J. Cent. South Univ. Technol. (2010) 17: 211-217
DOI: 10.1007/s11771-010-0032-4 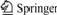
Synthesis and biochemical properties of fluorescent/magnetic bifunctional starch particles
WANG Feng-hua(王凤华)1, LIU Jun(刘俊)1, 2, TANG Dong-ying(唐冬英)1,
XUE Chang-gang(薛昌刚)1, XIAO Su-yao(肖苏尧)1, ZHENG Yuan-qing(郑元青)1,
TONG Chun-yi(童春义)1, WANG Ling-ling(王玲玲)3, LIU Xuan-ming(刘选明)1
1. College of Life Science and Biotechnology, Hunan University, Changsha 410082, China;
2. College of Materials Science and Engineering, Hunan University, Changsha 410082, China;
3. School of Physics and Microelectronics Science, Hunan University, Changsha 410082, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2010
Abstract: Magnetic starch particles (MSPs) were synthesized in water-in-oil microemulsion at room temperature. MSPs were characterized by transmission electron microscopy (TEM), Fourier transform infrared spectrometry (FTIR), zeta potential system, thermogravimetric analysis (TGA) and vibrating sample magnetometry (VSM). The average diameter of the MSPs was 220 nm, dispersed with well-proportioned size and magnetic resonance, the saturation magnetization was 3.64 A・m2/kg. MSP was coated with poly-L-lysine (PLL), and then the surface of PLL-MSP was combined with fluorescein isothiocynate (FITC). Results show that fluorescent/magnetic starch particles (FMSPs) are of stable photo-bleaching capability compared with free FITC, with low bio-toxicity and certain function of magnetic separation. It is expected that FMSPs are bifunctional nano-materials including fluorescence labelling and magnetic separation.
Key words: starch-particles; nano-materials; synthesis; fluorescent properties; magnetic properties; biochemical properties
1 Introduction
Iron oxide nanoparticles are of great interest for superparamagnetism, suitable for bio-compatible coatings and for applications in cell isolation and purification, immunoassay, targeting drug delivery and immobilization and transport of protein molecules [1-2]. But magnetic nanoparticles have no labelling capability themselves.
Fluorescent labelling is often employed in bioassays as a means to detect targeted biomolecules. This is partly due to the inherently high sensitivity of fluorescence- based technology and the relative accessibility of the technique [3]. Small molecule organic dye as fluorescent probes has been widely used in diagnose tests and molecular imaging. But most of the probes are not stable and easily disturbed by the background of fluorescent signal of the sample itself [4]. Nano-probe technology such as luminescent quantum dots and fluorescent latex nanoparticles could overcome some disadvantages of organic fluorescent probes [5]. But luminescent quantum dots are limited in application to bio-medicine because of low water-solubility, low quantum yield, and noxious compounds of Cd [6]. So, organic macromolecule and inorganic nanospheres as the base material loaded with fluorescent molecules have attracted much attention.
Thus, separation and labelling are two very important techniques in the modern bio-medicine engineering. If they can be accomplished simultaneously, imaging detection in life science will be developed more extensively.
As a natural biomaterial, starch is a traditional filling agent that has been used in curatorial field for a long time [7-9]. ALEXIOU et al [10] reported that ferrofluids coated with starch polymers could be used as biocompatible carriers and such magnetic particles could be enriched in a desired body compartment using an external magnetic field. HARRIS et al [11] obtained estimates of blood-brain barrier permeability following spontaneous reperfusion, subsequent to forebrain ischemia by unilateral carotid injection of ferrofluids starch microspheres. As starch-nanoparticle is biocompatible and biodegradable [12-15] fluorescent/ magnetic bifunctional starch-nanoparticle is synthesized for the application in bio-medicine.
In this work, magnetic starch particles (MSPs) were synthesized in water-in-oil microemulsion at room temperature, and their biochemical properties, such as structure character, grain identity, thermal stability and magnetic resonance property were investigated. Based on these researches, poly-L-lysine (PLL) and fluorescein isothiocynate (FITC) were combined on the surface of MSPs (St-Fe2O3NP:MSP) to synthesize fluorescent/ magnetic bifunctional starch particle (FMSP) probes. FMSPs are of stable fluorescence property, perfect anti-photobleaching capability, nontoxic, clear imaging and certain function of magnetic separation in cells.
2 Experimental
2.1 Materials and equipment
Starch (St) was obtained from China Medicine (Group) Shanghai Chemical Reagent Corp. PLL, FITC and fluram were obtained from Sigma Corp, USA. Other regents were of analytical grade. Tumor cell lines MCF-7 and Hela were supplied by our laboratory.
Transmission electron microscope (TEM, JEOL-1230, JEOL, Japan), Zetasizer Nano ZS (Malvern, UK), Fourier transform infrared spectrometer (FTIR, Beijing Rayleigh Analytical Instrument Corp, China), vibrating sample magnetometer (VSM, HH-15, Jiangsu, China), spectrofluorophotometer (F-2500, Hitach, Japan), thermal analyzer (STA449C, Netzsch, Germany); inverted fluorescence microscope (TE2000u, Nikon, Japan)), and ultrapure water systems (Millipore, USA) were used.
2.2 Synthesis of magnetic starch particles (St-Fe2O3NP: MSPs)
Firstly, 1 g of soluble starch was put into water, and then the acquired aqueous solution with content of 10% (mass fraction) was heated and hydrolyzed in the boiling water bath until the solution was clarified. Toluene and chloroform were mixed at volume fraction of 3:1, and at the same time, 2% of Span 80 was added and rotated at 1 000 r/min to form the oil phase. Starch solution and magnetic fluid were mixed to form the water phase and then put into the oil phase (V(oil)/V(H2O)=20/1) and mixed for a certain time to form microemulsion. Next, 0.05% cross-linker agent POCl3 was added and mixed for 1 h as follows: the microemulsion was broken by ethanol, washed with dilute hydrochloric acid solution once, and then with acetone and ethanol alternately three times. The useless liquor was removed by centrifugation. Eventually, the precipitation product was obtained by freeze-drying. In the same way, starch nanoparticles (StNP) were prepared while only starch solution was used as the water phase.
2.3 Fourier transform infrared spectrometry (FTIR) measurement
The chemical structure of freeze-dried sample (StNP and MSP) was measured by FTIR with pressing potassium bromide (KBr) troche.
2.4 Transmission electron microscopy (TEM) measurement
Fe2O3NP, StNP and MSP were suspended in ethanol, and then dripped on copper grid after ultrasonic dispersion. Subsequently, the copper grid was dried under incandescent lamp, and the morphology, size and distribution of the particles were determined by TEM.
2.5 Zetasizer nano ZS measurement
Fe2O3NP and MSP were suspended in demineralized water, and then their average diameter, distribution were determined by Zetasizer Nano ZS.
2.6 Determination of thermal properties
TGA curve of MSP, compared with that of starch nanoparticles (StNP), was determined in the same conditions of thermal analyzer.
2.7 Magnetism analysis
Saturation magnetization of MSP with diverse proportion starch solution and magnetic fluid was determined by VSM.
2.8 FITC labeled and determination of photo- bleaching property of FMSP
1 mg PLL-MSP was dispersed in 1 mL phosphate buffer saline (PBS) completely, and certain amount of FITC ethanol solution was added. After the mixture was vibrated for 4 h without light, the useless liquor was removed by centrifugation; the deposit was washed by demineralized water several times until there was no absorption at 480 nm by UV-spectroscopy and then lyophilized. The products were fluorescent/magnetic starch nanoparticles (FMSPs).
The powder of FMSP was determined in an inverted fluorescence microscope. 1 mg FMSP was dispersed in 1 mL PBS completely, and then determined on spectro- fluorophotometer every 5 min.
2.9 Determination of biological toxicity by 3-(4, 5-di- methythiazol-2-yl)-2,5-diphenyltetrazolium(MTT)
The prepared FMSP sample, natural starch and MSP were put in distilled water and treaded individually through filtering by germproofing filter. Mammary cancer cells were plated in 96-well culture dishes with a density of 1×104 cell/dish, and 90 μL complete culture medium was added to each dish. Cells were cultured for 24 h with 5% CO2 at 37 ℃. The pretreated particles were added to the culture medium, and the final concentrations were 5, 10, 50, 100 and 500 mg/L, respectively after the cell line being incubated with 5% CO2 for 24 h at 37 ℃. MTT method was used to detect the cell death ratio.
2.10 Experiment of live cell imaging
The prepared FMSP sample was put in distilled water and treaded individually through filtering by germproofing filter. Hela cells were plated in 3-well culture dishes with a density of 1×104 cell/dish. 1 mL complete culture medium was added to each dish. PLL-MSP was added to one dish. 0.3 mL pretreated FMSP was added to other well cells, and a piece of magnet was put on the right side of one well 4 h later, 3-well cells were washed by culture medium and then put on the inverted fluorescence microscope in order to observe the cell imaging.
3 Results and discussion
3.1 Synthesis principle of FMSP
The mixture of starch solution and magnetic fluid (Fig.1(a)) to form water phase was wrapped by the oil phase. The reaction liquid was stirred fast to form microemulsion, and then joined by POCl3 to obtain MSP (Fig.1(b)). PLL was coated on the surface of MSP through electrostatic-attraction (Fig.1(c)) to form PLL-MSP and thus there were a lot of primary amine groups on the surface. Then FMSPs could be obtained by the reaction between the isothiocyanate group of FITC and the primary amine groups of the PLL-MSP (Fig.1(d)). Fig.1(c) shows that FMSP is expected to be a kind of material that will have a wide range of applications in bio-medicine.
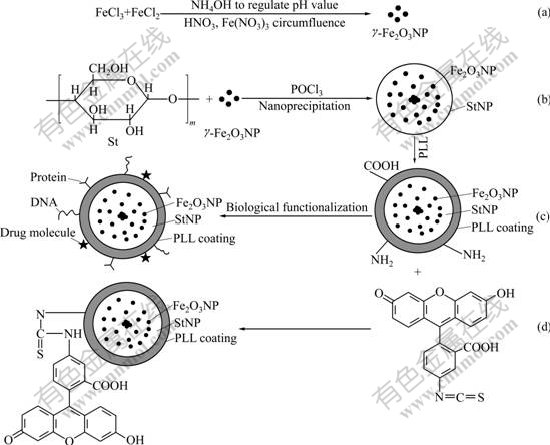
Fig.1 Schematic illustration of preparation and functionalization of FMSP: (a) Forming process of γ-Fe2O3NP; (b) Forming process of MSP; (c) Forming process of PLL-MSP; (d) Forming process of FMSP
3.2 FTIR spectra
As shown in Fig.2, there exists a single strong absorption at 579 cm-1 due to Fe2O3, indicating that magnetic particle is coated in StNP successfully. Simultaneously, StNP prepared by our laboratory is used as a contrast.
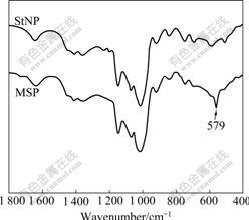
Fig.2 FTIR spectra of StNP and MSP
3.3 TEM images
The appearances of Fe2O3NP, StNP and MSP were checked by TEM (Fig.3). All the particles are well proportioned, and dispersed with average sizes of 15, 200, and 220 nm, respectively. Fig.3(d) shows that, compared with Fig.3(b), Fe2O3NP is enchased in StNP successfully.
Fig.3 TEM images of Fe2O3NP (a), StNP (b) and MSP (c, d)
3.4 Zetasizer Nano ZS measurement of MSP
Fig.4 shows the diameter and size distribution of Fe2O3NP and MSP determined by Zetasizer Nano ZS. The size of Fe2O3NP (Fig.4(a)) ranges from 5 to 20 nm, and the average diameter is 15 nm. The size of MSP (Fig.4(b)) ranges from 120 to 300 nm, and the average diameter is 220 nm.
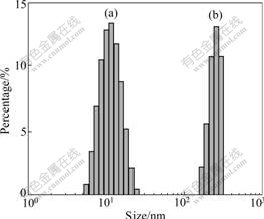
Fig.4 Size distribution of nanoparticles: (a) Fe2O3NP; (b) MSP
3.5 Thermal analysis
Fig.5 displays the TGA curves of MSP and StNP. StNP is found to be decomposed completely when temperature rises up to 900 ℃, and the content of Fe2O3NP residue can be detected. Fig.5 also shows that the mass fraction of Fe2O3NP in MSP is about 17.6%.
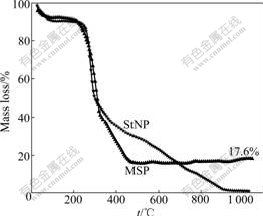
Fig.5 TGA curves of StNP and MSP
3.6 Magnetism analysis
Fig.6 demonstrates the hysteresis loop of MSP under the condition of diverse mass ratios of St to Fe2O3NP. MSP shows a superparamagnetic property, and the saturation magnetization varies with the mass ratio of St to Fe2O3NP (Table 1).
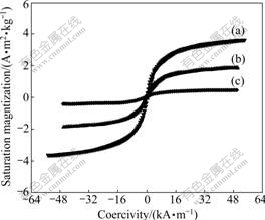
Fig.6 Hysteresis loop of MSP with different mass ratios of St to Fe2O3 NP: (a) 27: 1; (b) 50:1; (c) 200:1
Table 1 Results of magnetism parameter of MSP

3.7 FITC labeled and photo-bleaching property of FMSP
The excitation and emission spectra of FMSP in PBS are shown in Fig.7. The emission spectrum excited by 490 nm shows a typical wide band with a peak at about 520 nm. The positions of excitation and characteristic emission peaks of FMSP are similar to those of FITC. This proves that FITC is labeled on the surface of PLL-MSP successfully.
Fig.8 illustrates that FITC is decomposed easily when it is excited for 50 min, and the fluorescence intensity of FITC is 45% of the original, but that of FMSP is 85%, indicating that compared with FITC, FMSP has stronger anti-photobleaching property and higher photo-stability.
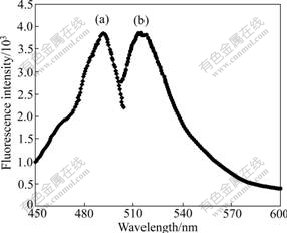
Fig.7 Excitation (a) and emission (b) fluorescence spectra of FMSP dissolved in PBS
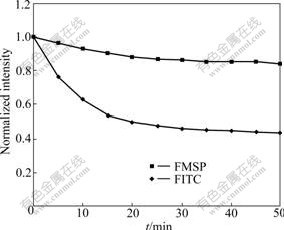
Fig.8 Stability test of FITC in different states
Fig.9 shows that FMSP is well dispersed and has good fluorescence property. In contrast, there is no green fluorescence of MSP that is not modified with FITC.

Fig.9 Image of FMSP under fluorescence microscope
3.8 Biological toxicity
As shown in Fig.10, the death ratio of the cells is close to 0 with 5 mg/L FMSP in cell culture, suggesting that FMSP is non-toxic in low concentration. When the concentration rises to 100 mg/L, the death ratio is up to 4%, and that of starch (control) is close to 3%. When the concentration rises to 500 mg/L, the death ratio for FMSP is 17%. In contrast, the death ratios of the cells for the starch (St) and MSP are 14% and 16%, respectively. Therefore, the biological toxicity of the FMSP resembles that of the natural starch, which can be a natural biomaterial and widely used as a drug and gene carrier.
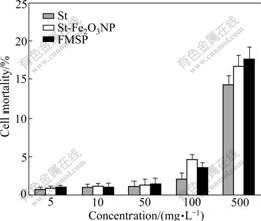
Fig.10 Biological toxicity of particles
3.9 Live cell imaging
Live cell images are shown in Fig.11. There is a bright green fluorescence in the tumor cells enclosed FMSPs (Figs.11(b) and (e)), whereas there is no green fluorescence in the tumor cells which phagocyted PLL-MSP (Figs.11(a) and (d)). These indicate that FMSPs can be phagocyted by the living cells. When a piece of magnet is put on the right side of a culture dish, the tumor cells will be attracted to the same side (Figs.11(c) and (f)). This shows that FMSP has certain function of magnetic separation.

Fig.11 Live cell images of PLL-MSP and FMSP phagocytosed by tumor cells (Images (a), (b) and (c) are taken in white light while images (d), (e) and (f) in blue light exciting): (a), (d) PLL-MSP phagocytosed; (b), (e) FMSP phagocytosed; (c), (f) FMSP phagocytosed with action of magnet
4 Conclusions
(1) Soluble starch and Fe2O3 nanoparticles are used as raw materials and POCl3 as a cross-linking agent, and the magnetic starch particles are prepared in water-in-oil microemulsion at room temperature. The particles possess obvious magnetic resonance property, and are uniform in size about 220 nm and well dispersed.
(2) PLL and FITC are coated on the surface of MSP to synthesize FMSPs. FMSPs are proved to have stable fluorescence property, perfect anti-photo-bleaching capability, low biological toxicity, and can be used as a fluorescence probe to label the targeted cells, trace the molecules metabolic process, or to localize the cell organelles.
(3) The residual primary amino groups and positive groups of the surface of FMSP can be combined with drug molecules, DNA, protein, RNA or other molecules. Due to the superparamagnetic property, FMSP can be used to separate or choose the specific cells, and direct sorts of molecules to targeted position in an external magnetic field. Therefore, FMSP will be a potential target and trace carrier in a wide range application.
References
[1] KATZ E, WILLNER I. Integrated nanoparticle-biomolecule hybrid systems: Synthesis, properties, and applications [J]. Angewandte Chemie: International Edition, 2004, 43(45): 6042-6108.
[2] GRANCHAROV S G, ZENG H, SUN S H, WANG S X, O'BRIEN S, MURRAY C B, KIRTLEY J R, HELD G A. Bio-functionalization of monodisperse magnetic nanoparticles and their use as biomolecular labels in a magnetic tunnel junction based sensor [J]. Journal of Physical Chemistry B, 2005, 109(26): 13030-13035.
[3] SWIFT J L, CRAMB D T. Nanoparticles as fluorescence labels: Is size all that matters? [J]. Biophysical Journal, 2008, 95(2): 865- 876.
[4] TRAUET D, YANG W J, SEYDACK M, CARUSO F, YU N T, RENNEBERG R. Nanoencapsulated microcrystalline particles for superamplified biochemical assays [J]. Analytical Chemistry, 2002, 74(21): 5480-5486.
[5] CHAN W C W, NIE S M. Quantum dot bioconjugates for ultrasensitive nonisotopic detection [J]. Science, 1998, 281(5385): 2016-2018.
[6] NIEMEYER C M. Nanoparticles, proteins, and nucleic acids: Biotechnology meets materials science [J]. Angewandte Chemie: International Edition, 2001, 40(22): 4128-4158.
[7] TANG R P, DU Y M, FAN L H. Dialdehyde starch-crosslinked chitosan films and their antimicrobial effects [J]. Journal of Polymer Science Part B: Polymer Physics, 2003, 41(9): 993-997.
[8] WANG H P, WEI R Q, SHEN B, LIU X N, WEI P, ZHOU H, OUYANG P K. Study on flexible immobilized papain with dialdehyde starch [J]. China Journal Bioprocess Engineering, 2004, 2(1): 25-29.
[9] ONISHI H, NAGAI T. Characterization and evaluation of dialdehyde starch as an erodible medical polymer and a drug carrier [J]. Journal of Pharmaceutics, 1986, 30(2/3): 133-141.
[10] ALEXIOU C, ARNOLDA W, HULINA P, KLEINB R J, RENZC H, PARAKD F G, BERGEMANNE C, LUBBEF A S. Magnetic mitoxantrone nanoparticle detection by histology, X-ray and MRI after magnetic tumor targeting [J]. Journal of Magnetism and Magnetic Materials, 2001, 225(1/2): 187-193.
[11] HARRIS N G, GAUDEN V, FRASER P A, WILLIAMS S R, PARKER G J. MRI measurement of blood-brain barrier permeability following spontaneous reperfusion in the starch microsphere model of ischemia [J]. Journal of Magnetic Resonance Imaging, 2002, 20(3): 221-230.
[12] XIAO S Y, TONG C Y, LIU X M, YU D M, LIU Q L, XUE C G, TANG D Y, ZHAO L J. Preparation of folate conjugated starch nanoparticles and its application to tumor-targeted drug delivery vector [J]. Chinese Science Bulletin, 2006,51(14): 1693-1697.
[13] XIAO S Y, LIU X M, TONG C Y, LIU J, TANG D Y, ZHAO L J. Studies of poly-L-lysine-starch nanoparticle preparation and its application as gene carrier [J]. Science in China, Series B: Chemistry, 2005, 48(2): 162-166.
[14] LIU J, LIU X M, XIAO S Y, TONG C Y, TANG D Y, ZHAO L J. Bioconjugated nanoparticle for DNA protection from ultrasound damage [J]. Analytical Sciences, 2005, 21(3): 193-195.
[15] LIU J, WANG F H, WANG L L, XIAO S Y, TONG C Y, TANG D Y, LIU X M. Preparation of fluorescence starch-nanoparticle and its application as plant transgenic vehicle [J]. Journal of Central South University of Technology, 2008, 15(6): 768-773.
Foundation item: Project(200501) supported by the “985” Program of China
Received date: 2009-08-05; Accepted date: 2009-12-10
Corresponding author: LIU Xuan-ming, PhD, Professor; Tel: +86-731-88821721; E-mail: sw_xml@hnu.cn
(Edited by CHEN Wei-ping)