Article ID: 1003-6326(2005)06-1407-07
Reduction mechanism of stainless steelmaking dust and carbon pellets
PENG Bing(�� ��), SONG Hai-chen(��),
CHAI Li-yuan(����Ԫ), WANG Ja(�� ��), WANG Yun-yan(������),
MIN Xiao-bo(��С��), HE De-wen(�ε���)
(School of Metallurgical Science and Engineering, Central South University,
Changsha 410083, China)
Abstract: The reduction mechanism of stainless steelmaking dust and carbon pellets was investigated. The metal oxides present in the dust were reduced by carbon with a new direct reduction technology. The direct reduction parameters were determined by measuring the rates of dust melting and reduction. The results show that the rate of reduction is faster than that of the melting. Both melting and reduction processes are accelerated by the direct transfer of heat from the smelting slag. The recovery of metals is improved while the pellets were added to argon oxygen decarburization(AOD) or vacuum oxygen decarburization(VOD) vessels in the late period of the first smelting stage. More carbon travels to the slag instead of to the steel because the diffusion coefficient of carbon, impacted by the viscosity of slag and surface tension between slag and melted steel, is larger in the slag than in the steel. The viscosity of slag is about 2.54Pa��s and the surface tension between slag and steel is about 490mN/m.
Key words: stainless steelmaking dust; direct recycling; diffusion; reduction CLC
number: TF741.5 Document code: A
1 INTRODUCTION
The smelting temperature generally reaches 1600�� or higher in stainless steelmaking operation. Under this condition, approximately 1%-2%(mass fraction) of the scrap charged into the furnace is converted to dust[1] as by-product of steelmaking process. It has been assigned as a hazardous waste and banned from landfills by various government regulatory agencies because it contains a lot of heavy metals such as zinc, lead, cadmium, chromium and nickel, which will be leached into groundwater or surface water when stockpiled and result in the pollution of environment[2]. Many dust treatment methods were developed in the past[3-5], while most of them only dealt with the recycling of zinc and lead from the dust[6, 7]. Solidification of the dust lost the valuable metal resources in it[8] and plasma treatment of the dust consumes too much electric power[9]. A new technology for directly recycling the stainless steelmaking dust was developed[10-13], which was a self-reduction process.
The improved method is aimed at recovering the metallic elements from the dust directly to the steel bath. In this process, the dust is mixed with carbon as reducing agent and then formed into pellets that are subsequently fed to the steelmaking furnace AOD or VOD. Under the thermal conditions that prevail in the furnace, the metal oxides in the dust are reduced by carbon. But the recovery of chromium from the dust is not so perfect as that of other metals.
This study focused on the mechanism of the direct reduction process by investigating the morphology and diffusion of carbonide chromium reduced in the new technology, the factors on reduction degree, the allocation of pellets in slag and steel, and the schedule for adding pellets. More-over, the viscosity and surface tension of slag were also calculated. There were only a few reduction mechanisms on the direct recycling of stainless steelmaking dust even though some of them have been undertaken on the iron composites and chromite fines[14, 15]. However, there were few studies about the mechanism of the direct reduction in electric furnace, AOD or VOD, especially in practice.
2 EXPERIMENTAL
2.1 Materials and apparatus
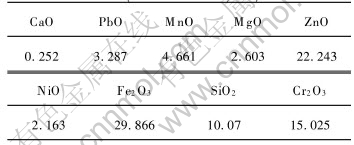
The stainless steelmaking dust used in the experiments was taken from a house-bag. The range of particle sizes in the dust samples was very wide from less than 0.074mm to larger than 0.48mm and the diameters of it are given in Table 1. Elemental analysis of the dust samples was performed using X-ray Fluorescence(XRF) and its composition is listed in Table 2. Most of the elements within the dust are oxidized because it was formed in air at high temperature. X-ray diffraction(XRD) analysis indicated that the main phases present in the dust were Fe2O3, Cr2O3, CaO, NiO and ZnO and the phase compositions are given in Table 3. The carbon used as the reducing agent was finer than 0.034mm and the CaO used as the binder in the pellets were finer than 0.45mm.
Table 1 Distribution of dust particle sizes

Table 2 Element composition of stainless steelmaking dust(mass fraction, %)
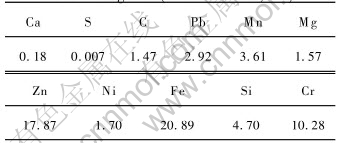
Table 3 Main phases of stainless steelmaking dust(mass fraction, %)
The pellets were made from the mixtures of stainless steelmaking dust, carbon and CaO. They were agglomerated by hand using a semi-ball shaped ceramic bowl. Water droplets were added to the 4-8g dust/carbon/CaO (some of them with additive Fe powder) mixture gradually. The ball was rolled for several minutes after the pellet was completely formed and then dried at 120�� for 3h. The diameter of pellets varied from 12 to 20mm. Four types of carbon content of 10%, 15%, 20% and 25% (mass fraction) were chosen. In each case, the binder content maintained as 5%.
The schematic diagram of experimental apparatus is shown in Fig.1. In this system, an electric furnace was used to heat the pellets and an electrical microbalance to measure and record the mass changes of pellets at an interval of 30s. The mass of pellets in the thermal process were printed out automatically using the printer that was linked to the microbalance.
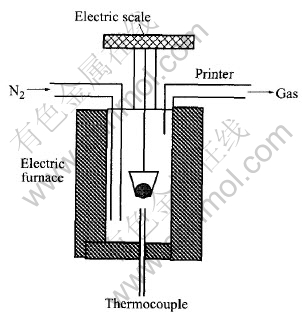
Fig.1 Schematic diagram of experimental apparatus
2.2 Experimental procedure
The furnace was heated to a certain temperature first and then nitrogen was introduced to the furnace at 1.5L/min in order to prevent the oxidation of carbon in the pellets by oxygen in the air during experiments. The pellet was put into the alumina crucible and then sent to the furnace. The microbalance recorded the mass changes of the pellet and the reduction degrees of pellet were calculated according to the ratio of the mass loss and original mass of pellets. The rates of smelting and reduction were detected by heating two exactly same pellets in the same way.
3 RESULTS
3.1 Reduction degree
It was found that the reduction degree (oxygen removed / maximal oxygen removed) of pellets was related to several factors such as temperature, reduction atmosphere and additive iron content. Fig.2 shows the profile of reduction degree of the pellets, which contain 15% (mass fraction) carbon and bear 19mm of the measured diameter, reduced at different temperatures of 1200, 1300, 1400 and 1500��. The figure clearly shows that the reduction degree of pellets increases with temperature, but the effect of reduction temperature on the reduction degree is not obvious at relatively low temperature. Fig.3 shows the effect of atmosphere on reduction degree. Application of a nitrogen atmosphere in the thermal process is very important for the pellet reduction. Fig.4 shows the relationship between reduction degree and amount of additive iron. It can be seen from Fig.4 that the reduction
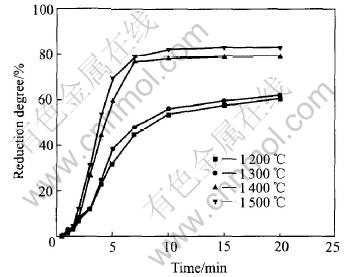
Fig.2 Profiles of reduction degree atdifferent temperatures
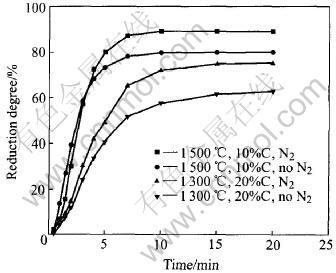
Fig.3 Reduction degrees in different atmospheres
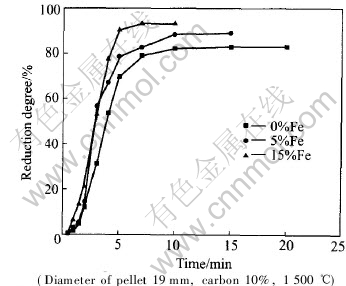
Fig.4 Relationships between reduction degree and time for pellets with different Fe contents
degree increases with more additive iron powders because they diminish the activation energy during metals being reduced from oxides. However it is not suitable to add much more iron powders because carbonization will happen and more reducing agent will be consumed, which will be discussed in the following part.
3.2 Reduction and melting rates of pellets
In practical direct recycling operation, the process of reduction can be classed into two stages after the pellets are added to AOD or VOD furnace at high temperature over 1600��. The metal oxides are reduced directly by carbon due to the contact between the particles in the first stage. In the second stage, the reduction depends on the Boudouard Reaction and the metal oxides are reduced by carbon monoxide produced in the first stage. However, the experimental temperature in this study was lower than that of practical operation so that the rates of reduction and melting were much slow. According to the observation in the run, the effect of temperature on the rate of melting is much great. Fig.5 shows the thermal process of stainless steelmaking dust pellets by using TGA and DTA. A heating rate of 15��/min for TGA and DTA was chosen up to 1300�� under argon atmosphere, as it was sufficiently slow to allow for separation between the reactions that take place during heating up. Fig.6 gives the profile of the effect of diameter on reduction degree at 25% (mass fraction) of carbon content. Fig.7 gives the experimental result on the smelting rate of pellets at 1500��.
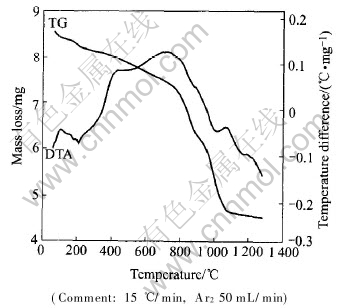
Fig.5 TGA��DTA curves of pellets
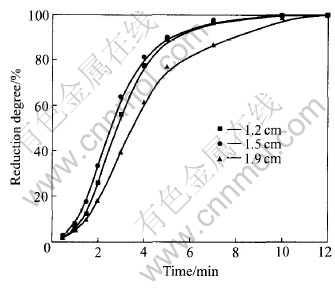
Fig.6 Reduction degrees of pellets at 1500��
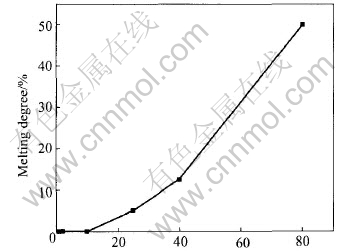
Fig.7 Melting rate of pellet at 1500��
From Fig.5, the time for heating pellets was attained by the heating ratio. Except for the evaporation of water from pellets at initial heating, it took about 13min for the mass loss of pellets in reduction process and this was similar to the reduction time in Fig.6. Because the experimental temperature in Fig.6 is higher than that in TGA-DTA test, the former reduction time was much short. It is very clear by comparison of Figs.5, 6 and 7 that the reduction rate is much quicker than that of the molten. Therefore, metal oxides in pellets will be reduced first, and then the reduced metals travel from slag to the liquid steel in the direct recycling operation.
4 DISCUSSION
4.1 Allocation of pellets in steel bath
The specific volume of slag ��t can be gotten in the way as follows[16, 17]:
��t=��1400��+0.07(1400-t/1400)(1)
��-11400��=0.45MSiO2+0.286MCaO+
0.204MFeO+0.35MFe2O3+0.237MMnO+
0.367MMgO+0.48MP2O5+0.402MAl2O3
=0.45��0.16+0.286��0.34+0.204��0.22+
0.35��0.10+0.237��0.05+0.367��0.07+
0.48��0.02+0.402��0.02
=0.304(2)
so, ��1400��=3.289g/cm3.
where �� stands for the density of steel or slag.
According to the Eqn.(1), the density of slag was obtained at 1550�� as
��1550��=��1400��+0.07(1400-t/1400
=3.289+0.07��(1400-1550)/1400
=3.282(g/cm3)
The density of slag may be different during the smelting process because the composition of slag is not always constant, but the differences are not so great. Although the density of the pellets varies with different carbon content, it is very similar to each other due to the small difference. Taking a certain carbon content pellet as an example, its density could be gotten by Eqn.(3).
��=mp/Vp=3.663g/cm3(3)
On the other hand, the density of liquid steel is about 7.78g/cm3.
After comparing the densities of slag, liquid steel and pellets, the allocation of pellets in the smelting bath can be determined in the place between slag and liquid steel as shown in Fig.8. It is approximately 1/10 of the pellets present in the liquid steel and the others in the slag. The diffusion of metals reduced from pellets mostly occurred in slag and the slag property also played an important role in the diffusion process.
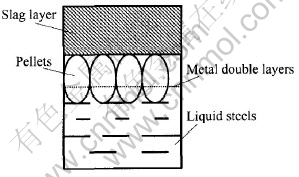
Fig.8 Allocation of pellets in steelmaking bath
4.2 Schedule for pellet adding
In general, there are three stages in stainless steels refinement by AOD furnace. The mass ratio of argon to oxygen is 1��2 in the first stage and 2��1 in the second stage for 15min, then additions and reducing agents are added in the last stage. The chance for pellets adding depends on the property of dust and slag. If pellets are added in the first stage, the reduction degree of the metal oxides is not high enough because the atmosphere in the furnace is oxidizability. If pellets are added between the end of the first stage and the beginning of the second stage, there are 15min for full reduction and element transfer. Furthermore, the volatilization of CO, Zn and Pb, which created in the process of reduction, accelerates pellets decomposition and melting in favor of recovery of reduced chromium and iron.
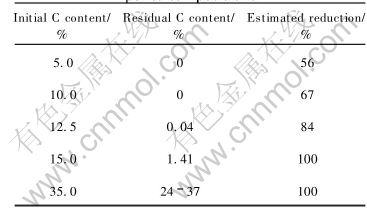
The component of the slag will not be changed so much in the direct recycling operation because the amount of added pellets is small. The quantity of added silicon and aluminum as reductant for the reduction of Cr2O3 is similar to no-joined pellets. The relationship of schedule and metal recovery ratio is given in Table 4. Based on Table 4, the highest recovery of chromium and other metals can be achieved when pellets with 15% carbon are added into electric furnace in the end of the first smelting stage. Some other relationships between the estimated reduction and residual carbon and initial carbon from TGA test are also given in Table 5.
Table 4 Relationship between joined time and recycling ratio
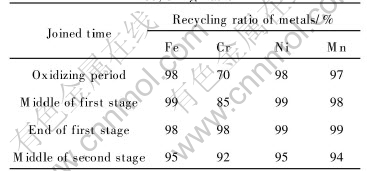
Table 5 TGA test results using different pellet compositions
4.3 Carbon diffusion
Carbon diffusion is related to the schedule of pellets adding. If some carbon doesn��t attend the reduction reaction, they probably move into the liquid steel through metal double layers. If the metals and carbon presents in the form of CrxCy and FexCy in the steels, it would make stainless steel products corroded in the way of intercrystalline and severely impair the quality of stainless steels[18]. While carbon moves to the slag, it scarcely damages the quality of stainless steels and it is also beneficial to the reduction of metal oxides in slag. In order to prevent carbon transferring into steel, two ways can be taken in the direct recycling practice. One is to reduce the carbn content in pellets. The other is to change the schedule for pellets adding to the furnace.
The main reactions of carbon and metals are as follows:

The diffusion coefficient of carbon in liquid steel is D1=6��10-5-7��10-5cm2/s, and in slag D2>2.4cm2/s at 1550��. Some carbon without involving in the reduction will form carbonides through Eqns.(4), (5) and (6). On the other hand, the carbonides will be oxidized again through Eqns.(7) and (8). It is because most pellets lie in the slag that the carbon entered slag will react with metal oxides in the slag. In order to reduce the time period of operation and not affect the quality of steels, the amount of the pellets added to the furnace should be 5% (mass fraction) in AOD furnace. Moreover, the diffusion rate of carbon in slag is much quicker than in steel. So the content of carbon in steels will not rise after pellets are put into the furnace.
Table 6 is the result of experiment with an induction furnace. The residual carbon in ingot is checked and the ratio of recycled metals in different direct recycling parameters.
4.4 Surface tension and viscosity of slag
The surface tension between slag and liquid steel affects the melting of pellets and the element transfer. It was estimated by some researchers with about 490mN/m at 1550��[17] and decreased at higher temperature.
Viscosity of slag can be calculated by using Eqns.(9) and (10).
log��=7.368+2.068MSiO2+1.985y(9)
y=8-{2(MFeO+MCaO+MNa2O+MK2O+
2MCaF2+2MSiO2+3MAl2O3)}/(MSiO2+2MAl2O3)
=8-{2��[(0.22+0.1)+0.34+0.02+
2��0.16+3��0.02]}/(0.16+2��0.02)(10)
so, log��=2.54Pa��s.
Under this viscosity, the balance content of Cr2O3 in slag is approximately 3% at 1550�� and will decrease at higher temperature. According to this, Cr2O3 will be easily reduced and move into the liquid steels.
Table 6 Summary of induction furnace tests

5 CONCLUSIONS
1) High temperature, inert atmosphere and addictive iron are favored for high recovery of metals.
2) After the stainless steelmaking and carbon pellet is added to the furnace, it will lie in the interface of slag and steel. 1/10 of it will be in the liquid steel and the others in slag. The reduced metals transfer from slag to steel. The rate of reduction is much quicker than that of melting and the slag will enhance both rates.
3) The recovery of metals will be increased when the pellets are added in the end of the first reduction stage of smelting practice, especially Cr reduction degree could reach as high as 98%.
4) The carbon remained in the pellets will move into slag. It depends on the viscosity of slag and surface tension between slag and steel. The viscosity of slag is 2.54Pa��s and the surface tension is 490mN/m.
REFERENCES
[1]Zunkel D. What to do with your EAF dust [J]. Steel Times International, 1996, 20(4): 46-50.
[2]US Environment Protection Agency. Land Disposal Restriction for Electric Arc Furnace Dust [S]. Federal Register, 5641164, 1991.
[3]Mastsuoka S. New technology for treating electric arc furnace dust [J]. Iron and Steel Engineer, 1991(2): 37-40.
[4]Jenson J T. Reduction of EAF dust emissions by injecting it into the furnace [J]. MPT international, 1997(3): 58-62.
[5]Geldenhuis J M A. Recovery of valuables from flue dust fines [J]. Minerals Engineer, 2002(15): 95-98.
[6]Labee C J. Update on electric arc furnace dust treatment [J]. Iron and Steel Engineer, 1992(5): 48-49.
[7]Floyd J. EAF dust treatment in an ausmelt furnace system [J]. Seaisi Quarterly, 1993(4): 60-65.
[8]PENG Bing, ZHANG Chuan-fu, PENG Ji. Treatment of steelmaking dust in electric are furnace by vitrification [J]. Journal of Central South University of Technology, 2000, 31(2): 124-126.(in Chinese)
[9]LI Ding-yi. Application of plasma technology to treat EAF dust [J]. Iron and Steel, 1995, 30(3): 72-74.
[10]PENG Bing, Lobel J, Kozinski J A, et al. Non-isothermal reduction kinetics of EAF dust -based pellets [J]. CIM Bulletion, 2001, 94(1049): 64-70.
[11]Lobel J, PENG Bing, Kozinski J A, et al. Pilot-scale direct recycling of flue dust generated in electric arc furnace stainless steelmaking [J]. Iron and Steelmaker, 2000, 27(1): 41-45.
[12]ZHANG Chuan-fu, PENG Bing, PENG Ji, et al. The direct recycling of electric arc furnace stainless steelmaking dust [A]. The 6th International Symposium on East Asian Resources Recycling Technology [C]. Gyeongiu, Korea: The Korean Institute of Resources Recycling, 2001. 404-408.
[13]PENG Bing, PENG Ji, Kozinski J A, et al. Thermodynamic calculation on the smelting slag of direct recycling of electric arc furnace stainless steelmaking dust [J]. Journal of Central South University of Technology, 2003, 10(1): 20-26.
[14]Tsomondo B M C, Simbi D J, Navara E. Isothermal reduction of chromite fines by carbon saturated chromium melt [J]. Ironmaking and Steelmaking, 1997, 24(5): 386-391.
[15]Goswami M C, Prakash S, Sarkar S B. Kinetics of smelting reduction of fluxed composite iron and pellets [J]. Steel Research, 1999, 70(2): 41-46.
[16]CHNEG Jia-xiang. Handbook of Icon and Data in Steelmaking [M]. Beijing: Metallurgical Industry Press ,1984. 183, 215.(in Chinese)
[17]ZHANG Jian. Theory and Practice of Secondary Refining[M]. Beijing: Metallurgical Industry Press, 1999. 77-78.(in Chinese)
[18]XIAO Ji-mei. Metal Problem of Stainless Steel [M]. Beijing: Metallurgical Industry Press, 1983. 38.(in Chinese)
(Edited by LONG Huai-zhong)
Foundation item: Project(50274073) supported by the National Natural Science Foundation of China
Received date: 2005-02-24; Accepted date: 2005-06-30
Correspondence: PENG Bing, Professor, PhD; Tel: +86-731-8830875; E-mail: pengyu@public.cs.hn.cn