含砷石灰铁盐渣的理化特性及其浸出行为
来源期刊:中国有色金属学报(英文版)2017年第5期
论文作者:彭兵 雷杰 闵小波 柴立元 梁彦杰 游洋
文章页码:1188 - 1198
关键词:含砷固废;石灰铁盐渣;理化特性;浸出行为;BCR连续提取法
Key words:arsenic-bearing sludge; lime-ferrate sludge; physicochemical properties; leaching behavior; BCR sequential extraction
摘 要:以典型石灰铁盐法处理二种含砷废水产生的污泥(污酸渣和砷酸钙渣)为研究对象,采用ICP-AES、SEM-EDS、XRD、XPS和化学物相分析等检测手段对其化学组成、形貌特征、物相结构及砷的赋存状态进行研究,采用毒性浸出实验和BCR三步连续提取法考察污泥中砷的浸出行为。研究结果表明,污酸渣和砷酸钙渣中砷的含量分别为2.5%和21.2%,主要组成物相为砷酸盐及砷氧化物,均以无定型的颗粒均匀分散或团聚在污泥中。砷的浸出毒性超出TCLP标准规定限值的119和1063倍,浸出率分别为47.66%和 50.15%。砷以酸可提取态和 可还原态为主,两者共占90%左右,而稳定的残渣态含量相对较低,这是含砷石灰铁盐渣浸出毒性大、环境活性高的直接原因。本研究为含砷石灰铁盐渣无害化处理技术提供了大量有用的基本数据。
Abstract: Physicochemical properties and leaching behaviors of two typical arsenic-bearing lime-ferrate sludges (ABLFS), waste acid residue (WAR) and calcium arsenate residue (CAR), are comprehensively described. The chemical composition, morphological features, phase composition and arsenic occurrence state of WAR and CAR are analyzed by ICP-AES, SEM-EDS, XRD, XPS and chemical phase analysis. The toxicity leaching test and three-stage BCR sequential extraction procedure are utilized to investigate arsenic leaching behaviors. The results show that the contents of arsenic in WAR and CAR are 2.5% and 21.2% and mainly present in the phases of arsenate and arsenic oxides dispersed uniformly or agglomerated in amorphous particles. The leaching concentrations of arsenic excess 119 and 1063 times of TCLP standard regulatory level with leaching rates of 47.66% and 50.15% for WAR and CAR, respectively. About 90% of extracted arsenic is in the form of acid soluble and reducible, which is the reason of high arsenic leaching toxicity and environmental activity of ABLFS. This research provides comprehensive information on harmless disposal of ABLFS from industrial wastewater treatment of lime-ferrate process.
Trans. Nonferrous Met. Soc. China 27(2017) 1188-1198
Bing PENG1,2, Jie LEI1, Xiao-bo MIN1,2, Li-yuan CHAI1,2, Yan-jie LIANG1,2, Yang YOU1
1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. Chinese National Engineering Research Center for Control & Treatment of Heavy Metal Pollution, Central South University, Changsha 410083, China
Received 30 June 2016; accepted 11 January 2017
Abstract: Physicochemical properties and leaching behaviors of two typical arsenic-bearing lime-ferrate sludges (ABLFS), waste acid residue (WAR) and calcium arsenate residue (CAR), are comprehensively described. The chemical composition, morphological features, phase composition and arsenic occurrence state of WAR and CAR are analyzed by ICP-AES, SEM-EDS, XRD, XPS and chemical phase analysis. The toxicity leaching test and three-stage BCR sequential extraction procedure are utilized to investigate arsenic leaching behaviors. The results show that the contents of arsenic in WAR and CAR are 2.5% and 21.2% and mainly present in the phases of arsenate and arsenic oxides dispersed uniformly or agglomerated in amorphous particles. The leaching concentrations of arsenic excess 119 and 1063 times of TCLP standard regulatory level with leaching rates of 47.66% and 50.15% for WAR and CAR, respectively. About 90% of extracted arsenic is in the form of acid soluble and reducible, which is the reason of high arsenic leaching toxicity and environmental activity of ABLFS. This research provides comprehensive information on harmless disposal of ABLFS from industrial wastewater treatment of lime-ferrate process.
Key words: arsenic-bearing sludge; lime-ferrate sludge; physicochemical properties; leaching behavior; BCR sequential extraction
1 Introduction
Arsenic pollution both in aqueous solutions [1-8] and contaminated soils [9-14] aroused a significant environmental concern. Various treatment processes such as coagulation, precipitation, ion exchange and adsorption were extensively employed for the removal of arsenic from water [15,16]. Lime-ferrate process is one of the most widely used methods for arsenic-containing wastewater treatment in practice of non-ferrous smelting industry because of its low cost and simple operation. However, this process generates a large amount of toxic arsenic-rich neutralization sludges, which are classified as a hazardous waste due to their high arsenic and other heavy metals contents [17]. The harm of arsenic-bearing lime-ferrate sludge (ABLFS) on health is mentioned by many researchers [18,19]. As one of the two most representative ABLFS, waste acid residue (WAR) is generated in traditional lime-ferrate process for the treatment of acidic wastewater from sulfuric acid production, while calcium arsenate residue (CAR) is from high arsenic wastewater treatment in less common and precious metal smelting, respectively. The huge quantities of these residues result in environmental pollution due to heavy metals leached out in disposal.
The physicochemical properties of sludge, including the chemical composition, structural feature and mineralogical phase composition, along with its leaching behaviors play a crucial role in treatment of solid wastes. In recent decades, various treatment techniques are developed to stabilize and recover heavy metals from solid waste, including direct extraction, roasting, crystallization and solidification based on its characteristics [20-23]. LI et al [24] determined the mineralogy characterizations of zinc leaching residue and provided information to develop new technology for separating iron and zinc from zinc leaching residue. MIN et al [25] accessed the environmental activity and potential ecological risks of heavy metals in zinc leaching residue by four different methods. CHAI et al [26] determined the behavior, transportation and distribution of arsenic during pyrometallurgical process in typical lead smelter. However, there is no detailed and complicated study on the characterization of ABLFS, which is an essential basis of proposing the most appropriate disposal method. Some critical mineralogy information about ABLFS is still unclear, especially phase composition, structural feature and grain morphology of arsenic in the sludge.
The transportation and transformation of heavy metals in environment were reported by many researchers. As an important accumulation of arsenic contaminant, arsenic-bearing solid waste becomes one of the hot issues on treatment and disposal. The potential for arsenic remobilization of arsenic-bearing solid wastes is assessed by TCLP, which is the current USEPA protocol for determining whether the waste is hazardous. PHENRAT et al [27] tested the leaching behaviors of arsenic-iron hydroxide sludge by TCLP. SHAW et al [28] studied the mobility of arsenic in arsenic-bearing solid residuals after stabilization by TCLP and CA-WET. However, it is well known that the TCLP poorly predicts the leaching of oxyanions, especially in arsenic species because the TCLP has a tendency to underestimate the leaching of arsenic from the residuals [29]. To efficiently evaluating various extraction methods, the standard sequential extraction method designed by BCR is used [30]. Such a sequential technique is also employed in several heavy metal and sediment-heavy metal studies [31-33]. Although the studies above are important to the pollution control and waste management, fewer relationships between physicochemical properties and leaching behaviors of ABLFS are clear. The lacking of basic knowledge on ABLFS can necessarily lead to difficulties in its treatment and disposal.
The objective of this work is to determine the relationship between physicochemical properties and arsenic leaching behaviors of ABLFS, and detect the reasons of its high leaching toxicity. Most significantly, the research will put forward reasonable proposals to minimize arsenic leaching concentration and provide information to develop appropriate technologies for the harmless treatment and disposal of this type of sludge.
2 Experimental
2.1 Materials and analysis
Two typical arsenic-bearing lime-ferrate sludges, waste acid residue (WAR) and calcium arsenate residue (CAR), were used as raw materials in this study. Both materials were generated from the process of arsenic-containing wastewater treatment by lime-ferrate process. WAR was supplied by a lead and zinc smelter in Chenzhou city, China and CAR was obtained from an antimony smelter in Huaihua city, China. All the samples were dried at 60 °C for 24 h and sieved below 0.15 mm.
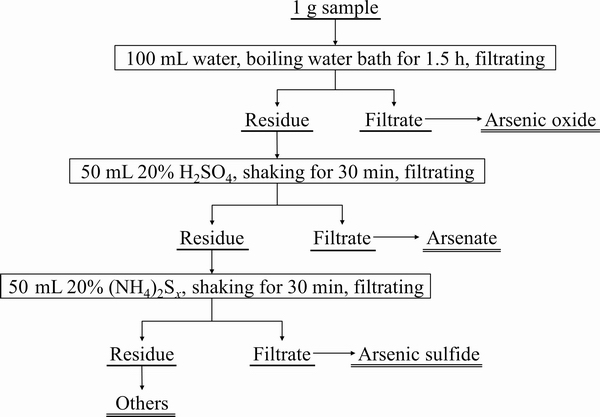
Fig. 1 Methodology used for chemical phase analysis of arsenic-bearing residues
The chemical composition of samples was detected firstly by inductively coupled plasma atomic emission spectrometry (ICP-AES, IRIS Intrepid II XSP), and then the main metal elements were analyzed by chemical analysis based on ICP-AES result. The main phases presented in ABLFS were detected by X-ray powder diffraction (Rigaku, TTR-III). The arsenic phase compositions were quantified by chemical analysis as described by ZHANG [34], and the analytical procedure is shown in Fig. 1. Morphological feature and micro-area chemical analysis of milled samples were observed by scanning electron microscope with energy spectrometer (SEM-EDS, Nova Nano SEM 230). The particle size distribution was determined by laser particle size analyzer (OMEC LS-POP VI). The grain morphology and mineralogical surface composition of WAR and CAR were examined by X-ray photoelectron spectroscopy (XPS, Kratos Ltd., XSAM800).
2.2 Leaching toxicity test
The toxicity characteristic leaching procedure (TCLP USEPA) [35] was used to simulate the leaching of contaminated materials by organic acid. The overall purpose of this analysis was to classify waste according to its ultimate hazard and to predict the long-term behavior of contaminants. The extraction solution was prepared by adding 5.7 mL of glacial acetic acid and distilled water to 1 L. The pH of this stock leaching solution was 2.88±0.05 and it was added at liquid-to- solid ratio of 20:1 (L/kg). The extracting process was conducted under rotary of end-over-end at (30±2) r/min for (18±2) h. Following the extraction, the leachate was filtered through a 0.45 μm glass fiber filter and acidified to pH<2 with nitric acid before being analyzed by ICP-AES.
The synthetic precipitation leaching procedure (SPLP, USEPA) [36] was performed in a similar fashion as the TCLP, but with a different leaching solution. The SPLP leaching fluid was a simulated acid rain, in which a mixture of H2SO4 and HNO3 with a volume ratio of 3:2 diluted to a pH of 4.20±0.05 was used as the extractant.
In the consideration of actual acid rain conditions in China, a Chinese standard sulfuric acid & nitric acid extraction procedure (CN-SNEP, Chinese EPA, HJ/T 299-2007) [37] was conducted in the determination of leaching behavior of solid waste in contrast to TCLP and SPLP. According to CN-SNEP protocol, the extraction solution was prepared by adding 2-3 drops of mixture of concentrated sulfuric acid and nitric acid (mass ratio 2:1) to 1 L distilled water to adjust pH value to 3.20±0.05. The leaching procedure was the same as TCLP except the liquid-to-solid ratio of 10:1. All the experiments were performed at (25±1) °C in triplicate.
2.3 Sequential extraction procedure
Arsenic speciation in WAR and CAR was examined by sequential extraction according to the modified three-step BCR extraction method, which was to evaluate the environmental availability of heavy metals in soils, sediments, mine tailings, sewage sludge and solid residues. Different solvents, extraction liquids and conditions taken during the extraction procedure are shown in Table 1. Each step was carried out in triplicate and the analysis procedures are as follows [38].
Step 1 (acid soluble, bound to carbonate and cation exchange site): 40 mL of HOAc (0.11 mol/L) is added to 1 g of dry sample in a 100 mL polypropylene wide-mouthed bottle. The bottle is shaken for 16 h at room temperature. The extract is separated from solid residue by centrifugation at 3000 r/min for 20 min, then decanted into a polyethylene container and stored at 4 °C for analysis. The residue is washed with 10 mL distilled water by shaking for 60 min, centrifuged and the washings are discarded.
Table 1 Modified three-step BCR sequential extraction procedure
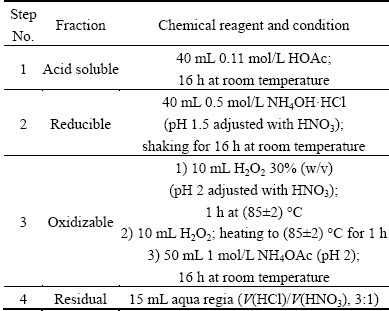
Step 2 (reducible, bound to the Fe-Mn oxides): 40 mL of 0.5 mol/L NH4OH・HCl (pH 1.5 adjusted with HNO3) is added to the residue of step 1 and shaken for 16 h at room temperature, then centrifuged at 3000 r/min for 20 min. The extraction procedure is performed as described above.
Step 3 (oxidizable, bound to organic matter and sulphides): 10 mL H2O2 (8.8 mol/L, pH 2 adjusted with HNO3) is added to the residue of step 2 at room temperature with occasional manual shaking for 1 h. Digestion is continued by heating the tube to 85 °C in a water bath for 2 h. A further 10 mL H2O2 is added and the digestion procedure is repeated. The cool moist residue is then returned to a 100 mL bottle and 50 mL NH4OAc (1 mol/L, pH 2 adjusted with HNO3) is added. The sample is shaken, centrifuged and separated as described in step 1.
Step 4 (remaining residue, bound to mineral matrix): The solid residue in the centrifuge tubes from step 3 is transferred to the beakers and digested with 15 mL of aqua regia solution on a heat plate without boiling. Later, it is filtered and diluted with 0.5 mol/L HNO3 to 25 mL.
3 Results and discussion
3.1 Physicochemical properties
3.1.1 Chemical compositions of ABLFS
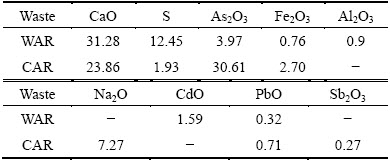
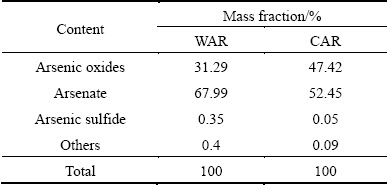
The elemental compositions of WAR and CAR samples analyzed by ICP-AES and chemical analysis are given in Tables 2 and 3, respectively. The results show that As and Ca are major elements in WAR and CAR. In addition, there are high contents of S and Cd in WAR and a certain amount of Na and Fe in CAR. Consequently, WAR is mainly composed of 31.28% CaO, 12.45% S, 3.97% As2O3 and 1.59% CdO. CAR is also enriched in 23.86% CaO, which is from lime cream used for adjusting pH and precipitation of heavy metals. A large quantity of As (equivalent to 30.61% As2O3) was precipitated and stored into CAR. Besides, 7.27% Na2O and 2.70% Fe2O3 are also included in CAR. The chemical compositions of WAR and CAR are mainly dependent on content of wastewater and the treatment process. Generally, the main heavy metals and metalloids in acidic wastewater are As and Cd while the elements in high-arsenic wastewater are As, Na, and Fe, which are precipitated by lime cream and ferric salt and then enter into ABLFS.
Table 2 Chemical compositions of WAR and CAR obtained by ICP-AES (mass fraction, %)
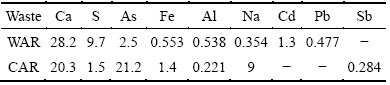
Table 3 Chemical compositions of WAR and CAR obtained by chemical analysis (mass fraction, %)
3.1.2 Phase and arsenic composition
Figure 2 shows the XRD patterns of ABLFS. The XRD pattern of WAR exhibits sharp and strong diffraction peaks, confirming its crystalline nature. The major constituent of WAR is calcium sulfate, with the dominant species of gypsum (CaSO4・2H2O) and hemihydrate gypsum (CaSO4・0.5H2O) revealed by XRD. A significant fraction of crystal water presents in the crystallites during the generating processes. However, there is no obvious absorption peak of arsenic phases in XRD pattern. The XRD pattern of CAR shows low diffraction intensity with a few peaks of CaCO3, Ca3(AsO4)2 and Na2SO4. This feedback indicates that the constituents in CAR are primarily in an amorphous phase, specifically for arsenic. The formation of amorphous phase is reasonable based on the principle of lime- ferrate process.
Since XRD pattern cannot determine the amorphous arsenic phases, chemical phase analysis was carried out to ascertain arsenic composition of WAR and CAR and the results are given in Table 4. It is seen from Table 4 that the arsenic mainly presents in the phases of arsenate and arsenic oxides, which occupy more than 99% of arsenic compounds in both WAR and CAR. According to the chemical compositions and its production process, the arsenate is calcium arsenate in WAR, and calcium arsenate, sodium arsenate and ferric arsenate in CAR. The arsenic oxides in WAR and CAR are arsenic trioxide or diarsenic pentoxide.
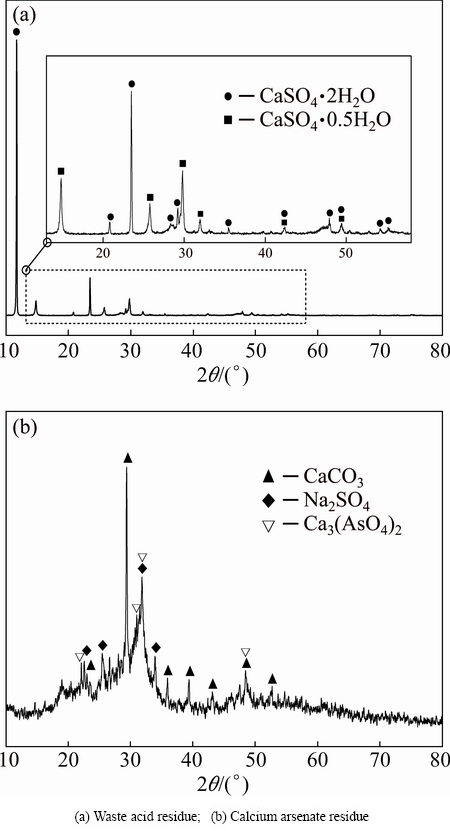
Fig. 2 XRD patterns of WAR and CAR
Table 4 Arsenic contents in WAR and CAR obtained by chemical analysis
3.1.3 Morphological features
The morphological feature plays an important role in treatment of solid waste. The micrographs and particle size distribution of WAR and CAR were detected by SEM-EDS and laser particle size analyzer. Based on these microphotographs as shown in Fig. 3, the general particles of WAR in flakiness and rod shape with clear edges and smooth surfaces are obtained. It is also clearly seen that some amorphous tiny particles are adsorbed on the surface of crystalline substance. The largest particle is 30-40 μm in length and 2-5 μm in depth. The EDS result indicates that the main micro-area elements of WAR are Ca and S, which is in agreement with ICP-AES result. Combined with the XRD and phase composition, the flakiness and rod crystal structure are calcium sulfate while arsenic is mainly presented in the amorphous tiny particles. Figure 4 shows that most particles of WAR are smaller than 20 μm and the median particle size (d50) is 6.03 μm. The SEM image of CAR reveals the general particles in spherical and irregular bulk shapes. The largest particle is about 50 μm and the smallest one is below 5 μm. The agglomerated morphology is predominantly with many tiny independent particles covered on the surface of larger spherical particles. The micro-area elements as shown in EDS are Ca and As. Larger spherical particles are crystal calcium arsenate and the agglomerated tiny particles are amorphous calcium arsenate and arsenic oxide. As seen from Fig. 4, most of these particles are smaller than 50 μm and the median particle size is 18.30 μm. The relatively large particle sizes are caused primarily due to the coprecipitation and coagulation by Fe(OH)3 colloid during the lime-ferrate process. Based on above analysis, the change of morphology and particle sizes is beneficial to reducing the leaching rate of heavy metals.
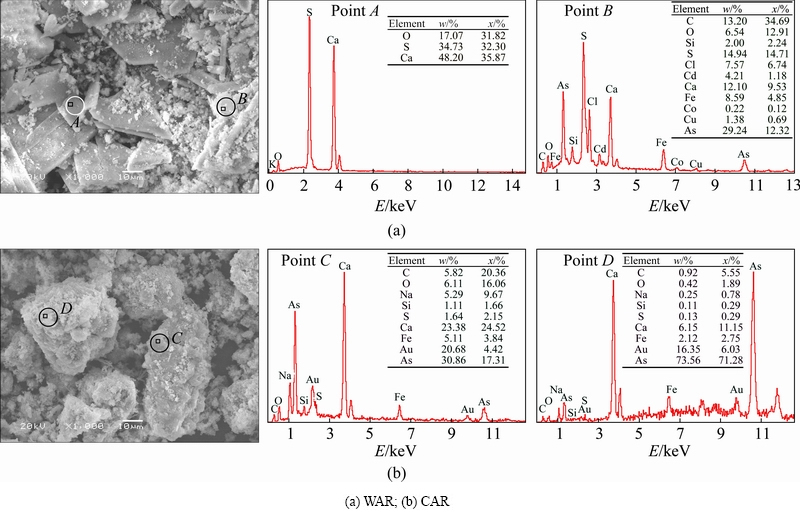
Fig. 3 SEM-EDS images of ABLFS

Fig. 4 Particle size distribution of WAR and CAR
3.1.4 Grain morphology and mineralogical surface composition
X-ray photoelectron spectroscopy (XPS) is a versatile surface analysis technique to detect the elemental composition and chemical oxidation states of surface and near-surface species [39]. It is very important to investigate the surface performance of WAR and CAR as most solid waste treatments are initially controlled by surface chemical reactions. XPS analyses were conducted to elemental composition and oxidation states on the surface of WAR and CAR, and the wide scan spectra are displayed in Fig. 5. Seven clear peaks at binding energies of 1326.63, 685.28, 532,15, 405.71, 348.36, 285.04 and 169.46 eV designated for the As 2p3, F 1s, O 1s, Cd 3d, Ca 2p, C 1s and S 2p, respectively, are observed in WAR (Fig. 5(a)), and six clear peaks 1326.07, 1071.21, 711.22, 530.77, 346.82 and 284.83 eV designated for the As 2p3, Na 1s, Fe 2p3, O 1s, Ca 2p, and C 1s in CAR, respectively (Fig. 5(b)). Table 5 shows the mole fractions measured in the XPS analyses of WAR and CAR. The most intense As XPS peak is As 2p3 line at 1326.63 and 1326.07 eV, which are used to quantitate the mole fractions of As being 17.38% and 37.84% in WAR and CAR, respectively.
As reported, the binding energy of As 3d indicates oxidation state, a value of 44 eV for As(III) and 45 eV for As(V), although differentiation challenges the line width and presence of multiple species on the surface [40]. In this study, as shown in Fig. 6(a), the results analyzed by XPSPEAK41 indicates that the As 3d spectrum of WAR is fitted with two components with binding energies of 45.02 and 44.14 eV designated for As(V) and As(III), respectively. The mole fractions show that 78.81% As(V) and 21.19% As(III) are distributed on the surface and near-surface of WAR. The As 3d spectrum of CAR is disassembled into two different individual component peaks, which are originated from the arsenic atom of different valence states and overlapped with each other (Fig. 6(b)). Quantitative analysis of arsenic on the surface and near-surface of CAR shows that 67.59% As(V) at binding energy of 44.82 eV and 32.41% of As(III) at binding energy of 44.16 eV. Moreover, these results combined with chemical phase analysis also indicate that the primary element chemical states As(V) in both WAR and CAR are arsenate and As(III) are mainly in the form of arsenic trioxide.
3.2 Leaching behaviors
3.2.1 Leaching toxicity analysis
The purpose of this analysis is to classify waste as to its ultimate hazard. The TCLP, SPLP and CN-SNEP tests were applied to investigating the leaching toxicity of ABLFS and the results are shown in Table 6. In TCLP test, the As leachate concentrations of WAR and CAR are 595.75 and 5315 mg/L, 119 and 1063 times higher than the standard level (5 mg/L for As). The leaching rates of arsenic are 47.66% and 50.15%, respectively. In addition, Cd leaching toxicity of WAR is 540.25 mg/L with the leaching rate of 83%, being 540 times higher than the standard level (1 mg/L for Cd). Sb leaching concentration of CAR is 3.55 mg/L, which excesses the regulatory level of 0.6 mg/L (The regulatory level is 100 times the level of the National Primary Drinking Water Standards established by the US EPA), even though the leaching rate is only 2.5%. Beyond that, other heavy metals in leachate are considerably lower than regulatory level. In summary, both wastes were classified as hazardous waste, specifically because As exceeded a thousand times of the regulatory level.

Fig. 5 XPS wide scan spectra of WAR (a) and CAR (b)
Table 5 Mole fractions measured in XPS analysis of WAR and CAR


Fig. 6 XPS spectra of As 3d for WAR (a) and CAR (b)
Table 6 Concentrations of heavy metals extracted by TCLP, SPLP and CN-SNEP (mg/L)
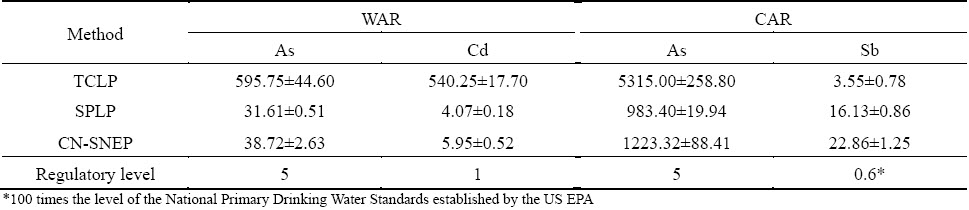
In some cases, the extraction of a specific heavy metal by different leaching methods is significant. For example, As and Cd concentrations in WAR are 595.75 and 540.25 mg/L by TCLP, but only 31.61 and 4.07 mg/L by SPLP. This is most likely due to the higher acidity (pH=2.88) used in the TCLP than that in the SPLP (pH=4.20). However, Sb concentration in CAR is 3.55 mg/L by TCLP, less than 16.13 mg/L by SPLP. The results of CN-SNEP are only a little higher than the consequences of SPLP. Both methods are appropriate for the wastes generated in mineral processing because they used a leachant that simulated the composition of acid rain [41]. According to the actual situation of acid rain in China, the pH of CN-SNEP leachant (pH=3.20) is set to be lower than that of SPLP.
This comparison indicated that the extractant pH was not the unique factor influencing the potential leachability of heavy metal in the residues. The leachability was determined by a variety of other factors, such as the composition and stability of the mineral species in the wastes.
3.2.2 BCR sequential extraction
Sequential extraction was performed to evaluate the environmental risk of ABLFS and to predict the long-term behavior of the main contaminants. The absolute concentrations are shown in Table 7. The amount of acid soluble fractions (F1) from BCR is roughly equivalent to the sum of exchangeable and carbonate phase of metals from the residue [42]. The carbonate form is a loosely bound phase and liable to change under environmental conditions, so this phase is susceptible to changing along with pH value of mild acid. It is widely believed that the acid soluble form of heavy metals is the direct phase that pollutes the environment [43].
Table 7 Contents of heavy metals in WAR and CAR extracted by BCR procedure (mg/kg)
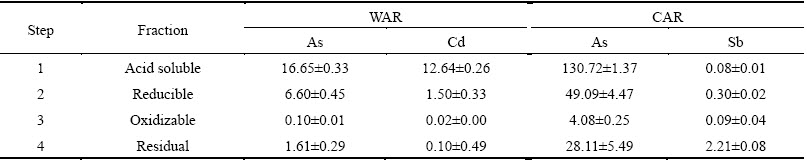
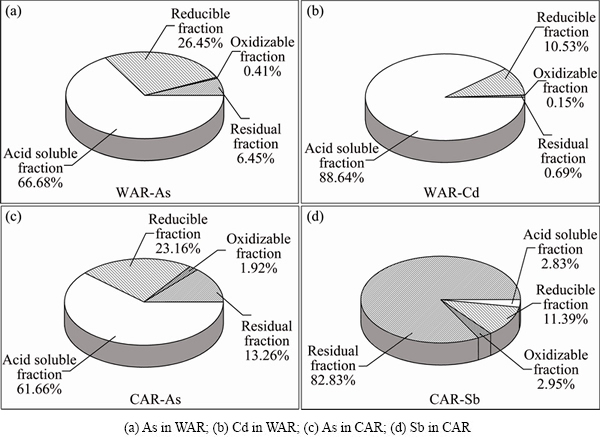
Fig. 7 Ratios of heavy metals fraction in ABLFS
The relative percentages of heavy metals extracted in different steps of BCR test are presented in Fig. 7. The relative contents of As in WAR and CAR were obtained in decreasing order: acid soluble > reducible > residual > oxidizable (Figs. 7(a) and (c)). Both sludges have a significant high content of As existing in acid soluble fraction (66.68% for WAR and 61.66% for CAR) so that As is easily released to groundwater in weak acidic environment under acid rain or regional acid deposition in China. Besides, a considerable amount of Cd in WAR (88.64% in acid soluble form) presents a significant environmental risk due to its high direct eco-toxicity and bioavailability. The amount of reducible fractions (F2) is roughly equivalent to the content of Fe-Mn phase in the sludge. The contents of reducible form of As in WAR and CAR are 26.45% and 23.16%, respectively. These two fractions (F1 and F2) are classified as direct effect phases. It is revealed that the direct effect phases of As in WAR and CAR are 23253 and 179818 mg/kg, respectively. The results show that both wastes have high environmental activity.
The amount of oxidizable fractions (F3) from BCR is roughly equivalent to the sum of organic matter and sulfide combination state of heavy metals. Under oxidizing conditions, metals bound to organic matter or sulfur are also liberated or transformed into F1 and F2. Thus, F3 is identified as a potential effect fraction. The results show F3 in WAR or CAR accounts for only a small proportion (0.15%-2.95%). The residual fraction (F4) is recognized as a stable fraction because it contains primary and secondary minerals hold metals within their crystal structures [38]. Sb in CAR exists in residual form (82.83%), which indicates that Sb is transformed into a more stable form in the production process of wastewater treatment. The contents of residual As in WAR and CAR are 6.45% and 13.26% respectively (Figs. 7(a) and (c)). This result shows that a part of As retained within the crystal lattices of minerals in crystallized oxides during the lime-ferrate process treatment of arsenic containing wastewater. This kind of metal would steadily exist in the sludge for a long period of time [44]. Both wastes show high environmental activity because of higher direct acid soluble fractions of As and cause great harm to surrounding environment.
3.3 Implications for appropriate disposal
According to the researches above, it is clearly seen that the leaching behavior of arsenic in ABLFS is mainly related to its properties and phase composition determined in the lime-ferrate treatment process of arsenic-containing wastewater. Arsenic mainly in the presence of amorphous arsenate and arsenic oxides phase, occupies 52.45%-67.99% and 31.29%-47.42%, respectively. The extracted species of amorphous arsenate are in the form of acid soluble and reducible. The percentage of these two forms occupies about 90%, whereas remained residue form just takes a percentage of 6.45%-13.26%. This is the main cause of high arsenic leaching toxicity and environmental risk of ABLFS.
Developing effective harmless disposal of ABLFS is an urgent requirement. Based on a lot of calcium and alkali added in the lime-ferrate process, arsenic could be immobilized in ABLFS by transforming into a stable calcium arsenate compound. Since As(V) is more stable than As (III) in high alkaline conditions, trivalent arsenic should be oxidized to pentavalent arsenic firstly. However, the process of trivalent arsenic oxidized to pentavalent arsenic in the natural environment is very slow. Many advanced oxidizing agents such as KMnO4-Fe(II) are reported to convert As(III) to As(V) [45]. When only As(V) is present, considerations should be controlled to transform amorphous arsenate to well-crystalline arsenic mineral structure. In the system of Ca-As-H2O, various calcium arsenate compounds are synthesized at various Ca/As molar ratio and pH [46]. Ca4(OH)2(AsO4)2・4H2O and Ca5(AsO4)3OH are found to be well crystallized and stable with low solubility and leachability under the conditions of high alkalinity [47,48]. Thus, immobilization of ABLFS by in-situ conversion of As into stable Ca4(OH)2-(AsO4)2・4H2O and Ca5(AsO4)3OH crystals is promoted to low arsenic leaching toxicity and environmental risk.
4 Conclusions
1) A large quantity of Ca is incorporated into ABLFS in species of calcium sulfate or calcium arsenate. The heavy metals presented in CAR are primarily composed of As, Cd and Sb, which are determined by the composition of wastewater.
2) Arsenic phases are mainly in arsenate and arsenic oxides dispersed uniformly in amorphous particles. Pentavalent arsenic is mainly in the form of calcium arsenate, while trivalent arsenic is in arsenic trioxide. The amorphous structure determines its instability.
3) The arsenic leachate concentrations of WAR and CAR are 119 and 1063 times higher than the TCLP standard level, while other toxic metals such as Cd and Sb show extraordinarily hazardous too, which indicate that ABLFS is classified as hazardous waste. The leaching behavior of arsenic in ABLFS is related to its properties and phase composition. Amorphous arsenate and arsenic oxides along with their acid soluble and reducible extraction species are the reasons of high arsenic leaching toxicity and environmental risk. The necessary work and problems to make ABLFS harmless are to adjust and control the crystal structure and occurrence state of heavy metals.
4) The development of advanced oxidizing agents to convert As(III) to As(V) species and immobilization of ABLFS by in-situ conversion of As into stable Ca4(OH)2(AsO4)2・4H2O and Ca5(AsO4)3OH crystals are promoted to achieve low arsenic leaching toxicity and environmental risk.
References
[1] ZHANG Q L, GAO N Y, LIN Y C, XU B, LE L S. Removal of arsenic(V) from aqueous solutions using iron-oxide-coated modified activated carbon [J]. Water Environment Research, 2007, 79(8): 931-936.
[2] PENG Liang, CHEN Ya-ping, DONG Hong, ZENG Qing-ru, SONG Hui-juan, CHAI Li-yuan, GU Ji-dong. Removal of trace As(V) from water with the titanium dioxide/ACF composite electrode [J]. Water Air and Soil Pollution,2015, 226(7):203-230.
[3] ZENG Fan-fu, WEI Wei, LI Man-sha, HUANG Rui-xue, YANG Fei, DUAN Yan-ying. Heavy metal contamination in rice-producing soils of Hunan province, China and potential health risks [J]. International Journal of Environmental Research and Public Health, 2015,12(12): 15584-15593.
[4] WU C, ZOU Q, XUE S G, PAN W S, HUANG L, HARTLEY W, MO J Y, WONG M H. The effect of silicon on iron plaque formation and arsenic accumulation in rice genotypes with different radial oxygen loss (ROL) [J]. Environmental Pollution, 2016, 212: 27-33.
[5] WU Chuan, LI Hui, YE Zhi-hong, WU Fu-yong, WONG M H. Effects of As levels on radial oxygen loss and As speciation in rice [J]. Environmental Science and Pollution Research, 2013, 20: 8334-8341.
[6] PAN Wei-song, WU Chuan, XUE Sheng-guo, HARTLEY W. Arsenic dynamics in the rhizosphere and its sequestration on rice roots as affected by root oxidation [J]. Journal of Environmental Sciences, 2014, 26: 892-899.
[7] LI Y C, MIN X B, CHAI L Y, SHI M Q, TANG C J, WANG Q W, LIANG Y J, LEI J, LIYANG W J. Co-treatment of gypsum sludge and Pb/Zn smelting slag for the solidification of sludge containing arsenic and heavy metals [J]. Journal of Environmental Management, 2016, 181: 756-761.
[8] WANG Zhen-xing, CHAI Li-yuan, WANG Yun-yan, YANG Zhi-hui, WANG Hai-ying, WU Xie. Potential health risk of arsenic and cadmium in groundwater near Xiangjiang River, China: A case study for risk assessment and management of toxic substances [J]. Environmental Monitoring and Assessment, 2011, 175: 167-173.
[9] CHAI L Y, MUBARAK H, YAN Z H, YONG W, TANG C J, MIRZA N. Growth, photosynthesis, and defense mechanism of antimony (Sb)-contaminated Boehmeria nivea L [J]. Environmental Science and Pollution Research, 2016, 23(8): 7470-7481.
[10] YANG Sheng-xiang, LIAO Bin, YANG Zhi-hui, CHAI Li-yuan, LI Jin-tian. Revegetation of extremely acid mine soils based on aided phytostabilization: A case study from southern China [J]. The Science of the Total Environment, 2016, 562: 427-434.
[11] WANG Y, CHAI L Y, YANG Z H, MUBARAK H, TANG C J. Chlorophyll fluorescencein leaves of Ficus tikoua under arsenic stress [J]. Bulletin of Environmental Contamination and Toxicology, 2016, 97(4): 576-581.
[12] WU Chuan, ZOU Qi, XUE Sheng-guo, MO Jing-yu, PAN Wei-song, LOU Lai-qing, WONG Ming-hung. Effects of silicon (Si) on arsenic (As) accumulation and speciation in rice (Oryza sativa L.) genotypes with different radial oxygen loss (ROL) [J]. Chemosphere, 2015, 138: 447-453.
[13] YANG Zhi-hui, LIU Lin, CHAI Li-yuan, LIAO Ying-ping, YAO Wen-bin, XIAO Rui-yang. Arsenic immobilization in the contaminated soil using poorly crystalline Fe-oxyhydroxy sulfate [J]. Environmental Science and Pollution Research, 2015, 22: 12624-12632.
[14] WANG Zhen-xing, CHAI Li-yuan, YANG Zhi-hui, WANG Yun-yan, WANG Hai-ying. Identifying sources and assessing potential risk of heavy metals in soils from direct exposure to children in a mine-impacted city, Changsha, China [J]. Journal of Environmental Quality, 2010, 39: 1616-1623.
[15] CHAI Li-yuan, CHEN Yun-nen, YANG Zhi-hui. Kinetics and thermodynamics of arsenate and arsenite biosorption by pretreated spent grains [J]. Water Environment Research, 2009, 81(9): 843-848.
[16] CAO Hua-zhen, ZHONG Yang, WU Lian-kui, ZHANG Yu-feng, ZHENG Guo-qu. Electrodeposition of As-Sb alloy from high arsenic-containing solutions [J]. Transactions of Nonferrous Metals Society of China, 2016, 26(1): 310-318.
[17] ZHU Jian-yu, ZHANG Jing-xia, LI Qian, HAN Tao, HU Yue-hua, LIU Xue-duan, QIN Wen-qing, CHAI Li-yuan, QIU Guan-zhou. Bioleaching of heavy metals from contaminated alkaline sediment by auto- and heterotrophic bacteria in stirred tank reactor [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(9): 2969-2975.
[18] XU Su-juan, ZHENG Na, LIU Jing-shuang, WANG Yang, CHANG Shou-zhi. Geochemistry and health risk assessment of arsenic exposure to street dust in the zinc smelting district, Northeast China [J]. Environmental Geochemistry and Health, 2013, 35(1): 89-99.
[19] LIANG Y J, MIN X B, GHAI L Y, WANG M, LI Y W J, PAN Q L, OKIDO M. Stabilization of arsenic sludge with mechanochemically modified zero valent iron [J]. Chemosphere, 2017, 168: 1142-1151.
[20] YU Guo-lin, ZHANG Ying, ZHENG Shi-li, ZOU Xing, WANG Xiao-hui, ZHANG Yi. Extraction of arsenic from arsenic-containing cobalt and nickel slag and preparation of arsenic-bearing compounds [J]. Transactions of Nonferrous Metals Society of China, 2014, 24(6): 1918-1927.
[21] LI Mi, PENG Bing, CHAI Li-yuan, PENG Ning, YAN Huan, HOU Dong-ke. Recovery of iron from zinc leaching residue by selective reduction roasting with carbon [J]. Journal of Hazardous Materials, 2012, 237: 323-330.
[22] MIN Xiao-bo, LIAO Ying-ping, CHAI Li-yuan, YANG Zhi-hui, XIONG Shan, LIU Lin, LI Qing-zhu. Removal and stabilization of arsenic from anode slime by forming crystal scorodite [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(4): 1298-1306.
[23] KE Yong, CHAI Li-yuan, MIN Xiao-bo, TANG Chong-jian, ZHOU Bo-sheng, CHEN Jie, YUAN Cui-yu. Behavior and effect of calcium during hydrothermal sulfidation and flotation of zinc-calcium-based neutralization sludge [J]. Minerals Engineering, 2015, 74: 68-78.
[24] LI Mi, PENG Bing, CHAI Li-yuan, PENG Ning, XIE Xian-de, YAN Huan. Technological mineralogy and environmental activity of zinc leaching residue from zinc hydrometallurgical process [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(5): 1480-1488.
[25] MIN Xiao-bo, XIE Xian-de, CHAI Li-yuan, LIANG Yan-jie, LI Mi, KE Yong. Environmental availability and ecological risk assessment of heavy metals in zinc leaching residue [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(1): 208-218.
[26] CHAI Li-yuan, SHI Mei-qing, LIANG Yan-jie, TANG Jing-wen, LI Qing-zhu. Behavior, distribution and environmental influence of arsenic in a typical lead smelter [J]. Journal of Central South University, 2015, 22(4): 1276-1286.
[27] PHENRAT T, MARHABA T F, RACHAKORNKIJ M. Leaching behaviors of arsenic from arsenic-iron hydroxide sludge during TCLP [J]. Journal of Environmental Engineering, 2008, 134(8): 671-682.
[28] SHAW J K, FATHORDOOBADI S, ZELINSKI B J, ELA W P, SAEZ A E. Stabilization of arsenic-bearing solid residuals in polymeric matrices [J]. Journal of Hazardous Materials, 2008, 152(3): 1115-1121.
[29] GHOSH A, MUKIIBI M, ELA W. TCLP underestimates leaching of arsenic from solid residuals under landfill conditions [J]. Environmental Science & Technology, 2004, 38(17): 4677-4682.
[30] KARCZEWSKA A, GERSZTYN L, GALKA B, JUSZCZYSZYN M, KANTEK K. Effects of sewage sludge application on arsenic species in polluted soils [J]. Fresenius Environmental Bulletin, 2013, 22(4): 962-967.
[31] DUNDAR M S, ALTUNDAG H, EYUPOGLU V, KESKIN C S, TUTUNOGLU C. Determination of heavy metals in lower Sakarya river sediments using a BCR-sequential extraction procedure [J]. Environmental Monitoring and Assessment, 2012, 184(1): 33-41.
[32] CHAI Li-yuan, WU Jian-xun, WU Yan-jing, TANG Chao-bo, YANG Wei-chun. Environmental risk assessment on slag and iron-rich matte produced from reducing-matting smelting of lead-bearing wastes and iron-rich wastes [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(10): 3429-3435.
[33] POYKIO R, MAKELA M, WATKINS G, NURMESNIEMI H, DAHL O. Heavy metals leaching in bottom ash and fly ash fractions from industrial-scale BFB-boiler for environmental risks assessment [J]. Transactions of Nonferrous Metals Society of China, 2016, 26(1): 256-264.
[34] ZHANG Hui-bin. Chemical phase analysis of ore and industrial product [M]. Beijing: Metallurgical Industry Press, 1992. (in Chinese)
[35] SW-846, 1311. USEPA. Test method for the evaluation of solid waste, physical/ chemical methods. Method 1311, toxicity characteristic leaching procedure [S]. Washington DC, 1992.
[36] SW-846, 1312. USEPA. Test method for the evaluation of solid waste, physical/ chemical methods. Method 1312, synthetic precipitate leaching procedure [S]. Washington DC, 1994.
[37] HJ/T299-2007. Chinese EPA. Solid waste-extraction procedure for leaching toxicity―Sulphuric acid & nitric acid method [S]. Beijing, China, 2007. (in Chinese)
[38] XIE Xian-de, MIN Xiao-bo, CHAI Li-yuan, TANG Chong-jian, LIANG Yan-jie, LI Mi, KE Yong, CHEN Jie, WANG Yan. Quantitative evaluation of environmental risks of flotation tailings from hydrothermal sulfidation-flotation process [J]. Environmental Science and Pollution Research, 2013, 20(9): 6050-6058.
[39] FAN Jian-xin, WANG Yu-jun, LIU Cun, WANG Li-hua, YANG Ke, ZHOU Dong-mei, LI Wei, SPARKS D L. Effect of iron oxide reductive dissolution on the transformation and immobilization of arsenic in soils: New insights from X-ray photoelectron and X-ray absorption spectroscopy [J]. Journal of Hazardous Materials, 2014, 279: 212-219.
[40] GROENEWOLD G S, AVCI R, FOX R V, DELIORMAN M, SUO Z Y, KELLERMAN L. Characterization of arsenic contamination on rust from ton containers [J]. Industrial & Engineering Chemistry Research, 2013, 52(4): 1396-1404.
[41] CAO X, DERMATAS D. Evaluating the applicability of regulatory leaching tests for assessing lead leachability in contaminated shooting range soils [J]. Environmental Monitoring and Assessment, 2008, 139(1-3): 1-13
[42] MUKHERJEE D, MUKHERJEE A, KUMAR B. Chemical fractionation of metals in freshly deposited marine estuarine sediments of sundarban ecosystem, India [J]. Environmental Geology, 2009, 58(8): 1757-1767.
[43] RIEUWERTS J S, FARAGO M E, CIKRT M, BENCKO V. Differences in lead bioavailability between a smelting and a mining area [J]. Water Air and Soil Pollution, 2000, 122(1-2): 203-229.
[44] SINGH S, TACK F, VERLOO M. Heavy metal fraction and extractability in dredged sediment derived [J]. Water Air and Soil Pollution, 1996, 87: 313-328.
[45] GUAN Xiao-hong, MA Jun, DONG Hao-ran, JIANG Li. Removal of arsenic from water: Effect of calcium ions on As(III) removal in the KMnO4-Fe(II) process [J]. Water Research, 2009, 43: 5119-5128.
[46] BOTHE J V, BROWN P W. Arsenic immobilization by calcium arsenate formation [J]. Environmental Science & Technology, 1999, 33: 3806-3811.
[47] ZHU Y N, ZHANG X H, XIE Q L, WANG D Q, CHENG G W. Solubility and stability of calcium arsenates at 25 °C [J]. Water Air and Soil Pollution, 2006, 169: 221-238.
[48] LEI J, PENG B, MIN X B, LIANG Y J, YOU Y, CHAI L Y. Modeling and optimization of lime-based stabilization in high alkaline arsenic-bearing sludges with a central composite design [J]. Journal of Environmental Science and Health: Part A, 2017, 52(5): 449-458.
彭 兵1,2,雷 杰1,闵小波1,2,柴立元1,2,梁彦杰1,2,游 洋1
1. 中南大学 冶金与环境学院,长沙 410083;
2. 中南大学 国家重金属污染防治工程技术研究中心,长沙 410083
摘 要:以典型石灰铁盐法处理二种含砷废水产生的污泥(污酸渣和砷酸钙渣)为研究对象,采用ICP-AES、SEM-EDS、XRD、XPS和化学物相分析等检测手段对其化学组成、形貌特征、物相结构及砷的赋存状态进行研究,采用毒性浸出实验和BCR三步连续提取法考察污泥中砷的浸出行为。研究结果表明,污酸渣和砷酸钙渣中砷的含量分别为2.5%和21.2%,主要组成物相为砷酸盐及砷氧化物,均以无定型的颗粒均匀分散或团聚在污泥中。砷的浸出毒性超出TCLP标准规定限值的119和1063倍,浸出率分别为47.66%和 50.15%。砷以酸可提取态和可还原态为主,两者共占90%左右,而稳定的残渣态含量相对较低,这是含砷石灰铁盐渣浸出毒性大、环境活性高的直接原因。本研究为含砷石灰铁盐渣无害化处理技术提供了大量有用的基本数据。
关键词:含砷固废;石灰铁盐渣;理化特性;浸出行为;BCR连续提取法
(Edited by Wei-ping CHEN)
Foundation item: Project (201509050) supported by Special Program on Environmental Protection for Public Welfare, China; Projects (51474247, 51634010) supported by the National Natural Science Foundation of China; Project (2015CX001) supported by Grants from the Project of Innovation-driven Plan in Central South University, China
Corresponding author: Xiao-bo MIN; Tel: +86-731-88830577; E-mail: mxbcsu@163.com
DOI: 10.1016/S1003-6326(17)60140-7

