
Acid leaching of vanadium from roasted residue of stone coal
ZHU Yang-ge(朱阳戈), ZHANG Guo-fan(张国范), FENG Qi-ming(冯其明),
LU Yi-ping(卢毅屏), OU Le-ming(欧乐明), HUANG Si-jie(黄思捷)
School of Resources Processing and Bioengineering, Central South University, Changsha 410083, China
Received 6 July 2009; accepted 30 December 2009
____________________________________________________________________________________________
Abstract: Through leaching from residue directly and leaching after a roasting treatment, respectively, the experimental research on sulfuric leaching of vanadium from residue of stone coal that came from power generation was conducted. Factors which influence the leaching of vanadium such as concentration of sulfuric acid, leaching temperature, leaching time and liquid-to-solid ratio were investigated in both processes. In the process of direct leaching, to achieve a leaching rate of 74.49%, H2SO4 concentration of up to 5.4%, leaching temperature of 90 ℃ and leaching time of 8 h were necessary reaction factors. The results show that after a roasting treatment at the optimum condition of 950 ℃ at 1 h, 76.88% vanadium can be leached under the experimental condition of 0.45% of H2SO4, 30 ℃ for 1 h with a liquid-to-solid ratio of 2 mL/g. Leaching after an oxidation roasting treatment is an efficient way to leach vanadium from the residue of stone coal, which has some advantages, such as high recovery, low economic cost and less impurities in leaching solution.
Key words: stone coal; roasting; acid leaching; vanadium
____________________________________________________________________________________________
1 Introduction
Vanadium-bearing stone coal is a type of carbonaceous shale with low calorific value. Currently, stone coal is widely used as a kind of fuel for power generation in south China. Vanadium element is enriched in the waste residue resulted from the combustion of carbon, which can be exploited as a type of vanadium resource[1-4]. The gross reserve of vanadium in stone coal is 1.18×108 t in terms of V2O5 in China, which accounts for more than 87% of the domestic reserve of vanadium[5-7]. Therefore, the effective utilization of such a great resource of vanadium is very important for the vanadium industry in China. Some companies have begun to extract vanadium from stone coal by the classical technology since the 1970s. The brief flow of such technology includes chloridizing roasting, water leaching, deposition, alkali melting, and thermal decomposition[8]. These classical technologies have two main problems, namely, low recovery of vanadium (<50%) and serious environment pollution[9]. Nowadays, efficient and environmental-friendly methods of vanadium extraction from stone coal are intensively investigated, and many novel technologies are developed to get higher recovery[10-12]. To a great extent, the leaching degree determines the recovery of vanadium, and a high leaching degree leads to a high vanadium recovery[13]. Many researchers found that the roasting treatment exerted significant influence on the leaching process. Various roasting processes were investigated in order to get high vanadium leaching rate, such as microwave roasting[14] and calcified roasting[15]. The acid leaching after oxidation roasting without additives is a promising technique to leach vanadium from stone coal in an environmental-friendly way, but the related research is relatively less[16-17]. This investigation was laboratory study for leaching vanadium from the residue of stone coal that came from power generation using the process of acid leaching after the oxidation roasting without additives (RL). To make the comparison, the process of leaching directly from residue without the roasting treatment (DL) was also studied. The purpose of the research is to identify the effect of oxidation roasting on leaching process and to realize the comprehensive utilization of vanadium from the waste residue.
2 Experimental
2.1 Materials
The raw material used in this study was taken from Shuangxi Coal Company, Hunan Province, China. The material consists of the stone coal residue from a fluidized-bed roasting process for power generation. The main chemical compositions of the material are as follows: 0.93% V2O5, 88.78% SiO2, and 1.70% C.
2.2 Methods
As shown in Fig.1, part of the raw material (residue from a fluidized-bed roasting process for power generation) is used in Leaching directly (DL), and the rest part of it is oxidation roasted in a rotary kiln, then the oxidation roasted residue is used in Leaching after oxidation roasting (RL). Both of the leaching materials are crushed and grinded to <0.074 mm of 75%-80%, which is suitable for leaching. The residue is leached with sulfuric acid in a conical flask using magnetic stirring. After being leached for the specified time, the slurry is filtered. The leach residues are dried, weighed and subjected to atomic absorption spectroscopy analysis to determine the content of vanadium.
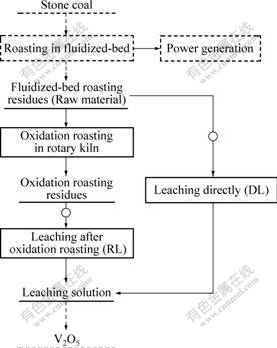
Fig.1 Flowsheet of roasting and leaching
3 Results and discussion
3.1 Effect of roasting time and temperature in RL
A series of experiments were carried out to investigate the effect of oxidation roasting on leaching degree of raw material in RL, and the result is shown in Fig.2. Roasting temperature varied from 800 to 950 ℃ and roasting time ranged from 0.5 to 1.5 h. All of the leaching experiments were conducted with fixed parameters including sulfuric acid concentration of 0.68% (mass fraction, all sulfuric acid concentrations in this work are mass fraction), liquid-to-solid ratio (L/S) of 2 mL/g, leaching temperature of 15 ℃ and leaching time of 1 h. As shown in Fig.2, the leaching degree improves with the increase of roasting temperature. Longer roasting time does not bring a higher leaching degree at 800-850 ℃, but at 900-950 ℃, leaching degree increases significantly with the increase of roasting time. When the roasting time increases from 1 h to 1.5 h at 950 ℃, the increase in leaching degree is not significant. So, the residue roasted for 1 h at 950 ℃ is chosen as the material for RL in all the subsequent leaching experiments. The main chemical composition of the oxidation roasted residue transforms to 1.02% V2O5, 88.30% SiO2, and 0.06% C.

Fig.2 Effect of roasting temperature and time on leaching degree of vanadium in RL
3.2 Effect of sulfuric acid concentration
To study the role of sulfuric acid concentration on the leaching degree of vanadium, the sulfuric acid concentration varied from 0 to 1.35% in RL and 0.9% to 27% in DL. The results are shown in Fig.3. It can be seen from Fig.3 that the leaching degree of vanadium increases with the increase of sulfuric acid concentration in both processes. Although a much lower leaching temperature and shorter leaching time are used in RL, the necessary sulfuric acid concentration needed in RL is much lower than that in DL. To obtain a leaching degree above 60% under their respective conditions, sulfuric acid concentration should reach 5.4% in DL, but in RL, necessary sulfuric acid concentration is only 0.45%, which indicates 1/10 consumption of sulfuric acid than that used in DL.
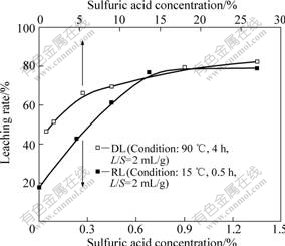
Fig.3 Effect of sulfuric acid concentration on leaching degree of vanadium
3.3 Effect of leaching temperature
The effect of temperature on leaching degree was investigated in the range from 15 to 90 ℃. The result is shown in Fig.4. From Fig.4, it can be concluded that the leaching temperature exerts an obvious influence on the leaching degree in DL. When keeping other reaction conditions fixed and increasing the reaction temperature, the leaching degree improves in the range from 40 to 90 ℃. In RL, the leaching degree increases rapidly under the range of the temperature from 15 to 30 ℃, but little improvement to the leaching degree is observed for a further increase of the temperature. Thus, 30 ℃ is the optimum leaching temperature for RL, and 90 ℃ is necessary for DL.

Fig.4 Effect of leaching temperature on leaching degree of vanadium
3.4 Effect of liquid-to-solid ratio
Fig.5 presents the effect of liquid-to-solid ratio on the leaching degree. It can be seen from Fig.5 that the leaching degree goes up with the increase of liquid-to-solid ratio in RL, while it does not reveal a significant increase in DL. Not only does a higher liquid-to-solid ratio increase the amount of sulfuric acid available, the vanadium concentration in the leach liquor decreases with an increase of the liquid-to-solid ratio. Too low concentration of vanadium in the leach liquor is harmful to the solvent extraction process. Therefore, the liquid-to-solid ratio should not be too high. Moreover, at a liquid-to-solid ratio of 1.5 mL/g, the leach solution is too thick, which is detrimental to the leaching process. Hence, a liquid-to-solid ratio of 2 mL/g is appropriate.
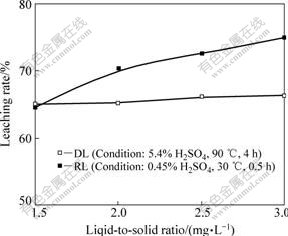
Fig.5 Effect of liquid-to-solid ratio on leaching degree of vanadium
3.5 Effect of leaching time
The effect of leaching time on leaching degree was studied from 0.5 to 8 h. It can be seen from Fig.7 that the leaching degree increases with the increase of the leaching time from 1 to 8 h in DL, and the leaching degree reaches 74.49% after 8 h leaching with the fixed parameters of sulfuric acid concentration of 5.4%, liquid-to-solid ratio of 2 mL/g and leaching temperature of 90 ℃. In RL, after leaching for 1 h, a further increase in the leaching time results in less influence over the leaching degree, and about 76.88% of the vanadium can be leached at reaction time of 1 h, sulfuric acid concentration of 0.45%, reaction temperature of 30 ℃, and liquid-to-solid ratio of 2 mL/g.

Fig.6 Effect of leaching time on leaching degree of vanadium
3.6 Discussion
Generally, it is difficult to realize leaching vanadium from stone coal without roasting. Three reasons can be concluded as follows. Firstly, the valence of vanadium in stone coal is mostly V(Ⅲ), which is very hard to be leached by H2SO4; secondly, V(Ⅲ) replaces Al(Ⅲ) in crystal lattice of clay minerals, consequently, clay lattices must be destroyed in order to leach this part of vanadium[18]; in addition, there is a large amount of carbon (usually >12%) covering the surface of mineral particles in stone coal, which can depress the reaction between clay lattices and H+[19].
During the process of roasting in a fluidized-bed for power generation, heat energy is utilized due to the combustion of carbon (C reduces to 1.70% in roasted residue). Without being covered with carbon, minerals can react with H+, and many pores which form during the carbon combustion can provide channels for the diffusion of H+. Therefore, vanadium can be leached by H2SO4. Nevertheless, clay lattices is not significantly destroyed because of the short roasting time, and the oxidization of vanadium is hindered because of the participation in the reaction of reducing substances, such as organic carbon and pyrite[18, 20], thus, high acid consumption, high temperature and long leaching time must be satisfied in DL due to the result of bad roasting.
Without the depression of reducing substances, the following reactions massively occur during oxidation roasting:
2V2O3+O2=4VO2 (1)
4VO2+O2=2V2O5 (2)
Most of V(Ⅲ) is oxidized to V(Ⅳ) and V(Ⅴ); vanadium is released because of the destroy of clay lattices and almost no carbon exists (0.06% in this work). It is much easier to leach vanadium from residues due to the oxidation and transformation of vanadium. Besides, the acid consumption is largely reduced; the leaching temperature is lower; and the necessary leaching time is shorter. In the leaching process, the main chemical reactions for vanadium in the leaching process are as follows[13]:
MVO3+H+=VO2++M++H2O (3)
V2O5+H2SO4=(VO2)2SO4+H2O (4)
V2O4+H2SO4=2VOSO4+2H2O (5)
In Eq. (1), M is Al, Mg and Fe; and M+ is Al3+, Mg2+, Fe2+ and Fe3+.
Compared with DL, RL can realize higher leaching rate of vanadium, greatly decrease the concentration of sulfuric acid, lower the leaching temperature and shorten the leaching time. According to Eq. (3), impurities such as Al, Fe and Mg are leached along with the vanadium, and Si are leached plentifully. These impurities will influence the preparation of vanadium pentoxide. A lower sulfuric acid concentration means less impurities existing in the leaching solution, which is beneficial to the purification of leaching solution and obtaining high quality vanadium pentoxide. Therefore, it is necessary to get a good roasting effect to lower the sulfuric acid concentration. Moreover, the good roasting effect is obtained by oxidation roasting process without additives, which is friendly to environment.
4 Conclusions
1) In the process of direct leaching from residue, to achieve a leaching rate of 74.49%, H2SO4 concentration of up to 5.4%, leaching temperature of 90 ℃ and leaching time of 8 h are necessary reaction factors. After a roasting treatment at the optimum condition of 950 ℃ at 1 h, as much as 76.88% vanadium can be leached under the experiment condition of H2SO4 of 0.45%, liquid-to-solid ratio of 2 mL/g, reaction temperature of 30 ℃, and reaction time of 1 h.
2) Leaching after a roasting treatment is an efficient way to leach vanadium from residue of stone coal that comes from power generation, which has some advantages such as high recovery, low economic cost and less impurities in leaching solution.
3) The combustion of carbon, oxidation and transformation of vanadium contribute to the change of leaching conditions.
References
[1] ALFANTAZI A M. Hydrometallurgical processing of Egyptian black shale of the Quseir-Safaga region[J]. Hydrometallurgy, 1994, 36(1): 95-107.
[2] HABASHI F. Two hundred years of vanadium[J]. Metall, 2006, 60(12): 804-808.
[3] REN Qiang, LIU Jian-zhong, ZHOU Jun-hu, YE Lin, CEN Ke-fa. Dynamic characteristic of sulfur release during stone coal combustion [J]. Journal of China Coal Society, 2006, 31(1): 99-103. (in Chinese)
[4] LIU Jian-zhong, ZHANG Bao-sheng, ZHOU Jun-hu, YE Lin, CEN Ke-fa. Combustion characteristics and classify attribute of stone coal [J]. Proceedings of the CSEE, 2009, 27(9): 17-22. (in Chinese)
[5] BIN Zhi-yong. Progress of the research on extraction of vanadium pentoxide from stone coal and the market of the V2O5 [J]. Hunan Nonferrous Metals, 2006, 22(1): 16-20. (in Chinese)
[6] LAN Yao-zhong, LIU Jin. Review of vanadium processing in China [J]. Engineer Sciences, 2005, 3(3): 58-62.
[7] MOSKALYK R R, ALFANTAZI A M. Processing of vanadium: A review [J]. Minerals Engineering, 2003, 16(9): 793-805.
[8] QI Ming-jian. The status and prospects of vanadium leaching from stone coal [J]. Hydrometallurgy of China, 1999, 72(4): 1-10. (in Chinese)
[9] CAI Jin-qing, BA Ling. New technology of extraction vanadium from stone coal [J]. Conservation and Utilization of Mineral Resources, 1993(5): 30-33. ( in Chinese)
[10] HE Dong-sheng, FENG Qi-ming, ZHANG Guo-fan, OU Le-ming, LU Yi-ping. An environmentally-friendly technology of vanadium extraction from stone coal [J]. Minerals Engineering, 2007, 20(12): 1184-1186.
[11] LI Cun-xiong, WEI Chang, LI Min-ting, FAN Gang, DENG Zhi-gan. Process optimization of vanadium extraction from black shale by acidic oxidizing pressure leaching [J]. The Chinese Journal of Nonferrous Metals, 2008, 18(S1): 84-87. (in Chinese)
[12] HE Dong-sheng, FENG Qi-ming, ZHANG Guo-fan, OU Le-ming, LU Yi-ping, SHAO Yan-hai. The study on leaching vanadium from stone coal with alkali [J]. Nonferrous Metals: Extractive Metallurgy, 2007(4): 15-17. (in Chinese)
[13] HE Dong-sheng, FENG Qi-ming, ZHANG Guo-fan, OU Le-ming, LU Yi-ping. Study on leaching vanadium from roasting residue of stone coal [J]. Minerals and Metallurgical Processing, 2008, 25(4): 181-184.
[14] OUYANG Guo-qiang, ZHANG Xiao-yun, TIAN Xue-da, LI Yi, XIE Sen. Effect of microwave roasting on vanadium extraction from stone coal [J]. The Chinese Journal of Nonferrous Metals, 2008, 18(4): 750-754. (in Chinese)
[15] MA Sheng-fang, ZHANG Guang-xu. Leaching process of vanadium from argillaceous vanadium ore using calcified roasting(1): Study on roasting technology [J]. Chinese Journal of Rare Metals, 2007, 31(6): 813-817. (in Chinese)
[16] ZOU Xiao-yong, OUYANG Yu-zhu, PENG Qing-jing, TIAN Ren-guo. Study on the process of producing vanadium pentoxide from bone coal vanadium mine by no-salt-roasting and acid leaching [J]. Chemistry World, 2001(3): 117-119. (in Chinese)
[17] ZOU Xiao-yong, OUYANG Yu-zhu, ZHANG Ai-hua, TIAN Renguo. Study on the no-salt-roasting process and equipment of bone coal vanadium mine [J]. Inorganic Chemicals Industry, 2000, 32(1): 32-34. (in Chinese)
[18] LIN Hai-ling, FAN Bi-wei. Study on mechanism of phase transformation during roasting and extracting vanadium from fangshankou bone coal [J]. Chinese Journal of Rare Metals, 2001, 25(4): 273-277. (in Chinese)
[19] HE Dong-sheng, FENG Qi-ming, ZHANG Guo-fan, OU Le-ming, LU Yi-ping. Mechanism of oxidizing roasting process of vanadium containing stone coal [J]. The Chinese Journal of Nonferrous Metals, 2009, 19(1): 195-200. (in Chinese)
[20] CHEN Tie-jun, QIU Guan-zhou, ZHU De-qing. Experiment study on new technology of extracting vanadium from stone coal with cyclic oxidation [J]. Journal of China Coal Society, 2008, 33(4): 454-458. (in Chinese)
______________________________
Foundation item: Project(50974133) supported by the National Natural Science of China; project(08zxgk06) supported by the Opening Foundation of Key Laboratory of Solid Waste Treatment and Resource Recycle (SWUST), Ministry of Education, China
Corresponding author: ZHANG Guo-fan; Tel: +86-731-88830913; E-mail: zhangguofan2002@163.com
(Edited by CHEN Can-hua)