
Synthesis of tetrapod-shaped ZnO whiskers and microrods in one crucible by thermal evaporation of Zn/C mixtures
DU Guo-ping(杜国平)1, LI Wang(李 旺)1, FU Min-gong(付敏恭)2, CHEN Nan(陈 楠)1,
FU Xue(付 雪)3, WAN Yi-qun(万益群)2, YAN Ming-ming(严明明)1
1. School of Materials Science and Engineering, Nanchang University, Nanchang 330031, China;
2. Center for Analysis and Testing, Nanchang University, Nanchang 330047, China;
3. Department of Systems Science, Jiangxi University of Finance and Economics, Nanchang 330013, China
Received 21 March 2007; accepted 25 July 2007
Abstract: Tetrapod-shaped ZnO whiskers and microrods were synthesized in one crucible by thermal evaporation of Zn/C mixtures at 930 ℃ in air without any catalyst. The digital camera, optical microscopy, scanning electron microscopy, energy dispersive X-ray spectroscopy, and X-ray diffraction techniques were used to study the morphologies and crystal structures of these tetrapod-shaped ZnO microcrystals. The results show that these two types of ZnO tetrapods are grown at different heights within the same crucible. The legs of these tetrapod-shaped ZnO crystals are hexagonally faceted. Some tetrapod-shaped ZnO whiskers show hierarchical structures. A short button-like hexagonal ZnO microcrystal is observed at the triple junctions of some tetrapod-shaped ZnO whiskers. The tetrapod-shaped ZnO microrods are capped by two sets of hexagonal pyramids with two different groups of crystal planes for the surfaces. These two types of tetrapod-shaped ZnO microcrystals have different side faces and aspect ratio, which are believed to be the result of their different growth behaviors. The octa-twin model was used to discuss the different growth behaviors of these two types of ZnO tetrapods. The crystal planes of the legs and the pyramids were determined.
Key words: ZnO; tetrapod-shaped whiskers; tetrapod-shaped microrods; growth from vapor; octa-twin model
1 Introduction
ZnO is one of the promising materials with a variety of application potentials[1]. ZnO has a direct wide bandgap of 3.4 eV and a large excitation binding energy of 60 meV. It is therefore considered to be a candidate for next generation optoelectronic material.
ZnO microcrystals have been synthesized in different morphologies, such as microtubes[2-4] and microrods[5-10]. HU et al[2] synthesized ZnO microtubes by first evaporating ZnS/C mixtures in Ar at 1 100 ℃ and then introducing O2. CHENG et al[3] grew ZnO microtubes by heating ZnO over 1 300 ℃ using microwave. SUN et al[4] synthesized ZnO microtubes by using Zn powder as the source and Ar as the carrier gas. ZnO microrods were generally grown by wet chemical method[5-10]. Tetrapod-shaped ZnO nano- and micro-crystals have also been fabricated by different researchers. DAI et al[11] and WANG et al[12] synthesized tetrapod-shaped ZnO nano- and micro- crystals by thermal evaporation of Zn powder in a horizontal tube furnace. WANG et al[12] used Si wafers as substrate and placed them in the tube, and then tetrapod-shaped ZnO grew on these substrates. Iwanaga et al[13] proposed an octa-twin growth model for tetrapod-shaped ZnO crystals, and it has been generally accepted[1,4,11].
In this work, we used a simple method to grow ZnO microcrystals by thermal evaporation of Zn/C mixtures in a crucible at 930 ℃ in air without using any catalyst. Different from the published method in Refs.[2,4,11,12], in which Zn powder or zinc compound was mixed with C and then simply put in a crucible, we added a layer of C powder at the bottom of the crucible before the Zn/C mixtures were put into the crucible. By adding the layer of amorphous C at the crucible bottom, we believed that C will react with O2 to generate extra CO/CO2 from the bottom and then carry the evaporated Zn/ZnO vapor to the upper area of the crucible. We therefore hoped to observe the possible new growth behaviors of ZnO crystals by this setup. We obtained tetrapod-shaped ZnO whiskers and microrods with quite different morphologies in the same crucible.
2 Experimental
High purity Zn powder and activated carbon were used as the source materials. The Zn powder was mixed with activated carbon in a mass ratio of 2?1. The Zn/C mixtures of 2-4 g were put in an alumina crucible of 30 mL, which contained a layer of C powder at the bottom of the crucible. The crucible without being covered by a lid was then placed inside a box furnace, and heated to 930 ℃ in air. The furnace was kept at 930 ℃ for about 30 min and then naturally cooled down to room temperature. X-ray diffractometry(XRD) (Bruker D8 Focus, Cu Kα1) was used to study the crystal structures of the as-synthesized products. Optical microcopy (Keyence AHX-100 Digital Microscope) and scanning electron microscopy (SEM) (FEI Quanta 200 equipped with Oxford Inca energy-dispersive X-ray spectroscopy) were used to observe the morphologies of these products. For SEM studies, the products were ultrasonically dispersed in ethanol for about 10 min before the observation. The composition of the products was investigated with energy-dispersive X-ray spectroscopy(EDXS).
3 Results and discussion
The as-synthesized products marked with A in Fig.1(a) grow from the inner walls of the crucibles at a certain height from the crucible bottom, and extend horizontally towards the central area. Their length is as long as 3 mm with a branch-like morphology. One can note in Fig.1(a) that two pieces (marked with short arrows) of product A fall to the crucible bottom. Fig.1(b) shows the optical micrograph of one of these branch-like pieces (A in Fig.1(a)). In Fig.1(a), the product marked with B is the as-synthesized white products, which is piled at the crucible bottom above the carbon ash (marked as C).
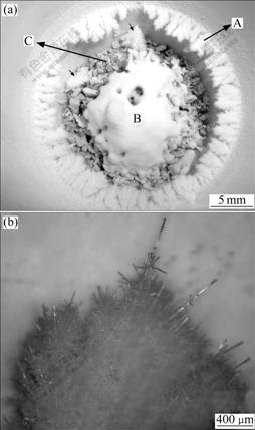
Fig.1 Digital photograph (a) of products (branch-like products grown from inner walls and products at bottom) taken with crucible; Optical micrograph (b) of a piece of branch-like products
Fig.2 shows the XRD results for the as-synthesized products (products A and B in Fig.1(a)). All diffraction peaks are readily indexed as the hexagonal wurtzite crystal structure of ZnO. The sharp peaks in Fig.2 indicate that these products are highly crystallized ZnO.
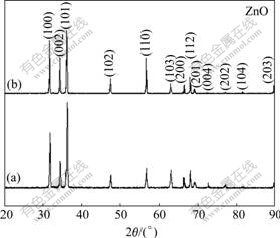
Fig.2 XRD patterns of branch-like products (a) (A in Fig.1(a)), and products at crucible bottom (b) (B in Fig.1(a)).
It is noted that the boiling point of Zn is 907 ℃, so Zn vapor can be easily formed under the synthesis conditions, and react with O2 to form ZnO molecules. The carbon in the Zn/C mixture can react with O2 to form CO/CO2. The consumption of O2 by the carbon can possibly ensure that some of the vapor atoms are Zn atoms. The carbon at the crucible bottom can react with O2 to generate CO/CO2, which will carry the Zn/ZnO vapor to the upper area of the crucible. The Zn atoms will be oxidized to form ZnO molecules. The formation of the branch-like ZnO products (A in Fig.1(a)) is a result of the presence of ZnO molecules in the upper area of crucible.
The ZnO particles at the crucible bottom (B in Fig.1(a)) are mainly tetrapod-shaped ZnO whiskers with high aspect ratio. Figs.3 and 4 show the SEM micrographs of these tetrapod-shaped ZnO whiskers at the crucible bottom. EDX results confirm that these tetrapod-shaped units are composed of elements Zn and O only (Fig.5). One can see a large number of tetrapod- shaped ZnO whiskers with different dimensions (up to 75 μm in length, and 8 μm in thickness) in Fig.3(a). The four legs on each unit taper down in size along their length-wise direction away from their central part, and this is in agreement with the published results[1,13]. Some units taper into wires (marked with arrows in Fig.3(a)) at the tips of their legs. These legs are hexagonally shaped. This can be clearly seen on the broken leg of a unit of tetrapod-shaped ZnO particle in Fig.4(a).

Fig.3 SEM micrographs of products B in Fig.1(a): (a) A number of tetrapod-shaped ZnO whiskers; (b) Magnified image of unit marked as A in Fig.3(a); (c) Magnified image of unit marked as B in Fig.3(a)

Fig.4 SEM micrographs of products B in Fig.1(a): (a) Four units of tetrapod-shaped ZnO whiskers; (b) Magnified image of unit pointed by thick white arrow in Fig.4(a); (c) Magnified image of leg indicated by thin white arrow in Fig.4(a), showing hierarchical structure; (d) Magnified image of two impinging legs pointed by black arrow in Fig.4(a)

Fig.5 EDX result for tetrapod-shaped whiskers in Figs.3 and 4
In Fig.3(a), it is interesting to see that several units of tetrapod-shaped ZnO whiskers have short button-like hexagonal ZnO microcrystals (marked with A) at their triple junctions, and this can be better viewed in Fig.3(b) in higher magnification. The presence of the button-like hexagonal ZnO crystals at the triple junctions in Fig.3(b) will be discussed later in this section. Differently, some units show clean triple junctions (marked with B in Fig.3(a)), and this can be clearly seen on the enlarged unit in Fig.3(c). In Figs.3(b) and 3(c), the three arrowed side faces of the three legs meet together at the triple junctions. If we extend the three arrowed side faces of the three legs at the triple junctions, they will then form three surfaces of an inverted trigonal pyramid.
Four units of tetrapod-shaped ZnO whiskers are present in Fig.4(a), a larger one and three smaller ones. As marked with a white thick arrow in Fig.4(a), one can also see the presence of a button-like hexagonal ZnO crystal at the triple junction region of the tetrapod-shaped ZnO whisker, and this can be better seen in Fig.4(b). The zones indicated by white thin arrows in Fig.4(a) are the two hierarchical legs of a unit of tetrapod-shaped ZnO whisker, while the other two legs are broken at their tips. Such type of hierarchical legs can be better viewed in Fig.4(c).
As indicated by the black arrow in Fig.4(a), one leg of a smaller unit of tetrapod-shaped ZnO whisker impinges into the leg of the much larger unit, and the enlarged micrograph is shown in Fig.4(d). This can be explained as follows. Firstly, the leg (marked as A in Fig.4(a)) of the smaller unit grows along its length-wise direction towards the surface of the leg (marked as B in Fig.4(a)) of the larger unit, and then the former is in direct touch with the surface of the latter. Subsequently, the length-wise growth of leg A is therefore blocked by the latter, and further growth is therefore stopped. The other three legs on the smaller unit however grow freely. This explains why leg A is shorter than the other legs as shown in Fig.4(a). It is believed that the thickness-wise growth of leg A should not be disturbed because the deposition of ZnO molecules on its surfaces is not blocked, and this is supported by the fact that the thickness of leg A is comparable with that of the other legs, as can be seen in Fig.4(a). In the meantime, leg B grows thicker and thicker, and the tip of leg A is then embedded into the body of leg B. Such impinging behavior in Fig.4(a) suggests that the growth mechanism is vapor-solid(VS) rather than the vapor-liquid-solid (VLS) model. This is also supported by the fact that no catalyst is used in this study.
Similar to Figs.3(a) and 3(b), a short button-like hexagonal ZnO microcrystal is also observed at the triple junction in Fig.4(b). In addition, three black arrowed side faces in Figs.4(a) and (b) meet together in the triple junction area. After being extended, these three side faces will also form the three surfaces of an inverted trigonal pyramid.
Fig.6 shows the SEM micrographs of a number of tetrapod-shaped ZnO microrods that were collected from products A in Fig.1. These particles were ultrasonically dispersed away from the branch-like products (A in Figs.1(a) and (b)). Their composition was confirmed to be ZnO by EDX (not shown here). One can note from Fig.6 that these tetrapod-shaped ZnO microrods are capped by hexagonal pyramids. Each leg of these tetrapod-shaped ZnO microrods is hexagonally faceted. Compared with the tetrapod-shaped whiskers shown in Figs.3 and 4, the aspect ratio of these tetrapod-shaped microrods is much smaller. In some cases the thickness of the legs of these tetrapod-shaped microrods is comparable to their length. In Fig.6(b), the legs of the tetrapod-shaped ZnO particle are 5 μm in length, and 4.5 μm in thickness. Another major difference from the tetrapod-shaped whiskers (Figs.3 and 4) is that the three arrowed edges (Fig.6) of each unit of these tetrapod-shaped microrods meet together at the central point of the triple junction.

Fig.6 SEM micrographs of products A in Fig.1(a): (a) A number of tetrapod-shaped ZnO microrods with hexagonal pyramidal caps; (b) Tetrapod-shaped ZnO microrods with hexagonal pyramidal caps different from Fig.6(a)
The octa-twin model was generally used to explain the formation mechanism of tetrapod-shaped ZnO crystals[13], and the details of this model were explained in a review paper[1]. ZnO nucleates in the form of octa-twins, each containing eight tetrahedral nanocrystals [1]. The side faces of each tetrahedral nanocrystal are three facets and one (0001) basal facet, as shown in Figs.7(a) and (b). The octa-twin has eight surfaces which are the basal planes. The eight surfaces of the octa-twin are alternately opposite in terms of their polarities, as shown in Fig.7(b), with four Zn-(0001) surfaces (+c) and four
facets and one (0001) basal facet, as shown in Figs.7(a) and (b). The octa-twin has eight surfaces which are the basal planes. The eight surfaces of the octa-twin are alternately opposite in terms of their polarities, as shown in Fig.7(b), with four Zn-(0001) surfaces (+c) and four  surfaces (-c). The growth on the four Zn-(0001) surfaces along [0001] direction is the fastest [1, 13], while no growth will be initiated on the
surfaces (-c). The growth on the four Zn-(0001) surfaces along [0001] direction is the fastest [1, 13], while no growth will be initiated on the surfaces according to Ref.[1]. This therefore will lead to the formation of tetrapod- shaped ZnO crystals. The growth of both the tetrapod- shaped ZnO whiskers (Figs.3 and 4) and microrods (Fig.6) should be explained by this octa-twin model.
surfaces according to Ref.[1]. This therefore will lead to the formation of tetrapod- shaped ZnO crystals. The growth of both the tetrapod- shaped ZnO whiskers (Figs.3 and 4) and microrods (Fig.6) should be explained by this octa-twin model.

Fig.7 Tetrahedral model of ZnO (a); Octa-twin model (b); Base portion (c) of Fig.7(a), showing two sets of facets (Copyright 2004 for (a) and (b), Wiley-VCH Verlag, Weinheim, Germany, image courtsey Prof. Z. L. Wang, Georgia Tech.)[1]
If the four surfaces of an octa-twin do not initiate any growth as indicated in Ref.[1], the triple junctions of all these tetrapod-shaped ZnO crystals should be sharp and clear, as shown in Fig.3(c). However, the presence of short button-like hexagonal ZnO micro-crystals (Figs.3(b) and 4(b)) at the triple junctions of the tetrapod-shaped ZnO whiskers may suggest that at least some octa-twins can still possibly initiate crystal growth on the
surfaces of an octa-twin do not initiate any growth as indicated in Ref.[1], the triple junctions of all these tetrapod-shaped ZnO crystals should be sharp and clear, as shown in Fig.3(c). However, the presence of short button-like hexagonal ZnO micro-crystals (Figs.3(b) and 4(b)) at the triple junctions of the tetrapod-shaped ZnO whiskers may suggest that at least some octa-twins can still possibly initiate crystal growth on the  surfaces, but with a much lower growth rate compared with the Zn-(0001) surfaces.
surfaces, but with a much lower growth rate compared with the Zn-(0001) surfaces.
As the hexagonal legs of a tetrapod-shaped ZnO crystal grow, each leg can have either six  facets or six
facets or six  facets[14]. This is illustrated in Fig.7(c), which is the base portion of Fig.7(a). It can be seen from Fig.7(c) that either of the two sets of facets can guarantee the growth of legs in hexagonal shape. The tetrapod- shaped ZnO whiskers should have the
facets[14]. This is illustrated in Fig.7(c), which is the base portion of Fig.7(a). It can be seen from Fig.7(c) that either of the two sets of facets can guarantee the growth of legs in hexagonal shape. The tetrapod- shaped ZnO whiskers should have the facets (Fig.7(c)), because this ensures that the three arrowed side faces of the three legs (Figs.3(b), 3(c) and 4(b)) meet together at their triple junctions. Differently, the tetrapod-shaped ZnO microrods should have the
facets (Fig.7(c)), because this ensures that the three arrowed side faces of the three legs (Figs.3(b), 3(c) and 4(b)) meet together at their triple junctions. Differently, the tetrapod-shaped ZnO microrods should have the  facets, ensuring that the three arrowed edges of each unit of these tetrapod-shaped microrods meet together at the central point at the triple junction (Fig.6). Due to such different facets associated with these two types of tetrapod-shaped ZnO crystals, they should have different growth rates in the thickness-wise directions of their legs, and this will result in a different aspect ratio. The higher aspect ratio of the tetrapod-shaped ZnO whiskers suggests that the thickness-wise growth rate on the
facets, ensuring that the three arrowed edges of each unit of these tetrapod-shaped microrods meet together at the central point at the triple junction (Fig.6). Due to such different facets associated with these two types of tetrapod-shaped ZnO crystals, they should have different growth rates in the thickness-wise directions of their legs, and this will result in a different aspect ratio. The higher aspect ratio of the tetrapod-shaped ZnO whiskers suggests that the thickness-wise growth rate on the  facets is lower than that on the
facets is lower than that on the facets of the tetrapod-shaped microrods.
facets of the tetrapod-shaped microrods.
It can be noted in Fig.6 that the hexagonal pyramidal caps on the tetrapod-shaped microrods (A in Fig.6(a) for an example) have different facets from these on the tetrapod-shaped microrods in Fig.6(b) (marked as A and B for example). From the above discussion, it is known that the legs underneath the hexagonal pyramidal caps have  facets (Fig.7(c)), so it is possible to use the ZnO lattice cell (Fig.8) to determine the crystal planes of the surfaces of the pyramidal caps. The bold lines in Fig.8 define two hexagonal pyramids based on the lattice structure of ZnO. The pyramid in Fig.8(a) has six surfaces with
facets (Fig.7(c)), so it is possible to use the ZnO lattice cell (Fig.8) to determine the crystal planes of the surfaces of the pyramidal caps. The bold lines in Fig.8 define two hexagonal pyramids based on the lattice structure of ZnO. The pyramid in Fig.8(a) has six surfaces with  crystal planes, and the angle between the two opposite edges is calculated (ZnO lattice constants, a=0.324 nm and c=0.52 nm) to be about 102.5?. In Fig.6(a), the angle between the two opposite edges of the pyramidal cap on the leg marked with A is measured to be about 105?. When ignoring the measurement error due to the slight tilt on the unit in Fig.6(a), this is in very good agreement with the angle in Fig.8(a), indicating that the surfaces of the pyramids in Fig.6(a) are in fact
crystal planes, and the angle between the two opposite edges is calculated (ZnO lattice constants, a=0.324 nm and c=0.52 nm) to be about 102.5?. In Fig.6(a), the angle between the two opposite edges of the pyramidal cap on the leg marked with A is measured to be about 105?. When ignoring the measurement error due to the slight tilt on the unit in Fig.6(a), this is in very good agreement with the angle in Fig.8(a), indicating that the surfaces of the pyramids in Fig.6(a) are in fact  crystal planes. The pyramid in Fig.8(b) has six surfaces with
crystal planes. The pyramid in Fig.8(b) has six surfaces with  crystal planes, and the angle between the two opposite edges is calculated to be about 64?. In Fig.6(b), the angle between the two opposite edges of the pyramidal cap on the leg marked with A is measured to be about 65?. By comparing this angle with the one in Fig.8(b) as calculated above, one can find that the surfaces of the pyramids in Fig.6(b) are
crystal planes, and the angle between the two opposite edges is calculated to be about 64?. In Fig.6(b), the angle between the two opposite edges of the pyramidal cap on the leg marked with A is measured to be about 65?. By comparing this angle with the one in Fig.8(b) as calculated above, one can find that the surfaces of the pyramids in Fig.6(b) are  crystal planes. The agreement above, in turn, strongly supports the discussion in the previous paragraphs.
crystal planes. The agreement above, in turn, strongly supports the discussion in the previous paragraphs.
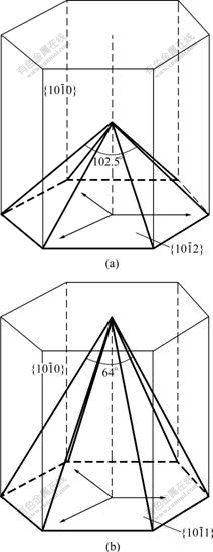
Fig.8 Two types of pyramids illustrated by bold lines in ZnO crystal cell: (a) Pyramid with six surfaces of 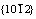 crystal planes with angle about 102.5? between two opposite edges; (b) Pyramid with six surfaces of
crystal planes with angle about 102.5? between two opposite edges; (b) Pyramid with six surfaces of 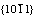 crystal planes with angle about 64? between two opposite edges
crystal planes with angle about 64? between two opposite edges
4 Conclusions
Tetrapod-shaped ZnO whiskers and microrods were synthesized in one crucible by thermal evaporation of Zn/C mixtures at 930 ℃ in air without catalyst. These two types of tetrapod-shaped microcrystals are found to be grown at different heights within the same crucible. The legs of these two types of tetrapod-shaped ZnO microcrystals are hexagonal. A short button-like hexagonal ZnO microcrystal is observed at the triple junctions of some tetrapod-shaped ZnO whiskers. Some of the tetrapod-shaped ZnO whiskers are found to have hierarchical structures. The tetrapod-shaped ZnO microrods are capped by hexagonal pyramids, and have a smaller aspect ratio than the whiskers. The surfaces of these hexagonal pyramids have two different groups of crystal planes,  and
and 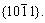 Three side faces of the tetrapod-shaped ZnO whisker meet together at the triple junction, while three edges meet together at the triple junction for the tetrapod-shaped ZnO microrod. This is well explained by the octa-twin model. The tetrapod-shaped ZnO whiskers and microrods are found to have different side facets, with
Three side faces of the tetrapod-shaped ZnO whisker meet together at the triple junction, while three edges meet together at the triple junction for the tetrapod-shaped ZnO microrod. This is well explained by the octa-twin model. The tetrapod-shaped ZnO whiskers and microrods are found to have different side facets, with  for the former, and
for the former, and  for the latter. The different aspect ratio between the tetrapod-shaped ZnO whiskers and microrods is due to the different growth rate on the side faces.
for the latter. The different aspect ratio between the tetrapod-shaped ZnO whiskers and microrods is due to the different growth rate on the side faces.
References
[1] WANG Z L, XIANG K Y, DING Y, GAO P, HUGHES W L, YANG R, ZHANG Y. Semiconducting and piezoelectric oxide nanostructures induced by polar surfaces [J]. Adv Funct Mater, 2004, 14: 943-956.
[2] HU J Q, BANDO Y. Growth and optical properties of single crystalline ZnO whiskers [J]. Appl Phys Lett, 2003, 82: 1401-1403.
[3] CHENG J, GUO R, WANG Q M. Zinc oxide single-crystal microtubes [J]. Appl Phys Lett, 2004, 85: 5140-5142.
[4] SUN X H, LAM S, SHAM T K, HEIGL F, Ju1rgensen A, WONG N B. Synthesis and synchrotron light-induced luminescence of ZnO nanostructures [J]. J Phys Chem B, 2005, 109: 3120-3125.
[5] Vayssieres L, KEIS K, Lindquist E E, Hagfeldt A. Purpose-built anistropic metal oxide material: 3D highly oriented microrod array of ZnO [J]. J Phys Chem B, 2001, 105: 3350-3352.
[6] Henley S J, Ashfold M N R, Nicholls D P, Wheatley P, Cherns D. Controlling the size and alignment of ZnO microrods using ZnO thin film templates deposited by pulsed laser ablation [J]. Appl Phys A, 2004, 79: 1169-1173.
[7] Jiang C, Zhang W, Zou G, YU W, QIAN Y. Precursor-induced hydrothermal synthesis of flowerlike cupped-end microrod bundles of ZnO [J]. J Phys Chem B, 2005, 109: 1361-1363.
[8] Kuo C L, Kuo T J, Huang M H. Hydrothermal synthesis of ZnO microspheres and hexagonal microrods with sheetlike and platelike nanostructures [J]. J Phys Chem B, 2005, 109: 20115-20121.
[9] Yang Y H, Wang B, Yang G W. Growth mechanisms of one-dimensional zinc oxide hierarchical structures [J]. Nanotechnology, 2006, 17: 5556-5560.
[10] KIM Y J, LEE C H, HONG Y J, YI G C, KIM S S, CHEONG H. Controlled selective growth of ZnO nanorod and microrod arrays on Si substrate by a wet chemical method [J]. Appl Phys Lett, 2006, 89: 163128.1-163128.3.
[11] DAI Y, ZHANG Y, WANG Z L. The octa-twin tetraleg ZnO nanostructures [J]. Solid State Commun, 2003, 126: 629-633.
[12] WANG F, YE Z, MA D, ZHU L, ZHUGE F. Rapid synthesis and photoluminescence of novel ZnO nanotetrapods [J]. J Cryst Growth, 2004, 274: 447-452.
[13] Iwanaga H, Fujii M, Takeuchi S. Growth model of tetrapod zinc oxide particles [J]. J Cryst Growth, 1993, 134: 275-280.
[14] WANG Z L. Zinc oxide nanostructures: growth, properties and applications [J]. J Phys: Condens Matter, 2004, 16: R829-R858.
Foundation item: Project(0061) supported by the Doctorate Foundation of Nanchang University, China; Project(2006015) supported by the Center for Analysis and Testing, Nanchang University, China
Corresponding author: DU Guo-ping; Tel: +86-13755792586; E-mail: guopingdu@ncu.edu.cn
(Edited by YUAN Sai-qian)