Recovery of iron and rare earth elements from red mud through an acid leaching-stepwise extraction approach
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2019���2��
�������ߣ��ܿ��� ��ѩ�� ��ΰ ����Դ ��ӱ ������
����ҳ�룺458 - 466
Key words��red mud; iron; rare earth elements; leaching; solvent extraction
Abstract: A feasible approach to selectively recover iron and rare earth elements (REEs) from red mud through acid leaching-coordination-solvent extraction was proposed. The leaching efficiencies of Fe, Al, Ti, Sc, La, Ce, Nd and Y can reach up to 95.9%, 82.1%, 68.3%, 93.3%, 82.3%, 96.9%, 98.3% and 95.6%, respectively, under the optimal condition in the leaching process. Aliquat 336 showed excellent extraction performance of iron in chloride-rich solution, and the maximum extraction efficiency can reach over 96% in one time extraction while the loss of other metals was less than 10%, under the condition of Aliquat 336 concentration (v/v) of 30%, aqueous-organic ratio of 1.0 and extraction time of 20 min. Furthermore, P204 can effectively extract the scandium while Al and most other REEs remain in the aqueous phase. This approach may provide a new insight for the recovery of valuable resources from red mud.
Cite this article as: ZHANG Xue-kai, ZHOU Kang-gen, CHEN Wei, LEI Qing-yuan, HUANG Ying, PENG Chang- hong. Recovery of iron and rare earth elements from red mud through an acid leaching-stepwise extraction approach [J]. Journal of Central South University, 2019, 26(2): 458�C466. DOI: https://doi.org/10.1007/s11771-019-4018-6.

ARTICLE
J. Cent. South Univ. (2019) 26: 458-466
DOI: https://doi.org/10.1007/s11771-019-4018-6
ZHANG Xue-kai(��ѩ��), ZHOU Kang-gen(�ܿ���), CHEN Wei(��ΰ),LEI Qing-yuan(����Դ), HUANG Ying(��ӱ), PENG Chang-hong(������)
School of Metallurgy and Environment, Central South University, Changsha 410083, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Abstract: A feasible approach to selectively recover iron and rare earth elements (REEs) from red mud through acid leaching-coordination-solvent extraction was proposed. The leaching efficiencies of Fe, Al, Ti, Sc, La, Ce, Nd and Y can reach up to 95.9%, 82.1%, 68.3%, 93.3%, 82.3%, 96.9%, 98.3% and 95.6%, respectively, under the optimal condition in the leaching process. Aliquat 336 showed excellent extraction performance of iron in chloride-rich solution, and the maximum extraction efficiency can reach over 96% in one time extraction while the loss of other metals was less than 10%, under the condition of Aliquat 336 concentration (v/v) of 30%, aqueous-organic ratio of 1.0 and extraction time of 20 min. Furthermore, P204 can effectively extract the scandium while Al and most other REEs remain in the aqueous phase. This approach may provide a new insight for the recovery of valuable resources from red mud.
Key words: red mud; iron; rare earth elements; leaching; solvent extraction
Cite this article as: ZHANG Xue-kai, ZHOU Kang-gen, CHEN Wei, LEI Qing-yuan, HUANG Ying, PENG Chang- hong. Recovery of iron and rare earth elements from red mud through an acid leaching-stepwise extraction approach [J]. Journal of Central South University, 2019, 26(2): 458�C466. DOI: https://doi.org/10.1007/s11771-019-4018-6.
1 Introduction
Red mud is an alkaline solid waste generated in the process of alumina production [1]. Typically, 1.5�C2.5 t of red mud will be produced during the production of 1 t alumina [2], which leads to the total volume of red mud reached over 100 million tons annually all over the world [3]. Because of the large output of red mud and the difficulty in processing, there is no mature approach for dealing with such large quantities of red mud in industry [4�C6]. At present, red mud is mostly stockpiled by open-air damming, which not only takes huge amounts of land resource but also threats the ambient environment [7, 8]. On the other hand, red mud has also been considered a polymetallic resource rich in valuable metals including rare elements (Ti, V, Ga), rare earth elements (REEs, such as Sc, La, Ce, Y, Nd), iron and aluminum [9�C11]. Therefore, recovery of valuable metals from red mud not only brings economic benefits to the enterprises, but also helps to address the environmental issue, which is important for the sustainable development of alumina industry.
Researches about the utilization of red mud mainly focused on the production of construction materials [12, 13], preparation of new materials for environmental protection [14�C16], and recovery of valuable elements [17, 18]. However, the large- scale application of red mud as construction materials or environmental materials is restricted by the high alkalinity and salinity of red mud that may cause secondary pollution [7]. In the field of valuable metals recovery, most work only focused on the recovery of individual element, such as iron [19�C21], aluminum [22, 23], and scandium [24, 25]. Besides, problems remain to be solved, such as selective separation of iron, high recovery cost, and secondary treatment of waste residue/leachate. Therefore, the development of novel approaches for the recovery of multiple valuable metals from red mud is beneficial to the cost-effectiveness of the treatment strategy of red mud after addressing the problem of separation.
This work aimed at providing an economically feasible way to recover Fe, Al, Ti and REEs from the red mud. To achieve this, a coordination-solvent extraction process was proposed to selectively separate iron from red mud, and the recovery of iron and rare earth elements from red mud was explored through an acid leaching-stepwise extraction approach. Factors that may affect the leaching and extraction processes were investigated, and the optimal parameters were determined. Moreover, the leaching residues were also analyzed.
2 Materials and methods
2.1 Materials
Red mud was sampled from Guangxi Pinglu Group Co. Ltd., China, which was produced from Bayer process. The chemical composition of the red mud sample is presented in Table 1. The results indicated that the concentration of iron was thousand times higher than those of the rare earth elements such as lanthanum, cerium, neodymium, yttrium and scandium.
Aliquat 336, 2-Octanol, and P204 were purchased from Shanghai Macklin Biochemical Co. Ltd., China. Analytical grade HCl was purchased from Sinopharm Chemical Reagent Co., Ltd., China. Deionized water was used throughout the experiment.
2.2 Procedures and methods
2.2.1 Acid leaching
The experimental procedure is illustrated in Figure 1. According to the concentration of metals in red mud, the theoretical amount of hydrochloric acid for leaching a certain amount of red mud was calculated, and the effect of hydrochloric acid dosage was studied using the ratio of actual amount to theoretical amount.
Table 1 Chemical composition of red mud

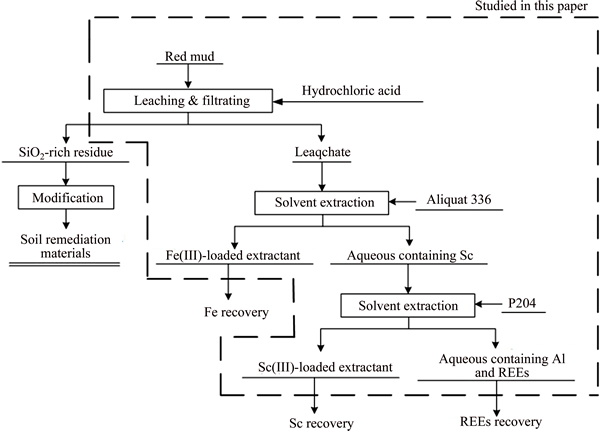
Figure 1 Experimental procedure for recovery of iron and REEs from red mud
Certain amounts of red mud and hydrochloric acid were added to a beaker and mixed with deionized water to the demanded liquid-to-solid ratio. Then the beakers were put in a magnetic stirrer at 300 r/min at different temperatures. The leaching time was fixed at 3 h to ensure leaching equilibrium based on our previous study. After leaching, the solution was filtered and the concentrations of metals were measured by inductively coupled plasma atomic emission spectrometry (ICP-AES, Thermo Scientific ICAP7400 Radial). The leaching efficiency (��) of metal was calculated as follows:
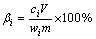 (1)
(1)
where m is the mass of red mud; V is the volume of leaching solution; ci and wi are the concentrations of metal (i) in the leaching solution and red mud, respectively.
The phase compositions of the red mud and the leaching residue were obtained by X�Cray diffraction (XRD, D/max 2500v/pc, Cu K�� radiation).
2.2.2 Solvent extraction
In the solvent extraction process, a certain amount of leaching solution was added to a separatory funnel with required volume of organic phase. In the extraction of iron, the organic phase included 15%(v/v) 2-Octanol to assist dissolving Aliquat 336 in kerosene. In the extraction of scandium, the organic phase consisted of P204 and kerosene. Then the separatory funnel was fixed and shaken in a vapour-bathing constant temperature vibrator for a certain time at room temperature (30 ��C). After phase disengagement, the aqueous phase was taken and analyzed by ICP-AES. The extraction efficiency (��) of metals was calculated according to Eq. (2).
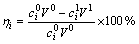 (2)
(2)
where 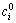 and
and 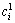 are the initial and equilibrium metal ion concentrations, respectively, and V 0 and V 1 are the volumes of aqueous phase before and after extraction, respectively.
are the initial and equilibrium metal ion concentrations, respectively, and V 0 and V 1 are the volumes of aqueous phase before and after extraction, respectively.
3 Results and discussion
3.1 Acid leaching of red mud
3.1.1 Effect of HCl dosage on leaching efficiencies of metals
The effect of HCl dosage on the leaching efficiencies of metals was studied under the following conditions: leaching temperature 85 ��C, leaching time 3 h and liquid-to-solid ratio 4 mL/g, and the results are shown in Figure 2. The results indicated that the leaching efficiencies of REEs increased with the HCl dosage increased from 80% to 130%. After that, the leaching efficiencies of REEs no longer changed with continuing increasing the HCl dosage. the maximum leaching efficiencies of Sc, La, Ce, Y and Nd were 98.6%, 90.5%, 98.8%, 94.5% and 94.8%, respectively, under 130% HCl dosage. On the other hand, the leaching efficiencies of Fe, Al and Ti increased from 41.0%, 72.5% and 42.9% to 93.2%, 82.1% and 75.6%, respectively, with the HCl dosage increased from 80% to 130%. The iron in the red mud mainly existed as hematite which was hard to be dissolved [26], so high concentration of HCl was required to achieve high leaching efficiency of iron. Therefore, the optimum HCl dosage was selected as 130%. At this point, the HCl acid concentration is 9.36 mol/L.
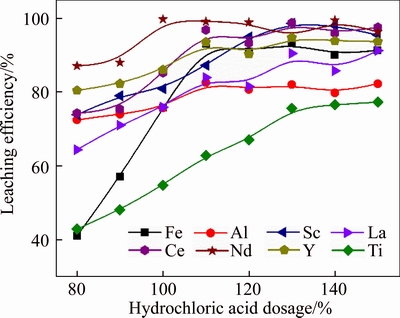
Figure 2 Effect of hydrochloric acid dosage on leaching efficiencies of metals
3.1.2 Effect of liquid-to-solid ratio on leaching efficiencies of metals
Liquid-to-solid ratio is a very important factor in a leaching process. Low liquid-to-solid ratio is beneficial for the enrichment of valuable metals and the reduction of the cost of installation. However, the slurry will be very viscous under low liquid-to- solid ratio condition which is difficult for agitating, and hydrochloric acid tends to volatilize at a high concentration. Therefore, the effect of liquid-to- solid ratio on the leaching efficiencies of metals was studied under the condition of HCl dosage of 130% for 3 h at 85 ��C, and the results are shown in Figure 3. The leaching efficiencies of all metals except Nd decreased with the increase of liquid-to- solid ratio, especially for the extraction of La and Ti, which decreased from 93.6% and 79.1% to 67.7% and 57.1%, respectively, with the liquid-to-solid ratio increased from 3 mL/g to 7 mL/g. While the HCl dosage was fixed, higher liquid-to-solid ratio will lead to lower HCl concentration, which will affect the leaching of metals. Therefore, the optimal liquid-to-solid ratio was 4 mL/g in full consideration of the metal leaching efficiency and the viscosity of the slurry.
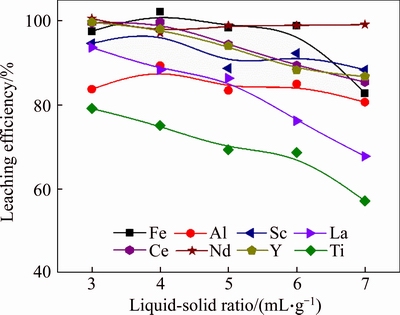
Figure 3 Effect of liquid-to-solid ratio on leaching efficiencies of metals
3.1.3 Effect of temperature on leaching efficiencies of metals
Figure 4 illustrates the effect of temperature on the leaching efficiencies of metals at HCl dosage 130% and liquid-to-solid ratio 4 mL/g. It was observed that temperature showed a significant effect on the leaching process. The leaching efficiencies of all metals increased with the increase of temperature, which may be due to the fact that high temperature will raise the chemical reactivity of the components in red mud [27]. However, higher temperature will increase the volatilization of hydrochloric acid, thus requiring corrosion- resistant equipment. At the leaching temperature 75 ��C, the leaching efficiencies of Fe, Al, Ti, Sc, La, Ce, Nd and Y were 95.9%, 82.1%, 68.3%, 93.3%, 82.3%, 96.9%, 98.3% and 95.6%, respectively.
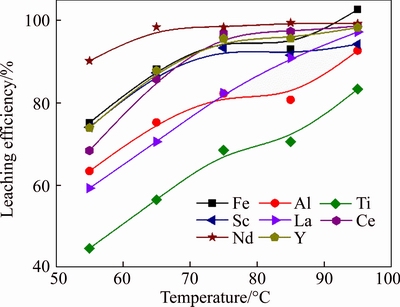
Figure 4 Effect of temperature on extraction efficiencies of metals
Further increasing temperature can still increase the leaching efficiencies, but with less extent, especially for Fe, Sc, Ce, Nd and Y. Therefore, the optimal temperature was 75 ��C.
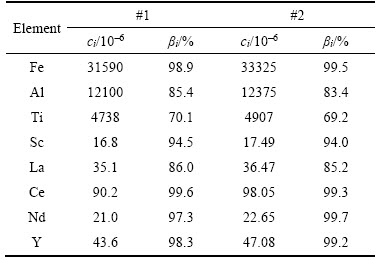
In order to verify the technological stability of the leaching process, two parallel extensive leaching experiments were conducted by using 800 g red mud under the optimal conditions: HCl dosage 130%, liquid-to-solid ratio 4 mL/g, leaching temperature 75 ��C and leaching time 3 h. The results are shown in Table 2, indicating that the leaching process was stable.
Table 2 Results of parallel extensive leaching experiments
The XRD patterns of the red mud and the leaching residue are presented in Figure 5.Figure 5(a) shows that hematite (Fe2O3), sodium iron sulfur (Na3FeS3), katoite (Ca3Al2(SiO4)(OH)3) and calcium aluminum iron silicate hydroxide (Ca3AlFe(SiO4)(OH)3) were the main crystal mineral phases in the red mud. After leaching, those four phases disappeared, corresponding well to the results presented in Table 2 that nearly all the iron and calcium were leached by HCl, while aluminum and titanium remaining in the residue mainly existed as diaspore (AlOOH) and anatase (TiO2), respectively (Figure 5(b)). Nearly all the silicon kept staying in the residue as quartz, which can be used as the silicon source for soil remediation materials [28].
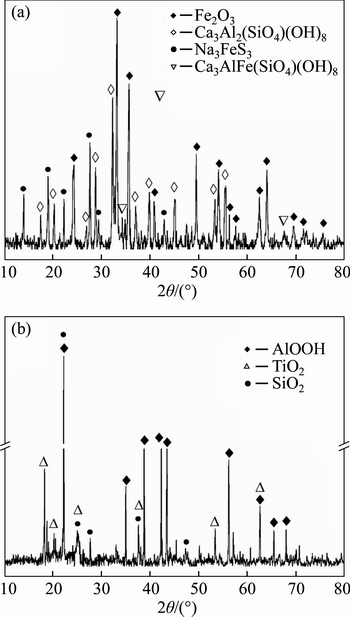
Figure 5 XRD patterns of red mud (a) and leaching residue (b)
3.2 Coordination-solvent extraction of iron
According to Table 2, valuable metals in red mud were mainly dissolved into the leachate after HCl leaching, but the concentrations of low-value- added metals, i.e., iron and aluminum, were thousand times higher than those of REEs. Therefore, how to separate metals from leachate, especially high-value-added REEs, is the key in recovering valuable metals from red mud. Considering that the concentrations of REEs were very low in the leachate, a solvent extraction process was proposed to enrich the REEs. However, the large amount of Fe(III) in the system would affect the extraction process since Fe(III) showed strong association ability with P204 and P507, both of which have been widely used as extractants for REEs. Therefore, separation of Fe(III) is necessary before the extraction of REEs.
Since a large amount of hydrochloric acid was used in the leaching process, the chloride concentration in the leachate was very high (over 8.0 mol/L). Studies have shown that Fe3+ is easily to coordinate with Cl�C to form a series of complexes, and Fe(III) mainly exists as FeCl3 and FeCl4�C in high chloride concentration [29�C31]. Meanwhile, other metals especially for REEs show weak coordination ability with Cl�C, and mainly exist as cation ions [32, 33]. Therefore, Aliquat 336, which is equipped with a quaternary ammonium and an exchangeable chloride ion, may be used to selectively extract the Fe(III) species while REEs remain in the liquid phase.
3.2.1 Effects of Aliquat 336 concentration on extraction efficiencies of metals
The effect of Aliquat 336 concentration (v/v) on the extraction efficiencies of metals in one extraction stage was studied under the condition of Aqueous/Organic (A/O) ratio of 1 and extraction time of 10 min, and the results are shown in Figure 6. The extraction efficiency of iron increased from 35.6% to 95.7% with the extractant concentration (v/v) increased from 10% to 30% and kept constant when continuing raise Aliquat 336 concentration. Meanwhile, Al and REEs showed a weak extraction ability with Aliquat 336, and the extraction efficiencies changed little with Aliquat 336 concentration (v/v), and less than 10% of REEs were extracted even with 50% of Aliquat 336 concentration. Therefore, the optimal concentration (v/v) of Aliquat 336 was selected as 30% for the subsequent experiments.
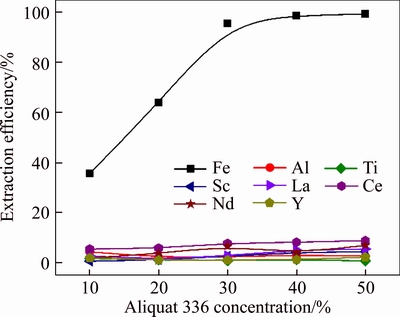
Figure 6 Effect of Aliquat 336 concentration on extraction efficiencies of metals in one extraction stage
3.2.2 Effect of phase ratio on extraction efficiencies of metals
Figure 7 illustrates the effect of A/O ratio on the extraction efficiencies of metals in one extraction stage at fixed Aliquat 336 concentration 30% and extraction time 10 min. The results indicated that A/O ratio could strongly affect the extraction efficiency of iron, which was up to 98.8% at A/O ratio 0.5, and decreased rapidly to 21.0% when the A/O ratio increased to 4. A/O ratio had little impact on the extraction of other metals (kept below 10%). Thus, the O/A ratio was fixed at 1, and the extraction efficiency of iron was over 96% in one extraction stage.
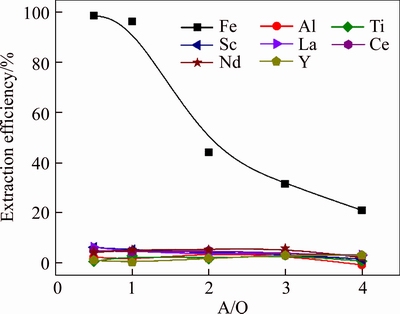
Figure 7 Effect of phase ratio on extraction efficiencies of metals in one extraction stage
3.2.3 Effect of extraction time on extraction efficiencies of metals
The effect of extraction time from 5 min to 25 min on the extraction efficiencies of metals was investigated under the conditions of Aliquat 336 concentration (v/v) 30% and A/O ratio 1, and the results are presented in Figure 8. Figure 8 shows that the extraction reaction of iron was very fast, and the extraction efficiency could achieve over 86% within 5 min, and increased slightly to 98.6% by continuing to extend the time to 20 min. Therefore, the extraction time was chosen as 20 min. The concentration of iron decreased from 32.3 g/L to 0.6 g/L, which will not affect the subsequent extraction of REEs. High concentration of iron in the organic phase could be stripped by deionized water and recovered to produce iron red or ferric chloride.
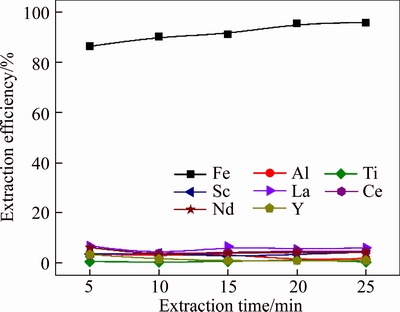
Figure 8 Effect of extraction time on extraction efficiencies of metals in one extraction stage
3.3 Solvent extraction of scandium
The compositions of the extraction raffinate after the extraction of iron are listed in Table 3. Among all the REEs, scandium was the most valuable element, so the feasibility of selective extraction of Sc was studied using P204 as the extractant. The effect of P204 concentration (v/v) on the extraction efficiencies of metals was studied under the condition of A/O ratio 2 and extraction time 10 min, and the results are presented in Figure 9. As shown in Figure 9, P204 can effectively extract Sc even with only 5% concentration, and the extraction efficiency in one stage can reach near 100%. Ti and Y can also be extracted by P204, and the extraction efficiencies increased from 78.6% and 66.1% to 96.9% and 98.9% when the P204 concentration increased from 5% to 25%, respectively. Al and other REEs showed a weak extraction ability with P204, which was helpful for the selective separation of Sc from Al and other REEs. The separation of Sc from Ti and Y in the organic phase can be achieved through different stripping agents [34, 35]. After the extraction of Sc, the leachate contains large amount of Al and small parts of REEs. Considering that Al(OH)3 can be dissolved in high concentration of NaOH solution to produce NaAlO2 while rare earth hydroxide will stay in the residue, a neutralization precipitation-alkali leaching process can be used to recover Al and other REEs in future studies.
Table 3 Chemical composition of extraction raffinate after extraction of iron
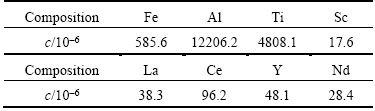
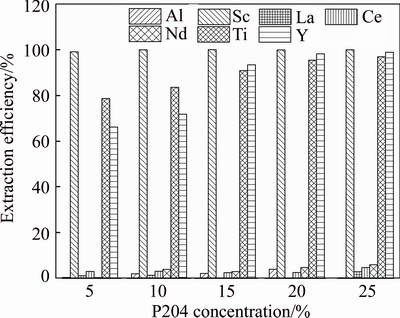
Figure 9 Effect of P204 concentration on extraction efficiencies of metals in one extraction stage
4 Conclusions
1) A novel approach of recovering iron and REEs from red mud was proposed by high concentration hydrochloric acid leaching followed with coordination-solvent extraction of iron by Aliquat 336, and P204 was used as the extractant to enrich scandium.
2) The leaching efficiencies of Fe, Al, Ti, Sc, La, Ce, Nd and Y were up to 95.9%, 82.1%, 68.3%, 93.3%, 82.3%, 96.9%, 98.3% and 95.6%, respectively, under the optimal conditions of HCl dosage of 130%, liquid-solid ratio of 4 mL/g, leaching temperature of 75 ��C and leaching time of 3 h.
3) Aliquat 336 showed strong extraction ability to iron since the Fe(III) existed as FeCl3 and FeCl4�C in the leachate. The extraction efficiency of iron can reach over 96% in one extraction stage under the condition of Aliquat 336 concentration (v/v) of 30%, O/A ratio of 1, leaching time of 20 min, while the loss of other metals was less than 10%. The concentration of iron was below 0.6 g/L in the aqueous phase which would not affect the subsequent extraction of REEs.
4) P204 can effectively extract Sc even with only 5% concentration, and the extraction efficiency in one stage was near 100%. Furthermore, the extraction efficiencies of Ti and Y increased with the increasing of P204 concentration (v/v) in the organic phase. Al and other REEs showed a weak extraction ability with P204.
References
[1] XUE Sheng-guo, KONG Xiang-feng, ZHU Feng, HARTLEY W, LI Xiao-fei, LI Yi-wei. Proposal for management and alkalinity transformation of bauxite residue in China [J]. Environmental Science and Pollution Research, 2016, 23(13): 12822�C12834. DOI: 10.1007/s11356- 016-6478-7.
[2] KONG Xiang-feng, TIAN Tao, XUE Sheng-guo, HARTLEY W, HUANG Long-bin, WU Chuan, LI Chu-xuan. Development of alkaline electrochemical characteristics demonstrates soil formation in bauxite residue undergoing natural rehabilitation [J]. Land Degradation & Development, 2018, 29(1): 58�C67. DOI: 10.1002/ldr.2836.
[3] WANG Meng, HU Hui-ping, LIU Jin-wei, CHEN Qi-yuan. Negative effect of dissolved organic compounds on settling behavior of synthetic monominerals in red mud [J]. Journal of Central South University, 2016, 23(7): 1591�C1602. DOI: 10.1007/s11771-016-3213-y.
[4] LIU Zhao-bo, LI Hong-xu. Metallurgical process for valuable elements recovery from red mud��A review [J]. Hydrometallurgy, 2015, 155: 29�C43. DOI: 10.1016/ j.hydromet.2015.03.018.
[5] KONG Xiang-feng, JIANG Xing-xing, XUE Sheng-guo, HUANG Ling, HARTLEY W, WU Chuan, LI Xiao-fei. Migration and distribution of saline ions in bauxite residue during water leaching [J]. Transactions of Nonferrous Metals Society of China, 2018, 28(3): 534�C541. DOI: 10.1016/ S1003-6326(18)64686-2.
[6] ZHU Feng, HOU Jing-tao, XUE Sheng-guo, WU Chuan, WANG Qiong-li, HARTLEY W. Vermicompost and gypsum amendments improve aggregate formation in bauxite residue [J]. Land Degradation & Development, 2017, 28(7): 2109�C2120. DOI: 10.1002/ldr.2737.
[7] XUE Sheng-guo, ZHU Feng, KONG Xiang-feng, WU Chuan, HUANG Ling, HUANG Nan, HARTLEY W. A review of the characterization and revegetation of bauxite residues (red mud) [J]. Environmental Science and Pollution Research, 2016, 23(2): 1120�C1132. DOI: 10.1007/s11356- 015-4558-8.
[8] ZHU Feng, LIAO Jia-xin, XUE Sheng-guo, HARTLEY W, ZOU Qi, WU Hao. Evaluation of aggregate microstructures following natural regeneration in bauxite residue as characterized by synchrotron-based X-ray micro-computed tomography [J]. Science of the Total Environment, 2016, 573: 155�C163. DOI: 10.1016/j.scitotenv. 2016.08.108.
[9] ZHU Feng, CHENG Qing-yu, XUE Sheng-guo, LI Chu-xuan, HARTLEY W, WU Chuan. TIAN Tao. Influence of natural regeneration on fractal features of residue microaggregates in bauxite residue disposal areas [J]. Land Degradation and Development, 2018, 29(1): 138�C149. DOI: 10.1002/ldr.2848.
[10] XUE Sheng-guo, YE Yu-zhen, ZHU Feng, WANHG Qiong-li, JIANG Jun, HARTLEY W. Changes in distribution and microstructure of bauxite residue aggregates following amendments addition [J]. Journal of Environmental Sciences, 2019, 78: 276�C286. DOI: 10.1016/j.jes.2018.10.010.
[11] LIU Wan-chao, YANG Jia-kuan, XIAO Bo. Review on treatment and utilization of bauxite residues in China [J]. International Journal of Mineral Processing, 2009, 93(3, 4): 220�C231. DOI: 10.1016/j.minpro.2009.08.005.
[12] XUE Sheng-guo, LI Meng, JIANG Jun, MILLAR G J, LI Chu-xuan, KONG Xiang-feng. Phosphogypsum stabilization of bauxite residue: Conversion of its alkaline characteristics [J]. Journal of Environmental Sciences, 2019, 77: 1�C10. DOI: 10.1016/ j.jes.2018.05.016.
[13] VINCENZO M S, RENZO C, STEFANO M, GIOVANNI C, MARZIO M, GEROLAMO B, GIORGIO C. Bauxite ��red Mud�� in the ceramic industry. Part 1: Thermal behaviour [J]. Journal of the European Ceramic Society, 2000, 20(3): 235�C244. DOI: 10.1016/S0955-2219(99)00088-6.
[14] LIU Jin-wei, HU Hui-ping, WANG Meng, CHEN Xiang-pan, CHEN Qi-yuan, DING Zhi-ying. Synthesis of modified polyacrylamide with high content of hydroxamate groups and settling performance of red mud [J]. Journal of Central South University, 2015, 22(6): 2073�C2080. DOI: 10.1007/ s11771-015-2731-3.
[15] LIAO Jia-xin, JIANG Jun, XUE Sheng-guo, CHENG Qing-yu, WU Hao, RAJENDRAN M, HARTLEY W, HUANG Long-bin. A novel acid-producing fungus isolated from bauxite residue: the potential to reduce the alkalinity [J]. Geomicrobiology Journal, 2018, 35(10): 840�C847. DOI: 10.1080/01490451.2018.1479807.
[16] ZHU Feng, ZHOU Jia-yi, XUE Sheng-guo, HARTLEY W, WU Chuan, GUO Ying. Aging of bauxite residue in association of regeneration: a comparison of methods to determine aggregate stability & erosion resistance [J]. Ecological Engineering, 2016, 92: 47�C54. DOI: 10.1016/ j.ecoleng.2016.03.025.
[17] LI Guang-hui, LIU Ming-xia, RAO Ming-jun, JIANG Tao, ZHUANG Jin-qiang, ZHANG Yuan-bo. Stepwise extraction of valuable components from red mud based on reductive roasting with sodium salts [J]. Journal of Hazardous Materials, 2014, 280: 774�C780. DOI: 10.1016/j.jhazmat. 2014.09.005.
[18] DENG Bo-na, LI Guang-hui, LUO Jun, YE Qing, LIU Ming-xia, PENG Zhi-wei, JIANG Tao. Enrichment of Sc2O3 and TiO2 from bauxite ore residues [J]. Journal of Hazardous Materials, 2017, 331: 71�C80. DOI: 10.1016/j.jhazmat.2017. 02.022.
[19] YANG Yang, WANG Xue-wen, WANG Ming-yu, WANG Hua-guang, XIAN Peng-fei. Recovery of iron from red mud by selective leach with oxalic acid [J]. Hydrometallurgy, 2015, 157: 239�C245. DOI: 10.1016/j.hydromet.2015.08.021.
[20] YANG Yang, WANG Xue-wen, WANG Ming-yu, WANG Hua-guang, XIAN Peng-fei. Iron recovery from the leached solution of red mud through the application of oxalic acid [J]. International Journal of Mineral Processing, 2016, 157: 145�C151. DOI: 10.1016/j.minpro.2016.11.001.
[21] LI Bo-ji, XIE Ying-bang, FAN Yan-jin, LIU Chen, HE Hang-jun. Recovery of aluminum from blast furnace slag of red mud ironmaking process [J]. Hydrometallurgy of China, 2015, 34(4): 328�C330. (in Chinese)
[22] WU Wen-yuan, LI Dong, ZHAO Zhi-hua, CHEN Jian-li, ZHANG Feng-yun, YIN Shao-hua, QIAN Mei-li, BIAN Xue. Formation mechanism of micro emulsion on aluminum and lanthanum extraction in P507-HCl system [J]. Journal of Rare Earths, 2010, 28(s1): 174�C178. DOI: 10.1016/S1002- 0721(10)60314-6.
[23] VACHON P, TYAGI R D, AUCLAIR J C, WILKINSON K J. Chemical and biological leaching of aluminum from red mud [J]. Environmental Science & Technology, 1994, 28(1): 26�C30. DOI: 10.1021/es00050a005.
[24] DITZE A, KONGOLO K. Recovery of scandium from magnesium, aluminum and iron scrap [J]. Hydrometallurgy, 1997, 44(1): 179�C184. DOI: 10.1016/S0304- 386X(96)00041-2.
[25] AKCIL A, AKHMADIYEVA N, ABDULVALIYEV R, ABHILASH, MESHRAM P. Overview on extraction and separation of rare earth elements from red mud: focus on scandium [J]. Mineral Processing and Extractive Metallurgy Review, 2017, 39(3): 1�C7. DOI: 10.1080/08827508.2017. 1288116.
[26] CHANG Yong-feng, ZHAI Xiu-jing, LI Bin-chuan, FU Yan. Removal of iron from acidic leach liquor of lateritic nickel ore by goethite precipitate [J]. Hydrometallurgy, 2010, 101(1, 2): 84�C87. DOI: 10.1016/j.hydromet.2009.11.014.
[27] LIU Zhi-xiong, YIN Zhou-lan, CHEN Yi-guang, XIONG Li-zhi. Leaching kinetics of molybdenum from Ni-Mo ore in sulfuric acid solution with sodium peroxodisulfate as oxidant [J]. Journal of Central South University, 2015, 22(3): 874�C879. DOI: 10.1007/s11771-015-2596-5.
[28] ZHU Qing. Stabilization on cadmium soil using modified silicon activated red mud based material [D]. Changsha: Hunan Agricultural University, 2016. (in Chinese)
[29] CUI Li, CHENG Fang-qin, ZHOU Jing-fang. Behaviors and mechanism of iron extraction from chloride solutions using undiluted Cyphos IL 101 [J]. Industrial & Engineering Chemistry Research, 2015, 54(30): 7534�C7542. DOI: 10.1021/acs.iecr.5b01546.
[30] HILL P S, SCHAUBLE E A, YOUNG E D. Effects of changing solution chemistry on Fe3+/Fe2+ isotope fractionation in aqueous Fe-Cl solutions [J]. Geochimica Et Cosmochimica Acta, 2010, 74(23): 6669�C6689. DOI: 10.1016/j.gca.2010.08.038.
[31] HILL P S, SCHAUBLE E A, SHAHAR A, TONUI E, YOUNG E D. Experimental studies of equilibrium iron isotope fractionation in ferric aquo-chloro complexes [J]. Geochimica Et Cosmochimica Acta, 2009, 73(8): 2366�C2381. DOI: 10.1016/j.gca.2009.01.016.
[32] LANGE N A, DEAN J A, Lange's Handbook of Chemistry (16th Edition) [M]. New York: McGraw-Hill, 2005: 687�C688.
[33] XU Guang-xian, YUAN Cheng-ye. Solvent extraction of rare earth elements [M]. Beijing: Science Press, 2010: 27�C28. (in Chinese)
[34] ZHU Zhao-wu, ZHANG Wen-sheng, CHENG Chu-yong. A literature review of titanium solvent extraction in chloride media [J]. Hydrometallurgy, 2011, 105(3, 4): 304�C313. DOI: 10.1016/j.hydromet.2010.11.006.
[35] LI Yu-hua, LI Qing-gang, ZHANG Gui-qing, ZENG Li, CAO Zuo-ying, GUAN Wen-juan, WANG Liu-pei. Separation and recovery of scandium and titanium from spent sulfuric acid solution from the titanium dioxide production process [J]. Hydrometallurgy, 2018, 178: 1�C6. DOI: 10.1016/j.hydromet.2018.01.019.
(Edited by YANG Hua)
���ĵ���
���-�ֲ���ȡ���ӳ����л�������ϡ��
ժҪ�������һ�֡����-��λ��ȡ��ѡ���Ի��ճ����е�����ϡ�����¹��ա��������̱��������Ż������£�Fe��Al��Ti��Sc��La��Ce��Nd��Y�Ľ����ʷֱ�ɴ�95.9%��82.1%��68.3%��93.3%��82.3%��96.9%��98.3%��95.6%��Aliquat 336�ڸ�����ϵ�±��ֳ����õ��������ܣ���30% Aliquat 336 +15%������+55%ú��Ϊ��ȡ�л���Խ���Һ�е���������ȡ�������Ϊ1����ȡʱ��Ϊ20 min�������£���������ȡ�ʿɴ�95.7%����ϡ��Ԫ����ʧ�ʲ���10%������ϡ���нϺõķ���Ч������P204��������Һ�е��ֽ��л��գ����Խ��ָ�������ȡ������Al�ʹ�ϡ��Ԫ�ط��롣
�ؼ��ʣ����ࣻ����ϡ��Ԫ�أ��������ܼ���ȡ
Foundation item: Project(21707167) supported by the Natural Science Foundation of China
Received date: 2018-10-26; Accepted date: 2018-12-12
Corresponding author: ZHOU Kang-gen, PhD, Professor; E-mail: zhoukg63@163.com; ORCID: 0000-0002-7914-6277

