���ڲⶨͭ�������к��������ѡ��������ȡ������������
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2020���10��
�������ߣ��Żݱ� ������ ��ݼݼ ����� �ܻ��� �ܿ� ֣����
����ҳ�룺2823 - 2835
�ؼ��ʣ��飻ͭ¯��������ȡ���̷Ϲ�����ͭұ��
Key words��arsenic; copper slag; sequential extraction; waste management; copper smelting
ժ Ҫ������һ���Ż���ѡ��������ȡ��������������ͭ�������к���������ɡ��÷�����ͭ�������еĺ����������Ϊˮ�����顢�����������Ρ������������Ρ����������ɰ�ͽ������Լ��̻��ڲ������е��顣���õ����ϵ�������ԭ�ӷ�����ס�ɨ��羵����羵��X��������ͺ�������ֶζԸ�����ȡҺ�������б��������õ�һ������ȡʵ��������ȡ�Լ���ѡ���Ժ�������ʡ����������¯���и������ı��������ױ������нϺõ�һ���ԣ�����������ʱ�����90%���ϡ�����������о��Ż���ѡ��������ȡ�����ܿɿ���Ӧ����ͭұ�����鸱�����к�������ijɷ����ۡ�
Abstract: An optimized selective sequential extraction (SSE) procedure was developed to assess the arsenic (As) partitioning in copper slag. The potential As species in copper slag are partitioned into the readily soluble As, dissolvable arsenates, sparingly soluble arsenates, As residing in sulfides, arsenopyrite and metal As, as well as As incorporated into glassy silicates. The inductively coupled plasma atomic emission spectrometry (ICP-AES), scanning electronic microscopy (SEM), transmission electron microscope (TEM), X-ray diffractometry (XRD) and Fourier transform infrared spectroscopy (FTIR) were used to characterize the leachates and residues produced from the operation scheme. The selectivity and recovery of extractants were evaluated through single-phase extraction procedures. Partitioning data of As in slag samples show good agreement with the reported works and the total As recovery of each operation is over 90%. This suggests that the optimized SEE scheme can be reliably employed for As partitioning in As-bearing byproducts from copper smelting.
Trans. Nonferrous Met. Soc. China 30(2020) 2823-2835
Hui-bin ZHANG1, Yu-zheng HE1, Jing-jing HU1, Ya-nan WANG1, Hua-zhen CAO1, Jun ZHOU2, Guo-qu ZHENG1
1. College of Materials Science and Engineering, Zhejiang University of Technology, Hangzhou 310014, China;
2. Tongling Nonferrous Metals Group, Tongling 244000, China
Received 27 January 2020; accepted 4 August 2020
Abstract: An optimized selective sequential extraction (SSE) procedure was developed to assess the arsenic (As) partitioning in copper slag. The potential As species in copper slag are partitioned into the readily soluble As, dissolvable arsenates, sparingly soluble arsenates, As residing in sulfides, arsenopyrite and metal As, as well as As incorporated into glassy silicates. The inductively coupled plasma atomic emission spectrometry (ICP-AES), scanning electronic microscopy (SEM), transmission electron microscope (TEM), X-ray diffractometry (XRD) and Fourier transform infrared spectroscopy (FTIR) were used to characterize the leachates and residues produced from the operation scheme. The selectivity and recovery of extractants were evaluated through single-phase extraction procedures. Partitioning data of As in slag samples show good agreement with the reported works and the total As recovery of each operation is over 90%. This suggests that the optimized SEE scheme can be reliably employed for As partitioning in As-bearing byproducts from copper smelting.
Key words: arsenic; copper slag; sequential extraction; waste management; copper smelting
1 Introduction
Arsenic (As), which is equivalently deemed as toxicants, is nowadays a common impurity in copper ores due to the exhaustion of high-quality copper ores [1,2]. The arsenic in copper ores usually appears as enargite (Cu3AsS4), tennantites (Cu12AsS13, (Cu,Fe)12As4S13) [3], arsenopyrite (FeAsS) [4] and sulfides (As2S3) [5-7]. During matte smelting and converting steps, a considerable proportion of arsenic would be incorporated into the copper slags [8,9]. According to a rough estimation, 1 t metallic copper product was generated together with about 2.2 t slags. The copper manufactures in China discharged approximately 8��106 t slags annually [10,11]. The presence of arsenic in copper slags not only increases the difficulty for recovering the valuable metals, but also causes the potential arsenic hazard to environment [2].
The first step of pyrometallurgical processes for treating copper concentrates is matte smelting. During this process, the associated As will be distributed into matte, slag and gaseous phase. According to the reported works [12-21], the possible arsenical compounds existed in the copper concentrate and smelting products are summarized in Table 1. It can be seen that various arsenic compounds such as arsenates, As oxides and sulfides, and metal As can be embedded or solubilized in copper slag; meanwhile, the As compounds in copper concentrate and flue dust may also remain in copper slag. In general, copper slags are either discarded as waste in slag dumps or sold as cement raw materials after recovering residual metal copper [9].
For the purpose of better disposal of As- bearing copper slags, As speciation and partitioning in copper slags are urged to be investigated. Spectroscopic methods including the X-ray diffractometry and vibrational spectroscopy, as well as analytical methods are two common kinds of methodologies employed for determining As species and identifying the chemical states [22]. However, the handy spectroscopic techniques, for example, X-ray fluorescence (XRF), X-ray photoelectron spectroscopy (XPS) and Raman spectroscopy (RS), generally suffer from the insufficient capability of accurately identifying As speciation at low concentrations [22]. The most fruitful X-ray absorption spectroscopy (XAS), which can supply detailed information on the speciation and coordination environment of As species, is also daunting due to the limited accessibility to instruments and the obscurity of data analysis technologies [23,24]. Selective sequential extraction (SSE) is a more operable approach used to determine the target phases by a careful selection of the suited extracting reagents [25]. It not only provides excellent quantitative elemental sensitivity, but also directly assesses the mobility of heavy metals [26,27]. Accordingly, it is widely used in the fields of mining, soil science, geochemistry and phytology.
In this work, the assessment and application of a proposed SSE procedure were described for As partitioning in copper flash smelting slags. This SSE procedure was principally designed based on the dissolution behaviors of different As species. The used extractants and extraction order are chosen and amended according to the testing methods of known pure As-bearing compounds and natural minerals [28-30]. Via the characterization of the leaching residues and leachates, various As fractions with different accessibilities can be determined.
2 Experimental
2.1 Samples from copper flash smelting
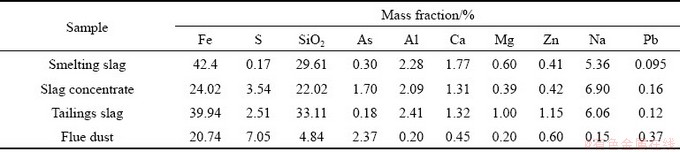
The slag and flue dust samples were collected from the flash smelting furnace at Tongling Nonferrous Metals Group, China. The slag samples include the smelting slag (before slag separation), tailings slag and slag concentrate (the recovered Cu-rich slag). The compositions of the samples used for this work are shown in Table 2.
2.2 Selective sequential extraction scheme
According to the results in Table 1, the potential arsenic compounds can be classified into six categories, that is, the readily soluble As, dissolvable arsenates, sparingly soluble arsenates, As residing in sulfides, arsenopyrite and metal As, as well as As incorporated into glassy silicates. Before the extraction experiments, the slag samples were firstly milled in an agate mortar in order to make sure that the sizes of the particles were less than 74 ��m, and then dried to constant mass. A polytetrafluoroethylene (PTFE) vessel and a thermostatically controlled heating block were used for chemical leaching. The used reaction vessels and glass wares were cleaned with high purity H2O with a resistivity higher than18.0 M����cm, and dried before use. All solutions were prepared with the high-purity water and analytical grade chemicals. The extractants and the operation details of SSE procedure are listed in Table 3. Each extraction started with 1 g of tailings slag. The residues after each leaching step were defined as Residues 1-5. The extracting processes were conducted with continuous agitation and the residues in each extraction step were charily rinsed. The leachates together with the washing liquids were diluted to a volume of 250 mL, and then the arsenic concentrations were determined. The averages were obtained from three duplicate runs. The recovery of As was assessed by comparing the sum of the six fractions of As with the initial As amount.
Table 1 Reported As-bearing compounds from copper smelting process
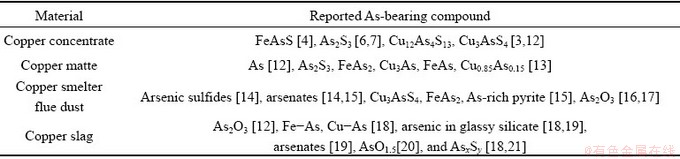
Table 2 Compositions of smelting slag, slag concentrate, tailings slag and flue dust
2.3 Single-phase extraction procedures
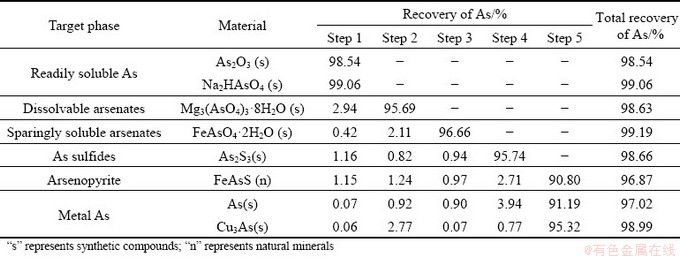
To assess the suitability and extraction efficiency of each selected leaching agent for target phases, some synthetic and natural arsenical minerals were subjected to the proposed SSE scheme. The synthetic methods of the arsenical minerals are shown in the supporting materials. As shown in Table 4, the readily soluble As includes As oxide and alkali metal arsenates, which are extracted by water; Mg3(AsO4)3��8H2O is selected as a representative of dissolvable arsenates and is basically leached in the second step; FeAsO4��2H2O is a well-known sparingly soluble arsenate and is indissolvable in previous steps, but can be dissolved with hot HCl; As sulfides are insoluble in acid, but can be extracted by NaOH; the arsenopyrite and metal As are dissolved and recovered in the 5th step. The As incorporated into glassy silicates is mostly immovable, so it is deemed to be extracted after completely digesting the glassy silicates in the last step. The As recovery of each portion in the SSE procedure is over 96.8%, indicating that the scheme is well designed.
2.4 Characterization methods
The inductively coupled plasma atomic emission spectrometry (ICP-AES IRIS Advantage-1000, Thermo Electron Corporation, America) was employed to determine the concentration of As in leachates. In this work, quality assurance measures included analytical duplicates, check standards and blanks. The primary As contents in slags and flue dust samples were also measured by ICP-AES after being completely digested with mixed acids. The morphologies of the materials were analyzed by field-emission scanning microscopy (SEM, Nova Nano 230) coupled with energy dispersive spectroscopy (EDS) and transmission electron microscopy (TEM, JEM-2010, JEOL). The phase compositions of the leaching residues were examined with an X-ray diffractometer (XRD, Dmax 2500VB) using Cu K�� source (��=0.1541 nm). Fourier transform infrared spectra (FTIR) of copper slags and the leaching residues were recorded on a Nicolet 6700 spectrophotometer (Thermo Nicolet, USA).
Table 3 Procedures of proposed selective sequential extraction
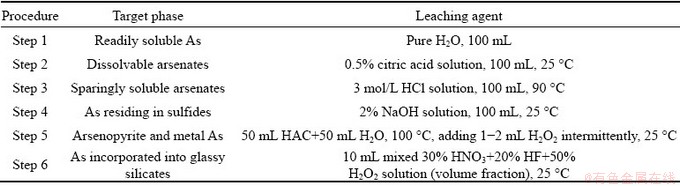
Table 4 Mean recoveries for dissolved As-bearing phases in single-phase extractions
3 Results and discussion
3.1 Characterization of tailings slag
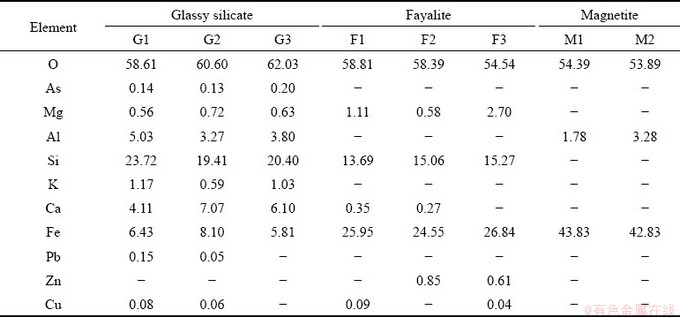
Figure 1(a) shows the XRD pattern of tailings slag. As can be seen, fayalite (Fe2SiO4, PDF No. 99-0049) and magnetite (Fe3O4, PDF No. 99-0073) are the major phases; a broadened XRD peak with the equivalent Bragg angle at 2��=23�� is also detected, which is related to the amorphous silicates. It is believed that fayalite was formed from the interaction of FeO and SiO2 and that the magnetite could be transformed from the oxidation-decomposition of fayalite and/or oxidation of Fe sulfides [10,31]. The gangue in copper concentrates can react with added SiO2, then producing the composite silicate melts which maintain the glassy feature after rapid cooling. Figures 1(b) and (c) show the cross-section SEM and BSE images, respectively. It is seen that the fayalite phase with rod-like shapes and the block magnetite are embedded in the glassy silicate substrates (Fig. 1(d)). The detailed element analysis results of the slag samples are displayed in Table 5. As can be seen, a trace amount of As is detected by EDS in the glassy silicates, while As is undetectable in fayalite phases and magnetite crystals. Other As-bearing minerals are hard to be investigated by XRD and EDS due to their relatively low detecting limit. Obviously, the distribution and category of As compounds determine the leaching kinetics of As, thus claiming necessary optimization for SSE procedures.
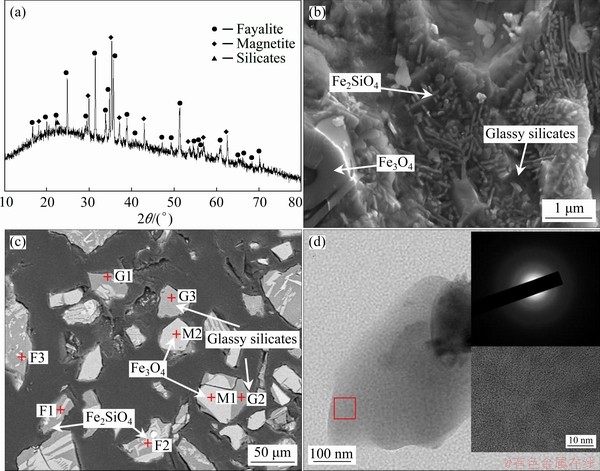
Fig. 1 XRD pattern (a), SEM (b), BSE (c) images of typical copper slags and TEM images of glassy silicates in copper slags (d)
Table 5 BSE-EDS analysis results of major phases in copper tailings slag (at.%)
3.2 Optimization of SSE procedure
3.2.1 Readily soluble As
Some researchers [32,33] have found that the water-soluble As can readily contaminate soil and ground water and then be assimilated by plants, so the portion of readily soluble As receives the most urgent concern. To our knowledge, As oxides and some As-bearing minerals such as sodium/kalium arsenates have great solubility in water. The use of mild neutral water as leaching agent would not change the pH and oxidoreduction conditions during leaching process [26]. In this leaching process, the solid/liquid (w/v) ratio is 1/100 (g/mL), which is sufficiently low to avoid the soluble As getting saturated in solution [29]. In general, this step is performed at room temperature. Considering the extracting efficiency, the approximately boiling water was used for extraction. As can be seen from Fig. 2(a), the concentration of extracted total As increases with time at 100 ��C, and an equilibrium can be achieved at ~100 min. For comparison, the extraction process carried out at room temperature costs about 600 min to reach the same equilibrium. Hence, the extraction operation at ~100 ��C with duration of 100 min was chosen for this step.
3.2.2 Dissolvable arsenates
In this step, an aggressive reagent citric acid used as a complexing agent was utilized. With the presence of citric acid, the dissolvable metal arsenates such as Mg, Ca, Al, and Zn can be readily released into solution [34]. The usage of citric acid is because its occurrence in natural environment and organisms is extensive [35]. This also suggests that the dissolvable arsenates might be set free into environment via natural citric acid leaching and become bioavailable. As shown in Fig. 2(b), increasing the mass fraction of citric acid from 0.2% to 0.8% leads to an increase of extracting efficiency of As. The citric acid solution with a mass fraction of 0.5% is sufficed to assure the rapid extraction in 80 min. Improving mass fraction of the citric acid up to 0.8% shows no obvious beneficial effect in leaching kinetics. Meanwhile, no re-adsorption phenomena are observed. In consequence, 0.5% citric acid and 80 min were selected as the optimal condition.
3.2.3 Sparingly soluble arsenates
The sparingly soluble ferric arsenates are excellent scavengers of As. This fraction of As is quite stable in water, low-molecular-mass inorganic acids, and inorganic acids like sulfuric acid (H2SO4), phosphoric acid (H3PO4), and hydrochloric acid (HCl) at room temperature [36]. Whereas, they are able to be dissolved in hot HCl solution with high acidity. To test this, the leaching behaviors of Residue 2 were investigated via optimizing the HCl concentration and contact time. As can be seen from Fig. 2(c), the contact time for full extraction is notably decreased with increasing the initial concentration of HCl solution. The leaching efficiency of 3 mol/L HCl is sufficiently high, which might be attributed to the complexing effect of chloridions under high acidity and chloridion concentration [37]. Regarding the effective and efficient extraction, the samples were subjected to 60 min leaching with 3 mol/L HCl.
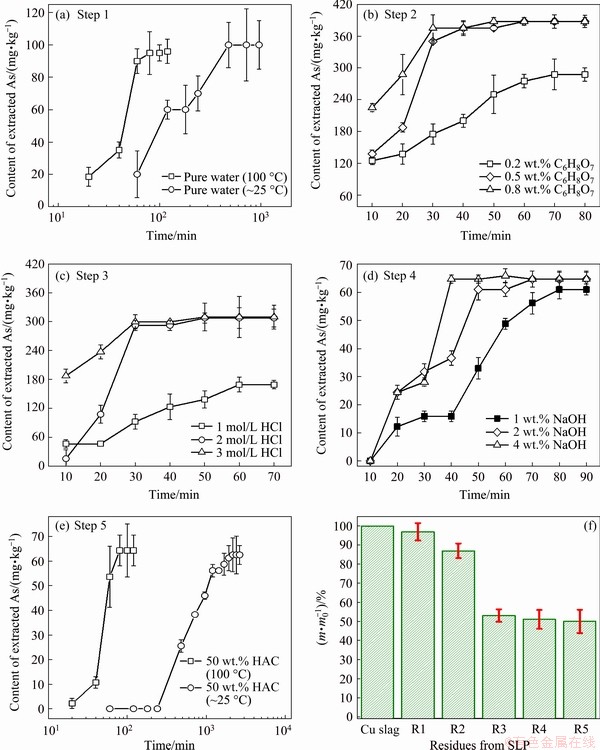
Fig. 2 Leaching kinetics of As at each step of SSE operations (a-e) and corresponding mass changes (f)
3.2.4 As residing in sulfides
During matter smelting, the sulfides of Cu, Pb, Fe, Zn, and As are all potential substances which might be generated from reactions with sulfur or remained from copper concentrates [38]. Some sulfides such as PbS, ZnS, and FeS could be dissolved in the previous leaching step [39], while the sulfides of As (i.e. As4S4 and As2S3) are difficult to be digested with non-oxidizing acids [40]. It is known that the As sulfides could be readily dissolved in sodium hydroxide (NaOH) solution, except arsenopyrite [41]. Figure 2(d) shows the leached As contents as a function of time in NaOH solutions with different concentrations. As can be seen, the curves of 2% and 4% NaOH reach the same As equilibrium concentration. Hence, it is possible to believe that the glassy silicates are inactive in the present dilute NaOH solution in limited reaction time. Meanwhile, a moderate leaching condition for the As residing in sulfides is 2% of NaOH and 80 min.
3.2.5 Arsenopyrite and metal As
Arsenopyrite and metal As are a potential portion of As species in copper slags. The extraction of this portion should change the leaching condition from reducing to oxidizing [41]. Therefore, a mixture of acetic acid (HAC) and hydrogen peroxide was used as the complexing agent and oxygenant for enhancing the extraction of As. Again, the leaching kinetics of two cases, which were performed at room temperature and boiling water bath, was respectively studied (Fig. 2(e)). It is seen that these two cases reach the same plateau but hot leaching contributes to much higher extraction efficiency, shortening the contact time from ~1400 to 100 min.
3.2.6 As incorporated into glassy silicates
A considerable portion of arsenic could be encapsulated in glassy silicate substrate because of the similar sizes of AsO43- ions and SiO44- units, making the vitrified As species with low leachability [42]. Obviously, the vitric silicates should be digested beforehand so as to liberate and determine this fraction of As. Hence, the mixed solution of HNO3+HF+H2O2 was utilized to completely degrade the glassy substrates. The obtained solution was used to determine the last fraction of As after diluting.
3.2.7 Mass loss during SSE process
Figure 2(f) shows the mass loss of the copper slag sample after each leaching step. As can be seen, although there is merely a trace amount of As in copper slag, the mass loss during leaching operations are considerable. Apparently, the selective extraction operations for As species are accompanied by the dissolution of other minerals. The leaching processes with hot water, citric acid and HCl respectively cause mass loss values of 3.0%, 10.1% and 34.1%, respectively, and the total mass change of the three operations can reach to 47.2%. The mass variations of the last two steps were slight, which also suggests that leaching reagents (NaOH and hot HAC) will not result in the noteworthy dissolution of glassy silicate substrate.
3.3 Evolution of solid phase during SSE procedure
Figure 3 shows the XRD patterns of the leaching residue samples. It has been shown that the detectable phases in Residues 1 and 2 are fayalite, magnetite and amorphous silicates, which has been discussed in Fig. 1(a). After leaching with HCl solution in Step 3, the magnetite phase turns out to be the dominant phase while the fayalite phase becomes undetectable. Considering the mass change curve shown in Fig. 2(f), it is facile to trace a line between the significant mass loss and the digestion of fayalite. After leaching by Steps 4 and 5, the XRD features of magnetite and silicate phases keep almost invariable.
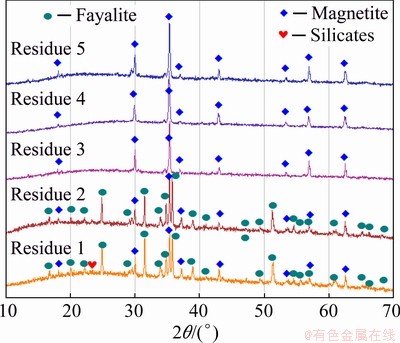
Fig. 3 XRD patterns of leaching residues obtained from Steps 1-5 (Residues 1-5)
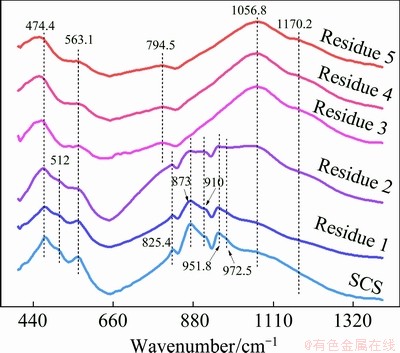
Fig. 4 FTIR spectra of leaching residues obtained at Steps 1-5 (Residues 1-5)
The overall FTIR spectra of copper slag and the resulting residues are shown in Fig. 4. As can be seen, there are two types of characteristic absorption bands which respectively correspond to the Si��O tetrahedral groups in fayalite and vitric silicates. The spectra of the underneath three samples present four absorption bands in 400- 1400 cm-1 range. In the low-frequency part, the peaks at 474.4, 512 and 563.1 cm-1 are attributed to the asymmetric Si��O bending vibration, which could be activated by both fayalite and silicates [43]. The weak bands at around 825.4 cm-1 are due to Si��O symmetric stretching vibration of [SiO4]4- tetrahedral groups [44]. Due to the discrepancy in bond strengths of the Si��O bondings in [SiO4]4-, the FTIR bands at 850-1000 cm-1 can be split into four peaks at around 873, 910, 951.8 and 972.5 cm-1 [44]. The last broad weak band at 1056.8 cm-1 is related to the Si��O asymmetric stretching of amorphous silicates [45]. In brief, before Step 3 the fayalite and amorphous silicates are the detected phases in FTIR spectra. After Step 3 the FTIR spectra of the residues (Residues 3, 4 and 5) change into the characteristic mode of amorphous silicates. Meanwhile, the intensities of the low-frequency bands at 400-600 cm-1 become weak and the peak widths get increased. Thissuggests that the superposed absorption peaks from the crystalline fayalite are removed, remaining the bands excited by amorphous silicates. Such result is in accordance with the XRD result.
The SEM and BSE morphological evolution of tailings slag during the SSE procedures is displayed in Fig. 5. Residue 1 shows no visible change on surface morphology (not shown). Figures 5(a) and (b) show the SEM image and the cross-sectional BSE image of Residue 2, respectively. It has been shown that some pitting and crevices newly emerge due to the dissolution by citric acid, suggesting a portion of dissoluble minerals in tailings slag are dissociated from the slag particles and dissolved in solution. From a section view of Residue 2, the outlines of the glassy silicates are continuous and smooth (Fig. 5(b)), indicating that the silicates can withstand the corrosion of citric acid. A radical change of the surface morphology takes place in Residue 3. As shown in Figs. 5(c) and (d), the platy fayalite crystals are corroded extensively, while the magnetite particles and some fayalite phases enclosed in the glassy substances keep intact. Over the subsequent leaching steps, the appearance of Residues 4 and 5 almost keeps unchanged (not shown), which also indicates that the glassy silicates are capable of resisting the relatively aggressive leaching using hot water, citric acid and even dilute HCl and NaOH. As shown in Fig. 6, particles in Residue 5 are composed of magnetite phase and glassy substrate with strip-like bar structures which are generated after the digestion of fayalite phases. According to the EDS mapping results, the glassy substrate is a composite silicate containing elements such as Fe, Al, Mg, Ca, and K. Meanwhile, a trace amount of As also exists in the glassy silicates.

Fig. 5 Surface SEM images (a, c) and cross-sectional BSE images (b, d) of Residues 2 and 4, respectively
3.4 SSE procedure assessment
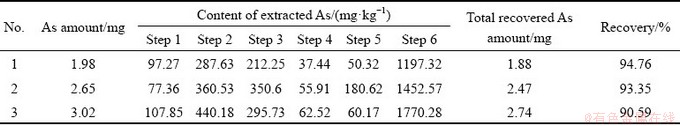
Three duplicate runs of SSE process for tailings slag are summarized in Table 6. As can be seen, the major arsenic species are extracted in Step 2, 3 and 6, which correspond to the dissolvable arsenates, sparingly soluble arsenates and As incorporated into glassy silicates, respectively. A minor portion of As oxides, sulfides, arsenopyrite and metal As may also exist in copper tailings slag. Meanwhile, Sample 2 may contain a considerable amount of arsenopyrite and metal As. It is noteworthy that the last step designed to extract the As incorporated into glassy silicates gets the largest fraction of As. Actually, this portion of As could be composed of two kinds of As species, that is, the AsO43- incorporated into the silicates at molecular-scale and the As compounds physically or structurally encompassed by glassy silicates. In the MO-SiO2 slag system [20], reactions of arsenates during slagging could be described as 3/2MO(l) + As(l) + 5/4O2(1) M3/2AsO4(l). Then, the formed
M3/2AsO4(l). Then, the formed  can be dissolved in silicate melts due to the similarity of [SiO4]4- units and AsO43- ions in size. The MO-SiO2 melts tend to produce polymeric species, with which AsO43- could be integrated to form chain-structures without free O2- ions. As such, it indicates that the arsenates are the major form of As consolidated in copper slags during copper smelting. In addition, partial tiny As compounds can be possibly enclosed in the glassy silicates, so they are released until the encapsulated glassy silicates are digested in Step 6. This portion of As is also quite stable upon chemical leaching, but may be affected by the size of copper slags. The recoveries of the three SSE operations are found to be acceptable with values between 90.59% and 94.76%.
can be dissolved in silicate melts due to the similarity of [SiO4]4- units and AsO43- ions in size. The MO-SiO2 melts tend to produce polymeric species, with which AsO43- could be integrated to form chain-structures without free O2- ions. As such, it indicates that the arsenates are the major form of As consolidated in copper slags during copper smelting. In addition, partial tiny As compounds can be possibly enclosed in the glassy silicates, so they are released until the encapsulated glassy silicates are digested in Step 6. This portion of As is also quite stable upon chemical leaching, but may be affected by the size of copper slags. The recoveries of the three SSE operations are found to be acceptable with values between 90.59% and 94.76%.

Fig. 6 BSE images of Residue 5 (a, b) and EDS mapping results of glassy silicates (c-i)
Table 6 As fractionations and recoveries of copper tailings slag in SSE processes
The As fractionation results with the proposed SSE method are depicted in Fig. 7. The percentages of As leached from Steps 1 and 2 account for 17.67%-20.44%. These As species are readily dissolved in water or organic acids, so they are considered to have the highest mobility. The remaining As partitions can reach up to 79.55%-82.32%, which are quite torpid in natural environment. It is noted that approximately 60% total As with low leachability is incorporated in the glassy silicates. Accordingly, the solidification/ stabilization treatment of copper slag should actually give special attention to the approximately 20% of relatively mobilizable As in copper slag.
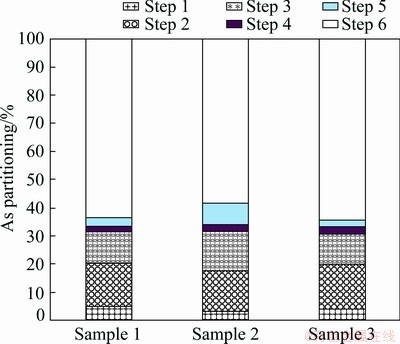
Fig. 7 As partitioning in studied tailing slag samples at different leaching steps
3.5 Application of SSE procedure to by-products from copper smelting
The proposed SSE method is also applied to determining the As fractionations of copper smelting slag, slag concentrate and flue dust (Table 7). As compared with tailings slag, the total As content in smelting slag is higher, and higher contents of As are extracted in Steps 1-5. It is noted that the contents of As in the leachates obtained in Steps 4 and 5 are high, indicating maybe there are more As sulfides and metal As in the smelting slag. Moreover, it is found that in the slag concentrate sample the Steps 4 and 5 extract particularly high proportions of As, suggesting that the As sulfides and metal As are the major As species in slag concentrate. The slag concentrate is actually the Cu-rich slag recovered from the smelting slag. Hence, during the slag separation these As species may be recovered together with Cu. The SSE procedure conducted for flue dusts reveals that a considerable content of As exists as oxides, arsenates and sulfides in flue dusts, which is in line with the reported results [14,15]. The recoveries of the three SSE experiments are also as high as 94.18%-96.90%, indicating good applicability and reliability.
As partitioning results shown in Fig. 8 reveals that the As redistribution takes place during slag separation. To be specific, most of As sulfides and metal As in smelting slags are separated with Cu and then enriched as the major phases of As in slag concentrate. As a result, the remained tailings slag has lower hazard. The slag concentrates with high content of As are usually returned to the copper smelting process. Likewise, the flue dusts containing high proportions of As oxides and arsenates should also be cautiously treated.
Table 7 As fractionations and recoveries of some by-products from copper smelting
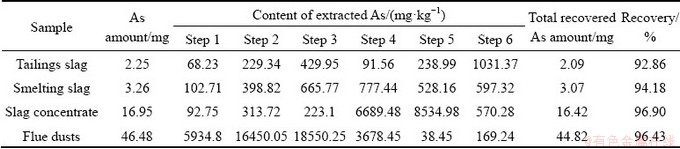
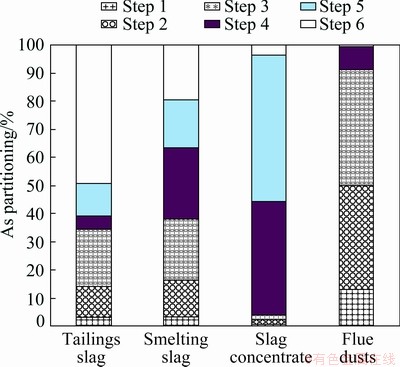
Fig. 8 As partitioning in some As-bearing by-products from copper smelting
4 Conclusions
(1) A selectively sequential extraction procedure, which partitions the As species into the readily soluble As, dissolvable arsenates, sparingly soluble arsenates, As residing in sulfides, arsenopyrite and metal As, as well as As incorporated into glassy silicates, was proposed for As partitioning in copper flash smelting slags.
(2) Single-phase extraction procedures using some synthetic and natural arsenical minerals show that the SSE method has high selectivity and recovery for various As species.
(3) The extraction kinetics of copper tailings slag mainly composed of fayalite, magnetite and glassy silicates was investigated. It is found that approximately 60% of total As with low leachability is incorporated in the glassy silicates. The total As recoveries of SSE procedure for tailing slag samples are in the range of 90.54%-94.76%.
(4) The SSE method was employed to reveal the As partitioning in copper smelting slags, slag concentrate and flue dusts and the corresponding As recoveries are as high as 94.18%-96.90%.
Supporting materials
Supporting materials to this article can be found online at http://qr.csupress.com.cn/Public/ ResourceList/Detail/22522.
References
[1] DUNG T T T, CAPPUYNS V, SWENNEN R, VASSILIEVA E, PHUNG N K. Leachability of arsenic and heavy metals from blasted copper slag and contamination of marine sediment and soil in Ninh Hoa district, south central of Vietnam [J]. Applied Geochemistry, 2014, 44: 80-92.
[2] SHIBAYAMA A, TAKASAKI Y, WILLIAM T, YAMATODANI A, HIGUCHIY, SUNAGAWA S, ONO E. Treatment of smelting residue for arsenic removal and recovery of copper using pyro-hydrometallurgical process [J]. Journal of Hazardous Materials, 2010, 181: 1016-1023.
[3] RUIZ M C, DAROCH F, PADILLA R. Digestion kinetics of arsenic removal from enargite�Ctennantite concentrates [J]. Minerals Engineering, 2015, 79: 47-53.
[4] CHANDRAPRABHA M N, NATARAJAN K A, SOMASUNDARAN P. Selective separation of pyrite from chalcopyrite and arsenopyrite by biomodulation using Acidithiobacillus ferrooxidans [J]. International Journal of Mineral Processing, 2005, 75: 113-122.
[5] NAKAZAW S, YAZAWA A, JORGENSEN F R A. Simulation of the removal of arsenic during the roasting of copper concentrate [J]. Metallurgical and Materials Transactions B, 1999, 30: 393-401.
[6] WATLING H R. The bioleaching of sulphide minerals with emphasis on copper sulphides��A review [J]. Hydrometallurgy, 2006, 84: 81-108.
[7] ZHANG Bao-hong. Experimental study on the separation of copper and arsenic by novel inhibitors [D]. Ganzhou: Jiangxi University of Science and Technology, 2013. (in Chinese)
[8] NAZARI A M, RADZINSKI R, GHAHREMAN A. Review of arsenic metallurgy: Treatment of arsenical minerals and the immobilization of arsenic [J]. Hydrometallurgy, 2016, 174: 258-281.
[9] GORAI B, JANA R K, PREMCHAND. Characteristics and utilisation of copper slag��A review [J]. Resources, Conservation and Recycling, 2003, 39: 299-313.
[10] CHUN T J, NING C, LONG H M, LI J X, YANG J L. Mineralogical characterization of copper slag from Tongling Nonferrous Metals Group, China [J]. JOM, 2016, 68: 2332-2340.
[11] ZHAO Z W, CHAI L Y, PENG B, LIANG Y, HE Y, YAN Z. Arsenic vitrification by copper slag based glass: Mechanism and stability studies [J]. Journal of Non-Crystalline Solids, 2017, 466: 21-28.
[12] WANG Q M, GUO X Y, TIAN Q H, CHEN M, ZHAO B J. Reaction mechanism and distribution behavior of arsenic in the bottom blown copper smelting process [J]. Metals, 2017, 7: 302-313.
[13] KARIMOV K A, NABOICHENKO S S, NEUSTROEV V I. Pressure leaching of copper arsenic-containing mattes with copper sulfate solutions [J]. Russian Journal of Non-Ferrous Metals, 2016, 57: 1-6.
[14] XU Zhi-feng, LI Qiang, NIE Hua-ping. Pressure leaching technique of smelter dust with high-copper and high-arsenic [J]. Transactions of Nonferrous Metals Society of China, 2010, 20: 176-181.
[15] JAROS KOV A, ETTLER V, MIHALJEVIC M, DRAHOTA P, CULKA A, RACEK M. Characterization and pH- dependent environmental stability of arsenic trioxide- containing copper smelter flue dust [J]. Journal of Environmental Management, 2018, 209: 71-80.
[16] MONTENEGRO V, SANO H, FUJISAWA T. Recirculation of high arsenic content copper smelting dust to smelting and converting processes [J]. Minerals Engineering, 2013, 49: 184-189.
[17] MORALES A, CRUELLS M, ROCA A, BERGO R. Treatment of copper flash smelter flue dusts for copper and zinc extraction and arsenic stabilization [J]. Hydrometallurgy, 2010, 105: 148-154.
[18] PIATAK N M, SEAL R R, HAMMARSTROM J M. Mineralogical and geochemical controls on the release of trace elements from slag produced by base- and precious-metal smelting at abandoned mine sites [J]. Applied Geochemistry, 2004, 19: 1039-1064.
[19] REDDY R G, FONT J M. Arsenate capacities of copper smelting slags [J]. Metallurgical and Materials Transactions B, 2003, 34: 565-571.
[20] CHEN C, JAHANSHAHI S. Thermodynamics of arsenic in FeOx-CaO-SiO2 slags [J]. Metallurgical & Materials Transactions B, 2010, 41: 1166-1174.
[21] WAN Xin-yu, QI Yuan-hong, GAO Jian-jun, WU Bing-qiang. Behavior of arsenic in arsenic-bearing copper slag by pyrolysis roasting process [J]. Nonferrous Metals Engineering, 2017, 7: 40-45. (in Chinese)
[22] WANG S, MULLIGAN C N. Speciation and surface structure of inorganic arsenic in solid phases: A review [J]. Environment International, 2008, 34: 867-879.
[23] KEON N E, SWARTZ C H, BRABANDER D, HARVEY J C, HEMOND H F. Validation of an arsenic sequential extraction method for evaluating mobility in sediments [J]. Environmental Science & Technology, 2001, 35: 2778-2784.
[24] ADE H, STOLL H. Near-edge X-ray absorption fine-structure microscopy of organic and magnetic materials [J]. Nature Materials, 2009, 8: 281-290.
[25] TESSIER A, CAMPBELL P G C, BISSON M. Sequential extraction procedure for the speciation of particulate trace metals [J]. Analytical Chemistry, 1979, 51: 844-851.
[26] LARIOS R, FERN NDEZMART NEZ R, RUCANDIO I. Assessment of a sequential extraction procedure for arsenic partitioning and application to samples from different pollution sources [J]. Analytical Methods, 2013, 5: 4096-4104.
[27] LARIOS R, FERNANDEZ-MARTINEZ R, LEHECHO I, RUCANDIO I. A methodological approach to evaluate arsenic speciation and bioaccumulation in different plant species from two highly polluted mining areas [J]. Science of the Total Environment, 2012, 414: 600-607.
[28] PAUL C J, FORD R G, WILKIN R T. Assessing the selectivity of extractant solutions for recovering labile arsenic associated with iron (hydr)oxides and sulfides in sediments [J]. Geoderma, 2009, 152: 137-144.
[29] DRAHOTA P, GRSSLOV Z, KINDLOV H. Selectivity assessment of an arsenic sequential extraction procedure for evaluating mobility in mine wastes [J]. Analytica Chimica Acta, 2014, 839: 34-43.
[30] COOPER D C, MORSE J W. Selective extraction chemistry of toxic metal sulfides from sediments [J]. Aquatic Geochemistry, 1999, 5: 87-97.
[31] PANDA S, MISHRA S, RAO D S, PRADHAN N. Extraction of copper from copper slag: Mineralogical insights, physical beneficiation and bioleaching studies [J]. Korean Journal of Chemical Engineering, 2015, 32: 667-676.
[32] MAJUMDER S, NATH B, SARKAR S, ISLAM S M, BUNDSCHUH J, CHATTERJEE D, HIDALGO M. Application of natural citric acid sources and their role on arsenic removal from drinking water: A green chemistry approach [J]. Journal of Hazardous Materials, 2013, 262: 1167-1175.
[33] ASH C, TEJNECK V, BORUVKA L, DRABEK O. Different low-molecular-mass organic acids specifically control leaching of arsenic and lead from contaminated soil [J]. Journal of Contaminant Hydrology, 2016, 187: 18-30.
[34] COLLINS R N. Separation of low-molecular mass organic acid�Cmetal complexes by high-performance liquid chromatography [J]. Journal of Chromatography A, 2004, 1059: 1-12.
[35] van HEES P A W, JONES D L, JENTSCHKE G, GODBOLD D L. Organic acid concentrations in soil solution: Effects of young coniferous trees and ectomycorrhizal fungi [J]. Soil Biology and Biochemistry, 2005, 37: 771-776.
[36] DOLD B. Dissolution kinetics of schwertmannite and ferrihydrite in oxidized mine samples and their detection by differential X-ray diffraction (DXRD) [J]. Applied Geochemistry, 2003, 18: 1531-1540.
[37] WILLEY G R, ASAB A, LAKIN M T, ALCOCK N W. Group 15�Cazamacrocyclic chemistry. Reactions of 1,4,8,11-tetramethyl-1,4,8,11-tetraazacyclotetradecane (L) with MCl3 (M=As or Sb): crystal and molecular structures of [H2L][As4O2Cl10] and [H2L][Sb2OCl6] [J]. J Chem Soc: Dalton Trans, 1993(3): 365-370.
[38] REDDY R G, BLANDER M. Modeling of sulfide capacities of silicate melts [J]. Metallurgical Transactions B, 1987, 18: 591-596.
[39] COOPER D C, MORSE J W. Extractability of metal sulfide minerals in acidic solutions: Application to environmental studies of trace metal contamination within anoxic sediments [J]. Environmental Science & Technology, 1998, 32: 1076-1078.
[40] CHAO T T, SANZOLONE R F. Chemical dissolution of sulfide minerals [J]. Journal of Research of the U.S. Geological Survey, 1977, 5: 409-412.
[41] CHANG F C, LO S L, LEE M Y, KO C H, LIN J D, HUANG S C, WANG C F. Leachability of metals from sludge-based artificial lightweight aggregate [J]. Journal of Hazardous Materials, 2007, 146: 98-105.
[42] VILLEGAS M A, NAVARRO J M F. Characterization of B2O3-SiO2 glasses prepared via sol-gel [J]. Journal of Materials Science, 1988, 23: 2464-2478.
[43] MACDONALD S A, SCHARDT C R, MASIELLO D J, Simmons J H. Dispersion analysis of FTIR reflection measurements in silicate glasses [J]. Journal of Non-Crystalline Solids, 2000, 275: 72-82.
[44] DALBY K N, KING P L. A new approach to determine and quantify structural units in silicate glasses using micro-reflectance Fourier-transform infrared spectroscopy [J]. American Mineralogist, 2006, 91: 1783-1793.
[45] DRAHOTA P, KNAPPOV M, KINDLOV H, Culka A, MAJZLAN J, MIHALJEVIC M, ROHOVEC J, VESELOVSKY F, FRIDRICHOVA M, JEHLICKA J. Mobility and attenuation of arsenic in sulfide-rich mining wastes from the Czech Republic [J]. Science of the Total Environment, 2016, 557-558: 192-203.
�Żݱ�1��������1����ݼݼ1�������1���ܻ���1���� ��2��֣����1
1. �㽭��ҵ��ѧ ���Ͽ�ѧ�빤��ѧԺ������ 310014��
2. ͭ����ɫ���ţ�ͭ�� 244000
ժ Ҫ������һ���Ż���ѡ��������ȡ��������������ͭ�������к���������ɡ��÷�����ͭ�������еĺ����������Ϊˮ�����顢�����������Ρ������������Ρ����������ɰ�ͽ������Լ��̻��ڲ������е��顣���õ����ϵ�������ԭ�ӷ�����ס�ɨ��羵����羵��X��������ͺ�������ֶζԸ�����ȡҺ�������б��������õ�һ������ȡʵ��������ȡ�Լ���ѡ���Ժ�������ʡ����������¯���и������ı��������ױ������нϺõ�һ���ԣ�����������ʱ�����90%���ϡ�����������о��Ż���ѡ��������ȡ�����ܿɿ���Ӧ����ͭұ�����鸱�����к�������ijɷ����ۡ�
�ؼ��ʣ��飻ͭ¯��������ȡ���̷Ϲ�����ͭұ��
(Edited by Wei-ping CHEN)
Foundation item: Projects (51874257, 51374185, 51801183) supported by the National Natural Science Foundation of China
Corresponding author: Guo-qu ZHENG, E-mail: zhenggq@zjut.edu.cn; Jun ZHOU, E-mail: zhouj@jgty.net
DOI: 10.1016/S1003-6326(20)65424-3

