
Microstructure evolution of nanostructured Ni3Al during annealing
REN Jiang-wei(任江伟)1, SHAN Ai-dang(单爱党)1, LIU Jun-liang(刘俊亮)2,
ZHANG Jun-bao(张俊宝)2, SONG Hong-wei(宋洪伟)2
1. Key Laboratory of Ministry of Education for High Temperature Materials and Tests,
School of Materials Science and Engineering, Shanghai Jiao Tong University, Shanghai 200030, China;
2. Baosteel Technology Centre, Baoshan Iron & Steel Co. Ltd, Shanghai 201900, China
Received 15 July 2007; accepted 10 September 2007
Abstract: A nanocrystalline layer was produced on the surface of Ni3Al intermetallic by means of surface mechanical attrition treatment. The surface nanocrystallites were annealed at 250-750 ℃ for 30 min. Microstructure evolution of nanocrystallites during annealing was studied by X-ray diffraction (XRD) and transmission electronic microscope (TEM). The experimental results show that long-rang order recovers rapidly when annealing temperature is below 250 ℃ and changes slowly at 350-550 ℃, and then it increases rapidly at 750 ℃. The grain size of nanocrystallites of Ni3Al keeps stable and crystal defects recover when they are annealed below 550 ℃. The grains grow normally in low temperature annealing and abnormal growth occurs at 750 ℃.
Key words: Ni3Al; intermetallic compound; nanocrystallite
1 Introduction
Nanomaterials have novel properties in physics, chemistry and mechanics. When the grains of intermetallics are refined from traditional coarse grains to nanocrystallites, they exhibit special properties such as higher hardness and lower temperature tensile superplasticity[1]. The intermetallic compound Ni3Al has attractive high temperature properties for its yield stress increases with the temperature increasing[2]. So far, it is possible to produce bulk nanocrystallites on intermetallic Ni3Al by means of severe plastic deformation methods such as high pressure torsion strain (HPTS)[3]. Surface mechanical attrition treatment (SMAT), which is a kind of severe plastic deformation method in essence, has been used to produce nanocrystalline layer on the surface of various materials including intermetallic Ni3Al[4-7].
Meanwhile, the thermal stability of nanostructured materials has been concerned, because nanostructured materials may be exposed to elevated temperature when they are in service, especially for nano- intermetallics[8-9]. Under this condition, the growth of nanocrystallites and phase transformation will alter the microstructure and properties of nanostructured materials. So, the thermal stability of nanostructured intermetallics has been subject of intensive study in recent years[10-15]. The researches on the thermal stability of nanostructured Ni3Al focus on the influence of annealing on the mechanical properties and microstructure, but the microstructure evolution during annealing is understood insufficiently[16-18].
In our previous work, a nanocrystalline layer was produced on directionally solidified intermetallic Ni3Al by means of surface mechanical attrition treatment[7]. In this work, a nanocrystalline layer was also produced on as-cast Ni3Al by means of SMAT. The nanocrystalline Ni3Al was annealed at 250-750 ℃ for 30 min under vacuum condition subsequently. The microstructure evolution of nanostructured Ni3Al during annealing was reported.
2 Experimental
Intermetallic compound Ni3Al was cast in vacuum condition and its composition was 75.9Ni-24.0Al-0.1B (mole fraction, %). The cast ingots were annealed at 1 200 ℃ for 8 h to get a homogenous and single-phase microstructure. The average grain size of as-cast Ni3Al is about 200 μm. Then, the as-cast Ni3Al samples were mechanically polished with 10 μm diamond paste and were subjected to surface mechanical attrition treatment. The principle of SMAT method was expatiated clearly in Ref.[19]. In this work, steel balls with 8 mm in diameter were used. The amplitude of vibration was 25 mm and vibration frequency was 50 Hz. Ni3Al samples were peened for 90 min under vacuum condition of 10-2 MPa. After SMAT, the deformed samples were annealed at 250, 350, 450, 550 and 750 ℃ for 30 min under vacuum condition. The corresponding annealed samples were labeled as NT1-NT5, respectively.
The microstructure of annealed samples was characterized by X-ray diffraction(XRD) and transmission electron microscope(TEM). XRD was performed on a Rigaku PINT 200/PC diffractometer. Cu Kα ray was used and the step angle was 0.02?. To compare the phase structures of the annealed samples with that of the deformed sample, the count intensity of diffraction peaks was normalized. The grain size and microstrain of the deformed and annealed samples were calculated from diffraction peaks of (111), (200), (220), (311) and (222) crystal planes of the Ni3Al phase using the Williamson-Hall technique[20]. The crystallites in the top surface of the deformed and annealed samples were observed on a JEOL JEM-200CX TEM. The accelerating voltage was 160 kV and beam current was 80 mA. To observe the crystallites in the top surface, TEM foils were mechanically polished and iron thinned only from the matrix side.
3 Results and discussion
X-ray diffraction spectra of the deformed and annealed Ni3Al samples are shown in Fig.1.

Fig.1 XRD patterns of deformed(a) and annealed Ni3Al sample (b)-(f)
It can be seen from Fig.1(a) that the disordering transformation happens in the top surface of Ni3Al after severe plastic deformation for the occurrence of (Ni, Al) solid solution. In addition, the Ni3Al disorders totally in the top surface for the disappearance of superlattice diffraction. After annealing above 250 ℃, the superlattice diffraction appears again, which means the disordered (Ni, Al) solid solution (Fig.1(b)-(f)) is reordered. Moreover, the diffraction intensity of (Ni, Al) solid solution changes with the annealing temperature. The relative content of (Ni, Al) solid solution is shown in Fig.2.
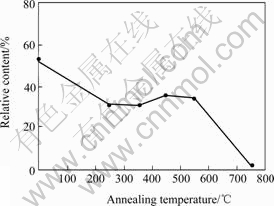
Fig.2 Relative content change of (Ni, Al) solid solution with annealing temperature
It is obviously that the content of (Ni, Al) solid solution changes hardly when the deformed Ni3Al sample is annealed at 250-550 ℃ and decreases sharply when the annealing temperature is elevated to 750 ℃.
The slow change of solid solution content at 250-550 ℃ annealing may result from the slow increase of long-rang order. It is noted that the content of solid solution in sample annealed at 250 ℃ is lower than that in deformed the sample, which may be induced by the reordering of solid solution during low temperature annealing. The reordering of disordered alloy during annealing occurs in Ni3Al nanocrystallites produced by HPTS and the temperature of reordering is in accordance with our results. The disordered nanocrystallites produced by HPTS whose grain size is about 20 nm are reordered at 317 ℃. Long-rang order increases slowly with the annealing temperature increasing and increases sharply when the disordered nanocrystallites annealed at 1 040 ℃[21].
It can also be seen from Fig.1 that the diffraction peaks of the deformed sample broaden and those of the annealed samples sharpen. The changes of diffraction peak shape mean the changes of grain size and microstrain. The changes of grain size and microstrain of Ni3Al nanocrystallites with annealing temperature are shown in Fig.3.
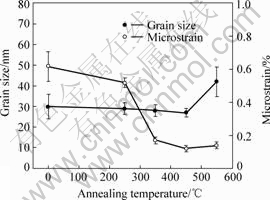
Fig.3 Changes of grain size and microstrain of Ni3Al nanocrystallites with annealing temperature
It can be seen from Fig.3 that the grain size does not increase markedly when the annealing temperature is lower than 450 ℃. However, microstrain decreases sharply when the deformed sample is annealed at 350 ℃ and keeps stable at 350-550 ℃. It should be pointed that the nanocrystallites annealed at 750 ℃ grow to above 100 nm that is not suitable to calculate grain size and microstrain from XRD data.
The annealing behavior of nanocrystalline Fe produced by mechanical milling is similar. The Fe nanocrystallites about 20 nm on average keep stable when they are annealed at 350-450 ℃ but they grow to 60 nm at 530 ℃[22]. The nanocrystallites of Ni3Al produced by HPTS grow to 0.7-2.0 mm and 2.6 mm when they are annealed at 560 ℃ and 740 ℃, respectively[21].
The stability of nanocrystallites of Ni3Al in this work may be attributed to the recovery of crystal defects. The disordered solid solution is unstable for its high free energy. So, reordering take place firstly in disordered solid solution and the ordered alloy grows secondly during annealing. When the nanocrystallites of Ni3Al is annealed at 250-550 ℃, the long-rang order increases slowly and the grain size of nanocrystallites keeps stable, so the recovery of crystal defects may be the main process of microstructure evolution.
The morphologies of nanocrystallites in the top surface of annealed Ni3Al are shown in Fig.4.

Fig.4 Morphologies of nanocrystallites in top surface of Ni3Al annealed at 350 ℃(a), 450 ℃(b), 550 ℃(c) and 750 ℃(d)
It is obviously that the crystallites in the top surface of all annealed samples are nanocrystallites and those nanocrystallites grow from the size of 10-20 nm in the sample annealed at 350 ℃ to 40-80 nm in the sample annealed at 750 ℃ gradually with the elevated annealing temperature. In these regions of nanocrystallites, the grain boundaries are blurry and curved.
However, the microstructure in the top surface is not uniform. Some regions with larger crystallites exist in all annealed samples. Some typical regions are shown in Fig.5.
It can be seen from Fig.5 that those larger crystallites in the samples annealed at 350-550 ℃ are still nanocrystallites for their grain size is less than 100 nm on average. But the surface crystallites in Fig.5(d) grow to 80-150 nm. In addition, the crystal defects inside of crystallites changes markedly. Alternating contrast can be seen in Fig.5(a), which means the existence of dislocations that produced in severe plastic deformation. In Fig.5(b), the grain boundaries become sharp and nanocrystallites become clear though there are alternating contrasts inside of nanocrystallites, which indicates the recovery of dislocations. When the annealing temperature is elevated to 550 ℃, the grain boundaries become sharp and flat. The trace of dislocations reduces distinctly inside of crystallites and exists only near the boundaries. So, the recovery of crystal defects is finished mostly. Moreover, the density of dislocations inside of crystallites is low in the sample annealed at 750 ℃. From the TEM observation, it can be concluded that the crystal defects recovers during the annealing at 350-550 ℃, which is in accordance with previous XRD data analysis. In the disordered Ni3Al produced by mechanical milling, the reordering begins at 200 ℃ annealing that is lower than the annealing temperature in this work[23]. This may be attributed to the high density grain boundaries induced by severe plastic deformation which can impede the recovery of long-rang order[23]. Some researches showed that the increase of long-range order and grain size during annealing depended on the enhanced mobility of atoms induced by crystal defects[24-25].
In the sample annealed at 750 ℃, a notable phenomenon is the abnormal growth of crystallites. A typical morphology is shown in Fig.6.
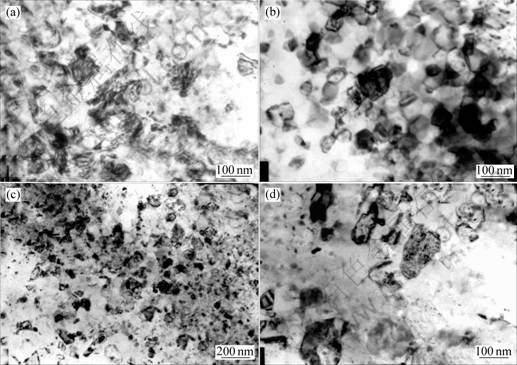
Fig.5 Morphologies of larger crystallites in top surface of Ni3Al annealed at 350 ℃(a), 450 ℃(b), 550 ℃(c) and 750 ℃(d)

Fig.6 TEM image of grains formed by abnormal growth at 750 ℃
The grains formed by abnormal growth are 0.5- 1.0 μm and no trace of defects inside is found. The abnormal growth of Ni3Al nanocrystallites is also found at 500 ℃ annealing in sputter-deposited Ni3Al thin films[26].
It can be seen from Figs.4 and 5 that the grain size of nanocrystallites increases gradually with the elevated annealing temperature, so normal growth is the main mechanism of grain growth at low temperature. The reduction of grain boundaries region is the drive force of the normal growth of nanocrystallites. The abnormal growth of crystallites at high temperature may be related to the recovery of mobility of some grain boundaries. Grain coalescence is an important mechanism of abnormal growth of grains[26].
4 Conclusions
1) The long-rang order increases rapidly when the annealing temperature is below 250 ℃ or above 750 ℃, but it changes slowly when the disordered Ni3Al is annealed at 350-550 ℃.
2) The grain size of nanocrystallites of Ni3Al keeps stable and the crystal defects recover when they are annealed below 550 ℃.
3) Grains grow normally at low temperature and abnormally grow occurs at 750 ℃.
References
[1] MISHRA R S, VALIEV R Z, MCFADDEN S X, MUKHERJEE A K. Tensile superplasticity in nanocrystalline nickel aluminide[J]. Materials Science and Engineering A, 1998, 252(2): 174-178.
[2] SAGAR P K, SUNDARARAJAN G, BHATIA M L. On the anomalous flow behaviour of nickel aluminide[J]. Scripta Metallurgica, 1990, 24(2): 257-262.
[3] VALIEV R Z, KORZNIKOV A V, MULYUKOV R R. Structure and properties of ultrafine-grained materials produced by severe plastic deformation[J]. Materials Science and Engineering A, 1993, 168(2): 141-148.
[4] TAO N R, WANG Z B, TONG W P, SUI M L, LU J, LU K. An investigation of surface nanocrystallization mechanism in Fe induced by surface mechanical attrition treatment[J]. Acta Materialia, 2002, 50(18): 4603-4616.
[5] ZHU K Y, VASSEL A, BRISSET F, LU K, LU J. Nanostructure formation mechanism of α-titanium using SMAT[J]. Acta Materialia, 2004, 52(14): 4101-4110.
[6] TAO N R, WU X L, SUI M L, LU J, LU K. Grain refinement at the nanoscale via mechanical twinning and dislocation interaction in a nickel-based alloy[J]. Journal of Materials Research, 2004, 19(6) 1623-1629.
[7] REN J W, SHAN A D, ZHANG J B, SONG H W, LIU J L. Surface nanocrystallization of Ni3Al by surface mechanical attrition treatment[J]. Materials Letters, 2006, 60(17/18): 2076-2079.
[8] SHAW L, LUO H, VILLEGAS J, MIRACLE D. Thermal stability of nanostructured Al93Fe3Cr2Ti2 alloys prepared via mechanical alloying[J]. Acta Materialia, 2003, 51(9): 2647-2663.
[9] LIU K W, M?CKLICH F. Thermal stability of nano-RuAl produced by mechanical alloying[J]. Acta Materialia, 2001, 49(3): 395-403.
[10] HU W Y, KATO T, FUKUMOTO M. Synthesis and characterization of nanocrystalline iron aluminide intermetallic compounds[J]. Materials Transactions, 2003, 44(12): 2678-2687
[11] MORRIS D G, GUTIERREZ-URRUTIA I, MUNOZ-MORRIS M A. Hardening and softening in milled nanostructured FeAl on annealing[J]. Scripta Materialia, 2007, 57(5): 369-372
[12] TANG W M, ZHENG Z X, TANG H J, REN R, WU Y C. Structural evolution and grain growth kinetics of the Fe-28Al elemental powder during mechanical alloying and annealing[J]. Intermetallics, 2007, 15(8): 1020-1026
[13] WANG L, QIN X Y, XIONG W, CHEN L, KONG M G. Thermal stability and grain growth behavior of nanocrystalline Mg2Si[J]. Materials Science and Engineering A, 2006, 434(1/2): 166-170
[14] SAM FROES F H, ZHANG F, LU L, LAI M O. Grain growth and recrystallization of nanocrystalline Al3Ti prepared by mechanical alloying[J]. Journal of Materials Science, 2003, 38(3): 613-619
[15] ZENG Q, BAKER I. Magnetic properties and thermal ordering of mechanically alloyed Fe-40at%Al[J]. Intermetallics, 2006, 14(4): 396-405
[16] JOZWIK P, BOJAR Z, BYSTRZYCKI J, PRZETAKIEWICZ W. Nanostructured Ni3Al intermetallic alloy processed by severe rolling at liquid nitrogen temperature and annealing[J]. Diffusion and Defect Data B, 2005, 101/102: 61-64
[17] KORZNIKOV A V, IDRISOVA S R, PAKIELA Z, KURZYDLOWSKI K J. Influence of long-ordering parameter and boron microalloying on mechanical properties of nanocrystalline intermetallic compound Ni3Al[J]. Diffusion and Defect Data A, 2002, 208/209: 223-228
[18] KHITOUNI M, NJAH N.  Microstructure evolution after thermal treatments of nanocrystalline Ni3Al and Ni3Al+B produced by filling[J]. Physica Status Solidi C, 2004, 1(7): 1981-1984
Microstructure evolution after thermal treatments of nanocrystalline Ni3Al and Ni3Al+B produced by filling[J]. Physica Status Solidi C, 2004, 1(7): 1981-1984
[19] LU K, LU J. Nanostructured surface layer on metallic materials induced by surface mechanical attrition treatment[J]. Materials Science and Engineering A, 2004, 375/377: 38-45.
[20] WILLIAMSON G K, HALL W H. X-ray line broadening from field aluminium and wolfram[J]. Acta Metallurgica, 1953, 1(1): 22-31.
[21] KORZNIKOV A V, TRAM G, DIMITROV O, KORZNIKOVA G F, IDRISOVA S R, PAKIELA Z. The mechanism of nanocrystalline structure formation in Ni3Al during severe plastic deformation[J]. Acta Materialia, 2001, 49(4): 663-671.
[22] NASU T, KOCH C C, EDWARDS A M, SAYERS D E. An EXAFS study of structural changes induced by mechanical milling of the Ni3Al ordered intermetallic compound[J]. Journal of Non-Crystalline Solids, 1992, 150(1/3): 491-496.
[23] GIALANELLA S, DELORENZO R, MARINO F, GUELLA M. Dilatometry of Ni3Al powder disordered by ball-milling[J]. Intermetallics, 1995, 3(1): 1-8.
[24] CLARK L C, DONALD I P. Recovery processes and ordering in Ni3Al[J]. Journal of Applied Physics, 1967, 38(10): 3894-3900.
[25] KORZNIKOV A V, DIMITROV O, KORZNIKOVA G F, DALLAS J P, IDRISOVA S R, VALIEV R Z, FAUDOT F. Thermal evolution of high-purity and boron-doped sub-microcrystalline Ni3Al produced by severe plastic deformation[J]. Acta Materialia, 1999, 47(11): 3301-3311.
[26] NG H P, NGAN A H W. An in situ transmission electron microscope investigation into grain growth and ordering of sputter-deposited nanocrystalline Ni3Al thin films[J]. Journal of Materials Research, 2002, 17(8): 2085-2094
Foundation item: Project(0452nm053) supported by the Nano-Materials Program of Shanghai City of China
Corresponding author: SHAN Ai-dang; Tel: +86-21-62932566; E-mail: adshan@sjtu.edu.cn
(Edited by ZHAO Jun)