Trans. Nonferrous Met. Soc. China 26(2016) 1019-1024
Preparation of micro-nanostructure on titanium implants and its bioactivity
Rui Zhang, Yi Wan , Xing Ai, Teng Wang, Bo Men
Key Laboratory of High Efficiency and Clean Mechanical Manufacture, Ministry of Education, School of Mechanical Engineering, Shandong University, Jinan 250061, China
Received 23 April 2015; accepted 25 February 2016
Abstract: Surface modification of medical implants was considered as an effective method to improve the cellular behaviors and the integration of tissue onto materials. The micro-nanostructured surface on the titanium alloy was prepared by laser treatment and multiple acid etching. The surface morphologies of different titanium alloy substrates were characterized by scanning electron microscopy (SEM). The effects of micro-nanostructured surfaces on the cellular responses were investigated in vitro by observing hydroxyapatite formation, cell morphology and cell adhesion. The results indicate that the micro-sized structure promoted the adhesion and proliferation of cultured osteoblasts. Furthermore, the micro-nanostructured surface was more conducive to cell adhension stretching compared with the micro-structured surface. All results suggest that the micro-nanostructured surface improved the biocompatibility and integration of tissue onto titanium alloy implants.
Key words: titanium alloy; micro-nanostructure; laser treatment; multiple acid etching; bioactivity
1 Introduction
Biomedical titanium alloys show good physical and mechanical performance, and are therefore widely used as surgical implant materials [1-3]. However, the bone could not be reconstructed efficiently on the surface of titanium and titanium alloys implants, which leads to poor integration with the surrounding bony tissue [4-6]. Therefore, to further improve the biocompatibility of titanium and titanium alloys implants, it is quite important to modify their surface with biotechnology, which will accelerate the integration between implanted materials and the interface of human tissue.
The current surface modification methods for titanium alloys include hydroxyapatite (HA)-coating, acid etching, sand blasting and anodizing. Among the various technologies for surface biomodification, the biomimetic topography of implant surface has attracted special attention of researchers [7-11]. A microstructure promotes secretion and mineralization of extracellular matrix [12,13], and promotes fast and steady bone integration [14]. A nanostructure also shows a positive effect on the osteoblast behavior [15], can accurately mimic the cellular growth environment [16], and promotes adhesion, proliferation and differentiation [17,18], which in turn promotes bone mineralization [19,20]. However, most of former methods focus on developing single scale structures on the implant surface, microstructure or nanostructure. There are few studies on the micro-nanostructured surface.
This work proposed a novel method to construct micro-nanostructure on bio-titanium alloy surface with laser treatment and multiple acid etching. The bioactivity of the samples was investigated in vitro by observing hydroxyapatite formation, cell morphology and cell adhesion. The new method for surface modification was expected to be widely applied in the biomedical field.
2 Experimental
2.1 Sample preparation
The sample material used was a titanium alloy (TC4), which is widely used in the field of biomedicine. Titanium alloy (TC4) samples with dimensions of 10 mm ЎБ 10 mm ЎБ 1.5 mm were used. The samples were polished by a polishing machine and further cleaned with acetone and water.
The polished samples were manufactured by a YAG-T80C laser-marking machine (Shenzhen HanЎҜs Laser Technology Limited Company, China), which has a wavelength of 1064 nm and a spot diameter of 30 ҰМm. The current and frequency of the laser machine were 13 A and 3 kHz, respectively, which were optimized in advance. The microstructure samples contained micro- pits with a diameter of 140 ҰМm and a depth of 35 ҰМm. Surface topography was characterized by scanning electron microscopy (SEM), as shown in Fig. 1. The clinkers produced by the laser on the inner surface and micro-pits edge can easily fall off, and were found to be harmful to the biocompatibility of the implant. Therefore, the clinkers were cleared in advance.
Multiple acid etching was used to clean off the laser clinkers and generate the micro-nanostructure, as shown in Fig. 2. Firstly, the samples were treated with a mixed solution containing 0.09 mol/L HNO3 and 0.11 mol/L HF at room temperature for 10 min. Secondly, the samples were treated with a mixed solution containing 4.5 mol/L H2SO4 and 2.9 mol/L HCl at 80 ЎгC for 25 min and then cleaned with water quickly. Thirdly, the samples were treated with a mixed solution of 98% H2SO4 and 30% H2O2 with volume ratio of 1:1 at room temperature for 70 min. Finally, the samples were washed with distilled water.
The laser clinkers were cleaned by multiple acid etching, as shown in Fig. 2. At the same time, the micro-nanostructure was obtained. After multiple acid etching, orderly ridge structures with widths of 0.5-2 ҰМm were observed in the inner micro-pits containing a cluster of nanopores with diameters of 20-100 nm.
The samples were divided into three groups: polished group, microstructure group and micro- nanostructure group.

Fig. 1 SEM images of titanium alloy (TC4) surfaces after laser treatment

Fig. 2 SEM images of titanium alloy (TC4) surfaces after multiple acid etching
2.2 Cell culture
MC3T3 osteoblasts (Oral Medicine Institute of Shandong University, China) were cultured in alpha-minimal essential medium (ҰБ-MEM) containing 10% fetal bovine serum and 1% penicillin-streptomycin (Gibco, USA) at 37 ЎгC under a 5% CO2 atmosphere in a saturated humidity incubator. The medium was refreshed every two days. The cultured cells were digested with 0.25% pancreatin for subsequent experiments.
2.3 Simulated body fluid (SBF) analysis
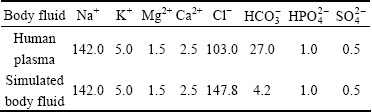
The bioactivity of unvaccinated titanium alloy implants was examined in SBF. The three groups of TC4 samples (polished, microstructure, micro-nanostructure) were soaked in 30 mL SBF in an Eppendorf tube, respectively. The pH value of the SBF was adjusted with tris (hydroxymethyl) aminomethane to 7.4 at 37 ЎгC. The samples were washed with distilled water and air-dried after soaking in the SBF for 15 d. At present, the SBF is usually pre-made by the Kokubo method. The concentrations of all kinds of ions in the SBF should be close to those of human plasma. The concentrations of the different types of ions in human plasma and SBF are shown in Table 1 [21].
Table 1 Ion concentrations in stimulated body fluid and human plasma (mmol/L)
2.4 Cell morphology
The three groups of sterilized samples were placed in a 48-well plate, and MC3T3 osteoblasts at a concentration of 3.3ЎБ104 cell/cm2 were inoculated in each 0.5 mL well. After culturing at 37 ЎгC under a 5% CO2 atmosphere for 3 d, the three groups were treated as follows. 1) Cell fixation: the samples were washed with phosphate-buffered saline (PBS) three times and then fixed with 2% glutaral pentanedial at 4 ЎгC for 20 min. 2) Dehydration: the samples were dehydrated in a gradient ethanol solution (15%, 30%, 45%, 60%, 75%, 90% and 100%, volume fraction) at room temperature for 10 min, respectively. 3) Drying: the samples were soaked in a mixed solution of hexamethyldisilazane (HMDS; Institute of Gangfu Fine Chemical Industry, Tianjin, China) and ethanol with volume ratios of 1:2, 1:1 and 2:1 for 10 min, respectively. Finally, the samples were soaked in pure HMDS for 10 min.
2.5 Cell adhesion
The three groups of sterilized samples were placed in a 48-well plate, and MC3T3 osteoblasts at a concentration of 3.3ЎБ104 cell/cm2 were inoculated in each 0.5 mL well. After culturing at 37 ЎгC under a 5% CO2 atmosphere for 6 h, the samples were moved to a new 48-well plate and washed with PBS to remove the unattached cells, and then digested with pancreatin for 2 min. The osteoblast suspension was centrifuged at 800 r/min for 8 min. Finally, the osteoblast suspension was counted on a counting board.
2.6 Data analysis
The standard deviation of the experimental data (cell adhesion) was determined through statistical analysis. The significant difference, p, between groups was analyzed by Student-Newman-Keuls test, where p<0.05 was determined to be a significant difference and p<0.01 was determined to be a very strong significant difference. Data analysis was performed using the Statistical Program for Social Sciences (SPSS) statistical software package.
3 Results and discussion
3.1 Measurement of hydroxyapatite deposition on sample surfaces
The three groups of samples were observed by SEM after soaking in the SBF for 15 d, and the representative images are shown in Fig. 3. A small amount of HA was observed on the surfaces of the polished samples (Fig. 3(a)) and microstructure samples (Fig. 3(b)), although a substantial amount was observed on the micro-nanostructure samples (Fig. 3(c)). The products on the surfaces of samples were identified as HA by energy dispersive spectroscopy (EDS). The distribution of HA on the micro-nanostructure surface was not uniform. The HA layer inside the micro-pits was thick and dense, whereas that on the edge of the micro-pits was thin and loose, which indicates that the inner micro-pits were more conducive to nucleation and HA growth. The possible reason for the increased amount of HA in the inner micro-pits is that all kinds of ions were gathered in the micro-pits, which results in easy nucleation. In addition, the three-dimensional structure formed with nanopores in the micro-pits was conducive to the further growth of the HA crystal nucleus. At the same time, this structure improved the stability of HA adhesion [22].
3.2 Cell morphology
The morphologies of osteoblasts after culturing for 3 d on different samples are shown in Fig. 4. The osteoblasts spread and proliferated on all surfaces. Oval cells with a small number of filopodia were observed on the polished (Fig. 4(a)) and microstructure (Fig. 4(b)) surfaces, and the cells were sparsely spread along these surfaces. By contrast, the osteoblasts were attached closely to the micro-nanostructure surface (Fig. 4(c)), and substantial cytoplasmic extensions and filopodia were observed. These results show that the micro- nanostructure has a positive effect on the adhesion, proliferation and differentiation of cultured osteoblasts.

Fig. 3 SEM images and EDS patterns of samples soaked in simulated body fluid for 15 d with polished (a), micro-pits (b) and micro-nanostructure (c,d) surfaces

Fig. 4 SEM images of cell morphologies on surfaces of different titanium alloy (TC4) samples
The micro-nanostructures not only increased the specific surface area, but also were beneficial to the aggregation of PO43-, Ca2+, OH-, HPO42- and so on [23]. Furthermore, the nanopores offered an appropriate environment for the cytoplasmic extensions and the filopodia to spread, which is conducive to initial cell adhesion. These results support the previous studies showing that nanostructure materials have a positive effect on the behavior of osteoblast behavior [15] to appropriately mimic the cellular growth environment [16] and to promote adhesion and proliferation [17.18].
3.3 Cell adhesion
A counting board was used to investigate the numbers of adherent cells. The adhesion rates of osteoblasts after culturing for 4 h on different samples are shown in Fig. 5. The adhesion rates were calculated as the ratio of the number of adherent cells to the total number of inoculated cells, and were 92.28%, 72.2% and 62.23% for the micro-nanostructure samples, microstructure samples, and polished samples, respectively.
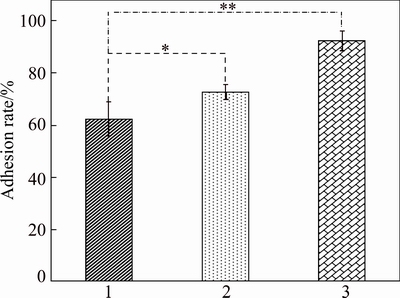
Fig. 5 Adhesion rates of osteoblasts after culturing for 4 h on different titanium alloy (TC4) samples with polished surface (1), microstructure surface (2), and micro-nanostructure surface (3), with histogram p-values of **p<0.01, *p<0.05
These results demonstrate that the adhesion of osteoblasts on the microstructure samples was better than that on the polished samples, and the micro- nanostructure could promote better osteoblastic adhesion compared with the microstructure. Cell adhesion is the first step required for a cell to grow on the implant surface, which plays an important regulatory role for subsequent cell behaviors such as proliferation, differentiation, protein production and gene expression [24,25]. These results confirm that a modified surface, especially a micro-nanostructured surface, can promote osteoblastic adhesion. An artificial nanostructure can affect the cell morphology, adhesion, proliferation and cell skeleton assembly, which controls the transcription activity and gene expression by regulating tyrosine kinase activity and intracellular signaling pathways [26]. In addition to the nanostructure (5 nm to the micron-level), the ordered surface structure can also regulate cell behavior [27].
In summary, conclusions can be drawn that a surface with micro-nanostructures is superior for inducing the formation of HA, and cell adhesion and growth on the surface with a micro-nanostructure were better than those on the surface with a microstructure or polished structure.
4 Conclusions
1) The micro-nanostructured surface was prepared by laser treatment and multiple acid etching. The micro-pit array was built by laser. The nanostructure was superimposed on micro-pit array by multiple acid etching. The clinkers generated by laser were cleaned completely by multiple acid etching.
2) The HA coating on the micro-nanostructured surface was denser than those on the polished and micro-textured surfaces. The micro-nanostructured surface could promote osteoblast adhesion and proliferation.
3) The micro-nanostructured surface displayed better ability for HA formation and cell activity, which increased the bioactivity of implants. Therefore, the combination of laser and multiple acid etching for the modification of a biomedical material is expected to have broad applications in the biomedical field.
References
[1] Long M, Rack H J. Titanium alloys in total joint replacementЎӘA materials science perspective [J]. Biomaterials, 1998, 19(18): 1621-1639.
[2] WANG Xiao-hua, LI Jin-shan, HU Rui, KOU Hong-chao. Mechanical properties and pore structure deformation behaviour of biomedical porous titanium [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(5): 1543-1550.
[3] XU Li-juan, XIAO Shu-long, TIAN Jing, CHEN Yu-yong. Microstructure, mechanical properties and dry wear resistance of ҰВ-type TiЁC15MoЁCxNb alloys for biomedical applications [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(3): 692-698.
[4] Nagano M, Nakamura T, Kokubo T, Tanahashi M, Ogawa M. Differences of bone bonding ability and degradation behaviour in vivo between amorphous calcium phosphate and highly crystalline hydroxyapatite coating [J]. Biomaterials, 1996, 17(18): 1771-1777.
[5] Liu X Y, Chu P K, Ding C X. Surface modification of titanium, titanium alloys, and related materials for biomedical applications [J]. Materials Science and Engineering R, 2004, 47(3): 49-121.
[6] Sporer S M, Paprosky W G. Biologic fixation and bone ingrowth [J]. Orthopedic Clinics of North America, 2005, 36(1): 105-111.
[7] XIAO Dong-ming YANG Yong-qiang, SU Xu-bin, WANG Di, LUO Zi-yi. Topology optimization of microstructure and selective laser melting fabrication for metallic biomaterial scaffolds [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(10): 2554-2561.
[8] Clem W C, Chowdhury S, Catledge S A, Weimerd J J, Shaikh F M, Hennessy K M, Konovalova V V, Hilla M R, Waterfeldg A, Bellisa S, Vohraa Y K. Mesenchymal stem cell interaction with ultra-smooth nanostructured diamond for wear-resistant orthopaedic implants [J]. Biomaterials, 2008, 29(24): 3461-3468.
[9] Brammer K S, Oh S, Cobb C J, Bjursten L M, Heyde H, Jin S. Improved bone-forming functionality on diameter-controlled TiO2 nanotube surface [J]. Acta Biomaterialia, 2009, 5(8): 3215-3223.
[10] de Oliveira P T, Nanci A. Nanotexturing of titanium-based surfaces upregulates expression of bone sialoprotein and osteopontin by cultured osteogenic cells [J]. Biomaterials, 2004, 25(3): 403-413.
[11] Kunzler T P, Drobek T, Schuler M, Spencer N D. Systematic study of osteoblast and fibroblast response to roughness by means of surface-morphology gradients[J]. Biomaterials, 2007, 28(13): 2175-2182.
[12] Abron A, Hopfensperger M, Thompson J, Cooper L F. Evaluation of a predictive model for implant surface topography effects on early osseointegration in the rat tibia model [J]. The Journal of Prosthetic Dentistry, 2001, 85(1): 40-46.
[13] WANG Xiao-hua, LI Jin-shan, HU Rui, KOU Hong-chao, ZHOU Lian. Mechanical properties of porous titanium with different distributions of pore size [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(8): 2317-2322.
[14] Zhao G, Schwartz Z, Wieland M, Rupp F, Geis-Gerstorfer J, Cochran D L, Boyan B D. High surface energy enhances cell response to titanium substrate microstructure [J]. Journal of Biomedical Materials Research Part A, 2005, 74(1): 49-58.
[15] Cavalcanti-Adam E A, Volberg T, Micoulet A, Kessler H, Geiger B, Spatz J P. Cell spreading and focal adhesion dynamics are regulated by spacing of integrin ligands [J]. Biophysical Journal, 2007, 92(8): 2964-2974.
[16] Webster T J, Ejiofor J U. Increased osteoblast adhesion on nanophase metals: Ti, Ti6Al4V, and CoCrMo [J]. Biomaterials, 2004, 25(19): 4731-4739.
[17] Ward B C, Webster T J. The effect of nanotopography on calcium and phosphorus deposition on metallic materials in vitro [J]. Biomaterials, 2006, 27(16): 3064-3074.
[18] Zhu Bang-shang, Lu Qing-hua, Yin Jie, Hu Jun, Wang Zong-guang. Alignment of osteoblast-like cells and cell-produced collagen matrix induced by nanogrooves [J]. Tissue Engineering, 2005, 11(5-6): 825-834.
[19] Geckili O, Bilhan H, Bilgin T. A 24-week prospective study comparing the stability of titanium dioxide grit-blasted dental implants with and without fluoride treatment [J]. The International Journal of Oral and Maxillofacial Implants, 2009, 24(4): 684-688.
[20] Lim J Y, Dreiss A D, Zhou Z, Hansen J C, Siedlecki C A, Hengstebeck R W, Cheng J, Winograd N, Donahue H J. The regulation of integrin-mediated osteoblast focal adhesion and focal adhesion kinase expression by nanoscale topography [J]. Biomaterials, 2007, 28(10): 1787-1797.
[21] CUI Fu-zhai, FENG Qing-ling. Biomaterials science [M]. Beijing: Tsinghua University Press, 2004. (in Chinese)
[22] Gao Ling, Feng Bo, Wang Jian-xin, Lu Xiong, Liu Da-li, Qu Shu-xin, Weng Jie. Micro/nanostructural porous surface on titanium and bioactivity [J]. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 2009, 89(2): 335-341.
[23] Oh S H, Finones R R, Daraio C, Chen L H, Jin S. Growth of nano-scale hydroxyapatite using chemically treated titanium oxide nanotubes [J]. Biomaterials, 2005, 26(24): 4938-4943.
[24] Dalby M J, Di Silvio L, Harper E J, Bonfield W. In vitro adhesion and biocompatability of osteoblast-like cells to poly (methylmethacrylate) and poly (ethylmethacrylate) bone cements [J]. Journal of Materials Science: Materials in Medicine, 2002, 13(3): 311-314.
[25] Grigoriou V, Shapiro I M, Cavalcanti-Adam E A, Composto R J, Ducheyne P, Adams C S. Apoptosis and survival of osteoblast-like cells are regulated by surface attachment [J]. Journal of Biological Chemistry, 2005, 280(3): 1733-1739.
[26] Stevens M M, George J H. Exploring and engineering the cell surface interface [J]. Science, 2005, 310(5751): 1135-1138.
[27] Curtis A S G, Gadegaard N, Dalby M J, Riehle M O, Wilkinson C D W, Aitchison G. Cells react to nanoscale order and symmetry in their surroundings [J]. IEEE Transactions on Nanobioscience, 2004, 3(1): 61-65.
оСәПҪрЦІИлМеұнГжОўДЙҪб№№№№ҪЁј°ЖдЙъОп»оРФ
ХЕ ИпЈ¬Нт мЪЈ¬°¬ РЛЈ¬Нх лшЈ¬ГЕ І©
ЙҪ¶«ҙуС§ »ъРө№ӨіМС§Фә ёЯР§Ҫаҫ»»ъРөЦЖФмҪМУэІҝЦШөгКөСйКТЈ¬јГДП 250061
ХӘ ТӘЈәТҪУГЦІИлОпөДұнГжёДРФКЗёДЙЖПё°ыРРОӘЎўМбёЯЦІИлМе№ЗХыәПөДТ»ЦЦУРР§·Ҫ·ЁЎЈНЁ№эјӨ№вјУ№Өј°¶аЦШЛбКҙФЪоСәПҪрұнГжЦЖұёіцОўДЙёҙәПҪб№№ЎЈІЙУГSEM¶ФоСәПұнГжРОГІҪшРРұнХчЎЈНЁ№э¶ФІ»Н¬ұнГжРОГІөДЦІИлМеҪшРРМеНвПё°ыЕаСшЈ¬№ЫІмфЗ»щБЧ»ТКҜөДРОіЙЎўПё°ыРОМ¬ј°Пё°ыХіёҪЈ¬·ЦОцІ»Н¬ұнГжРОГІөДЙъОп»оРФЎЈҪб№ыұнГчЈәОўГЧҪб№№ұнГжДЬ№»ҙЩҪшіЙ№ЗПё°ыХіёҪУлЖМХ№Ј»УлОўГЧҪб№№ұнГжПаұИЈ¬ОўДЙёҙәПҪб№№ұнГжёьУРАыУЪПё°ыХіёҪУлЙмХ№ЎЈОўДЙёҙәПҪб№№ДЬ№»МбёЯоСәПҪрЦІИлМеөДЙъОп»оРФј°№ЗХыәПДЬБҰЎЈ
№ШјьҙКЈәоСәПҪрЈ»ОўДЙҪб№№Ј»јӨ№вјУ№ӨЈ»¶аЦШЛбКҙЈ»ЙъОп»оРФ
(Edited by Mu-lan QIN)
Foundation item: Projects (51175306, 51575320) supported by the National Natural Science Foundation of China; Project (TS20130922) supported by the Taishan Scholar Foundation, China; Project (2014JC020) supported by the Fundamental Research Funds for the Central Universities of China
Corresponding author: Yi WAN; Tel: +86-531-88392539; E-mail: wanyi@sdu.edu.cn
DOI: 10.1016/S1003-6326(16)64217-6