pressure.
―T curve of gaseous boron oxides (BxOy) is declined downwards and opposite to that of SiO2. The reactions (8)-(12), given in Table 2, between SiO2 and [B] will occur at the determinate temperature and pressure.
Table 2 Reactions between SiO2 and [B]

The relationship between and T of the reactions (8)-(12) is shown in Fig.2.
and T of the reactions (8)-(12) is shown in Fig.2.

Fig.2 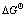 of reactions between SiO2 and [B] vs temperature
of reactions between SiO2 and [B] vs temperature
From Fig.2, the  values of reactions (8)-(12) are all positive below 2 300 K. It is possible to reach the negative
values of reactions (8)-(12) are all positive below 2 300 K. It is possible to reach the negative  values by changing system pressure. The impurity boron is then oxidized into gaseous oxides (BxOy) by the production of SiO2 and the reactions (8-12) are generally expressed as
values by changing system pressure. The impurity boron is then oxidized into gaseous oxides (BxOy) by the production of SiO2 and the reactions (8-12) are generally expressed as
(SiO2)+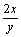 [B]=Si(l)+
[B]=Si(l)+ (BxOy)(g) (13)
(BxOy)(g) (13)
At the refining temperature of 1 685-2 500 K, the isothermal equation of reaction (13) is written as follows:
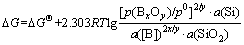 (14)
(14)
In the B-Si-O system, the activities of Si and SiO2 are regarded to be 1 (a(Si)=1, a(SiO2)=1). The silicon melt with low impurities concentration is considered to be a dilute solution and the element B is assumed as solute to obey Henry’s law. When the mass concentration w([B]) is expressed during the calculation, the standard state of w([B])/w0=1 for Henry’s law is selected. The activity a([B]) is expressed as (w([B])/w0)・f([B]) (coefficient of activity, f([B])), and a([B])=w([B])/w0 at which (w([B])/w0)→0, f([B])=1. Eq.(14) is then re- written as
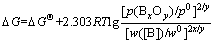 (15)
(15)
Supposing that the equilibrium is reached at which a([B])=w([B])/w0=2.0×10-4, when DG reaches zero (DG=0), Eq.(15) is expressed as
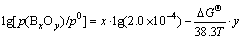 (16)
(16)
The relationship of equilibria partial pressure (BxOy) vs temperature is obtained and shown in Fig.3 according to Eq.(16). It is shown that the volatilities of gaseous boron oxides increase sharply at higher temperature and this is helpful for boron removal. Among all gaseous boron oxides, the volatility of BO is the highest and its equilibrium partial pressure is about 10-3-10 Pa at 1 685-2 500 K. The order of volatility is BO>B2O2>B2O3>B2O>BO2 (about 10-9-10-5 Pa). In a word, the boron in metallurgical grade silicon can be volatilized and removed by the oxidizing refining of O2 atmosphere to form mainly BO, plus minor B2O2, B2O3, BO2 and maybe B2O in the temperature range of 1 685-2 500 K.
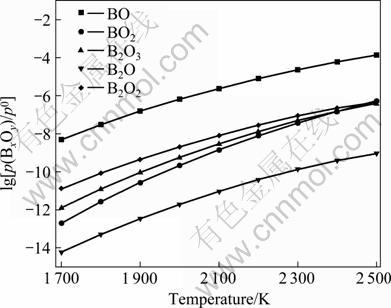
Fig.3 Partial pressure of BxOy vs temperature
2.2 Oxidizing refining with H2O-O2 mixture atmos- phere
The gaseous boron hydrates BHO, BHO2, BH2O2, BH3O3, B2H4O4, B3H3O3, B3H3O6 can be made according to the handbook of NIST-JANAF Thermochemical Tables[15]. The reactions (17-23) among B, H2O and O2 system are forecast and shown in Table 3.
Table 3 Reactions among B-H2O-O2 system

The relationship between  and temperature of the reactions is shown in Fig.4. As can be seen, except for BHO, the
and temperature of the reactions is shown in Fig.4. As can be seen, except for BHO, the  values of reactions are all negative and declined downwards in the temperature range of 1 685-2 000 K. It is concluded the boron gaseous hydrates (BxHzOy) can be made by B and H2O-O2 mixture gas, and the preferential order for the production is B3H3O3>BHO>BHO2>B3H3O6>BH3O3>B2H4O4>BH2O2 at lower temperature. The most easily formed productions for boron gaseous hydrates are B3H3O3 and BHO, respectively, in lower and higher temperature range.
values of reactions are all negative and declined downwards in the temperature range of 1 685-2 000 K. It is concluded the boron gaseous hydrates (BxHzOy) can be made by B and H2O-O2 mixture gas, and the preferential order for the production is B3H3O3>BHO>BHO2>B3H3O6>BH3O3>B2H4O4>BH2O2 at lower temperature. The most easily formed productions for boron gaseous hydrates are B3H3O3 and BHO, respectively, in lower and higher temperature range.
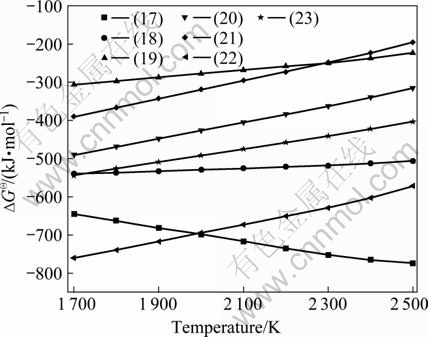
Fig.4  of chemical hydrates vs temperature (Reacting gas: H2O-O2 mixture)
of chemical hydrates vs temperature (Reacting gas: H2O-O2 mixture)
The reactions (17-23) are generally written as
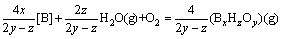
(24)
And the isothermal equation of reaction (24) is expressed as Eq.(25):
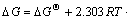

(25)
If the boron concentration (w([B])/w0) is still 2.0×10-6 (similarly to Eq.(14), a([B])=2.0×10-4) and the equilibria of reactions are reached. According to the reactions (17-23), the ratio ((2z/(2y-z))max=2 is chosen and it is supposed that the equilibrium partial pressures of H2O and O2 are kept constant in the system, at which p(H2O)/p0=10-2, p(O2)/p0=0.5×10-2, when DG reaches zero (DG=0), Eq.(25) is written as

 (26)
(26)
The logarithmic values of p(H2O)/p0 for the reactions (17-23) at the different refining temperatures are calculated according to Eq.(26) and the relationship between partial pressure of BxHzOy and temperature is shown in Fig.5. As can be seen, the partial pressure of gaseous boron hydrates (BxHzOy), except for BHO, is reduced at higher temperature and it is especially obvious for B3H3O6, which is opposite to the case of boron gaseous oxides (BxOy). So, in order to remove impurity boron much effectively, the refining temperature should be as low as possible but higher than the melting point of silicon when the H2O-O2 mixture gas is used. The partial pressure of gaseous boron hydrates is 105-1010 times that of gaseous boron oxides. According to the position of curves, the impurity element boron is removed mainly in form of the volatilization of B3H3O6 and BHO2 mainly, BH3O3, BHO, BH2O2, B2H4O4 subsidiarily and B3H3O3 hardly.
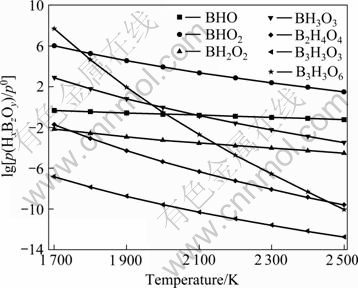
Fig.5 Partial pressure of BxHzOy vs temperature
boron is removed mainly in form of the volatilization of B3H3O6 and BHO2 mainly, BH3O3, BHO, BH2O2, B2H4O4 subsidiarily and B3H3O3 hardly.
From the thermodynamic equilibrium analysis, the impurity element boron in metallurgical grade silicon can be effectively removed respectively in form of gaseous boron oxides (BxOy) and gaseous boron hydrates (BHxOy) in O2 and H2O-O2 atmosphere, but the removing rate with the H2O-O2 mixture is absolutely higher than that of O2. Summarily, as for the oxidizing refining of metallurgical grade silicon, it is more advisable to use the H2O-O2 mixture gases at lower temperature.
3 Experiment of oxidizing refining in electric arc furnace
The metallurgical grade silicon (99.5% Si, 18×10-6 B, others for Fe, Al, Ca, Ti, Cu, V, C, O, etc.) was refined in the electric arc furnace in the Ar-H2O-O2 atmosphere (limited vacuum degree 10-4 Pa), as shown in Fig.6. The effect of refining time on boron removal was investigated and the boron content in silicon was measured by the inductively coupled plasma mass spectrometry (ICP-MS, Elan-5000A, USA).
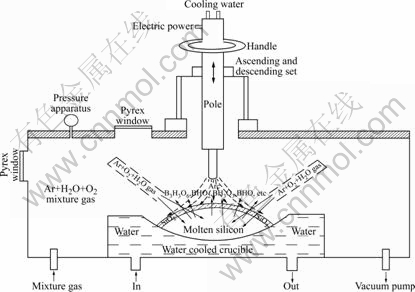
Fig.6 Schematic representation of electric arc furnace
4 Results and discussion
The metallurgical grade silicon sample was melted and refined in the electric arc furnace and the experimental results with the different refining time are given in Fig.7 (systematic total pressure, 10-3 Pa). It can be seen that the boron content in silicon sample reduces greatly and the removing rate increases sharply while prolonging the refining time. It is reduced from 18×10-6 to 2×10-6 when the refining time reaches 10 min but it is not valid for longer time.
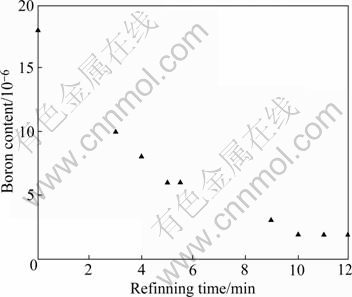
Fig.7 Effect of refining time on boron removal
5 Conclusions
1) The thermodynamic equilibrium calculation for the oxidizing refining of metallurgical grade silicon is performed to determine the results of boron removal in the temperature range of 1 685-2 500 K. In the O2 atmosphere, the impurity element boron is removed in form of gaseous boron oxides BO, B2O2, B2O3, B2O (BxOy) and their equilibrium partial pressures are about 10-3-10 Pa. When the H2O-O2 mixture is used under the same conditions, the gaseous boron hydrates are mainly B3H3O6, BHO2, BH3O3, BHO, BH2O2, B2H4O4 (BxHzOy) and their equilibrium pressures are 105-1010 times that of BxOy. The removing rate in the H2O-O2 mixture atmosphere is absolutely higher than that of O2 at lower
refining temperature.
2) In the electric arc furnace, the boron content in metallurgical grade silicon can be reduced from 18×10-6 to 2×10-6 by oxidizing refining in the Ar-H2O-O2 mixture atmosphere.
References
[1] US Department of Energy, National Renewable Energy Laboratory. Photovoltaics energy for the new millennium [R]. The National 2000-2004 Photovoltaics Program Plan. 2000.
[2] KHATTAK C P, JOYCE D B, SCHMID F. A simple process to remove boron from metallurgical grade silicon [J]. Solar Energy Materials & Solar Cells, 2002, 74(1/4): 77-89.
[3] ROUSSEAU S, BENMANSOUR M, MORVAN D, AMOUROUX J. Purification of MG silicon by thermal plasma process coupled to DC bias of the liquid bath [J]. Solar Energy Materials & Solar Cells, 2007, 91(20): 1906-1905.
[4] FLAMANTA G, KURTCUOGLUB V, MURRAYA J, STEINFELD A. Purification of metallurgical grade silicon by a solar process [J]. Solar Energy Materials & Solar Cells, 2006, 90(14): 2099-2106.
[5] MIKI T, MORITA K, SANO N. Thermodynamics of phosphorus in molten silicon [J]. Metallurgical and Materials Transactions B, 1996, 27(12): 937-941.
[6] MORITA K, MIKI T. Thermodynamics of solar-grade-silicon refining [J]. Intermetallics,2003, 11(11/12): 1111-1117.
[7] PEI Bin-xiong, ZHANG Hong-bin, CAO Mei-jiao, WANG Yuan-ming, HE Yun-hua, LI Shou-jun. Argumentation on the macrosegregation of boron in vacuum melting pouring ingot of GH901 alloy [J]. Special Steel Technology, 2006, 12(48): 5-13.
[8] TEIXEIRA LEANDRO A V, MORITA K. Thermo-dynamics of boron removal from molten silicon with CaO-SiO2 slag [J]. Current Advances in Materials and Processes, 2007, 20(1): 83-87.
[9] VEDDE J, TRONSTAD R. PV-FZ―A cost effective route to high efficiency solar cell [C]// Proceedings of 21st European Photovoltaic Solar Energy Conference. Germany: 2006. 976-978.
[10] TANAHASHI M, SHINPO Y, FUJISAWA T, YAMAUCHI C. Distribution behavior of boron between SiO2-saturated NaO0.5-CaO- SiO2 flux and molten silicon [J]. Journal of the Mining and Materials Processing Institute of Japan, 2002, 118(7): 497-505.
[11] WANG Xin-guo, DING Wei-zhong, SHEN Hong, ZHANG Jing-jiang. Oxidizing refining process of metallurgical grade silicon [J]. The Chinese Journal of Nonferrous Metals, 2002, 12(4): 827-831. (in Chinese)
[12] ALEMANY C, TRASSY C, PATEYRON B, LI K I, DELANNOY Y. Refining of metallurgical-grade silicon by inductive plasma [J]. Solar Energy Materials & Solar Cells, 2002, 72(1/4): 41-48.
[13] DELANNOY Y, ALEMANY C, LI K I, PROULX P, TRASSY C. Plasma-refining process to provide solar-grade silicon [J]. Solar Energy Materials & Solar Cells, 2002, 72(1/4): 69-75.
[14] SUZUKI K, SAKAGUCHI K, NAKAGIRI T, SANO N. Gaseous removal of phosphorus and boron from molten silicon [J]. J Japan Inst Melts, 1990, 54(2): 161-167.
[15] CHASE M W. NIST-JANAF thermochemical tables [M]. 4th ed. New York: American Chemical Society and the American Institute of Physics for the National Institute of standards and Technology, 1998: 221-1754.
Foundation item: Project(50674050) supported by the National Natural Science Foundation of China; Project(2006BAE01B08) supported by the Key Project of National Science and Technology Program of China
Corresponding author: MA Wen-hui; Tel: +86-871-5107208; E-mail: mwhui@kmust.edu.cn
DOI: 10.1016/S1003-6326(08)60296-4
(Edited by YANG Bing)