J. Cent. South Univ. Technol. (2007)06-0793-05
DOI: 10.1007/s11771-007-0151-8
Synthesis, oxygenation and catalytic performance of manganese complex with p-aminomethyl benzoic acid Schiff base
LIU Feng-liang(������)1, LI Yuan-jian(��Ԫ��)2, HUANG Ke-long(�ƿ���)1, XIAO Zhen(Ф ��)1
(1. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China)
(2. School of Pharmeutical, Central South University, Changsha 410013, China)
Abstract��The amino acid Schiff base complex (Sal-AMBA-Mn) was prepared with p-amino-methylbenzoic acid, salicylaldehyde and Mn(OAc)2��4H2O. Its structures was characterized with IR and UV spectra. Oxygenation mechanism of the complex in N, N-dimethylformamide solution was investigated. The results show that lower temperature is in favor of the oxygenation, and energy, enthalpy and entropy are -3.8 kJ/mol, -4.2 J/mol and -161.44 J/(mol��K), respectively. In the presence of the manganese complex, dehydroepiandrosterone acetate is effectively oxidized by molecular oxygen and the corresponding enone 7-keto- dehydroepiandrosterone acetate is obtained. The yield is 62.1% when the oxidation is carried out under the reaction conditions of 60 ��, 2 MPa of O2 pressure, C5H5N as a solvent and molar ratio of the substrate to the complex of 1:10.
Key words: amino acid Schiff base; manganese complex; oxygenation; catalysis; dehydroepiandrosterone acetate; aerobic oxidation
1 Introduction
There are many studies on amino acid Schiff base transition metal complexes mainly concentrated on aerobic oxidation of cyclohexene[1-7]. At present, 7-keto-dehydroepiandrosterone acetate(7-KDHEAAc) that has an intense anti-tumor activity as steroid drugs has been mainly synthesized by oxidation of dehydroepiandrosterone acetate with the involvement of chromium reagents or peracid and so on[8-14]. But the method has many disadvantages such as a large amount of post-processing reagent and unfriendly to the environment. In this study, the aerobic oxidation method was introduced in oxidation of DHEAAc to 7-KDHEAAc in order to overcome the above shortcomings. The manganese complex (Sal-AMBA-Mn) was prepared with salicylaldehyde (Sal), p-aminomethylbenzoic acid (AMBA) and Mn(OAc)2��4H2O, its dynamics and thermodynamics parameters of the oxygenation in N, N- dimethylformamide(DMF) solution were calculated. Three factors including temperature, solvent and partial pressure of molecular oxygen on yield of the oxidation were investigated using molecular oxygen as the oxidant, Sal-AMBA-Mn as the catalyst.
2 Experimental
2.1 Materials and equipment
DHEAAc (purity: 99%) and 7-KDHEAAc
(purity: 98%) were purchased from Changzhou Pharmaceutical Raw Materials Factory, Changzhou, Jiangshu Province, China. Other reagents commercially available, such as salicylaldehyde, AMBA, Mn(OAc)2��4H2O and DMF were all of the analytical grade.
IR spectra and UV spectra were recorded on a Nicolet AVATAR360 IR spectroscopy with KBr disks and on a Shanghai 756CRT UV spectrophotometer, respectively. Elemental analyses were performed by Vario EL III autoanalyzer and atom absorption spectrometry. The products of oxidation were determined and analyzed by using Shimadzu QP-5000 GC/MS system and LC2010A HPLC connected with the SPD-M10A VP detector. The latter was performed on a column packed with C-18 and the column temperature was set at 25 ��. The mobile phase was composed of methanol-water (volume ratio of 70:30) with flow rate of 0.5 mL/min and detection wavelength at 254 nm. The product 7-KDHEAAc was determined by comparing with the mass spectrometry and the retention time of the contrast.
2.2 Synthetic route
The synthetic route of 7-KDHEAAc from DHEAAc is shown in Fig.1.
2.3 Synthesis of Sal-AMBA
5 mmol of AMBA and 5 mmol of NaOH were dissolved in 40 mL ethanol solution, and then 5 mmol
of salicylaldehyde dissolved in 40 mL anhydrous ethanol was slowly added, the resulted mixtures were continuously stirred at 60 ��. After 1 h the solvent was gradually distilled off until all the ethanol was removed, the hot reaction mixture was cooled to the room temperature. The ligand was settled out, collected by filtration, and then washed with a small amount of ethanol and dried under vacuum to give Sal-AMBA. The yield was more than 75%.
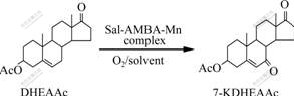
Fig. 1 Oxidation of DHEAAc to 7-KDHEAAc
2.4 Synthesis of Sal-AMBA-Mn
10 mmol of Sal-AMBA was dissolved in 25 mL ethanol solution, then 10 mmol of Mn(OAc)2��4H2O dissolved in 25 mL anhydrous ethanol was slowly added, the resulted mixtures were continuously stirred at room temperature for 6 h, and then left out for a night. The precipitate of manganese complex was collected by filtration, washed with a small amount of ethanol and water, and then dried under vacuum to give Sal-AMBA-Mn. The yield was more than 85%.
2.5 Oxygenation of Sal-AMBA-Mn complex
The stoichiometry of oxygen absorbed by the complex was measured in DMF solution according to the method in Ref.[14].
2.6 Oxidation of DHEAAc
0.61 mmol of DHEAAc, 0.061 mmol of Sal-AMBA-Mn and 50 mL C5H5N were added to the sealed autoclave in turn. The reactor was flushed twice with oxygen, and then oxygen was introduced to the definite pressure (2 MPa). After the solution was continuously stirred at 60 ��for 10 h, it was cooled to the room temperature and the excess of oxygen was deflated. The reactant solution was put into ice water, then a deposit was collected by filtration, washed with water, dried at 80 �� to afford the crude steroid. A partition was purified by alumina chromatography using ethyl acetate/cyclohexane (volume ratio of 3:10) as eluent to give 7-KDHEAAc, melting point 185-187 ��, IR (KBr) ��(cm-1): 1 742 (3-ester), 1 725 (17-ketone), 1 672 (��, ��-unsaturated ketone). MS: 344 (M+); 343 (M+ -1); 319 [M+-CH3]; 284 [M+-HOAc], the spectra data are the same as the contrast. The remains were analyzed by HPLC.
3 Results and discussion
3.1 Characterization of Sal-AMBA and Sal- AMBA- Mn
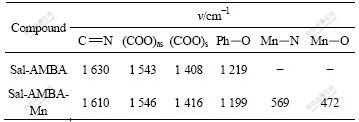
IR spectra (Table 1) show that stretching vibration of C=N appears at 1 630 cm-1, Ph-O at 1 219 cm-1 and the strong absorption of phenol hydrogen bonds near 3 442 cm-1 for Sal-AMBA. The stretching vibrations of C=N and Ph-O of complex at 1 610 cm-1 and 1 199 cm-1, respectively, are different because the backbones are influenced by the metal ion. ��as, COO and ��s, COO for the ligand are the same as those of the sal-AMBA-Mn complex. The hydrogen bonds disappear, the Mn��N bonds and Mn��O bonds appear in the complex.
Table 1 IR data of Sal-AMBA ligand and Sal-AMBA-Mn complex
The maximum absorption of UV spectra appear at 240 nm and 297 nm for Sal-AMBA. The two primary maximum absorptions shift respectively to a longer wavelength of 252 nm and 313 nm for the sal-AMBA-Mn complex. The calculated results of elements for C30H22N2O6NaMn are: C, 59.32; H, 3.65; N, 4.61; O, 15.8; Mn, 9.04. The found results of elements for C30H22N2O6NaMn are: C, 58.92; H, 3.50; N, 4.73; O, 16.05; Mn, 9.08.
So the structure of Sal-AMBA-Mn complex can be confirmed (Fig.2).
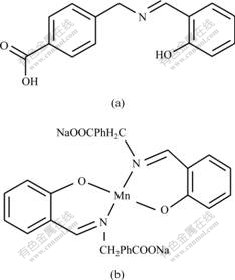
Fig.2 Structures of Sal-AMBA(a) and Sal-AMBA-Mn(b)
3.2 Oxygenation kinetics and thermodynamics of Sal-AMBA-Mn
The oxygenation result of the complex Sal-AMBA-Mn in DMF solution is shown in Fig.3. It can be seen that the rate of oxygenation increases quickly in the early stages at a constant temperature. Saturated oxygen absorption of the sal-AMBA-Mn complex decreases continuously with the increase in temperature range from 15 to 55 ��, which indicates that a lower temperature is in favor of the oxygenation.
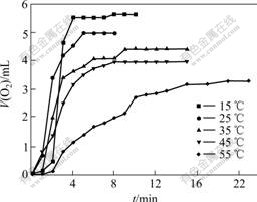
Fig.3 Plot of V(O2) vs t for oxygenation of sal-AMBA-Mn in DMF solution
The amount of absorbed oxygen was calculated by ideal gas equation pV=nRt. It is presumed that the rate of oxygenation is proportional to the concentration of the unreacted complex. The equation for the rate of oxygenation can be written as
d(c(O2))/dt = k1p(O2)c(A) (1)
where c(O2) is the concentration of the absorbed oxygen, c(A) is concentration of the unreacted complex, p(O2) is the pressure of oxygen and is a constant. Eqn.(1) can be also written as
d(c(O2))/dt=kc(A) (2)
where c(A)=c(A0)-c(O2), c(A0) is the concentration of the initial complex. Eqn.(2) can be written as:
dc(O2)/dt=k[c(A0)-c(O2)] (3)
The above equation can be written in an integral form as
-ln[c(A0)-c(O2)]=kt+C (4)
When t=0 and c(O2) = 0, C��-ln[c(A0)], so Eqn.(4) can be written as
-ln[1-c(O2)/c(A0)]=kt (5)
where c(O2)/c(A0)=V(O2)/Vth, V(O2) is the maximum volume of the absorbed oxygen at a moment and Vth is theoretical maximum volume of the absorbed oxygen. If the molar ratio of the complex to molecular oxygen is 1:1, the value of Vth calculated is 47.3 mL at 15 ��. Eqn.(5) can be written as:
-ln[1-V(O2)/Vth]=kt (6)
In Eqn.(6), a plot of -ln[1-V(O2) /Vth] vs t at 15 �� shows a straight line (Fig.4), from which the value of k is found to be 0.419, so we can deduce that the oxygenation is the first order reaction. The values of k at 25, 35, 45 and 55 �� are found to be 0.791, 0.427, 0.221 and 0.069, respectively. Thus, a plot of ln k vs 1/T gives a straight line (Fig.5).
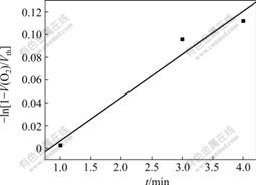
Fig. 4 Plot of -ln[1-V(O2) /Vth] vs t for Sal-AMBA-Mn at 15 �� in DMF solution
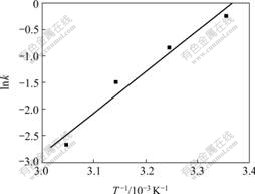
Fig.5 Plot of ln k vs 1/T for Sal-AMBA-Mn in DMF
The Arrhenius equation k=Aexp(�CE/RT) can be written in a logarithm form as:
ln k=ln A-E/(RT) (7)
The value of apparent activation energy E for the oxygenation can be calculated from the slope of the straight line in Fig.5, which is 3.8 kJ/mol, indicating that the oxygenation occurs easily.
Since the oxygenation is the first order reaction, the oxygenation equilibrium can be written as
MnL + O2 MnLO2
MnLO2
K =c(MnLO2)eq/[c(MnL)��p(O2)]=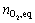 /[c(MnL)0-
/[c(MnL)0-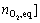 (8)
(8)
where K is the equilibrium constant.
From Fig.3 and Eqn.(8), c(MnL)0 is equal to c(A0), and 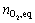 is the saturated oxygen uptake of the complex, and oxygenation equilibrium constants can be calculated, as shown in Table 2. A plot of ln K vs 1/T gives a straight line (Fig.6). The thermodynamics parameters, enthalpy ?Hf and entropy ?Sf were calculated: ?Hf =-14.16 J/mol and ?Sf =-161.44 J/(mol��K). Values of thermodynamic activation parameters indicate that of Sal-AMBA-Mn in the temperature range the oxygenation is an exothermic reaction.
is the saturated oxygen uptake of the complex, and oxygenation equilibrium constants can be calculated, as shown in Table 2. A plot of ln K vs 1/T gives a straight line (Fig.6). The thermodynamics parameters, enthalpy ?Hf and entropy ?Sf were calculated: ?Hf =-14.16 J/mol and ?Sf =-161.44 J/(mol��K). Values of thermodynamic activation parameters indicate that of Sal-AMBA-Mn in the temperature range the oxygenation is an exothermic reaction.
Table 2 Oxygenation equilibrium constants at different temperature
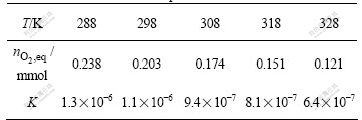

Fig.6 Plot of ln K vs 1/T of Sal-AMBA-Mn in DMF solution
3.3 Oxidation of DHEAAc
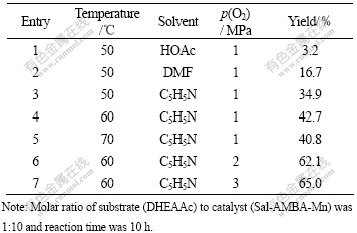
Influences of three factors including temperature, solvent and partial pressure of oxygen on yield of oxidation of DHEAAc to 7-KDHEAAc were investigated using molecular oxygen as the oxidant, Sal-AMBA-Mn as the catalyst. The results of the catalytic oxidation tests are given in Table 3.
3.3.1 Effect of solvent
It can be seen from Table 3 that yield of 7-KDHEAAc increases with the change in solvent from HOAc to DMF and from DMF to C5H5N. The results indicate that structure of active complex MnLO2 is probably destroyed by the acid solvent HOAc, whereas coordinate bonds probably exist between the axial base C5H5N and MnLO2, which can increase the electronics cloud density of center ion (Mn2+) and make Mn2+-O2 bonds more stable. This is favorable for the oxidation.
3.3.2 Effect of temperature
The effect of temperature on the yield of 7-KDHEAAc was studied in the temperature range of 50-70 ��, with C5H5N as the solvent and partial pressure of molecular oxygen at 1 MPa. The yield increases obviously with the increases of temperature from 50 to 60 �� but decreases from 60 to 70 ��. Saturated oxygen uptake of the complex decreases with the increase of temperature, which is not in favor of the oxidation, but the rate of oxidation increases with the increase of temperature, which is in favor of the oxidation.
Table 3 Yields of oxidation of DHEAAc to 7-KDHEAAc
3.3.3 Effect of partial pressure of molecular oxygen
It can be seen from Table 3 that the yield of 7-KDHEAAc increases quickly with the increase in partial pressure of molecular oxygen from 1 to 2 MPa, with C5H5N as the solvent and at 60 ��, but increases slowly when partial pressure of molecular oxygen increases from 2 to 3 MPa, which indicates that a higher partial pressure of O2 is favorable for the oxygenation and the oxidation.
4 Conclusions
1) The complex (Sal-AMBA-Mn) is synthesized, the activation parameters E, ?Hf and ?Sf are -3.8 kJ/mol, -14.16 J/mol and -161.44 J/(mol��K), respectively.
2) The optimized oxidation conditions are as follows: C5H5N is used as a solvent, reaction temperature is 60 ��, partial pressure of molecular oxygen is 2 MPa and molar ratio of the catalyst to substrate is 1:10. Under the optimized oxidation conditions the corresponding yield is 62.1%.
References
[1] FAN Qian, LI Yao-zhong, CHENG Pu-ming, et al. Synthesis of L-histidine Schiff base complex (Sal-His-Mn) and its catalytic behaviors in cyclohexene oxidation[J]. Journal of Sichuan University, 2001, 38(2): 230-234. (in Chinese)
[2] SUN Bin, CHEN Jun-ru, HU Jia-yuan, et al. Synthesis, oxygenation and catalytic performance of cobalt complexes with N,N'-bis(2-furaldehyde) diamine[J]. Acta Chimica Sinica, 2002, 60(9): 1613-1618. (in Chinese)
[3] NAN Guang-ming, CHEN Jun-ru, SHI Ying, et al. Synthesis, characterization and catalytic oxidation performance of didentate Schiff base and their Co(��), Mn(��) complexes[J]. Journal of Molecular Catalysis, 2002,16(3): 166-170. (in Chinese)
[4] WANG Rong-min, HAO Cheng-jun, WANG Yun-pu, et al. Amino acid Schiff base complex catalyst for effective oxidation of olefins with molecular oxygen[J]. Journal of Molecular Catalysis A: Chemical, 1999, 147: 173-178.
[5] ZHOU Zhi-ming, LI Lian-you, XU Qiao, et al. Bionic catalyst: Salen-metal complex catalyzed asymmetric epoxidation of the olefins[J]. Chinese Journal of Organic Chemistry, 2005, 25(4): 347-351. (in Chinese)
[6] MUKHERJEE S, SAMAN S, ROY B, et al. Efficient allylic oxidation of cyclohexene catalyzed by immobiled Schiff base complex using peroxides as oxidants[J]. Applied Catalysis A: General, 2006, 301(1): 79-88.
[7] LIU Feng-liang, LI Yuan-jian, HUANG Ke-long, et al. Synthesis, oxygenatin and catalytic behavior of manganese complex with bis(2-hydroxyphenyliminomethyl)[J]. Chinese Journal of Organic Chenistry, 2007, 27(9): 1106-1109. (in Chinese)
[8] PARISH E J, SUN H, KIZITO S A. Allylic oxidation of 5-steroids with pyridinium fluorochromate[J]. J Chem Res(Synopses), 1996(12): 544-545.
[9] BORA U, CHAUDHURI M K, DEY D, et al. 3,5- Dimethylpyrazolium fluorochromate (VI), C5H8N2H [CrO3F], (DmpzHFC): A convenient new reagent for oxidation of organic substrates[J]. Tetrahedron, 2001, 57: 2445-2448.
[10] MILLER R A, LI W, HUMPHREY G R. A ruthenium catalyzed oxidation of steroidal alkenes to enones[J]. Tetrahedron Letters, 1996, 37: 3429-3342.
[11] KIMURA M, MUTO T. On the reaction cholesteryl acetate with t-BuOOH in the presence of tri(acetonate) iron(��)[J]. Chem Pharm Bull, 1979, 27(1): 109-112.
[12] KIMURA M, MUTO T. Further studies on the oxy-functionlization of cholesteryl acetate with t-BuOOH and tri(acetonate) iron(��)[J]. Chem Pharm Bull, 1981, 29(7): 1862-1869.
[13] MARWAH P, LARDY HENRY A. Process for effecting allylic oxidation of allylic compounds using a combination of an alkali metal periodate and an alkyl hydroperoxide: WO, 9850409[P]. 1998-11-12.
[14] WAN Xue-shi, CHEN Xuan-hua. The kinetics of oxygen-adduct formation of Schiff base cobalt complex[J]. Journal of Central China Normal University, 1994, 28(3): 366-373. (in Chinese)
(Edited by YANG Hua)
Foundation item: Project(76132020) supported by the Post-Doctoral Science Fund of Central South University
Received date: 2007-03-02; Accepted date: 2007-04-18
Corresponding author: LIU Feng-liang, PhD; Tel: +86-13107210598; E-mail: lflcsu@yahoo.com.cn