J. Cent. South Univ. Technol. (2008) 15: 796-800
DOI: 10.1007/s11771-008-0147-z
Synthesis of mesoporous cerium-zirconium mixed oxides by
hydrothermal templating method
WANG Man-juan(王曼娟)1, GU Ying-ying(古映莹)1, QIN Li-ping(秦利平)2,
LI Jin-lin(李金林)3, XU Wen-yang(徐文泱)1
(1. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China;
2. Science and Technology Department, Guangxi University of Technology, Liuzhou 545006, China;
3. Key Laboratory of Catalysis and Materials Science of the State Ethnic Affairs Commission & Ministry of Education, Hubei Province, South-Central University for Nationalities, Wuhan 430074, China)
Abstract: Mesoporous cerium-zirconium mixed oxides were prepared by hydrothermal method using cetyl trimethyl ammonium bromide (CTAB) as template. The effects of amount of template, pH value of solution and hydrothermal temperature on mesostructure of samples were systematically investigated. The final products were characterized by XRD, TEM, FT-IR, and BET. The results indicate that all the cerium-zirconium mixed oxides present a meso-structure. At molar ratio of n(CTAB)/n((Ce)+(Zr))= 0.15, pH value of 9, and hydrothermal temperature of 120 ℃, the samples obtained possess a specific surface area of 207.9 m2/g with pore diameter of 3.70 nm and pore volume of 0.19 cm3/g.
Key words: cerium-zirconium mixed oxides; hydrothermal method; cetyl trimethyl ammonium bromide; mesopore; template
1 Introduction
Cerium-zirconium mixed oxide, serving as an excellent catalyst and promoter, has received widespread attention particularly in the application of automotive exhaust removal. It plays an important role in the preparation of three-way catalyst (TWC) with noble metal. Due to the participation of the cerium-zirconium mixed oxides, the amount of noble metal has been reduced tremendously, and the thermal stability of TWC has been highly improved[1-4].
As we all know, the oxidation-reduction reaction of TWC mainly takes place on the surface of the catalyst, therefore, high specific surface area and high thermal stability are essential to the improvement of the oxygen storage capacity. The preparation of catalytic materials with high specific surface area is somehow limited when only particle size is taken into consideration. So far, the nano-size cerium-zirconium oxides have been prepared by various methods. However, their specific surface area is low[5-7]. One approach has emerged in recent years with the discovery of mesoporous structure. It was reported mesoporous cerium-zirconium mixed oxides with high specific surface area were prepared using surfactant as template[8-12]. FENG et al[8-9] prepared mesoporous Ce0.5Zr0.5O2 mixed oxides with high specific surface area by hydrothermal method using anionic surfactant, sodium dodecylbenzenesulfonate (SDBS) or polyethyleneglycol (PEG4000) as template. YUAN et al[10] prepared hexagonal mesoporous Ce1-xZrxO2 with high specific surface area and regular structure by sol-gel method with block copolymer P123 as template. Cetyl trimethyl ammonium bromide (CTAB), a frequently-used cationic surfactant, can reduce the surface free energy and prohibit particle condensation, and it has been widely applied in the controlled synthesis of nano-size materials and molecular sieves[13-17]. LONG et al[14] prepared Pt-supported TWC by sol-gel method with CTAB as template. DU et al[15, 18] prepared cerium-zirconium mixed oxides with high specific surface area by precipitation method with CTAB as template. In these reports, the adding amount of CTAB was larger, which resulted in high cost and difficulty in its removal by washing. Hence, precipitation and sol-gel methods have been adopted as the main approaches, and the template can be removed in high-temperature calcinations.
In this work, hydrothermal method with CTAB as the template was used to synthesize the cerium- zirconium mixed oxides. Samples with high specific surface area, small pore diameter and narrow pore size distribution were obtained.
2 Experimental
2.1 Synthesis
All reagents and solvents used were analytical grade. In a typical synthetic route, the materials were prepared by adding an aqueous solution of CTAB to a mixed aqueous solution of cerium and zirconium ((c(Ce)+ c(Zr))=0.16 mol/L). The molar ratio of Ce to Zr was kept at 1.0. Ammonia (2.6 mol/L) was added into the solution with stirring to adjust pH value. The mixture was stirred for another 30 min, then sealed and placed for 4 h. After that process, the solution was transferred to a Teflon-lined stainless steel vessel (100 mL) and maintained at the desired temperatures for 72 h. Then, the mixture was cooled and filtered and washed repeatedly with deionized water for the removal of the surfactant. AgNO3 was used to examine the existence of Cl-. When there was no Cl- left, the filtrate was washed repeatedly with ethanol and water. The product was obtained after being dried at 120 ℃ for 12 h.
2.2 Characterization
Products were analyzed by X-ray powder diffraction (SIMENS-D500) using 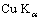 radiation, with a tube voltage of 36 kV and a current of 30 mA. Nitrogen adsorption-desorption isotherms were recorded with Micromeritics ASAP 2000 surface analyzer. Specific surface area was calculated by single-point method and the thickness of the pore wall was evaluated by the empirical formula[16]. It was supposed that the pores were cylindrical with uniform size, then
radiation, with a tube voltage of 36 kV and a current of 30 mA. Nitrogen adsorption-desorption isotherms were recorded with Micromeritics ASAP 2000 surface analyzer. Specific surface area was calculated by single-point method and the thickness of the pore wall was evaluated by the empirical formula[16]. It was supposed that the pores were cylindrical with uniform size, then
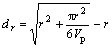 (1)
(1)
where dr is the thickness of the wall; r is the average pore diameter and Vp is the average pore volume. An NIC0-7199 infrared spectrometer was used to measure the Fourier infrared spectra of the samples, with scanning range from 400 to 4 000 cm-1.
3 Results and discussion
3.1 Effect of amount of template
The low-angle XRD patterns of the samples obtained under different amounts of CTAB are shown in Fig.1. The pH value of solution and hydrothermal temperature were kept at 9 and 120 ℃, respectively. It can be seen from Fig.1 that characteristic peak for mesostructure of each sample appears at 2θ=0.5?-1?, varying in intensities. When n(CTAB)?n(Ce+Zr) is 0.05, the peak is comparatively weak; when n(CTAB)? n(Ce+Zr) is 0.15, the intensity of the peak increases sharply, which indicates a good structural regularity of the samples. When n(CTAB)?n(Ce+Zr) continually increases to 0.25, the intensity and the shape of the peak decreases and broadens respectively. A possible interpretation is that micelle can hardly be obtained in solution due to small amount of template, which is disadvantageous for the formation of mesostructure. However, too much template causes high concentration of micelle in solution, offering little space between micelles, which results in insufficient interaction between the inorganic material and micelle, and therefore hampers the formation of mesostructure. It can be seen from Fig.1 that when n(CTAB)?n(Ce+Zr) is 0.15, cerium-zirconium mixed oxides with regular structure are obtained. LIU et al[17] prepared CeO2-ZrO2-Y2O3 at n(CTAB)?n(Ce+Zr)=0.34; ZOU et al[18] synthesized La-Ce-Co-Zr-O when n(CTAB)?n(Ce+Zr)=0.9; DU et al[15-16] synthesized cerium-zirconium mixed oxides when n(CTAB)?n(Ce+Zr) are 2 and 4, respectively. In comparison with these literatures, the amount of CTAB in this work is reduced considerably, which is beneficial to saving cost and simplifying the sequential treatment.
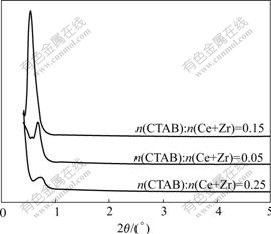
Fig.1 Low-angle XRD patterns of Ce0.5Zr0.5O2 synthesized at different amounts of template
3.2 Effect of pH value
Fig.2 shows the low-angle XRD patterns of the samples obtained at different pH values and the same hydrothermal temperature and amount of template (120 ℃, n(CTAB)?n(Ce+Zr)=0.15). It is observed that when pH value is between 8 and 11, the main peak of each sample appears at 2θ=0.5?-1?. The peak of sample obtained at pH 9 is stronger than that at other pH value, whereas no obvious peaks are observed at pH 8. With pH value rising, the characteristic peak becomes increasingly weaker. This is possibly because that the concentration of OH- at pH 8 is lower, which is disadvantageous for self-assembly between surfactant and inorganic material. While the concentration of OH- will be too high when the pH value is above 9, and absorption of OH- to the surface of cerium-zirconium mixed oxides will also hamper the formation of mesoporous samples with regular structure. Hence, the optimal pH value in this work is 9.

Fig.2 Low-angle XRD patterns of Ce0.5Zr0.5O2 synthesized at different pH values
3.3 Effect of hydrothermal temperature
Fig.3 shows the low-angle XRD patterns of the samples obtained at pH 9 and the molar ratio of n(CTAB)?n(Ce+Zr)=0.15 but different hydrothermal temperatures. It is demonstrated that obvious characteristic peak can be observed and becomes intensive at hydrothermal temperature 90 and 120 ℃, respectively. With temperature increasing, the intensity of peak decreases sharply, which reflects good correlation between hydrothermal temperature and structure regularity of samples. We can hypothesize that with temperature rising, regularity of crystalline structure of samples increases gradually. Then, further rising temperature will lead to damage to the regularity of crystal structure as de-gel reaction becomes main reaction, which is contributed to the decrease of the peak intensity in XRD patterns.
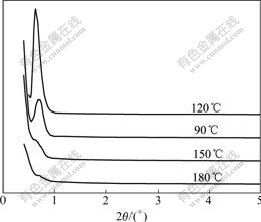
Fig.3 Low-angle XRD patterns of Ce0.5Zr0.5O2 synthesized at different hydrothermal temperatures
3.4 FT-IR analysis of samples
Fig.4 shows FT-IR patterns of samples prepared in the absence and the presence of template (n(CTAB)? n(Ce+Zr)=0.15) at pH 9 and hydrothermal temperature of 120 ℃. It can be seen from Fig.4 that the same main absorption peak can be observed in the two samples in the absence and presence of template. The bands at 3 400 and 3 500 cm-1 are the stretching vibration absorption spectra of H―O; the bands at 1 500-1 600 cm-1 are the bending vibration absorption spectra of HO―H; the band at 500 cm-1 is the stretching vibration absorption spectrum of Ce(Zr)―O. No peak of organic template has ever appeared in the pattern, which shows that CTAB template can be completely removed by repeatedly washed with water and ethanol. Without high- temperature calcinations, the preparation process is simplified, and agglomeration between particles owing to calcinations can be avoided.
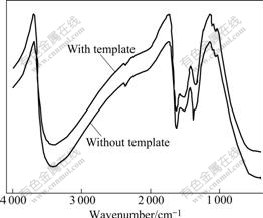
Fig.4 FT-IR patterns of Ce0.5Zr0.5O2
3.5 Thermal stability analysis of samples
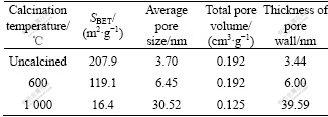
The nitrogen adsorption-desorption isotherms of samples before and after calcination are shown in Fig.5, where n(CTAB)?n((Ce)+(Zr))=0.15, pH=9, and hydrothermal temperature is 120 ℃. It is learned from Fig.5 that all the adsorption-desorption isotherms are type Ⅳ with distinct hysteresis loops, a typical adsorption type of mesostructure materials. Table 1 shows the pore structure parameters of these samples. It is clear that the specific surface area of samples calcined at 600 and 1 000 ℃ decrease considerably, especially for samples obtained at 1 000 ℃, and the average pore diameter and thickness of pore wall increase dramatically with the sharp decreasing of specific surface area .It can be inferred that growth of grain calcined at 600 ℃ gives rise to the decrease in specific surface area and increase in average pore diameter and thickness of pore wall. Moreover, the dramatic increase in average diameter and thickness of pore wall can be observed after samples are calcined at 1 000 ℃, which indicates the mesostructure has been ruined, leading to the sharp decrease in specific surface area. As known from the absorption-desorption isotherms, the hysteresis loops are not obvious, which indicates that there are few genuine mesopores in the samples, and the appearance of the loops is due to the pore structure resulting from the particle packing in samples.
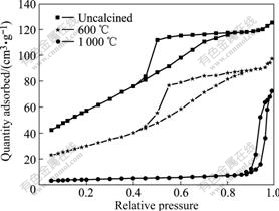
Fig.5 Nitrogen adsorption-desorption isotherms of Ce0.5Zr0.5O2
Table 1 Structural parameters of samples before and after calcination
The XRD patterns of samples obtained before and after calcination at 600 and 1 000 ℃ for 2 h are shown in Fig.6. In the experiment, n(CTAB)?n(Ce+Zr) was fixed at 0.15, pH value was 9, and hydrothermal temperature was 120 ℃. From low angle XRD patterns, it is found the intensity of characteristic peak of the sample at 2θ= 0.7? decreases with the calcination temperature rising. While the wide-angle XRD patterns show that the diffraction peak of the samples becomes sharper after calcination, which shows that the sample has undergone a crystalline growth process in the calcination, i.e. the small crystal aggregated to form a larger one during calcination. The curvature which plays a significant role in the formation of pore structure can not be obtained due to the crystallization, resulting in collapses.
3.6 TEM image of samples
TEM image of samples obtained at pH 9, hydro- thermal temperature of 120 ℃ and n(CTAB)?n(Ce+Zr)= 0.15 is shown in Fig.7. The elliptical particles with uniform size can be obtained. It can be seen from Fig.7 that white vermicular structure appears in the black area, which indicates the existence of mesoporous structure. The black area indicates the existence of the pore wall, whereas the well distributed white area shows a structure regularity of the samples, which is in good agreement with the low-angle XRD pattern.

Fig.6 XRD patterns of Ce0.5Zr0.5O2 before and after calcination: (a) Low angle; (b) Wide angle

Fig.7 TEM image of Ce0.5Zr0.5O2
4 Conclusions
1) Mesoporous cerium-zirconium mixed oxides with high specific surface area are synthesized by hydrothermal method using CTAB as template. The hydrothermal temperature, quantity of template and pH value of solution have great influences on formation of mesostructure. When the hydrothermal temperature, pH value and the amount of template (n(CTAB)?n(Ce+Zr)) are 120 ℃, 9, and 0.15, respectively, the obtained cerium-zirconium mixed oxides possess specific surface area of 207.9 m2/g , pore diameter of 3.70 nm and pore volume of 0.19 cm3/g.
2) Ce0.5Zr0.5O2 mixed oxides calcined at 600 and 1 000 ℃ maintain cubic structure. The growth of grain calcined at 600 ℃ gives rise to the decrease in specific surface area. After calcined at 1 000 ℃, the sharp decrease in specific surface area and increase in average pore diameter and thickness of pore wall are observed.
3) Owing to the low requirement on the template, it is easier to remove the template compared with the precipitation and sol-gel method when n(CTAB)? n(Ce+Zr) is 0.05-0.25, which could reduce the cost, simplify the process. Moreover, the agglomerations between particles due to high-temperature calcination are also avoided successfully.
References
[1] OZAWA M. Role of cerium-zirconium mixed oxides as catalysts for car pollution: A short review [J]. Journal of Alloys and Compounds, 1998, 275/277: 886-890.
[2] FALLY F, PERRICHON V, VIDAL H, KASPAR J, BLANCO G, PINTADO J M, BERNAL S, COLON G, DATURI M, LAVALLEY J C. Modification of the oxygen storage capacity of CeO2-ZrO2 mixed oxides after redox cycling aging [J]. Catalysis Today, 2000, 59(3/4): 373-386.
[3] FENG Chang-gen, HU Yu-cai, WANG Li-qiong. Influence of ceria-zirconia solid solution on the performance of palladium-only three-way catalyst [J]. Applied Chemistry, 2003, 20(2): 159-162. (in Chinese)
[4] WU Wen-hua, XIA Chang-qing. Microstructures and mechanical properties of Mg-Ce-Zn-Zr wrought alloy [J]. Journal of Central South University of Technology, 2004, 11(4): 367-370.
[5] FENG Chang-gen, ZHANG Jiang-shan, WANG Ya-jun. Synthesis and catalytic activity of nanometer scale Ce1-xZrxO2 polycrystals [J]. Journal of Chinese Rare Earth Society, 2004, 32(4): 502-506. (in Chinese)
[6] TROVARELLI A, ZAMAR F, LLORA J, de LEITENBURG C, DOLCETTI G, KISS J T. Nanophase fluorite-structured CeO2-ZrO2 catalysts prepared by high-energy mechanical milling [J]. Journal of Catalysis, 1997, 169(2): 490-502.
[7] de LEITENBURG C, TROVARELLI A, BINI G, LLORCA J, CAVANI F, BINI G. The effect of doping CeO2 with zirconium in the oxidation of siobutane [J]. Applied Catalysis A: General, 1996, 139(1/2): 161-173.
[8] FENG Sheng-sheng, GU Ying-ying, LI Jin-lin, SONG Feng-xuan. Preparation and characterization of mesoporous Ce0.5Zr0.5O2 mixed oxide [J]. Chinese Journal of Chemistry, 2007, 25(10): 1499-1502.
[9] GU Ying-ying, FENG Sheng-sheng, LI Jin-lin, GU Xiang-kui, WANG Man-juan. Preparation of mesoporous Ce0.5Zr0.5O2 mixed oxide by hydrothermal templating method [J]. Journal of Rare Earths, 2007, 25(12): 710-714.
[10] YUAN Quan, LIU Qiang, SONG Wei-guo, FENG Wei, PU Wan-li, SUN Ling-dong, ZHANG Ya-wei, YAN Chun-hua. Ordered mesoporous Ce1-xZrxO2 solid solutions with crystalline walls [J]. Journal of the American Chemical Society, 2007, 129(21): 6698-6699.
[11] MASUI T, OZAKI T, MACHIDA K, ADACHI G. Preparation of ceria-zirconia sub-catalysts for automotive exhaust cleaning [J]. Journal of Alloys and Compounds, 2000, 303/304: 49-55.
[12] CHEN Min, ZHANG Pei-zhuang, HUANG Zhi-bin, ZHENG Xiao-ming. Preparation of ultrafine Ce-Zr-Ba mixed oxide powder with high surface area [J]. Journal of Inorganic Materials, 2004, 19(3): 661-665. (in Chinese)
[13] FENG Ru-ming, YANG Xiu-juan, JI Wei-jie, AU Chak-tong. Hydrothermal synthesis of stable mesoporous ZrO2-Y2O3 and CeO2-ZrO2-Y2O3 from simple inorganic salts and CTAB template in aqueous medium [J]. Materials Chemistry and Physics, 2008, 107(1): 132-136.
[14] LONG Q, CAI M, ROGERS J D, RONG H L, LI J R, JIANG L. Synthesis of nanostructured Pt/CeO2-ZrO2-Al2O3 catalysts by a two-step sol-gel method [J]. Nanotechnology, 2007, 18(35): 355601.
[15] DU Yu-cheng, ZHANG Jiu-xing, HE Hong. High specific surface of Ce0.7Zr0.3O2 solid solution nanostructure by templated assemble [J]. Journal of the Chinese Rare Earth Society, 2003, 21(S1): 44-47. (in Chinese)
[16] LIU Xin-mei, XING Wei, YAN Zi-feng. Surface and pore structure modification of mesoporous nano-zirconium dioxide [J]. Journal of Inorganic Materials, 2005, 21(2): 191-196. (in Chinese)
[17] ZOU Zhi-qiang, MENG Ming, ZHA Yu-qing, LIU Yong. Dual templates assisted preparation and characterization of highly thermostable multicomponent mesoporous material La-Ce-Co-Zr-O used for low-temperature CO oxidation [J]. Journal Materials Science, 2008, 43(6): 1958-1965.
[18] ZHANG Yu-li, DU Yu-cheng, SUO Er-bin. Synthesis of rare earth compound oxides nano-crystal with high specific surface area [J]. Mining and Metallurgy, 2005, 14(1): 45-48. (in Chinese)
Foundation item: Project(CHCL0501) supported by Hubei Provincial Open Fund of Key Laboratory of Catalytic Material Science and Technology
Received date: 2008-03-07; Accepted date: 2008-05-11
Corresponding author: GU Ying-ying, Professor; Tel: +86-13467517387; E-mail: guyy02@163.com
(Edited by ZHAO Jun)