
Rheological and diffusion behaviours of Zr41Ti14Cu12.5Ni10Be22.5
bulk metallic glass around glass transition
WANG Jing-feng(王敬丰)1, HU Yao-bo(胡耀波)1, QIN Bin(覃 彬)2,
LIU Tian-mo(刘天模)1, PAN Fu-sheng(潘复生)1
1. College of Materials Science and Engineering, Chongqing University, Chongqing 400044, China;
2. College of Chemistry and Chemical Engineering, Chongqing University, Chongqing 400044, China
Received 26 October 2005; accepted 23 December 2005
Abstract:The viscosity of bulk metallic glass(BMG) Zr41Ti14Cu12.5Ni10Be22.5(Vit1) around glass transition was derived from the static extension method. In addition, the diffusion coefficient of the medium size atom Fe in the BMG Vit1 was calculated by using the Stokes-Einstein relation. The temperature dependence of both viscosity and diffusion coefficient of the atom Fe, could also be well fitted by two different Arrhenius equations at temperatures below and above the glass transition temperature. In the glass state, the effects of heating rate on viscosity and diffusivity were significant due to the structure relaxation. Besides, it was indicated that the linear plots between lnD0 and ED for above and below glass transition are just the same. The results suggest that the intrinsic diffusion of medium size atoms in BMGs, above and below glass transition, is possibly collective hopping processes.
Key words: bulk metallic glass; Zr41Ti14Cu12.5Ni10Be22.5; glass transition; viscosity; diffusivity.
1 Introduction
Since the Au-Si metallic glass were firstly prepared by rapid cooling technique in 1960 by DUWEZ et al[1], the study of structure and property of metallic glass has received much attention. The atom transportation behaviors (rheological and diffusion processes), which are closely linked to structure relaxation, phase separation and thermal stability, were investigated by a lot of theoretical and experimental scientists in the past two decades. The viscosity and diffusivity measurements can only be carried out in glass state, while in deeply undercooled liquid state they are not available because conventional metallic glasses have poor thermal stability. Up to recent decade, a series of multicomponent metallic alloys[2-5] with superior glass forming ability and superduper thermal stability against crystallization provide an experimental time scale which is sufficient to perform measurements of atomic transport phenomena like diffusion and viscosity in the supercooled liquid state.
The first viscosity study on BMG was reported by BAKKE et al[6]. This article reports the shear viscosity of Zr46.75Ti8.25Cu7.5Ni10Be27.5 (Vit4) in the vicinity of the glass transition using parallel plate rheometry. These data were supplemented by BUSH et al[7] using three-point beam bending in the same temperature region and can be described well with the Vogel-Fulcher-Tammann(VFT) relation. It is shown that the Vit4 BMG is a strong liquid, similar to sodium silicate liquid. The equilibrium viscosity of BMG Vit1 determined by WANIUK et al[8] using isothermal three-point beam-bending measurement and showed that this alloy is a strong liquid like other bulk metallic glasses. By static extension method, it was detected that the rheological behavior of BMG Vit1, with increasing temperature, changes from elasticity to anelasticity, finally to the Newtonian flow above Tg[9].
Most of the diffusion studies focus on the possible influence of the glass transition on the diffusivities and the diffusion mechanisms. GEYER et al[10] firstly studied the diffusion of atom Be in BMG Vit1, and argued that the diffusion of Be can be interpreted in terms of a single atomic jump diffusion in both glass and supercooled liquid states, although the temperature dependence of diffusion coefficient in the temperature regions below and above glass transition could not be described by the same Arrhenius equation. It was discovered with nuclear magnetic resonance(NMR) by TANG et al[11] that the relation between temperature and jump rate of the small atom Be obeys the same Arrhenius equation, unlike the Arrhenius behavior of diffusivity having a kink around glass transition. It was suggested that the glass transition did not cause visible change of atom jump rate. However, TANG et al[11] suggested that the diffusion of atom Be at below and above glass transition have two distinct diffusion processes, i.e. single atom hopping and collective motion. For medium size atoms, such as Co, Fe, Ni, the kink around glass transition in Arrhenius plot of diffusivity, was interpreted in terms of a single atomic jump diffusion in glass state, but collective hopping motion in undercooled liquid state [12,13]. The previous investigations have demonstrated that atomic diffusion behaviors in metallic glasses are quite complicated and still far from being understood. Further more, the available results do not provide a uniform picture of the diffusion mechanism and often seem to be conflicting. So the diffusion mechanism of BMGs deserves to be deliberated ulteriorly.
The diffusion coefficient of the medium size atom Fe in the BMG Vit1 was reported by FELITZ et al[14]. For contrast, in this article, the temperature dependence of diffusivity of the medium size atom Fe around glass transition was calculated by using Stokes-Einstein relation, and compared with experimental results reported in Ref.[14]. In this paper the correlation between viscosity and diffusion was studied, in order to educe the intrinsic atom diffusion mechanism in BMGs.
2 Experimental
The glassy bulk metallic ingots, with composition of Zr41Ti14Cu12.5Ni10Be22.5, were prepared by melting a mixture of elements of purity range from 99.5% to 99.9% under a titanium-gettered argon atmosphere, then the amorphous rods with diameter of 14 mm were prepared by water quenching in silica tube. The amorphous nature of the as-quenched rods was ascertained by X-ray diffractometry(XRD). The experimental samples 10 mm×1 mm×0.5 mm were cut from the amorphous rods. In order to eliminate the structure difference, the experimental samples were firstly heated above the glass transition to 633 K for 10 min. The amorphous nature of the preannealed samples was approved with the XRD again.
The static extension measurement of amorphous sample was carried out on Perkin Elmer Pyris DMA7e dynamic thermal mechanical analyzer(DMA) under flowing argon atmosphere. The sample with load of 800 mN was heated respectively at the rate of 2.5, 5, 10 and 15 K/min, the sample length and strain were recorded synchronously. The length accuracy of this experiment is ±50 nm. The furnace of the DMA was calibrated for temperature with high purity indium and zinc with accuracy of ±(1-2) K. The viscosity at different heating rate was derived from the method in Ref.[9].
 (1)
(1)
where F is the applied load, V0 is the original volume, l is the instantaneous length with the temperature change under the invariableness load, and is the strain rate.
is the strain rate.
The diffusivity, D, can be calculated based on the Stokes-Einstein relation as,
 (2)
(2)
where η is the viscosity, kB is the Boltzmann constant, and r is the atomic radius of Fe which is 0.124 nm.
3 Results and discussion
3.1 Influence of structure relaxation on viscosity
The metallic glass is a thermodynamic metastable state, the structure is going to transform continuously to equilibrious state, namely the structure relaxation takes place. All equilibrium viscosity data of the glass-forming systems can be described well with the VFT relation. However, often over a narrower temperature range, especially near Tg, the temperature dependence of viscosity can also be approximated very well by an Arrhenius equation[15], and is given by
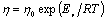 (3)
(3)
where the pre-factor, η0 is the viscosity when the temperature goes to infinity, Ev is an activation energy and R is the gas constant.
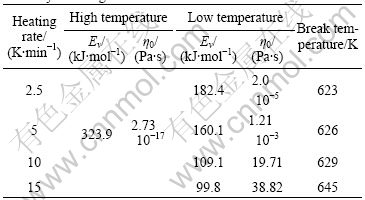
The viscosity and its Arrhenius relation of BMG Vit1 around glass transition at different heating rate are shown in Fig.1. It can be found that the temperature dependence of viscosity at every heating rate cannot be fitted by a single Arrhenius equation above and below the glass transition. Below Tg, the Arrhenius plots of viscosity at different heating rate are different, but above Tg they are the same. These parameters, effective activation energy Ev and pre-factor η0 of the Arrhenius relation of viscosity around glass transition of BMG Vit1, are collected in Table 1. The Ev and η0 above Tg are 323.9 kJ/mol and 2.73×10-17 Pa?s, respectively. Below Tg, Ev and η0 are 99.8 kJ/mol and 38.82 Pa?s, 109.1 kJ/mol and 19.71 Pa?s, 160.1 kJ/mol and 1.21×10-3 Pa?s, 182.4 kJ/mol and 2.0×10-5 Pa?s for 15, 10, 5 and 2.5 K/min, respectively. From the above-mentioned results, we found that the slower the heating rate is, the higher the viscosity and activation energy are, and the closer the Arrhenius equation in glass state is to that in supercooled liquid state. This attributes to structure relaxation.
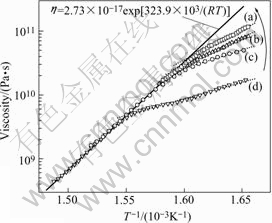
Fig.1 Viscosity and its Arrhenius relation of BMG Vit1 around glass transition at different heating rates: (a) 2.5 K/min, η=2.0×10-5 exp[182.4×103/(RT)]; (b) 5 K/min, η=1.21×10-3 exp[160.1×103/(RT)]; (c) 10 K/min, η=19.71exp[109.1×103/(RT)]; (d) 15 K/min, η=38.82exp[99.8×103/(RT)]
Table 1 Effective activation energy Ev and pre-factor η0 of viscosity around glass transition of BMG Vit1
The free volume model can be used to describe the temperature dependence of the viscosity[16]. Within the free volume model the viscosity
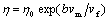 (4)
(4)
where vf is the average free volume per atom, vm is the atomic volume, bvm is the critical volume for flow. For the bulk metallic glass, the free volume decreased with the increase of the annealing time or temperature. During the continuous heating, the structure relaxation go with the decrease of the heating rate, and dispose of the atom was closer, namely the free volume was smaller, the viscosity gained according to the expression (4) was higher, thus the viscosity below Tg was closer to the extrapolation of the Arrhenius behaviour in the supercooled liquid down to lower temperature (Fig.1). The relaxation time was decreased rapidly because the movement ability of the atom was enhanced evidently above Tg. It is because that all atoms at different heating rate can achieve equilibrium configuration in the time scale of heating rate. Thus the temperature dependence curves of the viscosity were converged and accorded with a single Arrhenius relation.
3.2 Influence of structure relaxation on diffusivity
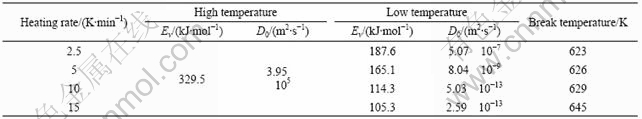
The diffusivity and its Arrhenius relation of Fe atom around glass transition calculated by the Eqn.(2) are shown in Fig.2. The Arrhenius behavior of diffusivity above and below glass transition was similar to that of viscosity. Above Tg, the temperature dependences of diffusivity at different heating rate follow the same Arrhenius equation, the ED and D0 are 329.5 kJ/mol and 3.95×105 m2/s. Below Tg, the Arrhenius behaviors are different for various heating rates, such as 15, 10, 5 and 2.5 K/min. The ED is 105, 114.3, 165.1 and 187.6 kJ/mol, respectively. The D0 is 2.59×10-13 m2/s, 5.03×10-13 m2/s, 8.04×10-9 m2/s and 5.07×10-7 m2/s, respectively (Table 2). The heating rate influences diffusivity in glass state markedly. The slower the heating rate is, the lower the diffusivity is, and the closer Arrhenius relation in glass state is with the extrapolation of the Arrhenius behaviour in the supercooled liquid down to glass state. The influence of structure relaxation on diffusivity in BMG Vit4 and Pd40Cu30Ni10P20 were investigated by THOMAS[17]. It is indicated that temperature dependence of diffusion coefficients in the non-relaxed glasses shows “non-linear” Arrhenius behaviour with a break near the glass transition, the diffusion in the long time relaxed glasses follows a uniform Arrhenius relation over the entire temperature range with considerably reduced diffusion coefficients below the glass transition.

Fig.2 Diffusivity and its Arrhenius relation of Fe atom around glass transition in BMG Vit1 calculated by Stokes-Einstein equation: (a) 2.5 K/min, D=5.07×10-7exp[-187.6×103/(RT)]; (b) 5 K/min, D=8.04×10-9exp[-165.1×103/(RT)]; (c) 10 K/min, D=5.03×10-13exp[-114.3×103/(RT)]; (d) 15 K/min, D=2.59×10-13exp[-105.0×103/(RT)]
3.3 Rheological and diffusion mechanisms
The temperature dependence of viscosity of BMG Vit1 was fitted by two different Arrhenius equations
Table 2 Effective activation energy ED and pre-factor D0 of diffusion coefficient deduced from the viscosity around glass transition of BMG Vit1
above and below Tg due to different rheological mechanism. In a narrower temperature range below Tg, the rheological behavior exhibited anelastic character with redistribution and annihilation of the free volume, as well as with local and short range movement of atoms. So activation energy corresponding with the process is lower. However, in undercooled liquid state above Tg, the rheological behavior showed Newtonian flow character with intermediate and long range movement. The process took place in supercooled liquid state above glass transition. Thus activation energy in process is higher, close to activation energy of glass transition.
For study of atom diffusion mechanism above and below glass transition, the correlation between the pre-factor D0 and activation energy ED of Arrhenius relation of diffusivity for Table 2 was studied (Fig.3). It was found that the same linear plot between lnD0 and ED, although the Arrhenius relation of diffusion coefficient was different for above and below glass transition. The results are similar to it reported by SHARMA and FAUPEL[18]. The results rather suggest that intrinsic diffusion above and below glass transition has a uniform mechanism. Furthermore, the diffusivity of Fe atom in BMG Vit1 calculated using Stokes-Einstein relation was well similar to experimental result reported by FIELITZ et al[14]. It is shown that the Stokes-Einstein relation is applicable for medium size-atoms in BMG Vit1, namely, the diffusion mechanism is similar to rheological mecha-

Fig.3 Correlation between lnD0 and ED for BMG Vit1 below and above glass transition temperature at different heating rates
nism. Therefore, intrinsic diffusion of medium size- atoms, proceeds possibly collective hopping processes in the whole temperature range. However, in many cases, the diffusivities in the glassy state are higher than expected from the extrapolation of the Arrhenius behavior in the undercooled liquid down to lower temperatures, which attributes to deficient structure relaxation. In non-relaxed glass state, the diffusion mechanism of medium size atoms is single-atom hopping mediated by vacancy defect with lower activation energy. After long time relaxation, the diffusion mechanism of medium size-atoms is cooperative motion of a group of several atoms because of the free volume annealing out.
4 Conclusions
The temperature dependence of viscosity in BMG Vit1 around glass transition cannot be fitted by a uniform Arrhenius relation. The slower the heating rate is, the closer the Arrhenius relation in glass state is with the extrapolation of the Arrhenius behaviour in the supercooled liquid down to glass state. It is indicated that structure relaxation influence the viscosity of BMGs markedly. The diffusion coefficient of the medium size-atom Fe in the BMG Vit1 was calculated by using the Stokes-Einstein relation. The temperature dependence of diffusion coefficient of the Fe atom, could also be well fitted by two different Arrhenius equations below and above the glass transition temperature. Besides, it indicated the same linear plots between lnD0 and ED for above and below glass transition. The results suggest that diffusion of medium size-atoms, substantially, proceeds collective hopping processes above and below glass transition.
References
[1] KLEMENT W, WILLENS R, DUWEZ P. Non-crystalline structure in solidified gold-silicon alloys [J]. Nature, 1960, 187(4740): 869-870.
[2] INOUE A, ZHANG T, MASURMOTO T. Al-La-Ni amorphous alloys with a wide supercooled liquid region [J]. Mater Trans JIM, 1989, 30: 965-972.
[3] INOUE A, NISHIYAMA N, MATSUDA T. Preparation of bulk glassy Pd40Ni10Cu30P20 alloy of 40 mm in diameter by water quenching [J]. Mater Trans JIM, 1996, 37(2): 181-184.
[4] INOUE A, ZHANG T. Fabrication of bulk glassy Zr55Al10Ni5Cu30 alloy of 30 mm in diameter by a suction casting method [J]. Mater Trans JIM, 1996, 37(2): 185-187.
[5] PEKER A, JOHNSON W L. A highly processable metallic glass: Zr46.75Ti8.25Cu7.5Ni10Be27.5 [J]. Appl Phys Lett, 1993, 63(17): 2342-2344.
[6] BAKKE E, BUSCH R, JOHNSON W L. The viscosity of the Zr46.75Ti8.25Cu7.5Ni10Be27.5 bulk metallic glass forming alloy in the supercooled liquid [J]. Appl Phys Lett, 1995, 67: 3260-3262.
[7] BUSCH R, BAKKE E, JOHNSON W L. Viscosity of the supecooled liquid and relaxation at the glass transition of the Zr46.75Ti8.25Cu7.5Ni10Be27.5 bulk metallic glass forming alloy [J]. Acta Mater, 1998, 46: 4725-4732.
[8] WANIUK A, BUSCH R, MASUHR A, et al. Equilibrium viscosity of the Zr41.2Ti13.8Cu12.5Ni10Be22.5 bulk metallic glass forming liquid and viscous flows during relaxation, phase separation, and primary crystallization [J]. Acta Mater, 1998, 46: 5229-5236.
[9] WANG J F, LIU L, PU J, XIAO J Z. The rheological behavior of bulk metallic glass Zr41Ti14Cu12.5Ni10Be22.5 [J]. Acta Physica Sinica, 2004, 53(6): 1916-1922. (in Chinese)
[10] GEYER U, SCHNEIDER S, JOHNSON W L. Atomic diffusion in the supercooled liquid and glassy states of the Zr41.2Ti13.8Cu12.5Ni10Be22.5 alloy [J]. Phys Rev Lett, 1995, 75(12): 2364-2367.
[11] TANG X P, GEYER U, BUSCH R, et al. Diffusion mechanisms in metallic supercooled liquids and glasses [J]. Nature, 1999, 402(11): 160-162.
[12] KNORR K, MACHT M P, FREITAG K, et al. Self-diffusion in the amorphous and supercooled liquid state of the bulk metallic glass Zr46.75Ti8.25Cu7.5Ni10Be27.5 [J]. J Non-Cryst Solids, 1999, 250-252: 669-673.
[13] EHMLER H, R?TZKE K, FAUPEL F. Isotope effect of diffusion in the supercooled liquid state of bulk metallic glasses [J]. J Non-Cryst Solids, 1999, 250-252: 684-688.
[14] FIELITZ P, MACHT M P, NAUNDORF V, et al. Diffusion in ZrTiCuNiBe bulk glasses at temperatures around the glass transition [J]. J Non-Cryst Solids, 1999, 250-252: 674-678.
[15] PERERA D N. Compilation of the fragility parameters for several glass-forming metallic alloys [J]. J Phys Condens Matter, 1999, 11: 3807-3812.
[16] MASUHR A, WANIUK T A, BUSCH R, et al. Time scales for viscous flow, atomic transport, and crystallization in the liquid and supercooled liquid states of Zr41.2Ti13.8Cu12.5Ni10Be22.5 [J]. Phys Rev Lett, 1999, 82: 2290-2293.
[17] THOMAS Z, VOLKMAR N, MICHAEL-PETER M, et al. Effect of reversible structure Relaxation on diffusion in bulk metallic glasses [J]. Ann Chim ScL Mat, 2002, 27(5): 55-60.
[18] SHARMA S K, FAUPEL F. Correlation between effective activation energy and pre-exponential factor for diffusion in bulk metallic glasses [J]. J Mater Res, 1999, 14(8): 3200-3203.
(Edited by LONG Huai-zhong)
Corresponding author: WANG Jing-feng; Tel: +86-23-65102466; E-mail: jfengwang@163.com