Leptosririllum ferrooxidans-sulfide mineral interactions withreference to bioflotation nad bioflocculation
来源期刊:中国有色金属学报(英文版)2008年第6期
论文作者:A. VILINSKA, K K. HANUMANTHA RAO
文章页码:1403 - 1409
Key words:bacteria; sulphide mineral; Zeta-potential; adhesion; FT-IR; flocculation; flotation
Abstract: The adhesion of ferrous ions grown Leptospirillum ferrooxidans cells on pyrite and chalcopyrite minerals was investigated through adsorption, Zeta-potential and diffuse reflectance FT-IR measurements. The influence of bacterial species on minerals floatability was determined by Hallimond flotation tests while the flocculation behaviour was examined by Turbiscan measurements. The minerals iso-electric point (pH 6.5-7.5) after interaction with bacterial cells shifted towards cells iso-electric point (pH 3.3), indicating the chemical nature of cells adsorption on mineral surfaces. The FT-IR spectra of minerals treated with bacterial cells showed the presence of all the cell functional groups signifying cells adsorption. The bacterial cells adsorption on chalcopyrite was higher compared with pyrite, which agreed with cells greater depression effect on chalcopyrite flotation and pronounced flocculation behaviour in comparison with pyrite.

Leptosririllum ferrooxidans-sulfide mineral interactions with reference to bioflotation nad bioflocculation
A. VILINSKA, K. HANUMANTHA RAO
Division of Mineral Processing, Lule? University of Technology, SE-971 87 LULE?, Sweden
Received 20 September 2008; accepted 5 November 2008
Abstract: The adhesion of ferrous ions grown Leptospirillum ferrooxidans cells on pyrite and chalcopyrite minerals was investigated through adsorption, Zeta-potential and diffuse reflectance FT-IR measurements. The influence of bacterial species on minerals floatability was determined by Hallimond flotation tests while the flocculation behaviour was examined by Turbiscan measurements. The minerals iso-electric point (pH 6.5-7.5) after interaction with bacterial cells shifted towards cells iso-electric point (pH 3.3), indicating the chemical nature of cells adsorption on mineral surfaces. The FT-IR spectra of minerals treated with bacterial cells showed the presence of all the cell functional groups signifying cells adsorption. The bacterial cells adsorption on chalcopyrite was higher compared with pyrite, which agreed with cells greater depression effect on chalcopyrite flotation and pronounced flocculation behaviour in comparison with pyrite.
Key words: bacteria; sulphide mineral; Zeta-potential; adhesion; FT-IR; flocculation; flotation
1 Introduction
Recent developments in biotechnology have given promise of not only aiding hydrometallurgical operations but also providing means for bioremediation of environmental problems generated by the mineral industries. Many other uses of microorganisms are potentially possible. These include the use of microorganisms in flocculation and flotation of minerals, where the adhesion of bacterium and/or extracellular polymeric substances(EPS) to minerals induces a change in surface properties. The biomodification of mineral surfaces involves the complex action of microorganisms on the mineral surface. There are three different mechanisms by means of which the bio-modification can occur: 1) attachment of microbial cells to the solid substrate[1-3], 2) oxidation reactions[4] and 3) adsorption and/or chemical reaction with the metabolite products [5]. Several types of autotrophic and heterotrophic bacteria, fungi, yeast and algae have been tested in minerals biobeneficiation. However, the interfaces between biological and geological materials, as well as means to design and manipulate that interface is not unexplored. It is necessary to study the biotic interfaces focusing on the mineral side of the interface, i.e., on response of chemical composition and structure, net charge/potential and wettability of the mineral surface to the bacterium presence.
Chemolitotrophic bacterial strains with natural abundance in mineral environment and mine water can be selectively attached to sulphides, thereby essentially modifying the surfaces and the follow-up adsorption of flotation reagents. Leptospirillum ferrooxidans used in this study is a chemolitotrophic acidophilic bacterium which is isolated from acid mine drainage water[6-7]. This bacterium is capable of iron oxidation similar to Acidithiobacillus ferrooxidans but cannot able to oxidise sulphur. Both strains of microorganisms were successfully used for bioleaching of sulphide ores and concentrates[8-9] while A. ferrooxidans was applied in some studies regarding bioflotation and bioflocculation of sulphide minerals. Studies showed that the xanthate flotation of pyrite was greatly reduced by the application of A. ferrooxidans[10-17] whereas the effect on chalcopyrite was found to be marginal, thereby selective flotation of chalcopyrite from pyrite has been suggested. Flocculation and settling behaviour of sulphide minerals is also enhanced in the presence of A. ferrooxidans with negligible effect on non-sulphide minerals[18]. A. ferrooxidans cells with a natural affinity towards sulphide minerals, mostly pyrite, can positively influence their surface properties and enhance the separation process. L. ferrooxidans isolated from same sources as that of A. ferrooxidans and originally assumed to be A. ferrooxidans, lacks sulphur oxidation ability. Therefore, the mechanism of L. ferrooxidans cells adhesion to mineral surface and their influence on surfaces properties are perceived to be different. This work presents the results on surface chemical studies of the adhesion of L. ferrooxidans cells on pyrite and chalcopyrite, and the changes in surface properties caused by bacterial interaction are discussed with reference to flotation and flocculation behaviour of minerals.
2 Experimental
2.1 Minerals
Pure natural pyrite and chalcopyrite crystals supplied by Gregory, Bottley and Lloyd, UK, were used in the present studies. The crystals were broken into small pieces and dry ground in an agate mortar. The product was sieved to obtain suitable size fractions of 38-106 μm for flotation experiments and <5 μm for zeta-potential, adsorption and FT-IR studies. Prepared samples were washed with HCl for cleaning the surface oxidised species and stored in a refrigerator until being used. The BET specific surface area of these coarse and fine samples was 0.06 m2/g and 1.02 m2/g for pyrite and, 0.17 m2/g and 1.90 m2/g for chalcopyrite, respectively.
2.2 Bacteria
The bacterial strain Leptospirillum ferrooxidans DSM2391 was grown in a modified Leptospirillum HH medium (40 g/L FeSO4?7H2O, 0.132 g/L (NH4)2SO4, 53 mg MgCl2?6H2O, 27 mg KH2PO4, 0.147 g CaCl2?2H2O, 62 μg MnCl2?2H2O, 68 μg ZnCl2, 64 μg CoCl2?6H2O, 31 μg H3BO3, 10 μg Na2MoO4, 67 μg CuCl2?2H2O) at pH 1.8 and at 30 ℃ on a rotary shaker maintaining 150 r/min. Collected cell mass was filtered and washed with pH 2 water to obtain cells devoid of possible precipitates and metabolites.
2.3 Zeta potential measurements
Zeta potential measurements were made with a Zeta Compact (Cad Instrumentation, France) equipped with video and image analysis system. The mineral concentration in the suspension was 0.25 g/L and a constant ionic strength of 0.01 mol/L was maintained with KNO3. Interaction time for cell-mineral system was 30 min and a cell concentration of 3×1010 mL-1 was used for both minerals. Measurements were performed as a function of pH adjusted with KOH and HNO3.
2.4 Adsorption measurements
The <5 μm size fraction of minerals was used for adsorption studies. The adsorption of bacterial cells on pyrite and chalcopyrite was carried out in 250 mL Erlenmeyer flasks. Tests were performed with 1 g of mineral in 100 mL water containing varied initial cell concentrations of 107, 5×107, 108, 5×108, 109 mL─1. After 30 min of interaction, the cells in liquid phase were estimated using a Neubauer counter under a microscope. The amount of cells adsorbed on mineral surfaces was determined by the difference in the cells concentration in liquid phase before and after adsorption.
2.5 Diffuse reflectance FT-IR measurements
The solid samples after bacterial cells adsorption measurements were filtered, air-dried and subjected to FTIR for recording the spectra. The spectra were obtained with a Perkin-Elmer 2000 spectrometer at 4 cm-1 resolution with a narrow band liquid N2-cooled MCT detector. The samples were prepared by dispersing the air-dried sample in KBr at a concentration of 2.5% for the cells and minerals. The absorbance units were defined by the decimal logarithm of the ratio of pure finely powdered KBr reflectance to the sample.
2.6 Settling measurements
Settling tests were conducted by using Turbiscan ma2000 instrument, which scans the entire height of the sample cell and measures the stability and instability of suspensions. The detectors in Turbiscan receives the transmitted and backscattered light from a cylindrical sample cell every 40 μm on a maximum height of 55 mm. The profile obtained characterise the suspension homogeneity, particle concentration and mean diameter. A solid concentration of 2.5 g/L of <5 μm size particles of pyrite and chalcopyrite was used to study the sedimentation behaviour of particles. Tests were made at different pH values of the mineral suspensions after bacterial conditioning of 4×1010 cells/g. A blank experi- ment in the absence of bacteria was always carried out.
2.7 Flotation tests
The single-mineral flotation tests were conducted in a Hallimond tube using 1 g of either pyrite or chalcopyrite of 38-106 μm size fraction. The mineral samples were first conditioned with a predetermined Leptospirillum ferrooxidans cells concentration in 100 mL of water at a specified pH for 30 min. Then potassium isopropyl xanthate collector was added and the sample was conditioned further for 5 min. The entire solution was transferred into a Hallimond tube and floated for 1 min. An airflow rate of 200 mL/min was applied during flotation. The influence of initial cell concentration on sulphides flotation was examined at 0.5×10-4 mol/L xanthate collector concentration.
3 Results and discussion
3.1 Zeta potential studies
The Zeta-potentials of pure Leptospirillum ferrooxidans cells and pyrite, and pyrite after interaction with cells as a function of pH are shown in Fig.1. Pure cells exhibited an iso-electric point(IEP) at pH 3.3 and beyond this pH the magnitude of negative potential increased with a rise in pH value. The magnitude of Zeta-potentials is relatively high compared to A. ferrooxidans cells grown under similar conditions [10-16, 18-19]. The presence of functional groups such as carboxyl, amino and hydroxyl, and their protonation and/or dissociation as a function of pH impart surface charge to the cells. The presence of ammonium containing polymers (proteins) and polysaccharides containing phosphate and/or carboxylic groups in the surface layers of bacteria and a charge balance of these anionic and cationic acid/base groups determine the IEP of bacterial cells. The infrared spectrum of cells showed the presence of these groups originating from cell wall components of lipo-polysaccharides, lipoprotein and bacterial surface proteins. The positive surface potential at low pH values is obviously due to the protonation of ammonium groups and the increase in negative charge beyond pH 3.3 is caused by the dissociation of anionic functional groups. The cells are thus expected to adsorb on mineral surface through electrostatic interactions and as well through specific chemical interaction of functional groups on surface metal ions besides metabolic reasons.
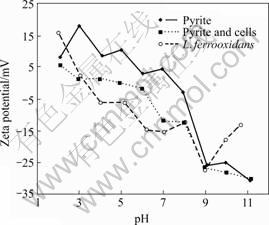
Fig.1 Zeta-potentials of L. ferrooxidans cells and pyrite, and pyrite treated with cells as function of pH
Pure pyrite exhibited an IEP at pH 7.5 and above this pH the negative potential of mineral increased with increase in pH. The reported IEP of pyrite as determined by electrophoresis fall between pH 3.5 and 7.5 [10-12, 14-18], and this variation could arise from several factors such as origin/mineralogy, sample preparation, surface oxidation and aging in water. After interaction with bacterial cells for 30 min, the IEP shifted to pH 5 and the Zeta-potential below pH 3 and above pH 7 nearly corresponds to that of the cells potential. The pyrite Zeta-potential decreased in the entire pH region and the curve moved towards the pure cells Zeta-potential, indicating the adsorption of cells on pyrite surface. However, higher Zeta-potentials between pH 3 and 7 compared to cells illustrate that either the pyrite surface is not fully covered at the present cell concentration of 3×1010 mL-1 or the presence of ferric ions on the surface due to bacterial oxidation of pyrite.
The Zeta-potential of chalcopyrite before and after interaction with bacterial cells are presented in Fig.2. The IEP of chalcopyrite displayed at pH 6.5 which moved to pH 3 after interaction with cells, coinciding to the IEP of cells. The Zeta-potentials between pH 3 and 6 are close to zero, which is seen similar to pyrite potentials after interaction with cells. Beyond pH 6, the Zeta-potentials of chalcopyrite interacted with cells are similar to pure chalcopyrite potentials. There is hardly any influence of cells on chalcopyrite potentials in the basic pH region. In general, the Zeta-potentials of mineral-bacteria system exhibit on the characteristic curves between pure cells and minerals [11-12,14,19-20].
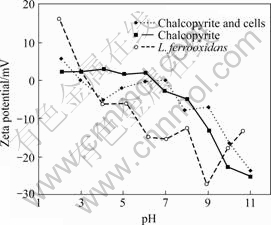
Fig.2 Zeta-potentials of L. ferrooxidans cells and chalcopyrite, and chalcopyrite treated with cells as function of pH
3.2 Adsorption studies
The Leptospirillum ferrooxidans cells adhesion on pyrite and chalcopyrite were carried out to determine the extent of cells adsorption on mineral surfaces. The adsorption isotherms of cells for pyrite and chalcopyrite at pH 4 are shown in Fig.3. The adsorption density increased with increasing equilibrium concentration of cells. At the beginning, when the initial cell concentration was 109 mL-1, nearly all the cells on pyrite and chalcopyrite are adsorbed corresponding to 80.6% and 99.9% of the initial cells, respectively. The isotherms display a linear increase in adsorption density from an equilibrium cells density of 104 mL-1 and there was no levelling off adsorption within the cell concentration range studied. While considering the geometrical cell dimensions of 1.65 μm in length and 0.35 μm in breadth, the cells adsorption density for full surface coverage in horizontal orientation corresponds to 1×1012 m-2. This approximate calculation reveals that the adsorption of cells at the highest adsorption data point in the isotherms of either mineral equals just 10% of the surface coverage. Adsorption measurements beyond 1×108 mL-1 equili- brium cells concentration are thus needed to establish the saturation level of cells adsorption on pyrite and chalcopyrite.
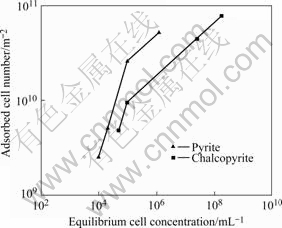
Fig.3 Adsorption isotherms of L. ferrooxidans cells on pyrite and chalcopyrite
The adsorption density of A. ferrooxidans cells on pyrite was found to be faster and higher compared to chalcopyrite[10,12,15-18]. It was also reported that the same species of bacterial cells arising from different strains had differences in adsorption behaviour[21]. Although the present adsorption isotherms are limited to lower surface coverage, L. ferrooxidans cells adsorption on chalcopyrite was higher than pyrite at any of the equilibrium cells concentration. Thiobacillus group bacteria are known to specifically adsorb on surface defects and imperfections[22]. Since chalcopyrite surface area is nearly twice that of pyrite, it is presumed to contain higher surface imperfections than pyrite and therefore higher adsorption of cells on chalcopyrite.
3.3 FT-IR studies
The diffuse reflectance FT-IR spectrum of Leptospirillum ferrooxidans cells depicts several absorbance bands (Fig.4) composing protein, lipid, extracellular polysacchrides and nucleic acids. The absorbance bands at 2 959, 2 925 and 2 856 cm-1 characterise asymmetric CH3 stretching, asymmetric CH2 stretching and symmetric CH2 stretching of hydrocarbon chains, respectively. The bands at 3 290, 3 070, 1 650 and 1 539 cm-1 represent the stretching and bending vibrations of ―NH groups of proteins (amideⅠand Ⅱ). The carbonyl absorption of carboxylic groups is identified with the bands at 1 730 and 1 712 cm-1[23]. These absorbance bands are comparable to the bands in the spectrum of Acidithiobacillus ferrooxidans cells [10-11,14,24], illustrating similar surface chemical composition of these two Thiobacillus group bacterial cells.
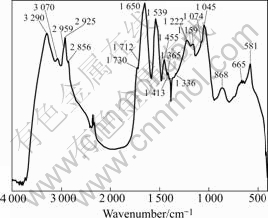
Fig.4 Diffuse reflectance FT-IR spectrum of Leptospirillum ferrooxidans cells
The spectra of pyrite and chalcopyrite treated with bacterial cells showed that with increasing cells concentration the presence of cells absorbance bands increased in intensity. Typical spectra in the case of pyrite are shown in Fig.5. These spectra characterise the adsorption of cells on pyrite with the presence of majority of the bacterial cells absorbance bands where the intensity of peaks is dependent on cell concentration. The spectra of chalcopyrite treated with cells are also similar, bearing most of the cells absorbance bands. However, the polysaccharide absorbance bands observed on pyrite are missing in the case of chalcopyrite, probably masked due to strong absorption character of chalcopyrite.
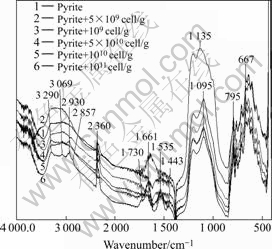
Fig.5 Diffuse reflectance FT-IR spectra of pyrite treated with increasing bacterial cell concentration
3.4 Flocculation and flotation studies
Transmission diagrams of the percentage of light transmitted through the pyrite and chalcopyrite suspensions in the presence and absence of L. ferrooxidans cells are shown in Figs.6 and 7 respectively. The light beam cannot transmit through a well dispersed suspension and therefore zero transmission represents stable suspension. When sedimentation occurs, the lower amount of dispersed particles allows the beam to pass through the sample and the transmittance increases. The suspensions were scanned covering the entire height of the sample holder for 10 min with one scan per minute. The diagrams show the state of stability or instability of suspensions with time and the higher transmittance in the presence of L. ferrooxidans indicates the sedimentation of particles. The adhesion of cells on minerals caused flocculation of both minerals at all pH values measured, ranging from acidic to basic. The cell surface polymers are thought to be responsible to bridge the particles and to cause flocculation. The transmission data shown for pyrite were at pH 6.5 and there is some natural sedimentation. Since this pH is close to pyrite iso-electric point, the particles are coagulated to some extent. The presence of cells caused flocculation of pyrite particles and therefore sedimentation was enhanced. The data shown for chalcopyrite were at pH 8 and the suspension is very stable in the absence of bacterial cells while their presence multiplied the sedimentation of particles. The higher flocculation of chalcopyrite compared with pyrite corresponds to the adsorption data where the bacterial cells adsorption density on chalcopyrite was also higher.
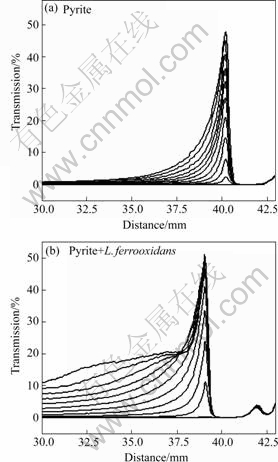
Fig.6 Transmission data of pyrite suspensions for 10 min settling time
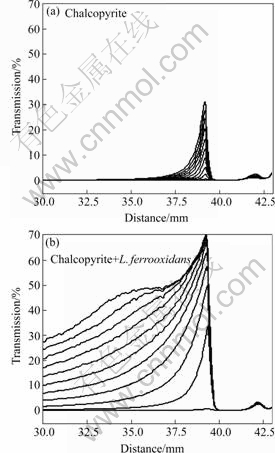
Fig.7 Transmission data of chalcopyrite suspensions for 10 min settling time
From the transmission data of Turbiscan, it is possible to evaluate the flocculation and settling phenomena of particles in the suspension. Accordingly, the recorded transmission data at a fixed time interval and fixed position for pyrite and chalcopyrite suspensions are shown in Fig.8. Bacterial cells addition increased the settling of both minerals, although the effect on pyrite is lower. The highest settling for cells treated pyrite was experienced at pH 5 and 6.5, and below pH 7 the suspension is 5-6 times more transparent in average, compared with untreated pyrite. Above pH 8 the pyrite suspensions alone and treated with cells are dispersed. Chalcopyrite exhibited higher effect of bacteria on settling behaviour and the particle size measured from transmission data (Fig.8) was found to increase significantly at all pH values studied. The increase of sedimentation is the highest at pH 8, where the settling can be considered to be 100%, due to the fact that the highest reachable transmission for this kind of experiment is about 50%-60%. When the suspension was stirred at high speed before the measurement, a further increase in the settling was observed. Pyrite flocculation and sedimentation are increased after A. ferrooxidans and A. thiooxidans addition has no effect on non-sulphide minerals[18] and moderate effect on other sulphide minerals[15,19-20].
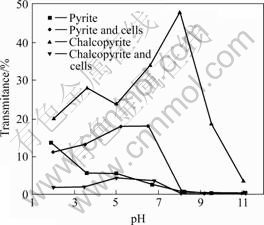
Fig.8 Transmission of pyrite and chalcopyrite suspensions as function of pH at fixed position after 10 min
Hallimond flotation tests were conducted according to previously established optimum reagents and pH conditions. The highest recovery of almost 100% for both minerals is obtained at pH 4 and 0.5×10-4 mol/L collector concentration, and these conditions were maintained for the cells interacted with pyrite and chalcopyrite. Flotation recoveries of both minerals are decreased (Fig.9) in the presence of cells but the depression of chalcopyrite is much higher than pyrite. A 95% chalcopyrite recovery is decreased to 25% at a cell concentration of 2.5×108 mL-1, while pyrite at the same conditions exhibited 67% recovery. The depression of minerals was found to depend on cell concentration. Several authors reported that flotation of pyrite interacted with A. ferrooxidans decreases while chalcopyrite still exhibits floatability at the same conditions; however, the cells adsorption on pyrite was found to be significantly higher [10,12,16-17] in these studies. CHANDRAPRA- BHA et al[16-17] observed that chalcopyrite keeps its flotation ability as a result of specific interaction between collector and mineral.
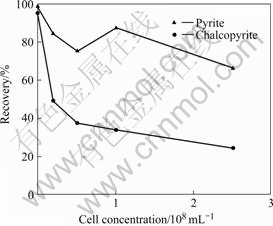
Fig.9 Xanthate flotation of pyrite and chalcopyrite in presence of L. ferrooxidans cells
4 Conclusions
Biobeneficiation studies on pyrite-chalcopyrite flotation/flocculation system using L. ferrooxidans cells showed greater impact on chalcopyrite than pyrite. The higher amount of adsorbed cells on chalcopyrite surface explains this phenomenon. The higher cell adsorption density on chalcopyrite corroborates with higher depression of chalcopyrite flotation and also higher enhancement of its settling behaviour compared with pyrite. Higher affinity of L. ferrooxidans to chalcopyrite is assigned to its higher surface defects and higher accessibility of surface Fe as an exclusive energy source because of higher surface area compared with pyrite.
Acknowledgements
The financial support from the EU BioMinE project (contract No. IP NMP2-CT-2005-500329) is gratefully acknowledged.
References
[1] ATTIA Y A, ELZEKY M. Enhanced separation of pyrite from oxidized coal by froth flotation using biosurface modification [J]. Int J Miner Process, 1993, 37: 61-71.
[2] BOTERO A E C, TOREM M L, MESQUITA L M S. Fundamental studies of rhodococcus opacus as a biocollector of calcite and magnesite [J]. Miner Eng, 2007, 20: 1026-1032.
[3] HOSSEINI T R, KOLAHDOOZAN M, TABATABAEI Y S M, OLIAZADEH M, NOAPARAST M, ESLAMI A, MANAFI Z, ALFANTAZI A. Bioflotation of Sarcheshmeh copper ore using Thiobacillus ferrooxidans bacteria [J]. Miner Eng, 2005, 18: 371-374.
[4] SANTHIYA D, SUBRAMANIAN S, NATARAJAN K A, HANUMANTHA RAO K, FORSSBERG KS E. Biomodulation of galena and sphalerite surfaces using Thiobacillus thiooxidans [J]. Int J Miner Process, 2001, 62: 121-141.
[5] NATARAJAN K A, DEO N. Role of bacterial interaction and bioreagents in iron ore flotation [J]. Int J Miner Process, 2001, 62: 143-157.
[6] MARKOSYAN G E. A new iron-oxidizing bacterium―Leptospirillum ferrooxidans nov. gen. nov. sp [J]. Biol J Armenia, 1972, 25: 26-29.
[7] BALASHOVA V, VEDENINA I Y, MARKOSYAN G E, ZAVARZIN G A. The autotrophic growth of Leptospirillum ferrooxidans [J]. Mikrobiologiya, 1974, 43: 491-494.
[8] SAND W, ROHDE K, SOBOTKE B, ZENNECK C. Evaluation of Leptospirillum ferrooxidans for leaching [J]. Appl Environ Microbiol, 1992, 58(1): 85-92.
[9] GIAVENO A, LAVALLE L, CHIACCHIARINI P, DONATI E. Bioleaching of zinc from low-grade complex sulfide ores in an airlift by isolated Leptospirillum ferrooxidans [J]. Hydrometallurgy, 2007, 98(1/2): 117-126.
[10] DAS A, HANUMANTHA RAO K, SHARMA P K, NATARAJAN K A, FORSSBERG K S E. Surface chemical and adsorption studies using Thiobacillus ferrooxidans with reference to bacterial adhesion to sulfide minerals [C]// International Biohyrdometallurgy Symposium 1999, Biohydrometallurgy and the Environment toward the Mining of the 21st Century, part 9A. Madrid, 1999: 697-707.
[11] SHARMA P K, DAS A, HANUMANTHA RAO K, FORSSBERG K S E. Surface characterisation of Thiobacillus ferrooxidans cells grown under different conditions [J]. Hydrometallurgy, 2003, 71: 285-292.
[12] SHARMA P K, DAS A, HANUMANTHA RAO K, FORSSBERG K S E. Thiobacillus ferrooxidans interaction with sulphide minerals and selective chalcopyrite flotation from pyrite [C]// SME Annual Meeting, Advances in Flotation Technology. Denver, 1999: 147-165.
[13] MISHRA M, BUKKA K, CHEN S. The effect of growth medium of Thiobacillus ferrooxidans on pyrite flotation [J]. Minerals Engineering, 1996, 9: 157-168.
[14] SHARMA P K, HANUMANTHA RAO K, NATARAJAN K A, FORSSBERG K S E. Bioflotation of sulphide minerals in the presence of heterotrophic and chemolitotrophic bacteria [C]// Proceedings of the XXI International Mineral Processing Congress, B8a. Rome, 2000: 94-103.
[15] CHANDRAPRABHA M N, NATARAJAN K A, SOMASUNDARAN P. Selective separation of ersenopyrite from pyrite by biomodulation in the presence of Acidithiobacillus ferrooxidans [J]. Journal of Colloid and Interface Science, 2004, 276: 323-332.
[16] CHANDRAPRABHA M N, NATARAJAN K A, MODAK J M. Selective separation of pyrite and chalcopyrite by biomodulation [J]. Colloids nad Surfaces B: Biointerfaces, 2004, 37: 93-100.
[17] CHANDRAPRABHA M N, NATARAJAN K A, SOMASUNDARAN P. Selective separation of pyrite from chalcopyrite and arsenopyrite by biomodulation using Acidithiobacillus ferrooxidans [J]. Int J Miner Process, 2005, 75: 113-122.
[18] NATARAJAN K A, DAS A. Surface chemical studies on Acidithiobacillus group of bacteria with reference to mineral flocculation [J]. Int J Mineral Processing, 2003, 72: 189-198.
[19] DEVASIA P, NATARAJAN K A, SATHYANARAYANA D N, RAMANANDA RAO G. Surface chemistry of Thiobacillus ferrooxidans relevant to adhesion on mineral surfaces [J]. Applied and Environmental Microbiology, 1993, 59: 4051-4055.
[20] BLAKE R C, SHUTE E A, HOWARD G T. Solubilization of minerals by bacteria: Electrophoretic mobility of Thiobacillus ferrooxidans in the presence of iron, pyrite and sulfur [J]. Appl and Environmental Microbioogy, 1994, 60: 3349-3357.
[21] GHAURI M A, OKIBE N, JOHNSON D B. Attachment of acidophilic bacteria to solid surfaces: The significance of species and strain variations [J]. Hydrometallurgy, 2007, 85: 72-80.
[22] ROHWERDER T, GEHRKE T, KINZLER K, SAND W. Bioleaching review (part A): Progress in bioleaching: fundamentals and mechanisms of bacterial metal sulfide oxidation [J]. Appl Microbiol Biotechnol, 2003, 63: 239-248.
[23] COLTHUP N B, DALY L H, WIBERLEY S E E. Introduction to infrared and Raman spectroscopy [M]. Boston: Academic Press, 1990: 547.
[24] VILINSKA A, HANUMANTHA RAO K, FORSSBERG K S E. Selective coagulation in chalcopyrite/pyrite mineral system using Acidithiobacillus group bacteria [J]. Advanced Materials Research, 2007, 20/21: 366-370.
Corresponding author: K. HANUMANTHA RAO; Tel: +46-920-491705; E-mail: hanumantha.rao@ltu.se
(Edited by YANG Bing)

