J. Cent. South Univ. Technol. (2008) 15: 64-68
DOI: 10.1007/s11771-008-0014-y
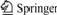
Flame retardancy and its mechanism of polymers flame retarded by DBDPE/Sb2O3
ZUO Jian-dong(左建东)1, LI Rong-xun(李荣勋)2, FENG Shao-hua(冯绍华)2,
LIU Guang-ye(刘光烨)2, ZHAO Jian-qing(赵建青)1
(1. College of Materials Science and Engineering, South China University of Technology, Guangzhou 510640, China;
2. Key Laboratory of New Materials of Qingdao, Qingdao University of Science and Technology,
Qingdao 266042, China)
Abstract: The flammability characterization and thermal composition of polymers flame retarded by decabromodiphenylethane (DBDPE) and antimony trioxide (Sb2O3) were studied by cone calorimeter and thermogravimetry (TG). The results show that ABS/DBDPE/Sb2O3 has the similar flammability parameters and thermal composition curves to ABS/DBDPO/Sb2O3. It suggests that DBDPE/Sb2O3 has the similar flame retardant behavior to DBDPO/Sb2O3. The heat release rate (HRR) and the effect heat combustion (EHC) curves of polymers flame retarded by DBDPE/Sb2O3 all decrease, but the mass loss rate (MLR) curve slightly increase. It shows that the decrease of HRR is not due to the increase of char formation ratio but the generation of incombustible gases. The major flame retardant mechanism of DBDPE/Sb2O3 is gas phase flame retardant mechanism. Increasing content of Sb2O3 in DBDPE/Sb2O3 can improve the flame retardant property and thermal stability of acrylonitrile butadiene styrene. Sb2O3 has a good synergistic effect with DBDPE.
Key words: decabromodiphenylethan (DBDPE); thermogravimetry; gas phase flame retarding
1 Introduction
It is well known that brominated flame retardants can effectively improve the fire retardant property of many polymer materials. Decabromodiphenyl oxide (DBDPO) has high bromine amount and excellent thermal stability, and its thermal decomposition temperature is close to that of many polymers. Unfortunately, it has been pointed out that DBDPO can release polybrominated dibenzo-p-dioxine (PBDD) and polybrominated dibenzofuranes (PBDF) that are toxic and carcinogenic gases. Therefore it has been forbidden to use by some countries recently[1]. Decabrom- bromodiphenylethane (DBDPE 8010) is also called 1, 2 bis (pentabromophenyl) ethane (SAYTEX 8010) developed by American Albemarle Corporation. It is equivalent with DBDPO in the molecular mass, thermal stability and bromine amount. International organization has evaluated the human health and environmental risk for DBDPE[2-3]. It cannot release PBDD and PBDF during combustion[4], and its light stability and anti-dialysis are better than DBDPO. Especially, polymers flame retarded by DBDPE can be recycled without great loss in the flame retarding. In China, bromine-containing flame retardants as the important flame retardants are widely used and DBDPE will be the most promising substitute of DBDPO.
Studies mainly focus on the photochemical reaction onto the hydrated surfaces[5], toxicity on the environment and health[6-7] and degradation productions on thermogravimetric analysis (TGA) and pyrolysis-gas chromatography-mass spectra (Py-GC-MS)[8], and its basic flame retardant behavior and mechanism have seldom been reported. It is necessary to study whether DBDPE has the same flame retardant behavior and mechanism with DBDPO. In this work, the flammability parameters of some polymers flame retarded by DBDPE/Sb2O3 were measured in the cone calorimeter, such as heat release rate (HRR), effect heat combustion (EHC) and mass loss rate (MLR). The effects of DBDPE/Sb2O3 content and Sb2O3 content on the thermal composition of several polymers were discussed by TGA, and the basic flame retarding mechanism of DBDPE/Sb2O3 was deduced.
2 Experimental
The polymer materials used in this work were acrylonitrile butadiene styrene(ABS) (L815, MFI=18 g/10 min, 220 ℃, 10 kg, Ningbo Lejin Yongxing Chemical Co., Ltd), LDPE (DNDA-7145, MFI=12 g/10 min, China Petrochemical Qilu Co. Ltd ), HIPS (466F, MFI=4 g/10 min, Yangzi-BASF Styrenics Co. Ltd), polypropylene (EPF-302R, MFI=13 g/10 min, China Petrochemical Qilu Co. Ltd). DBDPE was supplied by Weifang Haitian Yanhua Company, Sb2O3 (particle size: 4-6 μm) was manufactured by Shanghai Shiji Fourth Company.
DBDPE and Sb2O3 were mixed with different polymers at a certain proportion(Table 1) to granulate in a two-screw extruder. And the grains were dried in the vacuum drying oven. The grains were compression-molded into sheets of 4 mm by a hot press at 175 ℃ and 20 MPa for 8 min. These sheets were the samples used for the cone calorimeter tests.
The flammability parameters were obtained by the cone calorimeter made by England FTT Company. The samples were put on a horizontal sample holder in the cone calorimeter and then fired at a radiant flux of 50 kW/m2 according to ISO 5660-1[9]. The limit oxygen index (LOI) test had been carried out according to ASTM2863-77. Samples (120 mm×6 mm, 3 mm thick) were held vertically in an oxygen index measurement system.
Thermogravimetry tests were carried out on a Perkin-Elmer TG thermal system TGS2 in nitrogen atmosphere under a gas flow of 50 cm3/min at a heating rate of 20 ℃/min from 50 to 700 ℃.
Table 1 Formulas of flame retarded polymers

3 Results and discussion
3.1 Flame retardancy of flame retarded polymers
Cone calorimetric technique has been used in the fire test and researching[10-11]. In the experiment of cone calorimeter, heat release rate(HRR) was considered by HIRSCHLER[12] to be the most important parameter for evaluating fire behavior of materials. Usually, the peak value of HRR (pkHRR) is used to determine the intensity of a fire[13]. In Fig.1, the curves of ABS flame retarded by DBDPE/Sb2O3 and DBDPO/Sb2O3 almost coincide, and their pkHRR decrease by about 57% and 68%, respectively, in comparing with that of the virgin one. It shows that DBDPE/Sb2O3 has the same flammability as DBDPO/Sb2O3 and they have the same effect on the exothermic reaction processes of ABS. The effect heat combustion (EHC) reflects the burning degree of the combustible and volatile gas in the gas phase. The average EHC values of ABS with two flame retardant formulations decrease by about 63% than that of pure ABS, and their curves also overlap. With the same content of flame retardant, ABS-1 can give out the similar amount of combustible gases as ABS-2. DBDPE/Sb2O3 and DBDPO/Sb2O3 have the same effect on the chain decomposition reaction of ABS.

Fig.1 HRR(a) and EHC(b) curves of ABS and flame retarded ABS
Fig.2 displays the HRR curves of PS and PS-1. The average pkHRR and EHC values of PS-1 are much lower than those of pure PS. The same things happen on PP and PP-1 in Fig.3. It can also be found that DBDPE/Sb2O3 has better flame retarding effect on ABS and PS than PP. The reason is most likely that DBDPE contains benzene ring and has the good compatibility with ABS and PS. In a word, DBDPE/Sb2O3 has good flame retardant property for the polymers and makes the average values of pkHRR and EHC of the polymers decrease.
LOI of the flame retarded polymers is listed in Table 2. The LOI value of ABS/DBDPE/Sb2O3 is similar to that of ABS/DBDPO/Sb2O3. It reveals that the flame retardancy of DBDPE is same as DBDPO on ABS. Moreover, the LOI values of flame retarded polymers all obviously increase compared with those of the pure polymers.
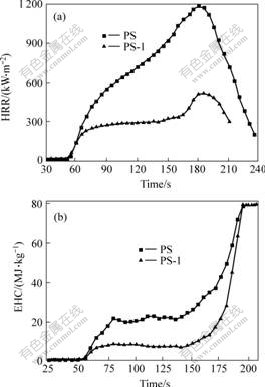
Fig.2 HRR(a) and EHC(b) curves of PS (PS-0) and PS/ DBDPE/Sb2O3 (PS-1)
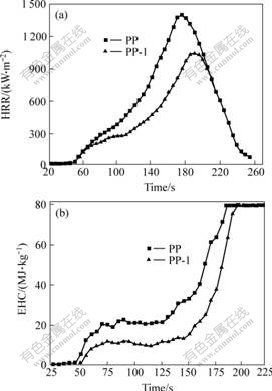
Fig.3 HRR(a) and EHC(b) curves of PP (PP-0) and PP/ DBDPE/Sb2O3 (PP-1)
Table 2 LOI of pure polymers and flame retarded polymers

3.2 Effect of DBDPE/Sb2O3 on thermal decomposition of polymers
TG curves and derivative thermogravimetry (DTG) curves of ABS with different formulations are shown in Fig.4. TG curve of ABS can fall into two steps and flame retarded ABS decompose in three steps due to the addition of DBDPE/Sb2O3. The first decomposition step of flame retarded ABS is in the temperature range of 335-370 ℃. The initial decomposition temperatures of ABS-1 and ABS-2 are lower by about 50 ℃ than that of ABS because of the decomposition of DBDPE (about 335 ℃). The second step is the main stage of polymer chain decomposition, which starts at about 370 ℃. In this step the average mass loss ratio of ABS-1 and ABS-2 are about 50% of that of ABS. The third step (from 470 to 700 ℃) is the thermal degradation of residual char, and the mass loss ratio of ABS decreases because the most of ABS decomposes. It shows that DBDPE/Sb2O3 makes the mass decompose rate of ABS decrease in the main degradation step of ABS. The TG and DTG curves of ABS-1 and ABS-2 nearly overlap based on the same content of flame retardants. It shows that DBDPE/Sb2O3 and DBDPO/Sb2O3 both can slow down the mass decompose rate of ABS, and DBDPE has the same effect on the pyrolysis reaction of polymers as DBDPO.

Fig.4 TG(a) and DTG(b) curves of different ABS
Fig.5 displays the TG and DTG curves of PP and flame retarded PP. It is different that the chain degradation reaction of PP usually happens at about 300 ℃. The thermal decomposition temperature of PP reduces by about 30 ℃ compared with those of flame retarded PP. Moreover, PP-1 and PP-2 have only two steps of degradation. It can attribute to the low residual char ratio of PP. With increasing the content of DBDPE/Sb2O3, there is a further decrease of the average mass loss rate. The average mass loss ratio of PP-1 and PP-2 are about 40% of that of PP, and the temperatures at the maximum mass loss ratio increase by about 70 ℃. The results show that thermal stability of PP is improved with increasing content of DBDPE/Sb2O3 and DBDPE has an excellent effect on the mass degradation of PP.
Fig.5 TG(a) and DTG(b) curves of PP and PP/DBDPE/Sb2O3
3.3 Flame retarding mechanism of DBDPE/Sb2O3
From the flammability and thermal stability of flame retarded polymers, it is evident that DBDPE has the similar effect on the thermal degradation of flame retarded polymers with DBDPO. It can be concluded that DBDPE has the same flame retarding mechanism as DBDPO. It can be further proved the conclusion from the following MLR curves of fire retarded polymers in the cone calorimeter tests.
MLR curves of ABS and PP are shown in Fig.6. The average MLR values of flame retarded ABS and PP are slightly higher than those of pure ABS and PP, and HRR and EHC values of fire retarded polymers are lower than those of pure polymers. HRR and EHC of flame retarded ABS and flame retarded PP all decrease, but MLR is bigger than that of pure polymers. It suggests that the decrease of HRR is not due to the increase of char formation ratio but the generation of incombustible gases. These incombustible gases cannot give out heat or the generated gases cannot fire completely. They dilute the concentration of the flammable degradation products covered the polymers surface and insulate the contact between the flammable degradation products and oxygen. It suggests that DBDPE/Sb2O3 mainly acts on the gas phase flame retarding mechanism in the combustion of polymers[14-15].
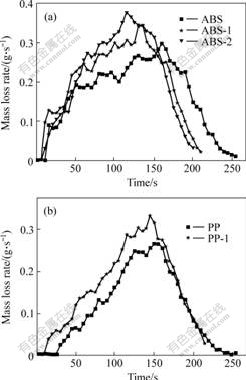
Fig.6 Mass loss rate(MLR) curves of ABS(a) and PP(b)
Sb2O3 is usually used in combination with halogen compounds and has synergistic effect with halogen-containing flame retardants. In Fig.7, the initial decomposition temperatures of ABS-1 and ABS-3 decrease and the mass loss rates become higher with increasing the content of Sb2O3 in the first step of thermal decomposing (335-380 ℃). It shows that increasing the content of Sb2O3 accelerates the degradation of DBDPE. Mass loss rates of flame retarded ABS gradually decrease, however, with increasing the content of Sb2O3 in the temperature range from 380 to 470 ℃. It suggests that the effect of DBDPE/Sb2O3 on the pyrolysis of polymer molecular chain becomes more obvious with increasing the content of Sb2O3. Some researches[14-15] had proved that HBr and SbBr3 were released when polymers flame retarded by DBDPO/Sb2O3 were burnt. They could catch active free radicals that transfer the degradation reaction of molecular chain and terminate the combustion reaction of polymers. It suggests that gas phase flame retarding is mainly flame retarding mechanism of DBDPE/Sb2O3.
The effect of the content of Sb2O3 on the HRR and EHC curves is shown Fig.8. It is similar that the increasing Sb2O3 content can improve the flame retarding of ABS and decrease the combustible gas of burning products. It further confirmed the synergistic effect of Sb2O3 with DBDPE in the simulated fire circumstance.

Fig.7 TG(a) and DTG(b) curves of different Sb2O3 content of ABS

Fig.8 HRR(a) and EHC(b) curves of ABS with different Sb2O3 contents
4 Conclusions
1) The HRR and EHC curves and TG curves of ABS/DBDPE/Sb2O3 are similar to those of ABS/DBDPO/Sb2O3. It suggests that DBDPE has the same effect on the flame retardant property and thermal stability as DBDPO.
2) DBDPE/Sb2O3 makes the average HRR and EHC values of fire retarded polymers greatly decrease, but makes the MLR values slightly increase. It shows that the decrease of HRR is not due to the increase of char formation ratio but the generation of incombustible gases.
3) The gas flame retarding mechanism is the main flame retarding mechanism of DBDPE/Sb2O3.
4) Increasing the content of Sb2O3, the flammability and thermal stability of fire retarded ABS are greatly improved. It can be concluded that Sb2O3 has good synergistic effect with DBDPE.
References
[1] STEVENS G C, MANN A H. Risks and benefits in the use of flame retardants in consumer products[R]. London: DTI Report, 1999.
[2] National Academy of Sciences, National Research Council. Subcommittee on flame retardant chemicals[M]. Washington DC: National Academy Press, 2000.
[3] European Chemicals Bureau. Institute for health and consumer protection[R]. 2002.
[4] HUA I, KANG N, JAFVERT C T, FABREGA-DUQUE J R. Heterogeneous photochemical reactions of decabromodiphenyl ether[J]. Environmental Toxicology and Chemistry, 2003, 22(4): 798-804.
[5] LI Xiang, YANG Liang, LIU Er-lie, XU Wen-guo. Research on risk characteristics of decabrominated diphenyl ethane[C]// Progress in Safety Science and Technology 4: Proceedings of the 2004 International Symposium on Safety Science and Technology, Part B, 2004, 4: 2164-2166.
[6] SEAN H, RICHARD B, RICHARD P. New information on decabromodiphenyl ether and how it changes our interpretations of risk[J]. Organohalogen Compounds, 2004, 66(9): 3800-3806.
[7] HARDY M L, MARGITICH D, ACKERMAN L. The subchronic oral toxicity of ethane, 1, 2-bis(pentabromophenyl) (Saytex 8010) in rats[J]. International Journal of Toxicology, 2002, 21(3): 165-170.
[8] TOMOYUKI I, MAKI IKUHIRO M, TOMOKAZU K, JUNIHIKO T. Thermal degradation and flame retardancy of polycarbonate as a flame retardant polymer[J]. Journal of the Society of Materials Science, 2004, 53 (12): 1301-1308.
[9] ISO Standard 5660-1, Heat release rate from building products(Cone calorimeter method)[R]. Geneva: The International Organization for Standardization, 1993.
[10] PRICE D, LIU Yan, HULL T R, MILNES G J, KANDOLA B K, HORROCKS A R. Burning behaviour of fabric/polyurethane foam combinations in the cone calorimeter[J]. Polymer International, 2000, 49(10):1153-1157
[11] GALLINA G, BRAVIN E, BADALUCCO C. Application of cone calorimeter for the assessment of class of flame retardants for polypropylene[J]. Fire and Materials, 1998, 22(1): 15-18.
[12] HIRSCHLER M M, BABRAUSKAS V, GRAYSON S J. Heat release in fires[M]. London: Elsevier Applied Science, 1992: 375-422.
[13] VANSPEYBROECK R, VAN H P, VANDEVELDE P. Combustion behaviour of fabric and polyurethane flexible foam mock-up combinations under cone calorimetry test conditions[J]. Cellular Polymers, 1992, 11(2): 118-128.
[14] MAMMEN H E. Role of antimony oxide in flame retardant formulations[J]. Plastics Design & Processing, 1978,18(7): 54-57.
[15] TAKASHI H, TOSHIAKI N. synergistic effects of sb2O3 and other metal oxide or hydroxide in plasticized flame retardant polypropylene and plasticized PVC[J]. Journal of Fire Retardant Chemistry, 1981, 8(4): 171-192.
(Edited by YANG Hua)
Foundation item: Project(20574020) supported by the National Natural Science Foundation of China
Received date: 2007-03-02; Accepted date: 2007-04-18
Corresponding author: ZUO Jian-dong, PhD; Tel: +86-20-22236818; E-mail: qingfeng_9709@126.com