Synthesis and upconversion luminescence of Lu2O3:Yb3+,Tm3+ nanocrystals
来源期刊:中国有色金属学报(英文版)2012年第2期
论文作者:李丽 曹雪琴 张友 郭常新
关键词:Lu2O3:Yb3+,Tm3+纳米晶;共沉淀法;上转换发光
Key words:Lu2O3:Yb3+,Tm3+ nanocrystal; co-precipitation method; upconversion luminescence
摘 要:
采用碳酸氢铵(NH4HCO3)为沉淀剂,用共沉淀法制备Yb3+ 和Tm3+ 共掺杂的Lu2O3:Yb3+,Tm3+纳米晶。研究Tm3+ 摩尔分数、Yb3+摩尔分数和煅烧温度对Lu2O3:Yb3+,Tm3+ 纳米晶的结构和上转换发光性能的影响。结果表明:所制备的纳米晶具有纯的Lu2O3相,结晶性较好。当掺杂的Tm3+浓度超过0.2%(摩尔分数)时,出现浓度淬灭效应。Tm3+ 和Yb3+的最佳掺杂比分别为0.2%和2%(摩尔分数)。在980 nm半导体激光器的激发下,样品发射出蓝光(490 nm)和红光(653 nm),分别对应Tm3+的1G4→3H6 和1G4→3F4跃迁。发射强度与激发功率的关系表明,Tm3+ 的1G4能级布居是三光子能量传递过程。随着煅烧温度的升高,上转换发光强度增强,这主要是因为随着温度的升高纳米晶表面的OH?减少和纳米晶尺寸增大。
Abstract:
Lutetium oxide nanocrystals codoped with Tm3+ and Yb3+ were synthesized by the reverse-like co-precipitation method, using ammonium hydrogen carbonate as precipitant. Effects of the Tm3+, Yb3+ molar fractions and calcination temperature on the structural and upconversion luminescent properties of the Lu2O3 nanocrystals were investigated. The XRD results show that all the prepared nanocrystals can be readily indexed to pure cubic phase of Lu2O3 and indicate good crystallinity. The experimental results show that concentration quenching occurs when the mole fraction of Tm3+ is above 0.2%. The optimal Tm3+ and Yb3+ doped molar fractions are 0.2% and 2%, respectively. The strong blue (490 nm) and the weak red (653 nm) emissions from the prepared nanocrystals were observed under 980 nm laser excitation, and attributed to the 1G4→3H6 and 1G4→3F4 transitions of Tm3+, respectively. Power-dependent study reveals that the 1G4 levels of Tm3+ can be populated by three-step energy transfer process. The upconversion emission intensities of 490 nm and 653 nm increase gradually with the increase of calcination temperature. The enhancement of the upconversion luminescence is suggested to be the consequence of reducing number of OH? groups and the enlarged nanocrystal size.

![]()

Trans. Nonferrous Met. Soc. China 22(2012) 373-379
LI Li1, 2, CAO Xue-qin2, ZHANG You1, GUO Chang-xin2
1. College of Mathematics and Physics, Chongqing University of Posts and Telecommunications,Chongqing 400065, China;
2. Department of Physics, University of Science and Technology of China, Hefei 230026, China
Received 21 January 2011; accepted 22 September 2011
Abstract: Lutetium oxide nanocrystals codoped with Tm3+ and Yb3+ were synthesized by the reverse-like co-precipitation method, using ammonium hydrogen carbonate as precipitant. Effects of the Tm3+, Yb3+ molar fractions and calcination temperature on the structural and upconversion luminescent properties of the Lu2O3 nanocrystals were investigated. The XRD results show that all the prepared nanocrystals can be readily indexed to pure cubic phase of Lu2O3 and indicate good crystallinity. The experimental results show that concentration quenching occurs when the mole fraction of Tm3+ is above 0.2%. The optimal Tm3+ and Yb3+ doped molar fractions are 0.2% and 2%, respectively. The strong blue (490 nm) and the weak red (653 nm) emissions from the prepared nanocrystals were observed under 980 nm laser excitation, and attributed to the 1G4→3H6 and 1G4→3F4 transitions of Tm3+, respectively. Power-dependent study reveals that the 1G4 levels of Tm3+ can be populated by three-step energy transfer process. The upconversion emission intensities of 490 nm and 653 nm increase gradually with the increase of calcination temperature. The enhancement of the upconversion luminescence is suggested to be the consequence of reducing number of OH- groups and the enlarged nanocrystal size.
Key words: Lu2O3:Yb3+,Tm3+ nanocrystal; co-precipitation method; upconversion luminescence
1 Introduction
In recent years, upconversion luminescence of lanthanide-doped nanocrystals have attracted much attention due to their unique optical properties and potential applications in such fields as infrared to visible upconversion lasers, fibre amplifiers, color displays, optical communications and biological ?uorescencelabels [1-3]. Many trivalent rare earth ions, such as Er3+ [4], Tm3+ [5] and Pr3+ [6], are doped as emission and absorption centres in these materials. Among the rare earth ions, Yb3+ becomes the most suitable sensitizer ion because of its special energy levels and long excited level lifetime. Tm3+ is a promising optical activator that opens the possibility for simultaneous blue and ultraviolet emission of laser action and various applications [7]. When a rare earth-doped optical device is developed, the host material is a very important factor to be considered. The host material with low phonon energy can result in a reduction of the multiphonon relaxation and help efficient upconversion occur. Therefore, choosing an appropriate host is very important for obtaining highly efficient upconversion luminescence. Rare earth oxides, as promising hosts for upconversion, possess relatively low phonon energy, high chemical durability and favorable physical properties [8]. During the past few years, extensive studies have been concentrated on the upconversion luminescence of Y2O3 host [9, 10]. The sesquioxide Lu2O3 is isostructural to Y2O3 and crystallizes in a cubic bixbyite structure. Lutetium could be a more favorable cation for RE3+ ions dopant emission [11]. Additionally, Yb3+ (86 pm) and Tm3+ (87 pm) have ionic radii similar to Lu3+ (85 pm), in favor of the dopant substitution. Therefore, Lu2O3 is an ideal host for the upconversion luminescent materials.
Co-precipitation method is one of the most promising techniques because of its advantages, such as the relatively simple synthetic route, low cost and ease of mass production. Although there are only some reports on the upconversion of rare earth ions-doped Lu2O3nanocrystals [12-14], there is no systematic study on the effect of Tm3+ content and calcination temperature on the structural and upconversion luminescent properties of Lu2O3:Yb3+,Tm3+ nanocrystals. In this work, lutetium oxide nanocrystals codoped with Tm3+ and Yb3+ were synthesized by a reverse-strike co-precipitation method. The effects of Tm3+, Yb3+ molar fractions and calcination temperature on the structure and the upconversion emission intensity of the Lu2O3:Yb3+,Tm3+ nanocrystals were investigated. The upconversion mechanisms were also discussed.
2 Experimental
Lu2O3 nanocrystals codoped with 2% Yb3+ (molar fraction, the same below) and 0.04%, 0.2%, 0.5%, 1%, and 2% Tm3+ were prepared by the reverse-like co-precipitation method, respectively. High pure Lu2O3 (99.99%), Yb2O3 (99.99%) and Tm2O3 (99.99%) were used as starting materials and ammonium hydrogen carbonate (NH4HCO3, A. R., 1 mol/L solution) was precipitant. Aqueous nitrate solutions were prepared by dissolving Lu2O3, Yb2O3 and Tm2O3 in HNO3 (A. R.) and distilled water under stirring and heating. The respective nitrate solutions were mixed with an appropriate proportion. The mixed salt solution was then added drop by drop to the excess NH4HCO3 solution at a speed of 2 mL/min under vigorous stirring at room temperature. After the process of titration, the precipitate was aged for 2 h with agitation by a electric stirrer to make the reaction proceed sufficiently. After filtering and washing with distilled water and anhydrous ethanol several times, the precipitates were dried at 80 °C for 12 h in an oven. The precipitates were further calcined in air at 900 °C for 2 h to obtain nanocrystals. Moreover, Lu2O3 nanocrystals codoped with 0.2% Tm3+ and 1%, 2%, 4%, 6%, 10%, and 15% Yb3+ were synthesized by the same procedure, respectively.
To investigate the effect of calcination temperature on the upconversion luminescence of Lu2O3:2% Yb, 0.2% Tm nanocrystals, the nanocrystals were obtained by calcining the precipitates at 800, 900, 1000 and 1100 °C for 2 h, respectively, using the same procedure.
The crystal structures were analyzed by a MXPAHF rotating anode X-ray diffractometer with Cu Ka radiation (l=0.154056 nm). The morphology of nanocrystals was examined on a FEI-Sirion 200 field-emission scanning electron microscope. The FT-IR transmittance spectra were recorded in the range of 400-4000 cm-1 on a Magna-IR 750 Fourier transformation infrared spectrometer. The upconversion luminescence spectrum was detected by R955 (Hamamatsu) from 400 to 700 nm, using an 980 nm diode laser Module (K98D08m-30W, China) as the excitation source. The excitation power was 455 mW. All spectra were collected at room temperature.
3 Results and discussion
3.1 Influence of Tm3+ molar fraction on structural and upconversion luminescent properties for Lu2O3:2%Yb3+, xTm3+ nanocrystals
Figure 1 shows the XRD patterns of Lu2O3 nanocrystals doped with 2% Yb3+ and x Tm3+(x=0.04%, 0.2%, 0.5%, 1%, 2%) and calcined at 900 °C and the data of JCPDS card No. 43-1021 for Lu2O3 were used as a reference. The patterns indicate that the nanocrystals were crystallized as cubic Lu2O3 with spatial group Ia3(206). No additional peaks of other phases have been found, indicating that the RE3+ ions are effectively doped into the host lattice. The high intensity of the diffraction peaks indicates good crystallinity of the nanocrystals. The average crystalline sizes can be estimated by using Scherrer equation [15]:
![]() (1)
(1)
where l stands for the wavelength of Cu Ka radiation; b is the corrected full width at half maximum (FWHM) of the diffraction peak; q denotes the diffraction angle; 0.89 is the characteristic of spherical particle. Using the Scherrer equation, the estimated crystalline size of Lu2O3: Yb3+, Tm3+ nanocrystals is 40-60 nm.
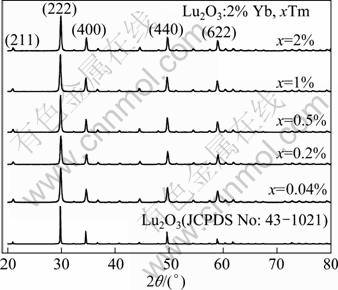
Fig. 1 XRD patterns of Lu2O3 nanocrystals doped with 2% Yb3+ and 0.04%, 0.2%, 0.5%, 1%, 2% Tm3+ and calcined at 900 °C and compared with standard pattern of JCPDS 43-1021
Figure 2 presents the typical FE-SEM image of Lu2O3:2% Yb3+, 0.2% Tm3+ nanocrystals calcined at 900 °C. As shown in Fig. 2, the obtained Lu2O3:2% Yb3+, 0.2% Tm3+ nanocrystals are aggregated, and have nearly spherical shape and an average diameter of 60 nm, which are consistent with the XRD results. The similar results were also observed for other Lu2O3:Yb3+, Tm3+ nanocrystals with various contents of Tm3+ (not shown here).

Fig. 2 FE-SEM image of Lu2O3:2% Yb3+, 0.2% Tm3+ nanocrystals
The typical upconversion spectrum of the Lu2O3:2% Yb3+, 0.2% Tm3+ nanocrystals calcined at 900 °C and excited by 980 nm diode laser excitation at room temperature is shown in Fig. 3. The strong blue emission around 490 nm easily seen by naked eye is assigned to the 1G4→3H6 transition of Tm3+. Weak red emission around 653 nm is attributed to the 1G4→3F4 transition. The experimental results show that the upconversion emission intensity ratio of the blue to red is approximately equal to 12 for the Lu2O3:2% Yb3+, 0.2% Tm3+nanocrystals.

Fig. 3 Upconversion emission spectrum of Lu2O3:2% Yb3+, 0.2% Tm3+ nanocrystals calcined at 900 °C under 980 nm excitation
Figure 4 shows the upconversion emission intensity of Lu2O3 nanocrystals doped with 2% Yb3+and xTm3+ and calcined at 900 °C as a function of Tm3+ content. As shown in Fig. 4, with increasing the Tm3+ content, the upconversion emission intensity increases at first, reaching a maximum value with 0.2% Tm3+ and then gradually decreases at higher doping concentrations. Concentration quenching occurs above 0.2% dopant content. When Tm3+ content is high, the self-quenching or cross-relaxation mechanisms between Tm3+ ions becomes active [16-18]. The energy transfer processes can be described as follows:
![]()
![]()
![]()
which can depopulate the1G4 levels but populate the 3H5 levels, leading to an increase in the extent of non-radiative transitions. Moreover, host may transfer the energy to lattice defects discussed later, which may trap energy, resulting in concentration quenching.
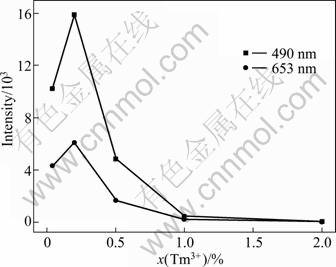
Fig. 4 Upconversion emission intensity of Lu2O3 nanocrystals doped with 2% Yb3+ and xTm3+ and calcined at 900 °C as function of Tm3+ mole fraction (The red emission intensity of 653 nm is magnified by a factor of 5)
3.2 Influence of Yb3+ molar fraction on structural and upconversion luminescent properties for Lu2O3:xYb3+, 0.2%Tm3+ nanocrystals
Figure 5 shows the XRD patterns of Lu2O3 nanocrystals doped with 0.2% Tm3+ and x Yb3+(x=1%, 2%, 4%, 6%, 10%, 15%) and calcined at 900 °C. The XRD results show that the diffracting peak positions of all the Lu2O3:xYb3+,0.2% Tm3+ nanocrystals are consistent with the standard powder diffraction pattern of Lu2O3 (JCPDS 43-1021). The crystalline size of Lu2O3: xYb3+, 0.2% Tm3+ nanocrystals is 40-60 nm using the Scherrer equation.
Figure 6 shows the upconversion emission intensity of Lu2O3 nanocrystals doped with 0.2% Tm3+ and xYb3+ and calcined at 900 °C as a function of Yb3+ content. It is noticed that the emission intensities of 490 nm and 653 nm first increase and approach a maximum at a doping content of 2% and then decrease with the increase of Yb3+ content. It is obvious that with the increase of Yb3+ content, more energy is transferred from Yb3+ to Tm3+. However, when Yb3+ ions are heavily doped, many factors such as increased amount of impunities, concentration-quenching of Yb3+, energy back transfer from Tm3+ to Yb3+ can be described as![]() , which effectively reduces upconversion emission intensity.
, which effectively reduces upconversion emission intensity.
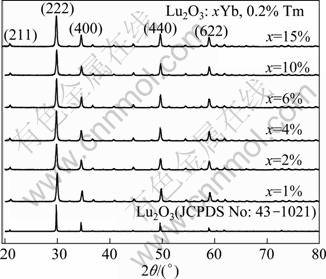
Fig. 5 XRD patterns of Lu2O3 nanocrystals doped with 0.2% Tm3+ and 1%, 2%, 4%, 6%, 10%, 15% Yb3+ and calcined at 900 °C and compared with standard pattern of JCPDS 43-1021
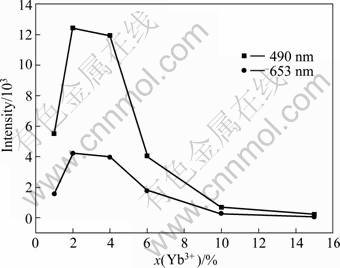
Fig. 6 Upconversion emission intensity of Lu2O3 nanocrystals doped with 0.2% Tm3+ and xYb3+ as function of Yb3+ mole fraction (The red emission intensity of 653 nm is magnified by a factor of 5)
3.3 Influence of calcination temperature on structural and upconversion luminescent properties for Lu2O3:2% Yb3+, 0.2% Tm3+ nanocrystals
Figure 7 shows the XRD patterns of Lu2O3:2% Yb3+, 0.2%Tm3+ nanocrystals calcined at various temperatures. The results reveal that all the prepared Lu2O3 nanocrystals are of cubic structure, and the diffracting peak positions are well consistent with the standard powder diffraction pattern of Lu2O3 (JCPDS 43-1021). The average crystalline size can be estimated by using Scherrer equation. In cubic crystal lattice [19], the lattice constant can be calculated from Eq. (2).
![]() (2)
(2)
where d, a, and ( h k l) are the interplanar distance, lattice constant and crystal indices (Miller indices), respectively. The strongest peaks (222) at 2q=29.775, 29.812, 29.807, 29.806° and d=3.0010, 2.9962, 2.9967, 2.9961 ? were used to calculate the average crystallite size (D) and lattice constants (a) of the Lu2O3: 2% Yb3+, 0.2% Tm3+ nanocrystals, respectively. Table1 presents the calculated crystallite sizes and lattice constants of Lu2O3:2% Yb3+, 0.2% Tm3+ nanocrystals at different calcination temperatures. The lower the calcination temperature is, the smaller the crystallite sizes are. The crystallite size increases from 32 to 70 nm with the increase of calcination temperatures from 800 to 1100 °C. The lattice constants are compatible with the literature values (10.390 ?) from the standard card (JCPDS 43-1021).
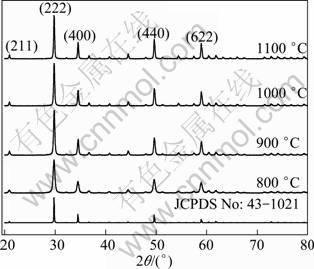
Fig. 7 XRD patterns of Lu2O3:2% Yb3+,0.2%Tm3+ nanocrystals calcined at various temperatures and compared with standard pattern of JCPDS 43-1021
Table 1 Calculated crystallite sizes and lattice constants of Lu2O3:2%Yb3+,0.2%Tm3+ nanocrystals calcined at various temperatures

Figure 8 shows the FT-IR spectra of Lu2O3:2%Yb3+, 0.2% Tm3+ nanocrystals calcined at various temperatures. The data show that the bands around 490 and 580 cm-1 are assigned to the Lu―O vibration of cubic Lu2O3 [20]. The absorption bands of OH- (around 3400 cm-1) become weaker with the increase of calcination temperature. OH- groups with high vibration frequency will increase the nonradiative relaxation rate and hence decrease upconversion efficiency [21]. This indicates that the enhanced upconversion intensity may come from the reducing of OH- groups, which are located on the surface of nanocrystals. From Table 1, it is known that the crystallite size of Lu2O3:2% Yb3+, 0.2% Tm3+ nanocrystals increases with increasing the calcination temperature. The decrease of surface-to-volume ratio can reduce the OH- groups on the surface of nanocrystals. Therefore, the reducing of OH- groups is suggested to be relevant to the enhancement of the upconversion emission.
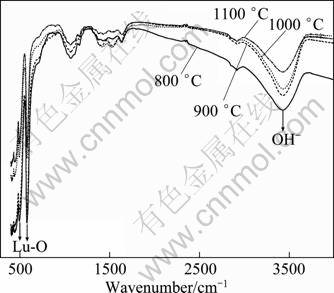
Fig. 8 FT-IR spectra of Lu2O3:2% Yb3+, 0.2% Tm3+ nanocrystals calcined at various temperatures
Figure 9 shows the influence of calcination temperature on the upconversion emission intensity of 490 nm and 653 nm under 980 nm laser excitation. It can be seen that the emission intensities of 490 nm and 653 nm become stronger gradually with the increase of calcination temperature. The increased nanocrystal size can also contribute to the upconversion emission enhancement, since the larger nanocrystal size has less defects [22]. Larger Lu2O3:2% Yb3+, 0.2% Tm3+ nanocrystals have lower surface-to-volume ratio which leads to lower surface scattering of the excitation light by the surface of the radiated Lu2O3 nanocrystals. As a result, the Lu2O3:2% Yb3+, 0.2% Tm3+ nanocrystals with larger diameter can absorb more efficiently the excitation light than Lu2O3:2% Yb3+, 0.2% Tm3+ nanocrystals with smaller diameter. It can be seen from Fig. 9 that the enhanced upconversion intensity may be due to the reduction of OH- groups [23]. Therefore, the enhancement of the upconversion luminescence is suggested to be the consequence of reducing number of OH- groups and the enlarged nanocrystal size.
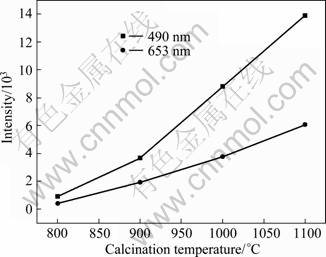
Fig. 9 Effect of calcination temperature on upconversion emission intensity of Lu2O3:2% Yb3+, 0.2% Tm3+ nanocrystals (The red emission intensity of 653 nm is magnified by a factor of 5)
3.4 Upconversion mechanisms
The energy level diagram of Tm3+ and Yb3+ shown in Fig.10 can be used to explain the upconversion mechanism. The pump photons of 980 nm laser only excite Yb3+ ion, because Tm3+ ion has no matched excited level above its ground state. The upconversion emission intensity depends on the population of 1G4 levels for Tm3+, which can be populated by three-step process [24]. Under 980 nm laser excitation, the 1G4 excited state can be populated by a three-step energy transfer (ET) process. Firstly, the 2F7/2→2F5/2 transition of Yb3+ occurs due to absorption of 980 nm photons. Then, Yb3+ in the 2F5/2 level transfers its energy to Tm3+, which is excited to the 3H5 level and relaxes nonradiatively to the 3F4 level. Tm3+ in the 3F4 level can be excited to the 3F2 level by energy transfer process after which nonradiative relaxation to 3H4 occurs. Subsequently, Yb3+ in the 2F5/2 level transfers its energy to Tm3+ and excites Tm3+ at 3H4 level to 1G4 level. Therefore, the Tm3+ decays radiatively to the 3H6 ground state and the 3F4 metastable state generating strong blue and weak red emissions around 490 nm and 653 nm from the 1G4 levels, respectively.
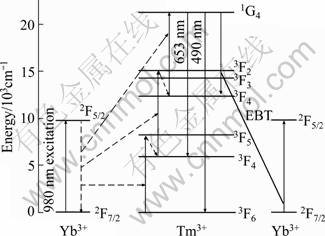
Fig. 10 Energy level diagram of Tm3+ and Yb3+ with excitation and emission process scheme (The full arrows pointing upwards represent energy absorption; full arrows pointing downwards represent emission; dotted arrows pointing downwards represent multiphonon relaxations (nonradiative decay); the diagonal line without arrows represents energy back transfer; dash-dotted arrows indicate energy transfer processes, respectively.)
The upconversion efficiency is principally governed by nonradiative processes in materials, which are dependent on the energy gap separating the upper and lower levels as well as the highest phonon energy in the materials. The multiphoton nonradiative decay rate can be expressed as [25]
![]() (3)
(3)
where T is the absolute temperature; Wn(T) is the rate at temperature T; W0(0) is the rate at 0 K; k is the Boltzmann constant; n=ΔE/(hv), n accounts for the number of phonons involved in the sensitizer excitation; hν is the maximum phonon energy of the host matrix; ?E is the energy gap involved. According to the energy gap law, multiphoton relaxation is dominant when n<5 [26]. The energy gap of Tm3+ between the levels 1G4 and the 3F2 is approximately equal to 5840 cm-1, which corresponds to 9 phonons of the highest phonon energy (the maximum phonon energy of 600 cm-1 in our experiments), so the probability of non-radiative transition from the level 1G4 to the level 3F2 can be negligible. This indicates that high population at the level 1G4 can be achieved.
To further understand the upconversion mechanisms, pump power (Ppump) dependences on the upconversion emission intensities (Iup) of Lu2O3:2% Yb3+,0.2% Tm3+ nanocrystals calcinated at 900 °C are shown in Fig. 11. According to the relationship between ![]() and
and ![]() , where n is the number of photons involved in the pump process [27], the typical upconversion luminescent intensity Iup depends on the incident pump power Ppump. The upconversion emission intensities of Lu2O3:2% Yb3+, 0.2% Tm3+nanocrystals were integrated at 490 and 653 nm for various pump powers. A plot of ln I versus ln P yields a straight line with slope n. On the basis of the experimental data in Fig. 11, the obtained n values are 2.597 and 2.712. The two slopes are close to 3, which indicates that the blue and weak red upconversion luminescence is indeed a three-photon upconversion process.
, where n is the number of photons involved in the pump process [27], the typical upconversion luminescent intensity Iup depends on the incident pump power Ppump. The upconversion emission intensities of Lu2O3:2% Yb3+, 0.2% Tm3+nanocrystals were integrated at 490 and 653 nm for various pump powers. A plot of ln I versus ln P yields a straight line with slope n. On the basis of the experimental data in Fig. 11, the obtained n values are 2.597 and 2.712. The two slopes are close to 3, which indicates that the blue and weak red upconversion luminescence is indeed a three-photon upconversion process.
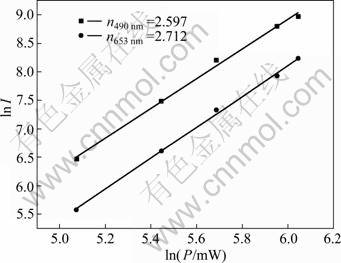
Fig. 11 Relationship between lnI and lnP of Lu2O3:2% Yb3+, 0.2% Tm3+ nanocrystals under 980 nm excitation
4 Conclusions
1) The Lu2O3 nanocrystals codoped with 2% Yb3+ and 0.04%, 0.2%, 0.5%, 1%, 2% Tm3+ were successfully synthesized by the reverse-like co-precipitation method. The further investigation reveals that the upconversion emission intensity depends on the Tm3+ and Yb3+ contents and calcination temperature.
2) The experimental results show that all the prepared Lu2O3:2% Yb3+, 0.2% Tm3+ nanocrystals can be readily indexed to pure cubic phase of Lu2O3. The strong blue and weak red emissions from the prepared nanocrystals were observed, and attributed to the 1G4→3H6 and 1G4 →3F4 transitions of Tm3+ ion, respectively. Concentration quenching occurs when the content of Tm3+ is above 0.2%. Power-dependent study reveals that the 1G4 level can be populated by three-step energy transfer process.
3) The upconversion emission intensities of 490 nm and 653 nm increase gradually with the increase of calcination temperature. The enhancement of the upconversion luminescence is suggested to be the consequence of reducing number of OH- groups and the enlarged nanocrystal size.
4) The data presented in this study might provide useful information for further development of upconversion ceramic laser associated with the1G4→3H6 transitions of Tm3+ ions.
References
[1] LIU F, MA E, CHEN D Q, YU Y L, WANG Y S. Tunable red-green upconversion in novel transparent glass ceramics containing Er: NaYF4 nanocrystals [J]. Journal of Physical Chemistry B, 2006, 110(42): 20843-20846.
[2] WANG M, MI C C, WANG W X, LIU C H, WU Y F, XU Z R, MAO C B, XU S K. Immunolabeling and NIR-excited fluorescent imaging of HeLa cells by using NaYF4:Yb,Er upconversion nanoparticles [J]. ACS Nano, 2009, 3(6): 1580-1586.
[3] WANG F, LIU X G. Recent advances in the chemistry of lanthanide-doped upconversion nanocrystals [J]. Chemical Society Reviews, 2009, 38(4): 976-989.
[4] EILER H. Effect of particle/grain size on the optical properties of Y2O3:Er, Yb [J]. Journal of Alloys and Compounds, 2009, 474(1-2): 569-572.
[5] LIM K S, BABU P, LEE S K, PHAM V T, HAMILTON D S, Infrared to visible up-conversion in thulium and holmium doped lutetium aluminum garnet [J]. Journal of Luminescence, 2003, 102-103: 737-743.
[6] NACCACHE R, VETRONE F, SPEGHINI A, BETTINELLI M, CAPOBIANCO J A. Cross-relaxation and upconversion processes in Pr3+ singly doped and Pr3+/Yb3+ codoped nanocrystalline Gd3Ga5O12: The sensitizer/activator relationship [J]. Journal of Physical Chemistry C, 2008, 112(20):7750-7756.
[7] AN L Q, ZHANG J, LIU M, WANG S W. Upconversion luminescence of Tm3+ and Yb3+ codoped lutetium oxide nanopowders [J]. Journal of Alloys and Compounds, 2008, 451(1-2): 538-541.
[8] POLIZZI S, BUCELLA S, SPEGHINI A, VETRONE F, NACCACHE R, BOYER J C, CAPOBIANCO J A. Nanostructured lanthanide-doped Lu2O3 obtained by propellant synthesis [J]. Chemistry of Materials, 2004, 16(7): 1330-1335.
[9] BAI Y F, WANG Y X, PENG G Y, YANG K, ZHANG X R, SONG Y L. Enhance upconversion photoluminescence intensity by doping Li+ in Ho3+ and Yb3+ codoped Y2O3 nanocrystals [J]. Journal of Alloys and Compounds, 2009, 478(1-2): 676-678.
[10] ZHANG Ming, LI Xin-ai, WANG Zhi-xing, HU Qi-yang, GUO Hua-jun. Synthesis of Y2O3:Eu3+ phosphors by surface diffusion and their photoluminescence properties [J]. Transactions of Nonferrous Metals Society of China, 2010, 20: 115-118.
[11] CAPOBIANCO J A, VETRONE F, BOYER J C, SPEGHINI A, BETTINELLI M. Visible upconversion of Er3+ doped nanocrystalline and bulk Lu2O3 [J]. Optical Materials, 2002, 19(2): 259-268.
[12] FIORENZO V, BOYER J C, CAPOBIANCO J A. NIR to visible upconversion in nanocrystalline and bulk Lu2O3:Er3+ [J]. Journal of Physical Chemistry B, 2002, 106(22): 5622-5628.
[13] AN L Q, ZHANG J, LIU M, WANG S W. Upconversion properties of Yb3+, Ho3+:Lu2O3 sintered ceramic [J]. Journal of Luminescence, 2007, 122-123: 125-127.
[14] YANG J, ZHANG C M, PENG C, LI C X, WANG L L, CHAI R T, LIN J. Controllable red, green, blue (RGB) and bright white upconversion luminescence of Lu2O3:Yb3+ /Er3+ /Tm3+ nanocrystals through single laser excitation at 980 nm [J]. Chemistry-A European Journal, 2009, 15(18): 4649-4655.
[15] WANG C N, ZHAO J B, LI Y, ZHANG W P, YIN M. Influence of dispersant on Y2O3:Eu3+ powders synthesized by combustion method [J]. Journal of Rare Earth, 2009, 27(6): 879-885.
[16] WANG X F, XIAO S G, BU Y Y, YANG X L, DING J W. b-Na(Y1.5Na0.5)F6: Tm3+―A blue upconversion phosphor [J]. Journal of Luminescence, 2009, 129: 325-327.
[17] PEI X J, HOU Y B, ZHAO S L, ZHENG X, FENG T. Frequency upconversion of Tm3+ and Yb3+ codoped YLiF4 synthesized by hydrothermal method [J]. Materials Chemistry and Physics, 2005, 90: 270-274.
[18] DEXTER D L, SCHULMAN J H. Theory of concentration quenching in inorganic phosphors [J]. The Journal of Physical Chemistry, 1954, 22(6): 1063-1070.
[19] JIA G, YOU H P, LIU K, ZHENG Y H, GUO N, ZHANG H J. Highly uniform Gd2O3 hollow microspheres: Template-directed synthesis and luminescence properties [J]. Langmuir, 2010, 26(7): 5122-5128.
[20] YANG J, LI C X, QUAN Z W, ZHANG C M, YANG P P, LI Y Y, YU C C, LIN J. Self-assembled 3D flowerlike Lu2O3 and Lu2O3:Ln3+(Ln =Eu, Tb, Dy, Pr, Sm, Er, Ho, Tm) microarchitectures: Ethylene glycol-mediated hydrothermal synthesis and luminescent properties [J]. Journal of Physical Chemistry C, 2008, 112(33): 12777-12785.
[21] GUO H, DONG N, YIN M, ZHANG W P, LOU L R, XIA S D. Visible upconversion in rare earth ion-doped Gd2O3 nanocrystals [J]. Journal of Physical Chemistry B, 2004, 108(50): 19205-19209.
[22] VETRONE F, BOYER J C, CAPOBIANCO J A, SPEGHINI A, BETTINELLI M. Concentration-dependent near-infrared to visible upconversion in nanocrystalline and bulk Y2O3:Er3+ [J]. Chemistry of Materials, 2003, 15(14): 2737-2743.
[23] CAPOBIANCO J A, VETRONE F, DALESIO T, TESSARI G., SPEGHINI A, BETTINELLI M. Optical spectroscopy of nanocrystalline cubic Y2O3:Er3+ obtained by combustion synthesis [J]. Physical Chemistry Chemical Physics, 2000, 2(14): 3203-3207.
[24] QIN G S, QIN W P, WU C F, HUANG S H, ZHAO D, ZHANG J S, LU S Z. Intense ultraviolet upconversion luminescence from Yb3+and Tm3+ codoped amorphous ?uoride particles synthesized by pulsed laser ablation [J]. Optics Communication, 2004, 242(1-3): 215-219.
[25] CHEN G Y, LIU H C, LIANG H J, SOMESFALEAN G, ZHANG Z G. Upconversion emission enhancement in Yb3+/Er3+-codoped Y2O3 nanocrystals by tridoping with Li+ ions [J]. Journal of Physical Chemistry C, 2008, 112: 12030-12036.
[26] WERMUTH M, RIEDENER T, GUDEL H U. Spectroscopy and upconversion mechanisms of CsCdBr3:Dy3+ [J]. Physical Review B, 1998, 57(8): 4369-4376.
[27] BALDA R, GARCIA-ADEVA A J, VODA M, FERNANDEZ J. Upconversion processes in Er3+-doped KPb2 C15 [J]. Physical Review B, 2004, 69(20): 12030-12036.
李 丽1, 2, 曹雪琴2, 张 友1, 郭常新2
1. 重庆邮电大学 数理学院,重庆 400065;2. 中国科学技术大学 物理系,合肥 230026
摘 要:采用碳酸氢铵(NH4HCO3)为沉淀剂,用共沉淀法制备Yb3+ 和Tm3+ 共掺杂的Lu2O3:Yb3+,Tm3+纳米晶。研究Tm3+ 摩尔分数、Yb3+摩尔分数和煅烧温度对Lu2O3:Yb3+,Tm3+ 纳米晶的结构和上转换发光性能的影响。结果表明:所制备的纳米晶具有纯的Lu2O3相,结晶性较好。当掺杂的Tm3+浓度超过0.2%(摩尔分数)时,出现浓度淬灭效应。Tm3+ 和Yb3+的最佳掺杂比分别为0.2%和2%(摩尔分数)。在980 nm半导体激光器的激发下,样品发射出蓝光(490 nm)和红光(653 nm),分别对应Tm3+的1G4→3H6 和1G4→3F4跃迁。发射强度与激发功率的关系表明,Tm3+ 的1G4能级布居是三光子能量传递过程。随着煅烧温度的升高,上转换发光强度增强,这主要是因为随着温度的升高纳米晶表面的OH-减少和纳米晶尺寸增大。
关键词:Lu2O3:Yb3+,Tm3+纳米晶;共沉淀法;上转换发光
(Edited by LI Xiang-qun)
Foundation item: Projects (10704090, 10774140, 11047147) supported by the National Natural Science Foundation of China; Projects (KJ090514, KJTD201016) supported by the Natural Science Foundation of Chongqing Municipal Education Commission, China
Corresponding author: LI Li; Tel: +86-23-62471721; E-mail: lilic@cqupt.edu.cn; lilic@mail.ustc.edu.cn
DOI: 10.1016/S1003-6326(11)61186-2

