
Electrochemical characteristics of Sn-Al-P-B composites synthesized by rheological technique
HE Ze-qiang(何则强), MA Ming-you(麻明友), XIAO Zhuo-bing(肖卓炳),
XIONG Li-zhi(熊利芝), CHEN Shang(陈 上), WU Xian-ming(吴显明)
College of Chemistry and Chemical Engineering, Jishou University, Jishou 416000, China
Received 20 April 2006; accepted 30 June 2006
Abstract: Sn-Al-P-B composites were synthesized by novel rheological technique. The microstructure, morphology, and electrochemical performance of the materials were investigated by X-ray diffraction, scanning electron microscopy and electrochemical methods. The particles of tin oxide-based materials form an interconnected network structure like mesoporous materials. The average size of the particles is about 150 nm. The material delivers a charge capacity of more than 570 mA?h/g. The capacity retention of the material is about 95.5% after being cycled 30 times. The good electrochemical performance indicates that this kind of tin oxide-based material is promising anode for lithium ion battery.
Key words: tin based composite materials; rheological phase reaction; lithium ion batteries; electrochemical properties
1 Introduction
Recently, there has been an increasing demand for electrode materials possessing high capacity for rechargeable lithium-ion batteries. Much research has been undertaken to search for new anode materials in place of carbon (theoretical maximum capacity of 372 mA?h/g) to improve energy density for rechargeable lithium ion batteries[1,2]. Notably, tin oxide-based materials, as possible candidates for the next generation anode materials for Li-ion batteries due to their high lithium storage capacity and low potential of lithium ion intercalation, have drawn considerable attention[3-9]. By in situ X-ray measurements COURTNEY et al[4, 6] have shown that the reversible Li-Sn alloy formation is responsible for the observed reversible capacity in such compounds. Accordingly, the tin-oxide is irreversibly reduced to metallic Sn and Li2O during the first discharge followed by the alloying of Sn with Li, which can be written as
SnOy+2yLi++2ye-→Sn+yLi2O (y=1 or 2) (1)
xLi++Sn+xe-?LixSn (x≤4.4) (2)
Reaction (1) leads to nanostructured Sn domains homogeneously dispersed in the inactive Li2O matrix. Reaction (2) is reversible and responsible for the capacity.
Conventional glass-melting techniques are normally used to process tin oxide-based glasses[3-6]. However these methods can suffer from the problems of volatilization and phase separation, generally leading to the formation of inhomogeneous glasses. These problems tend to be particularly severe in boron- and phosphorus- containing glasses because of the volatility of B2O3 and P2O5[10-12]. Therefore, it is necessary to develop a new method to prepare this kind of material.
Notably, studies on Bingham fluid, such as electrorheological fluid, are actively pursued[13,14]. Rheological techniques have been playing an important role in synthetic chemistry.
The rheological phase reaction method is the process of preparing compounds or materials from a solid-1iquid rheological mixture. That is, the so1id reactant is fully mixed in a proper molar ratio,and made up by a proper amount of water or other solvents to a Bingham body in which the so1id particles and liquid substance are distributed uniformly, so that the product can be obtained under suitable experiment conditions.
By means of this method, many functional materials and compounds with novel structures and properties have been obtained[15], such as amorphous MnO2 as an anodic material of lithium ion battery which can be got from KMnO4 and Mn(C6H5CO2)2. In the present study, we reported a rheological technique to synthesize tin oxide-based material for lithium ion battery. The microstructure and electrochemical properties of the tin oxide-based materials were studied.
2 Experimental
Analytical grade powders of SnO, Al(OH)3, H3BO3, NH4H2PO4 and H2C2O4・2H2O were mixed and ground at a molar ratio of x(Sn): x(Al): x(B): x(P): x(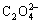 )= 1.0:0.4:0.6:0.4:1.0. Then a certain quantity of distilled water was added to the mixture under magnetic force stirring to obtain a mash. The mash was aged at 85 ℃ for 3 h, and an off-white precipitate obtained was dispersed with ultrasonic vibration and neutralized by ammonia. After the precipitate had been washed with distilled water and ethanol three times respectively, it was dried in vacuum at 80 ℃ for 24 h to obtain the precursor. Finally, the precursor was calcinated in Ar atmosphere at 400 ℃ for several hours to obtain the tin oxide-based materials.
)= 1.0:0.4:0.6:0.4:1.0. Then a certain quantity of distilled water was added to the mixture under magnetic force stirring to obtain a mash. The mash was aged at 85 ℃ for 3 h, and an off-white precipitate obtained was dispersed with ultrasonic vibration and neutralized by ammonia. After the precipitate had been washed with distilled water and ethanol three times respectively, it was dried in vacuum at 80 ℃ for 24 h to obtain the precursor. Finally, the precursor was calcinated in Ar atmosphere at 400 ℃ for several hours to obtain the tin oxide-based materials.
Phase identification and surface morphology studies of the precursor and heat-treated powders were carried out with X-ray diffractometer(Rigaku D/MAX-gA) and scanning electron microscope(JSM 5600LV).
A slurry containing 80% synthesized material, 10% acetylene black and 10% PVDF (polyvinylidene fluoride) was made using NMP (N-methylprrolidinone) as the solvent. Electrodes with an area of 1 cm2 for the measurements of electrochemical characterization were prepared by coating slurries (about 100 μm in thickness) on copper foils followed by drying in vacuum at 120 ℃ for 12 h.
Electrochemical tests were conducted using a conventional coin-type cell, employing lithium foil as a counter electrode and utilizing 1.0 mol/L LiPF6 in ethylene carbonate/dimethyl carbonate(EC/DMC) (with an EC to DMC volume ratio of 1:1) as the electrolyte. The assembly was carried out in an Ar-filled glove box. The discharge-charge tests were done under a constant current density of 0.10 mA/cm2 in the voltage range of 0-1.0 V.
3 Results and discussion
The XRD patterns of the precursor and the powders calcined at 400 ℃ for various times are shown in Fig.1. No diffraction peaks of oxides of Al, B and P can be seen in all the patterns, which indicates that the oxides of Al, B and P in the tin oxide-based materials are in the form of amorphous state. It is noted that there is a great difference between the XRD patterns of the precursor and the calcined powders. Only some diffraction peaks attributed to SnO can be seen for the precursor, however, when the precursor was calcined in Ar atmosphere at 400 ℃, not only the evolution of crystalline SnO, but also a few peaks of SnO2 and Sn are observed. With the increase of heat-treatment time, the peaks of SnO2 and Sn become more and more intense, while the peaks of SnO get weaker and weaker, which may be caused by the following disproportionating reaction[16,17]:
2SnO?SnO2+Sn (2)
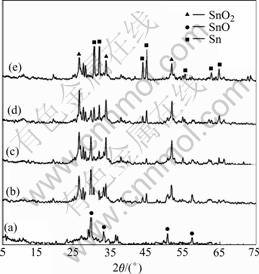
Fig.1 XRD patterns of precursor(a) and powders calcined at 400 ℃ for 2 h (b), 4 h (c), 6 h (d) and 8 h (e)
The scanning electron micrograph of the powder calcined at 400 ℃ for 6 h is shown in Fig.2. As seen from the micrograph, the particles form an interconnected network with mesoporous structure, which may be composed of the amorphous oxides of glass forming agents P, B and Al. The average size of the

Fig.2 SEM image of powder calcined at 400 ℃ for 6 h
particles is about 150 nm with some particles forming aggregates.
Fig.3 shows the initial two discharge-charge profiles of the electrode of the powder calcinated at 400 ℃ for 6 h. In the first discharge curve, two plateaus can be observed: a short plateau at about 1.42 V and a longer plateau at about 0.90 V. With the exception of the two irreversible plateaus in the first discharge curve, no other plateau can be observed in the discharge-charge curves, which may be related to the poor crystallinity of the active center in the tin-based composite oxides[18]. A discharge capacity of 1 094 mA?h/g and a charge capacity of 574 mA?h/g was achived, which suggests that tin oxide-based materials work as a high capacity anode for lithium ion batteries but also suffer from a great deal of the irreversible capacity in the first discharge-charge cycle. The large irreversible capacity in the first discharge-charge cycle may originate from two main sources[19]: the first is the reduction of tin oxide as shown in reaction (1); the second is the electrolyte decomposition to form SEI film on the surface of electrode.
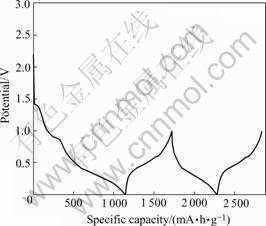
Fig.3 Initial two discharge―charge curves of powder calcined at 400 ℃ for 6 h at current density of 0.1 mA/cm2 and voltages between 0 and 1.0 V
The plots of differential capacity (dQ/dV) vs voltage for the tin oxide-based material are presented in Fig.4. Two sharp peaks at around 1.42 V and 0.89 V vs Li/Li+ are corresponding to the two plateaus of the first discharge curve. The smooth shape of the first-cycle curve indicates the formation of fine metallic Sn due to the reaction of tin oxide with Li[6]. Reduction in the peak intensity and shrinkage of the curves combined with the appearance of sharp peaks in the curve corresponding to the 30th cycle imply a fade in capacity and the aggregation of Sn clusters during cycling, respectively[6].
The relationship between charge capacity and cycle number for the electrode of the powder calcined at 400℃ for 6 h are displayed in Fig.5. The electrode shows a high charge capacity of more than 570 mA?h/g. The capacity retention is 95.5% after being cycled 30 times, which is smaller in comparison with either SnO2 or metallic Sn or the chemically derived tin oxide-material[12]. Although the coulombic efficiency for the first cycle is only 50%, it reaches 97.7% in the second cycle and keeps at 97%-99%. The results indicate that the tin oxide-based materials synthesized by the rheological technique as anode materials for lithium ion batteries have the excellent electrochemical properties. The excellent electrochemical properties of this kind of tin oxide-based materials are related to the porous network structure, which is formed by the amorphous oxides of B2O3, P2O5, Al2O3 in the tin oxide-based materials and Li2O formed in the first discharge. The porous network structure makes this kind of tin oxide-based host framework be able to provide good intercalation pathways for lithium ion to diffuse and is robust with respect to lithium intercalation and de-intercalation reaction, resulting in
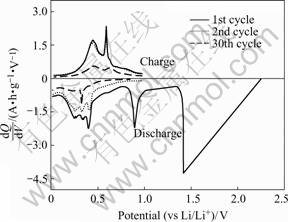
Fig.4 Plot of differential capacity vs voltage for Li/tin oxide- based material cell
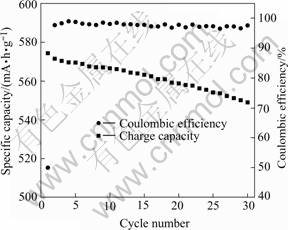
Fig.5 Relationship between charge capacity and cycle number for powder calcined at 400 ℃ for 6 h (Current density=0.1 mA/cm2)
higher reversible capacity and better cycling performance of the materials.
4 Conclusions
The tin oxide-based materials were prepared by rheological technique. The particles of tin oxide-based materials form an interconnected network structure like mesoporous materials. The average size of the particles is approximately 150 nm. Electrochemical characteriza- tion shows that the reversible capacity of the tin oxide-based materials is higher than 570 mA?h/g and the cycling performance is very good with a capacity retention of about 95.5% after cycling for 30 times. Consequently, this kind of tin-oxide based material has potential application as anodes for lithium ion batteries.
References
[1] POIZOT P, LARUELLE S, GRUGEON S, et al. Nanosized transition-metal oxides as negative-electrode materials for lithium-ion batteries [J]. Nature, 2000, 407: 496-499.
[2] KIM S S, IKUTA H, WAKIHARA M. Synthesis and characterization of MnV2O6 as a high capacity anode material for a lithium secondary battery [J]. Solid State Ionics, 2001,139: 57-65.
[3] IDOTA Y, KUBOTA T, MATSUFUJI A, MAEKAWA Y, MIYASAKA T. Tin-based amorphous oxide: A high-capacity lithium-ion-storage material [J]. Science, 1997, 276: 1395-1397.
[4] COURTNEY I A, DAHN J R. Electrochemical and in situ X-ray diffraction studies of the reaction of lithium with tin oxide composites [J]. J Electrochem Soc, 1997, 144(6): 2045-2052.
[5] COURTNEY I A, DAHN J R. Key factors controlling the reversibility of the reaction of lithium with SnO2 and Sn2BPO6 glass [J]. J Electrochem Soc, 1997, 144(9): 2943-2948.
[6] COURTNEY I A, MCKINNON W R, DAHN J R. On the aggregation of tin in SnO composite glasses caused by the reversible reaction with lithium [J]. J Electrochem Soc, 1999, 146(1): 59-68.
[7] HUGGINS R A. Lithium alloy negative electrodes formed from convertible oxides [J]. Solid State Ionics, 1998, 113-115: 57-67.
[8] MOHAMEDI M, LEE S J, TAKAHASHI D, et al. Amorphous tin oxide films: preparation and characterization as an anode active material for lithium ion batteries [J]. Electrochimica Acta, 2001, 46: 1161-1168.
[9] LI N, MARTIN C R. A high-rate, high-capacity, nanostructure Sn-based anode prepared using sol-gel template synthesis [J]. J Electrochem Soc, 2001, 148A: 164-170.
[10] HSU R, KIM J Y, KUMTA P N. Modified oxide sol-precipitation (MOSP) approach for synthesizing borophosphosilicate glasses and glass-ceramics [J]. Chem Mater, 1996, 8: 107-112.
[11] KIM J Y, KUMTA P N. Modified sol-gel based approaches for synthesizing borophosphosilicate glasses and glass-ceramics [J]. J Phys Chem B, 1998, 102(30): 5744-5753.
[12] KIM J Y, KING D E, KUMTA P N, et al. Chemical synthesis of tin oxide-based materials for Li- ion battery anodes: Influence of process parameters on the electrochemical behavior [J]. J Electrochem Soc, 2000, 147(12): 4411-4420.
[13] SIM I S, KIM J W, CHOI H J, et al. Preparation and Electrorheological characteristics of poly (p-phenylene)-based suspensions [J]. Chem Mater, 2001, 13(4): 1243-1247.
[14] CHOI H J, KIM J W, JOO J, et al. Synthesis and electrorheology of emulsion intercalated PANI-clay nanocomposite[J]. Synthetic Met, 2001, 121(1-3): 1325-1326.
[15] SUN J, XIE W, YUAN L, et al. Preparation and luminescence properties of Tb3+-doped zinc salicylates [J]. Mat Sci Eng B, 1999, 64(3): 157-164.
[16] TAO D P, YANG X W. Kinetics of disproportionation and reduction of stannous oxide and mechanism of reduction of stannic oxide [J]. The Chinese Journal of Nonferrous Metals, 1998, 3(1): 126-130. (in Chinese)
[17] MORENO M S, PUNTE G, RIGOTTI G, et al. Kinetic study of the disproportionation of tin monoxide [J]. Solid State Ionics, 2001, 144: 81-86.
[18] YANG J, TAKEDA Y, IMANISHI N, et al. Morphology modification and irreverisibility compensation for SnO anodes [J]. Journal of Power Sources, 2001, 97-98: 216-218.
[19] WACHTLER M, BESENHARD J O, WINTER M. Tin and tin-based intermetallics as new anode materials for lithium-ion cells[J]. J Power Sources, 2001, 94(2): 189-193.
(Edited by PENG Chao-qun)
Foundation item: Projects(04B016; 05C140) supported by the Scientific Research Fund of Hunan Provincial Education Department
Corresponding author: MA Ming-you; Tel: +86-743-8564088; E-mail: mamy_9589@163.com