
Preparation and elements analysis of porous fluorescent glasses
ZHU Dong-mei (Ц춬÷)1, LUO Fa (ВЮ ·ў)1, ZHOU Wan-cheng (ЦЬНтіЗ)1Ј¬
D. E. DAY2Ј¬C. S. RAY2
1. State Key Laboratory of Solidification Processing, Northwestern Polytechnical University, XiЎЇan 710072, China;
2. Graduate Center for Materials Research, University of Missouri-Rolla, Rolla, Missouri 65409-1170, USA
Received 10 April 2006; accepted 25 April 2006
Abstract: A large variety of porous fluorescent glasses were prepared and the concentration of different elements in these glasses was analyzed. The start porous glasses were soaked in a solution containing soluble salts and then heated at 650ЎЎЎж for 3 h to decompose the salts in the pores into oxides. Fluorescent agents, such as UO3, Eu2O3, were impregnated into the porous glasses to prepare the fluorescent glasses. The results show that soaking is a feasible method to prepare the glass sphere with compositions distinguishable from each other and easy to be located in a complicated background. Six or more components can be impregnated together into one glass sample and the concentration of them can be analyzed accurately. UO3 and Eu2O3 can be impregnated into porous glass to make the glass strongly fluorescent. Higher concentration of Eu2O3 produces stronger fluorescence while higher concentration of UO3 reduces the fluorescence intensity because of the concentration quenching effect.
Key words: fluorescent glasses; porous; fluorescent properties; variable composition
1 Introduction
Impregnation of substances into porous glasses has been widely studied for special applications, such as gradient index glasses to be used as lenses or wave guiding patterns [1], slow drug delivery [2]. Lots of ions have been successfully impregnated into a porous glass [3-8] and these works indicate that it is possible to impregnate multi-components into a porous glass.
The purpose of this work is to find a way to make a large variety of different glasses with compositions easy to control, and find a way to make the glasses easy to be located. In this paper the work on exploration of the possibility to make such glasses were described.
2 Experimental
Porous bulk glass and glass spheres (90-212 ¦Мm in diameter) were made from Vycor# glass (A trade name of Corning Inc) according to the reported method[9,10] . The glasses were heat-treated at 565 Ўж or 3 h for phase-separation, and then leached in 1 mol/L nitric acid to remove the boron-rich phase. The porous glasses prepared in this way are used as the base glass to make impregnated glasses. Solutions used to impregnate ions into the porous glasses were made by dissolving the corresponding salts into distilled water. Impregnation was done by soaking the porous glasses in a solution for 12 h at room temperature. The excess solution was removed by vacuum filtration. The impregnated spheres were dried through blowing with compressed air and then heated at 650 Ўж for 3 h to decompose the salts into oxides.
The concentration of the impregnated oxides in a glass sample was analyzed by energy dispersion spectra(EDS) and by X-ray fluorescence spectra (XRF). The fluorescence intensity was observed using a black box with both long wavelength ultra-violet (UV) light (365 nm) and short wavelength ultra-violet light (254 nm). The relative fluorescence intensity was estimated visually by comparing the brightness of the glasses in the black box with both short and long wav elength UV lights on.
3 Results and discussion
Table 1 lists the calculated and analyzed data of the amount of the oxides in different samples of impregnated glass beads. Since Mg was not detected by the XRF method, it can not be used as a component to identify the impregnated glass. The three concentration levels of the other components can be clearly distinguished by the XRF method except for the middle and low concentration levels of Fe2O3. The measured concentra-
tion of Fe2O3 in the low concentration sample is obviously higher than the calculated one, possibly because the base glass contains some amount of Fe2O3. Thus, all the oxides except MgO can be used as components to identify an impregnated glass. There are four concentration levels for Gd2O3, La2O3, and Nd2O3 considering the 0% concentration level, while there are three concentration levels for Fe2O3.
According to the analyzed amounts of the oxides, the sample with the high concentration level and higher Fe2O3 can be distinguished from the one with the high concentration level. Therefore, by increasing the concentration of one of the oxide and keeping the concentrations of the others un-changed, it is possible to have a higher concentration level for one of the oxides in each glass sample.
Table 2 lists the calculated and analyzed amounts of some other oxides in some more samples. Since we consider the amount of each oxide in the base glass to be one of the concentration levels, we have four concentration levels for each of the oxides in Table 2 except CdO and Y2O3. The amounts of CdO and Y2O3
Table 1 Calculated (Cal.) and analyzed (Ana.) amounts of oxides in impregnated glass beads(mass fraction, %)
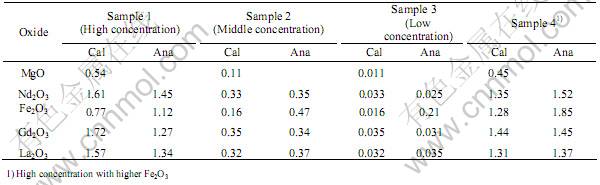
Table 2 Calculated and analyzed amounts of oxides in impregnated glass spheres(mass fraction, %)
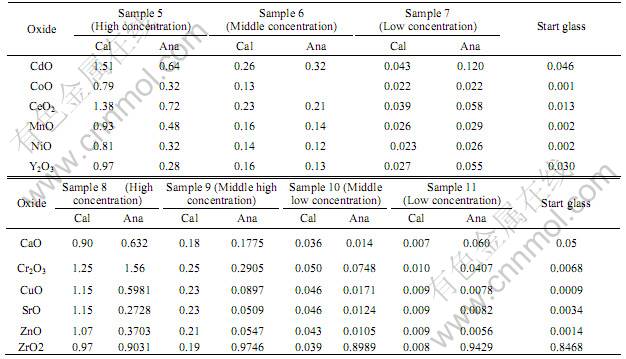
in the base glass are relatively high and are not distinguishable from the amount in the low concentration sample (sample 7). Therefore, we only have three concentration levels for these two oxides.
It can be seen from Table 2 that ZrO2 can not be used as a component to identify impregnated glasses because the amount of ZrO2 in the base glass is high. Since the CaO content in the base glass is higher than the calculated amount of CaO in the middle low concentration sample (sample 10), only three concentration levels exist in an impregnated porous glass.
The samples 8-11 were prepared in an attempt to make five concentration levels of oxides in porous glasses. The data for Cr2O3, CuO, SrO, and ZnO indicates that it is possible to make five distinguishable concentration levels for these oxides.
The relative fluorescence intensity of porous glass beads impregnated with fluorescent agents is shown in Fig.1. When UO3 is used as fluorescent agent, lower concentration in this experimental range gives stronger fluorescence intensity and the samples impregnated in the solution with the UO3 concentration from 0.08% to 0.40% have the highest intensity. Since there is a so-called concentration quenching effect, higher concentration of UO3 reduces the fluorescence intensity. For Eu2O3-impregnated beads, higher concentration gives stronger fluorescence intensity. Under the UV lights in this experiment, the relative fluorescence intensity of Eu2O3-impregnated beads is lower than that of the UO3-impregnated beads.
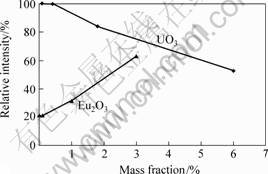
Fig.1 Relative fluorescence intensity of glass beads impregnated from fluorescent solutions with different concentrations (Fluorescence intensity of sample impregnated in solution of 0.40%UO3 is taken as 100%)
4 Conclusions
Porous glasses with compositions distinguishable from each other can be made through impregnating porous glass with a solution containing salts of the desired ions. Six or more components can be impregnated into a single glass sample and the concentration of them can be analyzed by XRF with reasonable accuracy. UO3 and Eu2O3 can be impregnated into porous glass to make the glass strongly fluorescent. Higher concentration of Eu2O3 produces stronger fluorescence while higher concentration of UO3 reduces the fluorescence intensity because of the concentration quenching effect.
References
[1] KOONE N D, ZERDA T W. Diffusion of solvents and cations in porous sol-gel glass[J]. J Sol-Gel Sci Technol, 1997, 8(1-3): 883-887.
[2] SIEMINSKA L, FERGUSON M, ZERDA T W, COUCH E. Diffusion of steroids in porous sol-gel glass: Application in slow drug delivery[J]. J Sol-Gel Sci Technol, 1997, 8(1-3):1105.
[3] KOONE N D, GUO J, ZERDA T W. Diffusion of Er3+ in porous sol-gel glass[J]. J Non-Cryst Solids, 1997, 211(1-2): 150.
[4] RYSIAKIEWICZ-PASEK E, MARCZUK K. Properties of porous glass after impregnation with carbon[J]. J Porous Mater, 1996, 3(1): 17.
[5] RYSIAKIEWICZ-PASEK E, MARCZUK K. Properties of porous glasses after impregnation with caesium and potassium ions[J]. Phys Chem Glasses, 1994, 35(4): 164.
[6] ROIZIN Y O, ALEXEEV-POPOV A, GEVELYUK S A, SAVIN D P, MUGENSKI E, SOKOLSKA I, RYSIAKIEWICZ-PASEK E, MARCZUK K. Luminescence of porous silica glasses with quantum sized silicon domains[J]. Physics and Chemistry of Glasses, 1996, 37(5): 196.
[7] TAGHAVINIA N, LERONDEL G, MAKINO H. Blue- and red-emitting phosphor nanoparticles embedded in a porous matrix[J]. Thin Solid Films, 2006, 503 (1-2): 190-195.
[8] PETUSHKOV A A, SHILOV S M, PUZYK M V. Adsorption and luminescence properties of porous glasses containing europium(III) chloride nanoparticles[J].Glass Physics And Chemistry, 2004, 30 (4): 333-336.
[9] ANTROPOVA T V, Kinetics of corrosion of the alkali borosilicate glasses in acid solutions[J]. Journal of Non-Crystalline Solids, 2004, 345: 270-275.
[10] GUO L P, LI Y X, CHEN Y X. The dynamics of acid treatment in the preparation of porous glass[J]. Journal of Wuhan University Of Technology-Materials Science Edition, 2003, 18 (4): 10-13.
(Edited by LONG Huai-zhong)
Corresponding author: ZHU Dong-mei; Tel: +86-29-88494574; Email: dzhunwpu@nwpu.edu.cn