J. Cent. South Univ. Technol. (2009) 16: 0563-0568
DOI: 10.1007/s11771-009-0094-3
Effect of stirring on preparation of hollow copolymer particles by alkali/cooling method
LI Hai-pu(李海普)1, OKUBO M2, SUZUKI T2
(1. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China;
2. Graduate School of Science and Technology, Kobe University, Kobe 657-8501, Japan)
Abstract: Hollow particles were prepared by the treatment of styrene-methacrylic acid copolymer particles with alkali/cooling method. The influences of stirring position (in aqueous phase or at the interface of O/W) and stirring speed (90, 110 and 240 r/min) on the formation of hollow particles were investigated. It is found that the soft stirring in aqueous phase at 90 r/min leads to the formation of monohollow particles, while the violent stirring at the interface of O/W and 240 r/min gives non-hollow products. In contrast, the weak stirring in aqueous phase at 110 r/min results in sterically heterogeneous dispersion of methacrylic acid-rich regions within the original particles, and hence the formation of multihollow particles. Further investigation indicates that the change of stirring efficiency provides a way to tune the diffusion behavior of monomer styrene, and therefore influences the distribution of methacrylic acid units in the original particles as well as the morphology of the treated particles.
Key words: hollow polymer particle; stirring effect; morphology; emulsion copolymerization
1 Introduction
Hollow polymer particles have attracted extensive attention for their unique core/shell structure and acquired interesting application in the coatings, ink, cosmetics, and paper industries [1-2]. Up to now, several methods have been developed to prepare hollow particles, such as W/O/W emulsion polymerization [2], Shirasu porous glass membrane emulsification [3], selective removal of core [4], self-assembly of phase-separated polymer method [5-7], alkali/acid method [8], solvent evaporation method [9], and controlled phase separation techniques [10-11]. Besides, an alkali/cooling method was proposed to prepare multihollow particles with facile operation [12], and related studies were carried out to examine the influences of molecular weight, particle diameter, cooling condition [13], partial neutralization condition [14], and emulsifiers [15] on the formation of multihollow particles. In addition, the results in Ref.[16] showed that the stirring condition also played a decisive role. However, so far there has been no detailed information about the stirring study on the formation of multihollow polymer particles.
It was very recently reported that the repeatable and reproducible stirring conditions could be well established by the rational design of distinct stirring profiles concerning the stirrer type, stirring position and stirring speed [16]. Similarly, in this work, a half-moon stirrer was applied and exactly placed in aqueous phase or at the interface of O/W, while the related stirring speed was set at 90, 110 and 240 r/min, respectively. The influences of different stirring profiles on the formation of multihollow styrene(St)-methacrylic acid (MAA) copolymer [P(St-MAA)] particles by alkali/cooling method were investigated.
2 Experimental
2.1 Materials
Commercial St and MAA were purified by distillation under reduced pressure in a nitrogen atmosphere. Potassium persulfate (KPS) of analytical grade (Nacalai Tesque Inc, Kyoto, Japan) was purified by recrystallization. Polyoxyethylene nonylphenyl ether nonionic emulsifier (Emulgen 911, HLB 13.7) in commercial grade with averages of 10.9 ethylene oxides per molecule was used as received (Kao Co., Tokyo, Japan). Deionized water was distilled prior to use.
2.2 Emulsion copolymerization
P(St-MAA) particles were prepared by batchwise emulsion copolymerization in a 1 L reactor equipped with an inlet of N2, a reflux condenser, and a half-moon type stirrer [16]. A solution of Emulgen 911 (4.0 g) in 10 mL water was charged into the reactor, and then purged with N2 at 70 ℃ for 30 min. St (55.0 g) and MAA (5.0 g) were added to this reactor.
The initial working status corresponding to three stirring profiles before starting emulsion was presented in Fig.1, assigned to soft stirring (Fig.1(c)), weak stirring (Fig.1(d)) and violent stirring (Fig.1(e)), respectively.
The resultant mixture was stirred for 30 min using pre-designed stirring profiles. After workup, the copolymerization was initiated by the addition of KPS aqueous solution under the previous stirring condition.
2.3 Post-treatments
The methanol treatment and alkali/cooling treatment were performed by the following procedures described in Refs.[12, 14, 16-17].
2.4 Measurements
The solid contents of emulsion were measured by gravimetry. Transmission electron microscope (TEM) observation was performed on a JEOL JEM-1230 electron microscope.
MAA conversions were determined by gas chromatography (GC-18A, Shimazu, Kyoto, Japan) with helium as carrier gas, N, N-dimethylformamide as solvent and p-xylene as internal standard. St conversions were calculated by subtracting the mass of MAA units from the total mass of the copolymer in the emulsion.
The concentration of St in growing particles was also determined by gas chromatography after the removal of monomer droplets from emulsion by centrifugation at 3 000 r/min for 3 min.
The amount of MAA units on the particle surface was estimated by measuring calorific value of the neutralization reaction between the carboxyl groups and an excess amount of KOH with isothermal titration calorimeter (ITC-4200, Calorimetry Science Corp, Utah, USA) at 25 ℃ [18]. The number of carboxyl groups on the particle surface was accordingly calculated based on the solid content, BET specific surface area and the result of ITC.
3 Results and discussion
3.1 Preparation of hollow particles
The alkali/cooling method was employed to prepare hollow particles at the initial pH values of 10 and 13, respectively. The morphologies of the original and the corresponding alkali/cooling treated P(St-MAA) particles are shown in Fig.2. In all three cases, original particles exhibit a solid core (Figs.2(a)-(c)), however, the differentiation among them after the treatment with alkali/cooling is noticeably observed.
It is seen from Figs.2(e) and (h) that the treated particles obtained via weak stirring are monodisperse and evidently multihollow. In particular, those with an initial pH value of 13 present an eyeball-like style (Fig.2(h)). However, as shown in Figs.2(d) and (g), the treated particles produced under soft stirring are polydisperse. The majority of the larger ones exhibit the low contrast region in the center, representing the inner mono-hollow. Again, monodisperse particles are obtained under violent stirring, but have a non-hollow structure (Figs.2(f) and (i)). It is also interesting to note that the surface of particles treated at an initial pH value of 10 is fairly smooth (Figs.2(d)-(f)).
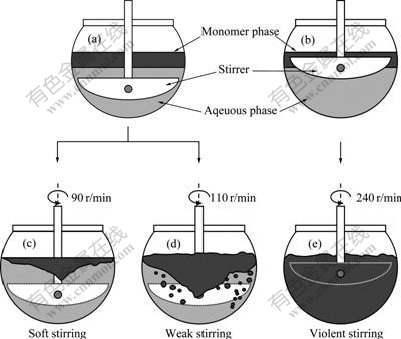
Fig.1 Stirring profiles in standstill ((a), (b)) and at initial working status before starting emulsion ((c)-(e)) (Monomer phase was dyed with oil blue)
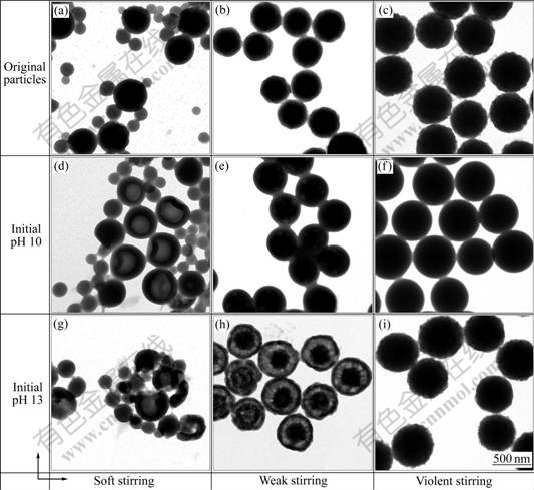
Fig.2 TEM images of original P(St-MAA) particles prepared under soft (a), weak (b), and violent (c) stirring and corresponding alkali/cooling treated particles with initial pH values of 10 (d)-(f) and 13 (g)-(i), respectively
3.2 Conversions of monomers St and MAA
It is well known that the mechanical stirring has a profound effect on the kinetics of copolymerization reactions [19-21], generally by prompting the mixing of reactants and improving the efficiency of reaction. In this work, simply changing the stirring efficiency even makes it possible to form mono-hollow, multihollow and non-hollow particles by the same treatment. To clarify this diversification, the conversions of monomers St and MAA were examined.
As shown in Fig.3, the conversions of St (η(St)) and MAA (η(MAA)) increase uniformly with the increase of time under all stirring conditions. A further examination reveals that the time for the completion of copolymerization of MAA is drastically prolonged with the increase of stirring efficiency in the order of 2 h (Fig.3(a)), 5 h (Fig.3(b)) and more than 8 h (Fig.3(c)).
The conversion difference of two monomers (η(St)-η(MAA)) is indicative of an obviously higher conversion of MAA than that of St, under soft/weak stirring, especially in the early stage of polymerization (Figs.3(a), and (b)), which is in accordance with previous observation [16]. Under violent stirring, the conversion of MAA exceeds that of St slightly in the early 3 h, and then becomes slower but is still comparable relative to that of its competitor St (Fig.3(c)). Taking the dynamic growth of P(St-MAA) particles into account, it can be proposed that the difference of conversions between monomers MAA and St in different stages of copolymerization can affect the steric distribution of these two components in P(St-MAA) particles. That is to say, the faster conversion of MAA in early stage of copolymerization possibly results in the MAA-rich core, while the faster conversion of MAA in final stage leaves MAA more chances to copolymerize on the surface of particles and therefore affords MAA-rich shell. This idea is supported by the determination of the distribution of MAA units on the surface and inside of P(St-MAA) particles at the completion of the polymerization.
3.3 MAA units on surface of particles
As listed in Table 1, the amounts of carboxyl groups
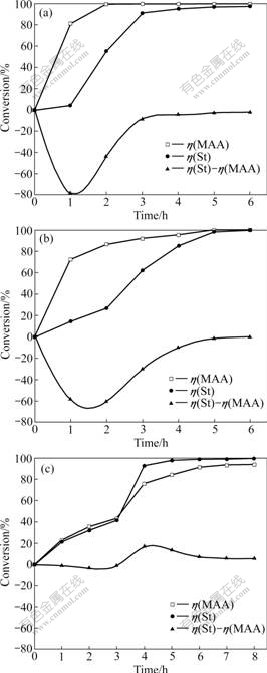
Fig.3 Conversions of MAA and St with time under soft (a), weak (b), and violent (c) stirring
Table 1 Carboxyl groups on surface of original particles prepared using different stirring profiles

of MAA on the surface of particles are found to increase with the increase of stirring efficiency.
This result provides adequate information to characterize the distribution of MAA in shell of particles. It can be deduced that under soft/weak stirring MAA units are relatively deficient in the shell due to the preceding enrichment of MAA in core, whereas under violent stirring MAA is prone to accumulate in the shell because of the retarded copolymerization afterward.
3.4 MAA distribution inside particles
The methanol treatment was conducted to examine the distribution of MAA units inside particles. The morphologies of methanol-treated particles are shown in Fig.4. The products obtained via soft stirring clearly displays the inner hollow (Fig.4(a)), which is caused by the removal of MAA-rich copolymer during the methanol treatment. As seen in Fig.4(b), the voids left by

Fig.4 TEM images of methanol-treated P(St-MAA) particles prepared under soft (a), weak (b) and violent (c) stirring
MAA-rich copolymer are dispersed in the treated particles prepared under weak stirring. In view of this characteristic, their original particles are highly suitable for the preparation of multihollow ones, as proved before (see Figs.2(e) and (h)).
In contrast, Fig.4(c) exhibits a homogeneity inside the treated particles, which is quite similar to the observation in their precursors produced under violent stirring (Fig.2(c)), so as to exclude the possibility for swelling into detectable voids during the post-treatment by alkali/cooling procedure.
The results from methanol treatment indicate that the soft/weak stirring favors the formation of the MAA-rich core, while the violent stirring usually gives the homogeneous core.
3.5 Concentration of St in growing particles
The fact that MAA units own a facile access to water but acquired a noticeable variation of steric distribution in P(St-MAA) particles under different stirring profiles promotes us to consider the role of monomer St during the copolymerization. As shown in Fig.5, the concentration of monomer St in the growing particles is significantly improved by increasing the stirring efficiency.

Fig.5 Concentration of monomer St in growing particles under different stirring conditions
Under violent stirring, the concentration of St is near to its equilibrium swelling (5.2 mol/L) when the conversions locate in the range of 20%-35%. This indicates that the violent stirring corresponds to efficient transfer of St and provides sufficient St in the early stage of its copolymerization with MAA, which results in a copolymer with relatively homogeneous MAA units in core. This deduction is consistent with the observation in Fig.4(c).
Under weak/soft stirring, the concentrations of St are far lower than its equilibrium swelling. As a result, the copolymerization proceeds under St-starved condition, so MAA is consumed in advance and MAA-rich copolymer is formed in the initial stage (Figs.4(a) and (b)).
It is evidenced that in the current system the increase of stirring efficiency most likely accelerates the diffusion speed of monomer St from monomer phase to copolymerization loci, which should in turn discourage the conversion of MAA to some degree and then vary the steric distribution of MAA inside or on the surface of the P(St-MAA) particles.
4 Conclusions
(1) The pre-profiled stirring proves to be feasible to affect the diffusion behavior of monomer St, so as to influence the distribution of MAA units in original P(St-MAA) particles as well as the morphologies of particles after alkali/cooling treatment.
(2) The soft stirring corresponds to less efficient diffusion of monomer St and then produces the original particles with the MAA-rich core and MAA-deficient shell, from which the monohollow particles are obtained.
(3) The violent stirring furnishes the monomer St with higher but comparable conversion relative to MAA, so as to afford the original particles with the homogeneous core and MAA-rich shell, which results in non-hollow particles.
(4) The original particles obtained via weak stirring with the proper stirring efficiency feature a sterically heterogeneous dispersion of MAA-rich regions inside, and practically lead to multihollow particles.
References
[1] KOWALSKI A, VOGEL M, BLANKENSHIP R M. Sequential heteropolymer dispersion and a particulate material obtainable therefrom, useful in coating compositions as a thickening and/or opacifying agent: US, 4468498 [P]. 1984.
[2] KIM B S, KIM J W, SUH K D. Poly (methyl methacrylate) multihollow particles by water in oil in water emulsion polymerization [J]. Journal of Applied Polymer Science, 2000, 76(1): 38-44.
[3] LIU R, MA G H, MENG F T, SU Z G. Preparation of uniform-sized PLA microcapsules by combining Shirasu porous glass membrane emulsification technique and multiple emulsion-solvent evaporation method [J]. Journal of Controlled Release, 2005, 103(1): 31-43.
[4] LI G L, LIU G, KANG E T, NEOH K G, YANG X L. pH-responsive hollow polymeric microspheres and concentric hollow silica microspheres from silica-polymer core-shell microspheres [J]. Langmuir, 2008, 24(16): 9050-9055.
[5] MINAMI H, OKUBO M, OSHIMA Y. Preparation of cured epoxy resin particles having one hollow by polyaddition reaction [J]. Polymer, 2005, 46(4): 1051-1056.
[6] ITO M, FURUKAWA Y, MINAMI H, OKUBO M. Preparation of micrometer-sized, monodisperse, hollow polystyrene/poly(ethylene glycol dimethacrylate) particles with a single hole in the shell [J]. Colloid and Polymer Science, 2008, 286(11): 1335-1341.
[7] MINAMI H, KOBAYASHI H, OKUBO M. Preparation of hollow polymer particles with a single hole in the shell by SaPSeP [J]. Langmuir, 2005, 21(13): 5655-5658.
[8] OKUBO M, ITO A, HASHIBA A. Production of submicron-sized multihollow polymer particles having high transition temperatures by the stepwise alkali/acid method [J]. Colloid and Polymer Science, 1996, 274(5): 428-432.
[9] OKUBO M, MINAMI H. Formation mechanism of micron-sized monodispersed polymer particles having a hollow structure [J]. Colloid and Polymer Science, 1997, 275(10): 992-997.
[10] OKUBO M, KONISHI Y, MINAMI H. Production of hallow particles by suspension polymerization of divinylbenzene with nonsolvent [J]. Aqueous Polymer Dispersions, 2004, 124: 54-59.
[11] YIN W S, YATES M Z. Effect of interfacial free energy on the formation of polymer microcapsules by emulsification/freeze-drying [J]. Langmuir, 2008, 24(3): 701-708.
[12] OKUBO M, ITO A, KANENOBU T. Production of submicron-sized multihollow polymer particles by alkali/cooling method [J]. Colloid and Polymer Science, 1996, 274(8): 801-804.
[13] OKUBO M, ITO A, NAKAMURA M. Effect of molecular weight on the production of multi-hollow polymer particles by the alkali/cooling method [J]. Colloid and Polymer Science, 1997, 275(1): 82-85.
[14] OKUBO M, OKADA M, SHIBA K. Heterogeneity among multihollow polymer particles prepared by the alkali/cooling method under partial neutralization conditions [J]. Colloid and Polymer Science, 2002, 280(9): 822-827.
[15] OKADA M, MATOBA T, OKUBO M. Influence of nonionic emulsifier included inside carboxylated polymer particles on the formation of multihollow structure by the alkali/cooling method [J]. Colloid and Polymer Science, 2003, 281(2): 193-197.
[16] OKUBO M, CHAIYASAT A, YAMADA M, SUZUKI T, KOBAYASHI H. Influence of hydrophilic-lipophilic balance of nonionic emulsifiers on emulsion copolymerization of styrene and methacrylic acid [J]. Colloid and Polymer Science, 2007, 285(15): 1755-1761.
[17] CHAIYASAT A, KOBAYASHI H, OKUBO M. Incorporation of nonionic emulsifier inside methacrylic polymer particles in emulsion polymerization [J]. Colloid and Polymer Science, 2007, 285(5): 557-562.
[18] OKUBO M, SUZUKI T, TSUDA N. Estimation of distribution state of carboxyl groups within submicron-sized, carboxylated polymer particle with isothermal titration calorimeter [J]. Colloid and Polymer Science, 2006, 284(11): 1319-1323.
[19] NOMURA M, HARADA M, EGUCHI W, NAGATA S. Effect of stirring on the emulsion polymerization of styrene [J]. Journal of Applied Polymer Science, 1972, 16(4): 835-847.
[20] ?ZDE?ER E, SUDOL E D, EL-AASSER M S, KLEIN A. Role of mixing in copolymerizations of styrene and n-butyl acrylate [J]. Journal of Applied Polymer Science, 1998, 69: 2277-2289.
[21] RAMIREZ J C, HERRERA-ORDONEZ J, MALDONADO- TEXTLE H. Kinetics of the styrene emulsion polymerization above CMC (II): Agitation effect on molecular weight [J]. Polymer Bulletin, 2005, 53(5): 333-337.
(Edited by CHEN Wei-ping)
Foundation item: Project supported by China Scholarship Council; Project(09JJ3100) supported by Hunan Provincial Natural Science Foundation of China
Received date: 2008-10-31; Accepted date: 2008-12-20
Corresponding author: LI Hai-pu, PhD; Tel: +86-731-88830603; E-mail: lihaipu@mail.csu.edu.cn