J. Cent. South Univ. Technol. (2009) 16: 0429-0433
DOI: 10.1007/s11771-009-0072-9

Effect of baking processes on properties of TiB2/C composite cathode material
L? Xiao-jun(������), LI Jie(�� ��), LAI Yan-qing(������), FANG Zhao(�� ��)
(School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China)
Abstract: Pitch and TiB2/C green composite cathode material were respectively analyzed with simultaneous DSC-TGA, and effects of three baking processes of TiB2/C composite cathode material, i.e. K25, K5 and M5, on properties of TiB2/C composite cathode material were investigated. The results show that thermogravimetric behavior of pitch and TiB2/C green composite cathode is similar, and appears the largest mass loss rate in the temperature range from 200 to 600 ��. The bulk density variation of sample K5 before and after baking is the largest (11.9%), that of sample K25 is the second, and that of sample M5 is the smallest (6.7%). The crushing strength of sample M5 is the biggest (51.2 MPa), that of sample K25 is the next, and that of sample K5 is the smallest (32.8 MPa). But, the orders of the electrical resistivity and electrolysis expansion of samples are just opposite with the order of crushing strength. The heating rate has a great impact on the microstructure of sample. The faster the heating rate is, the bigger the pore size and porosity of sample are. Compared with the heating rate between 200 and 600 �� of samples K25 and K5, that of sample M5 is slower and suitable for baking process of TiB2/C composite cathode material.
Key words: aluminum electrolysis; TiB2/C composite cathode material; baking process
1 Introduction
Many shortcomings of the present aluminum electrolysis process, including high energy consumption and release of greenhouse gas [1-2], have become main obstacles to sustainable development of aluminum industries. Carbon material, used as cathode of aluminum reduction cell, is non-wetting by liquid aluminum, necessitating a substantial pool of metal [3-4]. Although the aluminum pool to a certain extent protects the cathode bottom, the electric magnetic force creates movements and standing waves in liquid aluminum, and the wave-movements aggravate with reducing the height of aluminum. To avoid short circle between the metal and anode, the anode-cathode distance (ACD) has to be kept at 4-5 cm, and cell voltage increases. Thus, a mass of energy was consumed by ohm voltage drop between ACD, which led to the high energy consumption [5]. However, the inert wettable cathode, as a substitute of carbon cathode, has good wettability with liquid aluminium, and can form the stable aluminum liquid film on the surface of cathode, avoiding the interference of electric-magnetic force. So it may realize the vast saving energy through reducing the ACD, and save electrical energy of 1 800 kW?h per ton aluminum with reducing 1 cm of ACD [6].
TiB2/C composite cathode material has been considered the most promising inert wettable cathode
[7]. TiB2/C composite cathode is composed of TiB2 powder, carbon and organic binder. During baking, organic binder will generate small molecule volatilization and a series of polymerization and fasculation reaction. The change of mass and energy occur in the process of reaction, and the reaction rate has a great influence on cathode material performances [8]. LI et al [9] studied the influence of different solidification and carbonization programs on properties of TiB2/C coating, and the TiB2/C coating was solidified and carbonized with the shift and even heating process respectively. The result shows that the electrical resistivity and tensile strength of coating with shift heating process are better than those with even heating process. However, the influence of baking processes on properties of TiB2/C composite cathode material is rarely reported. In this work, three different baking processes based on differential thermal analysis were established, and the influence of baking processes on properties and microstructure of TiB2/C composite cathode material was investigated.
2 Experimental
2.1 Cathode composition and experimental materials
Components (mass fraction) of TiB2/C composite cathode are as follows: TiB2 powder (average grain size of 8 ��m) 60%, carbonic additive (average grain size of 50 ��m) 20%, and pitch 20%.
Components (mass fraction) of bath are as follows: K3AlF6 (analytically pure) 21.7%, Na3AlF6 (analytically pure) 50.5%, Al2O3 (analytically pure) 3.8% and AlF3 (commercially pure) 24.0%. The cryolite ratio is 1.6, and m(K3AlF6)/[m(K3AlF6)+m(Na3AlF6)]=0.3.
2.2 Preparation of TiB2/C composite cathode
TiB2 powder, carbonic additive and pitch were weighed according to certain mass ratio, and mixed in the three-dimension kinetic mixer adequately. The mixture was pressed into the specimen with size of d20 mm��40 mm on the omnipotent liquid crushing machine. Cathode samples were obtained under three different baking processes.
2.3 Properties test and analysis methods
The test details of the electrical resistivity and electrolysis expansion of TiB2/C composites cathode were the same as those in Refs.[10-11]. The test conditions of electrolysis expansion were as follows: current density 0.8 A/cm2, electrolysis time 1.5 h, superheat temperature 50 ��, and electrolysis temperature 873 ��. The whole test processes were performed under protective atmosphere with high pure argon.
The bulk density was measured in accordance with YB/T119��1997. The crushing strength of TiB2/C composite materials was tested in accordance with GB1431��85.
The microscopic analysis of TiB2/C composite cathode was implemented by using high/low vacuum scanning electron microscope (JSM-6360LV, Japan). SDT simultaneous DSC-TGA (SDTQ600, USA) was used for DTA-TGA analysis of TiB2/C green composite cathode and pitch binder. The test was performed under argon gaseous atmosphere. The test conditions were as follows: the heating rate 5 K/min, the airflow velocity 100 mL/min, the temperature range from room temperature to 1 000 ��.
3 Results and discussion
3.1 Selection of baking processes
Fig.1 shows the thermogravimetric analysis (TGA) curves of pitch binder and TiB2/C green composite cathode. No matter pure pitch or green composite cathode, before 200 �� the mass loss is very little, in this course, the moisture adsorbed in pitch is vented. The mass loss of pitch between 200 and 600 �� is the largest, because the liberation of the volatiles, and decomposition of pitch take place, and mosophase, then semicoke are formed gradually through polycondensation.
The mass loss of pitch has little change above

Fig.1 TGA curves of pitch binder (a) and TiB2/C green composite cathode (b)
600 ��. In this stage only volatiles are vented and semicoke is transformed into bonding coke. However, the mass of TiB2/C green composite cathode does not reduce but increases above 600 ��. The reason for this is that titanium diboride is oxidized at high temperature, resulting in an increase of mass for the presence of the moisture and oxygen in argon gas [12-13]. For only pitch binder, the mass loss rate reaches 47.2% from room temperature to 600 �� and 6.8% from 600 to 1 006 ��. However, for TiB2/C green composite cathode, the mass loss rate reaches only 28.0% from room temperature to 600 �� instead of increasing 1.3% from 600 to 1 006 ��. Anyway, since pitch is just the binder of TiB2/C green composite cathode, both pitch and TiB2/C green composite cathode exhibit similar thermogravimetric behavior, i.e. the largest mass loss rate between 200 and 600 ��. So the heating rate in this stage has a great influence on properties of TiB2/C composite cathode, and the heating rate should be as slow as possible in order to improve properties of material. But taking energy consumption, economic and baking time into account, and combining different thermal analytic results, three baking processes are shown in Fig.2.
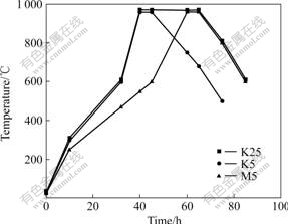
Fig.2 Baking processes of TiB2/C composite cathode
From Fig.2, it can be seen that the total baking time of sample K25 with holding time of 25 h at 960 ��, is the same to that of sample M5, but the heating rate of sample K25 is the same to that of sample K5. The heating rate of sample K5 is quicker than that of sample M5, and their holding time is 5 h at 960 ��.
3.2 Electrical resistivity, crushing strength and bulk density variation before and after baking of TiB2/C composite cathode
Cathode samples with the same components are baked with three baking processes mentioned above, and the experimental results (the mean values of three data measured) are listed in Tables 1 and 2. As can be seen from Table 1, the bulk densities of samples vary with the different baking processes. Density variation [(��f-��g)/��f] of sample K5 is the maximum, and its bulk density is reduced by 11.9%; that of sample K25 is the next; and that of sample M5 is the minimum, whose bulk density is reduced by 6.7%. Compared with samples K25 and M5,
Table 1 Effect of baking processes on bulk density of cathode material
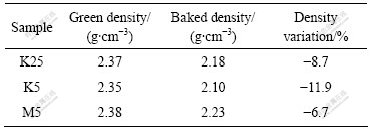
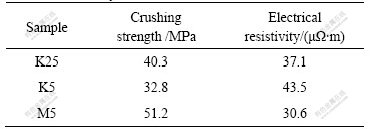
Table 2 Effect of baking processes on crushing strength and electrical resistivity of cathode
sample K5 has a shorter baking time and quicker heating rate, which correspondingly expedites its polycondensation reaction and liberates the volatile rapidly in large quantity, thus resulting in lower carbon yield and larger porosity [8]. On the other side, different stages of coking reaction will take place in the same sample simultaneously at the overquick heating rate, resulting in the asymmetrical shrinkage of inside and outside of the product, which induces inner stress and forms cracks of different sizes in the sample. Thereafter, the bulk density variation of the sample after and before baking is much larger. As seen from Fig.1, especially in the temperature range between 200 and 600 ��, the largest mass loss rate takes place, so the heating rate in this temperature range has a decisive influence on carbonization quality of the sample. For sample K25, though its heating rate is the same as that of sample K5, its heat holding time and baking time are longer than those of other samples. As the heating holding time prolongs, the chemical process and shrinkage of inside and outside complete further, so the bulk density variation of sample K25 after and before baking is less than that of sample K5. The heating rate of sample M5 is relatively slow, which facilitates the increase of carbon yield of pitch, and the density variation of the sample before and after baking is smaller.
It can be concluded from Table 2 that the crushing strength of sample M5 is the largest (51.2 MPa), that of sample K25 is the next, and that of sample K5 is the smallest (32.8 MPa). While the electrical resistivity shows the opposite order, sample M5 has the lowest electrical resistivity (30.6 �̦���m), and the electrical resistively of sample K5 (43.5 �̦���m) is higher than that of sample K25. When the compositions of the cathode materials are the same, the bulk density has a great impact on the cathode properties. Sample M5 has the largest bulk density and crushing strength while the lowest electrical resistivity; sample K5 has the smallest bulk density and crushing strength while the highest electrical resistivity.
3.3 Electrolysis expansion of TiB2/C composite cathode
It can be seen from Fig.3 that electrolysis expansions of samples M5, K25 and K5 are 1.3%, 1.8% and 2.1%, respectively, and the K/Na penetration rate of sample K5 is faster in the first 30 min after electrolysis, but those of samples M5 and K5 become lower. Under the same testing conditions, the K/Na penetration rate and electrolysis expansion are related to the porosity and pore size of cathode material and so on [14-15]. The resistance to K/Na penetration will reduce with the increase of porosity and pore size. Sample K5 has the largest porosity and pore size, so its K/Na penetration rate and electrolysis expansion are also the highest, followed by those of sample K25 and then sample M5.
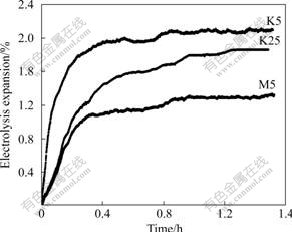
Fig.3 Electrolysis expansion of cathode materials for different baking processes

Fig.4 SEM images of different samples: (a) K5; (b) K25; (c) M5
3.4 Microstructure of TiB2/C composite cathode
Fig.4 shows the SEM images of samples K5, K25 and M5. It can be seen from Fig.4(a) that sample K5 has cracks and large pores. Compared with sample K5, it is obviously that sample K25 in Fig.4(b) has smaller pore and lower porosity. Fig.4(c) shows that sample M5 is the most compact, and its porosity and pore size are the smallest. This further proves that sample��s porosity and pore size increase greatly as reaction is quickened by the overquick heating rate, thereby reducing the bulk density, the crushing strength, the resistance to K/Na penetration while increasing the electrical resistivity of samples.
4 Conclusions
(1) Both pitch and TiB2/C green composite cathode have the similar thermogravimetric characteristics, and appear the largest mass loss rate in the temperature range from 200 to 600 ��.
(2) The baking processes have a great impact on the bulk density variation of samples after and before baking. The bulk density variation for sample K5 before and after baking is the largest, which is reduced by 11.9% compared with that before baking, the next is that of sample K25, and that of sample M5 is the smallest and reduces by 6.7%.
(3) The baking processes have a great influence on the crushing strength, electrical resistivity and resistance to K/Na penetration of samples. The crushing strength of sample M5 is the largest, reaching 51.2 MPa, that of sample K25 is the next, and that of sample K5 is the smallest (32.8 MPa). But the orders of electrical resistivity and electrolysis expansion are just opposite to the order of the crushing strength. The electrical resistivity and electrolysis expansion of sample M5 are the minimum of 30.6 �̦���m and 1.3%, respectively. Those of sample K25 are the next, and those of sample K5 are the maximum of 43.5 �̦���m and 2.1%, respectively. K/Na penetration rate of sample K5 is faster than those of samples M5 and K25 in the first 30 min after electrolysis.
(4) From the microstructures of samples, it is obtained that the faster the heating rate is, the larger the pore size and porosity are. The pore size of sample K5 is the biggest, and some cracks appear. The pore size and porosity of sample K25 are the second. Sample M5 is the most compact, whose pore size and porosity are the smallest. Compared to baking processes of samples K25 and K5, the slower heating rate between 200 �� and 600 �� of sample M5, is more suitable for baking of TiB2/C composite cathode material.
References
[1] SORLIE M, ?YE H A. Cathodes in aluminum electrolysis [M]. 2nd ed. Dusseldorf: Aluminum-Verlag, 1994: 162-163.
[2] LIU Ye-xiang. Research progress of inert anode and wettable cathode for aluminum electrolysis [J]. Light Metals, 2001(5): 26-29. (in Chinese)
[3] LI Qing-yu. Development and industrial application of wettable inert TiB2 cathodic composite coating for aluminum electrolysis [D]. Changsha: Central South University, 2003. (in Chinese)
[4] WELCH B J. Future materials requirements for the high-energy intensity production of aluminum [J]. Journal of Metals, 2001, 53(2): 13-15.
[5] LI Jie, L? Xiao-jun, LAI Yan-qing, LI Qing-yu, LIU Ye-xiang. Research progress in TiB2 wettable cathode for aluminum reduction [J]. Journal of the Minerals Metals & Materials Society, 2008, 60(8): 32-37.
[6] KVANDE H. Energy balance [C]// Fundamentals of Aluminum Production 2004. Trondheim, 2004: 197-216.
[7] LI Jie, L? Xiao-jun, LAI Yan-qing, LI Qing-yu, TIAN Zhong-liang, FANG Zhao. Effect of carbon fibre on properties of TiB2/C composite cathode coating for aluminum electrolysis [J]. Journal of Central South University of Technology, 2008, 15(4): 526-530.
[8] QIAN Zhen-fen. Carbon process [M]. Beijing: Metallurgical Industry Press, 2006: 121-126. (in Chinese)
[9] LI Qing-yu, LAI Yan-qing, LI Jie, LIU Ye-xiang. Solidification and carbonization program of TiB2 cathode coating for aluminum electrolysis [J]. The Chinese Journal of Nonferrous Metals, 2003, 13(1): 251-254. (in Chinese)
[10] LI Jie, FANG Jing, LI Qing-yu, LAI Yan-qing. Effect of TiB2 content on resistance to sodium penetration of TiB2/C cathode composites for aluminum electrolysis [J]. Journal of Central South University of Technology, 2004, 11(4): 400-404.
[11] LI Jie, L? Xiao-jun, LI Qing-yu, LAI Yan-qing, YANG Jian-hong. Electrical resistivity of TiB2/C composite cathode coating for aluminum electrolysis [J]. Journal of Central South University of Technology, 2006, 13(3): 209-213.
[12] XUE Ji-lai, ?YE H A. Sodium and bath penetration into TiB2-carbon cathodes during laboratory aluminum electrolysis [C]// CUTSHALL E R. Light Metals, 1992. Warrendale PA, TMS, 1992: 773-778.
[13] IBRAHIEM M O, FOOSNASS T, ?YE H A. Properties of pitch and furan-based TiB2-C cathodes [C]// DAVID H D. Light Metals 2008. New Orleans: TMS, 2008: 1013-1018.
[14] BRISSON P Y, SOUCY G. Revisiting sodium and bath penetration in the carbon lining of aluminum electrolysis cell [C]// KVANDE H. Light Metals 2005. Warrendale PA: TMS, 2005: 727-732.
[15] ZOLOCHEVSKY A, HOP J G, SERVANT G, FOOSNAES T, ?YE H A. Rapoport-Samoilenko test for cathode carbon materials I: Experimental results and constitutive modelling [J]. Carbon, 2003, 41(3): 497-505.
(Edited by YANG You-ping)
Foundation item: Project (2005CB623703) supported by the Major State Basic Research and Development Program of China; Project (2008AA030502) supported by the National High-Tech Research and Development Program of China
Received date: 2008-09-05; Accepted date: 2008-11-20
Corresponding author: L? Xiao-jun, Doctoral candidate; Tel: +86-731-8876454; E-mail: lxj8886@yahoo.com.cn