
Selective adsorption of bacteria on sulfide minerals surface
JIA Chun-yun(贾春云), WEI De-zhou(魏德洲), LIU Wen-gang(刘文刚),
HAN Cong(韩 聪), GAO Shu-ling(高淑玲), WANG Yu-juan(王玉娟)
School of Resource and Civil Engineering, Northeastern University, Shenyang 110004, China
Received 20 November 2007; accepted 10 March 2008
Abstract: The adsorption of bacteria on sulfide minerals surface was studied, and the selective adsorption mechanism of cells on the sulfide minerals was investigated by means of FTIR, UVS and XPS. The results show that the three strains of bacteria adsorbed more preferentially on pyrite than on other two sulfide minerals surface at neutral and alkaline pH conditions. FTIR and UVS of three strains of bacteria indicate that there are more functional groups on their surface, such as O―H, C=O, N―H, C―O, and the content of saccharide is more than that of protein. The state of every element on sulfide minerals surface was analyzed by XPS. The empty orbital number of electronic shell of metal ions on minerals surface is important in selective adsorption process, and some stable constants of metal coordinates can be used to explain the contribution of some groups in saccharide of cell wall to the selective adsorption.
Key words: selective adsorption; surface; bacteria; sulfide minerals; bio-flotation
1 Introduction
Bio-flotation technology combines biology with flotation, which has attracted much attention for several years. In the process of bio-beneficiation, adsorption of bacterial cell on the surface of minerals is dominant, and the selective adsorption of bacteria on different minerals is required to alter the surface properties so as to realize mineral separation[1-3]. Much work had been carried out focused on the adsorption quantity of bacteria onto mineral, and a few related to the selective adsorption behavior[4-6]. As for the mechanism of bacteria adsorption on mineral, many researchers suggested that different forces play a major role, such as static force, hydrophobic attraction, hydrogen bonding, and the component on cell surface is also important. Essentially, chemical force is probably the main factor making some bacteria have a selective adsorption on different minerals[7-11], in which the bonding capacity of functional groups on cell surface to metal ions on mineral surface plays an important role.
In the present work, the interactions of three strains of bacteria with pyrite, galena and sphalerite were investigated in order to reveal the selective adsorption behavior. The differences in the adsorption behavior of bacterial cells on these sulfide minerals were investigated by FTIR and UVS. The valency of every element and structure component of the minerals surface were detected by XPS, and then the selective adsorption mechanism of bacteria on different minerals was discussed briefly by biochemistry theory.
2 Experimental
2.1 Materials
Mineral samples of pyrite, galena and sphalerite used in this study were collected from a lead-zinc mine in northeast of China by means of manual picking. Mineralogical studies as well as X-ray powder diffraction analyses indicated that the samples are of high purity, and the contents of pyrite, galena and sphalerite are 98.63%, 98.48% and 99.99%, respectively. These samples were ground using a porcelain ball mill and then sieved through 74 ?m, 54 ?m BBS sieves. The selective adsorption of three strains of bacteria was carried out using the <74 ?m fraction of mineral samples, and the <54 ?m fraction was used for XPS analysis.
Three strains of bacteria used in this study were obtained from the Institute of Microbiology, Chinese Academy of Science(CAS). They are Gordona amarae, Mycobacterium Phlei and Bacillus mucilayinosus, and their corresponding symbols are 4.1126, 4.1180 and 1.231. The three strains of bacteria were subcultured in the laboratory using same sterile modified medium. The components of medium were 5 g/L sodium chloride, 5 g/L peptone and 3 g/L beef grease, and the pH was adjusted to 7.0-7.2.
2.2 Growth of bacteria
A pure active inoculum was added to the sterile modified medium and incubated at 28 ℃ on a rotary shaker at 150 r/min. The cell concentration was determined by cell wet mass per litre. During the bacterial growth, the cell culture was taken at intervals on every 100 mL culture and then centrifuged at 10 000 r/min for 10 min until the stationary phase was attained.
2.3 Preparation of metabolite-free cell suspension
The cultures of three strains of bacteria, which were fully grown in the sterile modified medium (after 60 h), were filtered through Whatman No.1 filter paper to remove all of the insoluble compounds. The filtrate was centrifuged at 10 000 r/min for 10 min and then washed several times with distilled water, and dispersed in distilled water at neutral pH. The cells harvested from the stationary growth period were used in experiment.
2.4 Adsorption test
The adsorption of three bacterial cells on sulfide minerals was estimated at the desired pH in the presence of 1.5 g/L bacterial cells. Distilled water was used as the supporting electrolyte for these tests. In all adsorption tests, 0.5 g each of the mineral powder was taken in 100 mL Bunsen beaker containing 50 mL bacterial cells suspension, and then agitated for 10 min with a magnetic stirrer at 400 r/min and 20 ℃. After equilibration, the suspension was then centrifuged at 2 000 r/min for 5 min to separate the mineral particles with adsorbed bacterial cells. The supernatant solution containing the unadsorbed cells was further filtered through Whatman No.1 filter paper, and the quantity of bacteria cells present in the supernatant after adsorption with different minerals was estimated using spectrophotometer. The corresponding bacteria concentration was calculated using normal bacterial calibration curve. The fraction of adsorbed cells to total cells is expressed by the following equation:
Q=(c0-c1)/c0×100% (1)
where c0 is the concentration of bacterial cells suspension before interaction, g/L; c1 is the concentration of bacterial cells in supernatant after interaction, g/L.
2.5 Detection method
Three strains of bacteria obtained from the stationary growth period were thoroughly washed using distilled water and vacuum dried. Fourier transform infrared (FTIR) spectra of bacterial cells were recorded using a Perkin-Elmer model FTIR spectrometer. Ultraviolet spectra(UVS) of bacterial cells were recorded using a SP-2000UV model ultraviolet spectrometer. For pyrite, galena and sphalerite, the X-ray photoelectron spectra (XPS) of them were recorded on a LAB250 model electron spectroscopy for chemical analysis.
3 Results and discussion
3.1 Growth of three strains of bacteria
The growth curves of three strains of bacteria are shown in Fig.1. It is observed from Fig.1 that, after a very short lag phase, the cell number of three strains of bacteria increases rapidly in logarithmic phase, and then comes to the stationary phase. For Gordona amarae and Mycobacterium Phlei, the logarithmic phase extends up to 60 h, and then the cell number decreases slightly to 5.5 g/L more or less. The growth curve of Bacillus mucilayinosus is similar to the former two strains of bacteria, and the logarithmic phase is up to 40 h. Beyond 40 h, the stationary period is observed. The difference is that the cell mass of Bacillus mucilayinosus (6.8 g/L) is much more than that of the former two strains of bacteria (5.5 g/L) in stationary phase. In the following studies, all the cells are obtained from the stationary phase (80-100 h) in order to ensure cells in good state.
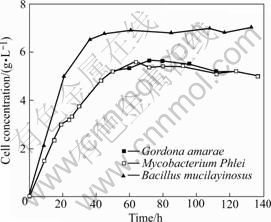
Fig.1 Growth curve of three strains of bacteria
3.2 Selective adsorption
The selective adsorption of Gordona amarae, Mycobacterium Phlei and Bacillus mucilayinosus on different sulfide mineral surfaces was studied as a function of pH. The results are shown in Figs.2-4.
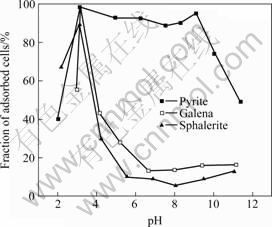
Fig.2 Adsorption of Gordona amarae on surface of minerals
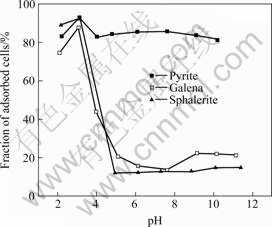
Fig.3 Adsorption of Mycobacterium Phlei on surface of minerals

Fig.4 Adsorption of Bacillus mucilayinosus on surface of minerals
`The influence of pH on the adsorption of Gordona amarae cells on the sulfide minerals is displayed in Fig.2. The cell adsorption is high from pH 3 to 10 and then it decreases in the case of pyrite. However, the adsorption of cells on galena and sphalerite is relatively low above pH 5. When pH is more than 5, the fraction of adsorbed cells on pyrite is about 90%, and that on other sulfide minerals is less than 20%.
The adsorption of Mycobacterium Phlei cells on pyrite is much higher than that on other two sulfide minerals when pH is more than 4 (see Fig.3). The adsorption behavior is almost the same for Mycobacterium Phlei cells on pyrite, galena and sphalerite, and is relatively high when pH is less than 3. The adsorption of cells on pyrite is always about 88% at pH 4-10, while the adsorption of bacterial cells on galena and sphalerite decreases rapidly as pH is higher than 3.0, and then remains low under pH 5-11.
Fig.4 shows the adsorption of Bacillus mucilayinosus cells on three sulfide minerals. It can be seen that the adsorption behavior of Bacillus mucilayinosus on pyrite, galena and sphalerite is similar to that of other two strains of bacteria, and the difference is that the fraction of adsorbed cells on pyrite is larger than that of the former two strains of bacteria when pH is more than 6. In other words, the selective adsorption of Bacillus mucilayinosus on pyrite, galena and sphalerite is a little stronger than that of the former two strains of bacteria under the same pH range.
3.3 FTIR spectra
Fig.5 shows the FTIR spectra of Gordona amarae, Mycobacterium Phlei and Bacillus mucilayinosus. It can be seen that the main peaks of three strains of bacteria are almost at the same sites. The assignments of various peaks have been made by comparing them with those available in Refs.[12-13]. A strong and broad peak near 3 430 cm-1 is assigned to the O―H stretching vibrations in or among the molecules or the free N―H stretching vibrations in saccharide, while the small twin-peaks around 2 963 cm-1 and 2 925 cm-1 are due to the asymmetric C―H stretching mode of the CH3 and CH2 groups respectively. The sharp peak around 1 650 cm-1 is due to an amide group (amide Ⅰ band) while the 1 540 cm-1 peak is assigned to N―H bending of the secondary amide group ―CONH (amide Ⅱ peak). The peak near 1 460 cm-1 can be ascribed to symmetric C―H
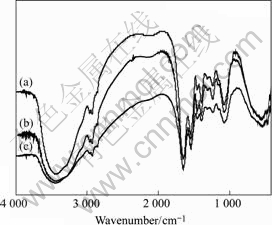
Fig.5 FTIR spectra of Mycobacterium Phle (a), Gordona amarae i(b) and Bacillus mucilayinosus(c)
distorted mode of CH2 and asymmetric distorted mode of CH3 in carbohydrate, including ―O―CH3. Similarly, the peaks at 1 400 cm-1 and 1 310 cm-1 are distorted vibrations of ―C―CH3. The peak around 1 240 cm-1 is due to the vibration contribution from C―O―S stretching of sulfate, the C―O stretching of carboxyl or P=O stretching of phosphate. Another small peak at 1 154 cm-1 is the stretching vibration of C―O on the structure of carbohydrates, the main component of the cell wall. The sharp peak at 1 080 cm-1 is due to primary alcoholic group (CH2OH) stretching. The peak at 550 cm-1 may be attributed to ―CH2 groups of polysaccharides.
3.4 UVS investigation
UVS spectra of Gordona amarae, Mycobacterium Phlei and Bacillus mucilayinosus are shown in Fig.6. Two obvious absorbed peaks (220, 275 nm) occur on the spectra of three strains of bacteria, and three curves have a similar trend. It is well known that the peak of saccharide is around 220 nm and that of tryptophan and tyrosine in most proteins is at 280 nm. It is clear from Fig.6 that three strains of bacteria cells all have saccharide and proteins, and the content of saccharide is larger than that of protein. Referred to some references about the absorbed peaks of typical groups[14], the peak near 220 nm is due to ―OH, ―CHO and ―COOR, and the peak around 275 nm is due to C=O, which are in agreement with the measurement of FTIR studies.

Fig.6 UVS spectra of Gordona amarae, Mycobacterium Phlei and Bacillus mucilayinosus
3.5 XPS studies
The electronic binding energy, valency and atom fraction of every element on the surface of three sulfide minerals are calculated according to the fine scanning spectra[15], and the results are shown in Table 1.
Table 1 Surface elements analysis of pyrite, galena and sphalerite
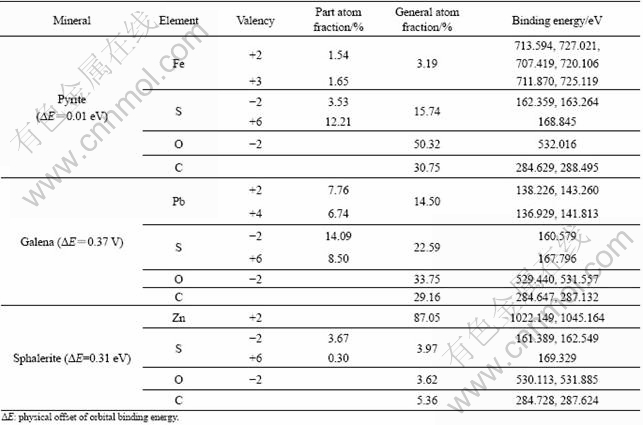
It is clear that elements C and O also exist on the surface of three sulfide minerals, and this confirmed the high purity of them. The partial oxidation of element S and metal ions of three sulfide minerals are also observed.
3.6 Mechanism of selective adsorption
From the above adsorption behavior of three strains of bacteria on the surface of pyrite, galena and sphalerite, it can be observed that there is a similarity in the trend with respect to the adsorption of three strains of bacteria onto sulfide minerals as a function of pH. They all have a selective adsorption on sulfide minerals at neutral and alkaline conditions. As well known, cell surfaces and sulfide mineral surfaces are negatively charged, resulting in electrostatic repulsion, but a number of cells still adsorb on pyrite, which suggests that other forces such as chemical interaction are the governing factors in the adsorption process.
Based on FTIR spectra and UVS spectra of three strains of bacteria, it is clear that the groups in cell walls of three strains of bacteria are almost the same. They are O―H, C=O, C―O, N―H, P=O etc, and there are more saccharide and less protein. It fits the components of gram-positive bacterial cell wall perfectly, and the content of peptidoglycan is larger than that of protein. Consequently, it can be concluded that some groups in saccharide of cell wall are important in the adsorption process, such as O―H, C=O and C―O.
For three sulfide minerals, element S is the common component, and elements Fe, Pb, Zn are the constituted metal ions. Under the same adsorption conditions, the difference of metal ions on three sulfide mineral surface is the main factor affecting selective adsorption for a strain of bacteria. That is to say, the bonding capacity of groups on bacterial cells surface to metal ions on different sulfide minerals is not same. The valence shell of Fe, Pb, Zn is 3d64s2, 6s26p2, 3d104s2 in turns, and that of Fe2+, Fe3+, Pb2+, Pb4+, Zn2+ is 3d64s0, 3d54s0, 6s26p0, 6s06p0, 3d104s0 respectively, so the empty orbital number of Fe2+, Fe3+ is more than that of other metal ions[16]. When chemical interaction occurs at the interface of bacterial cells and minerals, the acceptant capacity of lone pair electrons of Fe2+, Fe3+ is larger than other metal ions, and the lone pair electrons are offered by O, N, P in groups on bacterial cell surface.
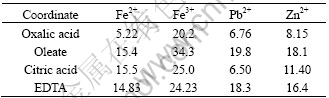
One cell can be seen as a unit whose surface is covered with many groups. Some cooperated atoms in groups will bind to metal ions when a cell interacts with the mineral surface. In fact, the selective adsorption is the result of many groups bonding to one metal ion together. For above three strains of bacteria, the bonding capacity of some cooperated atoms, like O, in saccharide of cell wall to different metal ions may be the key factor affecting the selective adsorption. In addition, the stable constants of some metal coordinates (Table 2) reveal the bonding capacity of some groups in saccharide to metal ions in a sense, and may explain the selective adsorption partially. These data provide new insight into the function of surface groups conferred to the selective adsorption of some bacteria to sulfide minerals.
Table 2 Stable constants of some metal coordinates
4 Conclusions
1) Adsorption experiments show that Gordona amarae, Mycobacterium Phlei and Bacillus mucilayinosus all have a selective adsorption on the surface of pyrite, galena and sphalerite respectively at neutral and alkaline conditions.
2) FTIR spectroscopy studies indicate that the main peaks of three strains of bacteria are almost the same, and the main groups are O―H, C=O, C―O, N―H, P=O etc.
3) UVS analyses show that the strains all have saccharide and proteins, and the content of saccharide is larger than that of protein.
4) From the analysis of XPS on pyrite, galena and sphalerite, it is concluded that the oxidation of some elements on three sulfide minerals surface occurs.
5) The empty orbital numbers of electronic shell of different metal ions on mineral surface play an important role for selective adsorption, and some stable constant of metal coordinates also can provide evidence in support of the function of some groups in saccharide of cell wall to the selective adsorption.
References
[1] PATRA P, NATAFAJAN K A. Microbially-induced flocculation and flotation for pyrite separation from oxide gangue minerals [J]. Minerals Engineering, 2003, 16(7): 965-973.
[2] CHANDRAPRABHA M N, NATAFAJAN K A, MODAK JAYANT M. Selective separation of pyrite and chalcopyrite by biomodulation [J]. Colloids and Surfaces B: Biointerfaces, 2004, 37(6): 93-100.
[3] CHANDRAPRABHA M N, NATAFAJAN K A, SOMASUNDARAN P. Selective separation of pyrite from chalcopyrite and arsenopyrite by biomodulation using Acidithiobacillus ferrooxidans [J]. Int J Miner Process, 2005, 75(8): 113-122.
[4] SHASHIKALA A R, RAICHUR A M. Role of interfacial phenomena in determining adsorption of Bacillus Polymyxa onto hematite and quartz [J]. Colloids and Surfaces B: Biointerfaces, 2002, 24(3): 11-20.
[5] ZHENG X P, ARPS PEGGY J, SMITH, ROSS W. Adhesion of two bacteria onto dolomite and apatite: Their effect on dolomite depression in anionic flotation [J]. Int J Miner Process, 2001, 62(7): 159-172.
[6] HARNEIT K, GOKSEL A, KOCK D, KLOCK J H, GEHRKE T, SAND W. Adhesion to metal sulfide surfaces by cells of Acidithiobacillus ferrooxidans, Acidithiobacillus thiooxidans and Leptospirillum ferrooxidans [J]. Hydrometallurgy, 2006, 83(5): 245-254.
[7] DEO N, NATAFAJAN K A, SOMASUNDARAN P. Mechanisms of adhesion of Paenibacillus polymyxa onto hematite, corundum and quartz [J]. Int J Miner Process, 2001, 62(7): 27-39.
[8] SHARMA P K, HANUMANTHA R K. Adhesion of Paenibacillus polymyxa on chalcopyrite and pyrite: Surface thermodynamics and extended DLVO theory [J]. Collids and Surfaces B: Biointerfaces, 2003, 29(10): 21-38.
[9] CHANDRAPRABHA M N, NATAFAJAN K A. Surface chemical and flotation behaviour of chalcopyrite and pyrite in the presence of Acidithiobacillus thiooxidans [J]. Hydrometallurgy, 2006, 83(5): 146-152.
[10] PATRA P, NATAFAJAN K A. Surface chemical studies on selective separation of pyrite and galena in the presence of bacterial cells and metabolic products of paenibacillus polymyxa [J]. Journal of Colloid and Interface Science, 2006, 298(2): 720-729.
[11] SANTHIYA D, SUBRAMANIAN S, NATARAJAN K A. Surface chemical studies on sphalerite and galena using Bacillus polymyxa [J]. Journal of Colloid and Interface Science, 2001, 235(9): 298-309.
[12] SOCRATES G. Infrared characteristic group frequencies [M]. New York: Wiley-Interscience, 1980.
[13] HERMAN A S. Infrared handbook [M]. New York: Plenum, 1963.
[14] GOKEL G W. Handbook of inorganic chemistry [M]. Beijing: Chemical Industry Press, 2006. (in Chinese)
[15] MOULDER J F, CHASTAIN J. Handbook of X-ray photoelectron spectroscopy [M]. Beijing: Chemical Industry Press, 1992. (in Chinese)
[16] Inorganic Chemistry Staff Room in Tianjing University. Inorganic chemistry [M]. Beijing: Higher Education Press, 1992. (in Chinese)
Foundation item: Project(50174014) supported by the National Natural Science Foundation of China; Project(2006223002) supported by the High-Tech Program of Liaoning Province, China
Corresponding author: JIA Chun-yun; Tel: +86-24-83679006; E-mail: jiachunyun@126.com
(Edited by YUAN Sai-qian)