
Effect of mechanical activation on alkali leaching of chromite ore
ZHANG Yang(张 洋)1, 2, ZHENG Shi-li(郑诗礼)1, DU Hao(杜 浩)1, XU Hong-bin(徐红彬)1, ZHANG Yi(张 懿)1
1. National Engineering Laboratory for Hydrometallurgical Cleaner Production Technology,
Key Laboratory of Green Process and Engineering, Institute of Process Engineering,
Chinese Academy of Sciences, Beijing 100190, China;
2. Graduate University of Chinese Academy of Sciences, Beijing 100049, China
Received 6 July 2009; accepted 13 January 2010
Abstract: Mechanical activation was used to improve the extraction of chromium in molten NaOH. It is observed that the extraction ratio reaches 97% after leaching for 200 min when chromite ore is mechanically activated for 10 min, but only 34% if not activated. Mechanical activation can decrease the particle size, increase the surface area, and enhance the lattice distortion. Further, the mechanisms for mechanical activation were exposed. The results show that the mechanical activation mainly focuses on chromite ore particle size decrease and the lattice distortion. The formation of aggregation weakens the strengthening effect of mechanical activation for releasing high surface energy.
Key words: mechanical activation; chromite ore; leaching
1 Introduction
Chromite ore is widely used in metallurgical and chemical industries for the production of ferrochrome and chromium chemicals, such as sodium dichromate[1]. The production of chromate from chromite ore usually suffers from low extraction ratio and high environmental pollution. Traditionally, the chromite ore is processed by roasting with sodium carbonate at 1 200 ℃ in a rotary kiln with the addition of limestone and dolomite, yielding only 75% of chromium extraction[2]. Further, the hazardous hexavalent chromium wastes released bring about significant environmental pollution problems. Two ameliorations are expected to be able to enhance the utilization ratio of chromite ore and to prevent the pollution of chromate production process[3-4]. One is the improvement of the decomposition ratio of chromite ore via outfield intensifying, and in this regard, mechanical activation is considered to be the most common method. It is well known that the utilization of mechanical activation in mineral processing leads to the decrease of decomposition temperature, the reduction of reagents consumption, and the promotion of the recovery of valuable components[5-11]. The other one is the improvement of the reactivity of reaction media, such as replacing sodium carbonate by alkali metal hydroxide. The mechanism and process of alkali metal hydroxide used in chromite processing were studied in previous work in detail[12-14].
In this work, mechanical activation was used to improve the decomposition of chromite ore in molten NaOH, and the strengthening effect was investigated by study on the changes of particle size, morphology and mineralogy phases.
2 Experimental
2.1 Chromite ore and reagents
The chromite ore used is Vietnamese chromite ore with the chemical composition and the mineralogical analysis presented in Table 1 and Fig.1, respectively. They both indicate that Vietnamese chromite ore consists mainly of magnesiochromite and quartz.
Table 1 Main chemical composition of Vietnamese chromite ore (mass fraction, %)


Fig.1 XRD pattern of Vietnamese chromite ore
The reagents used in the experiments were of analytical grade, and de-ioned water was used in all experiments.
2.2 Mechanical activation of chromite samples
The mechanical activation of chromite ore was performed in a planetary mill PuLuerisette 6 (FRITSH, Germany). 50 g of chromite ores were ground with 400 g of steel balls (10 mm in diameter) for 10 and 45 min, respectively, and revolution speed of the grinding shaft was set to be 550 r/min. After grinding, the activated samples were used for the following experiments.
2.3 Experimental setup
All leaching experiments were carried out in a stainless steel reactor which is illustrated schematically in Fig.2. The temperature of the reactor was controlled by a programmable temperature controller, with a precision of ±2 ℃. The oxygen was provided by an oxygen cylinder and the flow rate was controlled by a flowmeter. Sodium hydroxide and sodium nitrate were mixed with de-ioned water in the reactor. With the increase of temperature, the mixture formed a homogeneous liquid. When the temperature reached the designed value, the chromite ore sample was added to

Fig.2 Schematic diagram of experimental apparatus
the reactor, and continuous oxygen flow was passed through the reactor to facilitate the decomposition of the chromite ore. After reaction, oxygen was first replaced by N2 to protect the products, and dilute alkali solution was added to decrease NaOH concentration to 50%. Under ambient condition, the products were filtered to obtain chromite solution and solid residue for further analysis.
The chromite ore samples and residues were analyzed using inductively coupled plasma-optical emission spectrometer (ICP-OES, PE Optima 5300DV, Perkin Elmer). The mineralogy phases of chromite ore and the residues were examined by X-ray diffractometry (XRD, Phillips PW223/30), and the morphology was observed by scanning electron microscopy (JEOL SEM). The particle size distribution of chromite ore was measured by laser particle size analyzer (Mastersizer 2000).
3 Results and discussion
3.1 Effect of mechanical activation on chromite ore decomposition
The primary effect of mechanical activation mainly focuses on the comminution of mineral particles that resulted in various physicochemical properties. The disintegration by high-energy is accompanied by the increase of the number of particles and by the generation of fresh, previously unexposed surface[9]. Further, the lattice distortion of mineral is also the consequence of mechanical activation. The decrease of particle size and the lattice distortion can both enhance the decomposition of minerals.
To validate the strengthening effect of mechanical activation in chromite ore decomposition, the extractions of non-activated chromite ores and those activated for 10 min and 45 min were studied, respectively, with sodium hydroxide as the regent and the medium. The extraction ratios of chromium are shown in Fig.3. The results indicate that the extraction ratio of chromium is improved obviously when chromite ore is mechanically activated for 10 min; while a little increase of extraction ratio appears when the activation time is prolonged to 45 min. The extraction ratio reaches 97% after leaching for 200 min when chromite ore is mechanically activated for 10 min, and the extraction ratio is only 34% when the ore is not activated. The strengthening effect of mechanical activation is due to the change of physicochemical properties including particle size and lattice structures of the ores.
3.2 Effect of particle size
The decrease of particle size is considered to be an important aspect of mechanical activation. Fig.4 shows
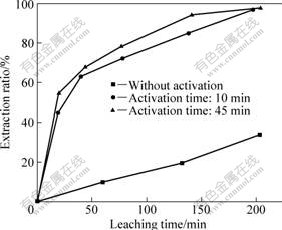
Fig.3 Chromium extraction for non-activated and activated chromite (Temperature 400 ℃and mass ratio of NaOH to chromite 4?1)
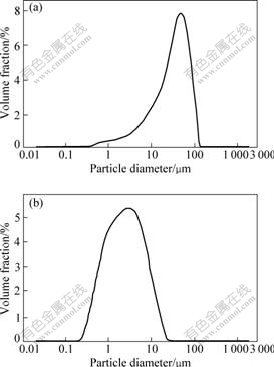
Fig.4 Particle size distribution of non-activated (a) and activated (b) chromite ores
decrease of chromite ore size when it is activated in the planetary mill for 10 min. The results show that the mean particle size decreases from 39.2 μm to 3.7 μm.
The decrease of chromite ore size mainly leads to the decrease in thickness of product layer and the increase of contact areas between liquid media and chromite ore.
3.3 Effect of lattice distortion
Lattice distortion is mainly analyzed by XRD. Lattice distortion can be concluded from the excursion and expansion of the characteristic peaks of the chromite. From Fig.5, the peak (220) of non-activated chromite locates at 30.46?; while the values of 30.62? and 30.64? are observed, when the chromite ore is activated for 10 min and 45 min, respectively. The high angle excursion of peak (220) shows the existence of inner-stress. The lattice distortion ratio of the mechanically activated chromite ores is calculated by Jade Software, and the values of mechanical activation for 10 min and 45 min are 0.006 54% and 0.010 05%, respectively. That is, lattice distortion increases when the activation time is prolonged.

Fig.5 XRD patterns of activated and non-activated chromite ore: (a) Non-activated ore; (b) Activated for 10 min; (c) Activated for 45 min
3.4 Effect of chromite ore surface
After mechanical activation, the chromite ore particle size is significantly smaller than that of non-activated ore, in which high surface energy is stored. However, the unstable surface energy may be easily released, due to the formation of aggregations from tiny particles.
The release of surface energy can be proved by the formation of aggregations from tiny particles. Therefore, the morphology of the activated chromite ore is studied, as shown in Fig.6. Fig.6 illustrates that compared with non-activated ores, the activated chromite ore particles agglomerate together. The formation of aggregation decreases the surface areas of activated chromite ore, leading to the decrease in chromium extraction ratio. Fig.7 shows the extraction ratio of chromium when the activated sample is stored in air for 45 h, and the extraction ratio is less than that stored for 1.5 h for the same leaching time. The decrease of extraction ratio is proposed to be attributed to the release of stored surface energy.

Fig.6 SEM images of non-activated (a) and activated (b) chromite ore
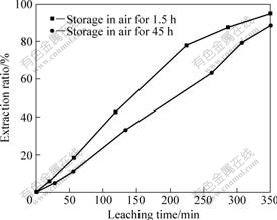
Fig.7 Effect of storage time on chromium extraction
4 Conclusions
1) Mechanical activation significantly accelerates the extraction of chromium in NaOH medium. The extraction ratio reaches 97% after leaching for 200 min when chromite ore is mechanically activated for 10 min, and the extraction ratio is only 34% when it is not activated.
2) The effect of mechanical activation mainly contributes to the decrease in particle size and the increase of lattice distortion. The mean particle size decreases from 39.2 μm to 3.7 μm when chromite ore is activated for 10 min. The lattice distortion ratios of activated chromite ore for 10 min and 45 min are 0.006 54% and 0.010 05%, respectively.
3) The high surface energy can be easily released to the environment by forming aggregations of tiny particles. The formation of aggregation weakens the strengthening effect of mechanical activation.
References
[1] CHENG Si-wei, DING Yi, YANG Chun-rong. Chromate production processes [M]. Beijing: Chemical Industry Press, 1988. (in Chinese)
[2] DING Yi, JI Zhu. Production and applications of chromium compounds [M]. Beijing: Chemical Industry Press, 2003. (in Chinese)
[3] ZHANG Yi, LI Zuo-hu, QI Tao. Green manufacturing process of chromium compounds [J]. Environmental Process, 2005, 24(1): 44-50.
[4] ZHANG Yi, LI Zuo-hu, WANG Zhi-kuan. Green chemistry and new revolution of chromic salts industry [J]. Progress in Chemistry, 1998, 10(2): 172-178.
[5] HEINICKE G. Tribochemistry [M]. Berlin: Akademie-Verlag, 1984.
[6] AVVAKUMOV E G. Mechanical methods of activation of chemical processes [M]. Novosibirsk: Nauka, 1986. (in Russian)
[7] TKACOVA K. Mechanical activation of minerals [M]// FUERSTENAU D W. Developments in Mineral Processing. vol. 11. Amsterdam: Elsevier, 1989.
[8] MOLCHANOV V I, YUSUPOV T S. Physical and chemical properties of finely ground minerals [M]. Moscow: Nedra, 1981. (in Russian)
[9] FERNANDEZ-BERTRAN J F. Mechanochemistry: An overview [J]. Pure Appl Chem, 1999, 71: 581-586.
[10] BAL?? P. Mechanical activation in hydrometallurgy [J]. Int J Miner Process, 2003, 72: 341-354.
[11] KALINKIN A M, KALINKINA E V, MAKAROV V N. Mechanical activation of natural titanite and its influence on the mineral decomposition [J]. Int J Miner Process, 2003, 69: 143-155.
[12] KASHIWASE K, SATO G, NARITA E, OKABE T. The kinetics of oxidation reaction of chromite by NaOH-NaNO3 molten salts [J]. Journal of the Chemical Society of Japan, Chemistry and Industrial Chemistry (Nippon Kagaku Kaishi), 1974(1): 54-59. (in Japanese)
[13] ARSLAN C, ORHAN G. Investigation of chrome(Ⅵ) oxide production from chromite concentrate by alkali fusion [J]. International Journal of Mineral Processing, 1997, 50: 87-96.
[14] SUN Zhi, ZHANG Yi, ZHENG Shi-li, ZHANG Yang. A new method of potassium chromate production from chromite and KOH-KNO3-H2O binary submolten salt system [J]. AIChE Journal, 2009, 55(10): 2646-2656.
(Edited by YANG Bing)
Foundation item: Project(2009AA06XK1485430) supported by the National Hi-tech Research and Development Program of China; Project(2007CB613501) supported by the National Basic Research Program of China
Corresponding author: ZHENG Shi-li; Tel: +86-10-62520910; E-mail: slzheng@home.ipe.ac.cn
DOI: 10.1016/S1003-6326(09)60231-4