J. Cent. South Univ. Technol. (2010) 17: 516-521
DOI: 10.1007/s11771-010-0516-2
Production and characterization of biosurfactant from Bacillus subtilis CCTCC AB93108
LIU Zhi-feng(刘智峰)1, 2, ZENG Guang-ming(曾光明)1, 2, ZHONG Hua(钟华)1, 2,
FU Hai-yan(傅海燕)1, 3, LIU Xiao-lan(刘小兰)1
1. College of Environmental Science and Engineering, Hunan University, Changsha 410082, China;
2. Key Laboratory of Environmental Biology and Pollution Control, Ministry of Education, Hunan University, Changsha 410082, China;
3. Department of Environmental Engineering, Xiamen University of Technology, Xiamen 361024, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2010
Abstract: The production and properties of the biosurfactant synthesized by Bacillus subtilis CCTCC AB93108 were studied. The maximum concentration of the surfactant is 1.64 g/L when the bacteria grow in a medium supplemented with glucose as carbon sources. The isolated biosurfactant is a complex of protein and polysaccharide without lipids. It reduces the surface tension of distilled water to 45.9 mN/m, and its critical micelle concentration (CMC) is 2.96 g/L. It can stabilize emulsions of several aromatic and aliphatic hydrocarbons, such as benzene, xylene, n-pentane, n-nonane, gasoline and diesel oil. It presents high emulsification activity and stability in a wide range of temperature (4-100 ℃) and a long period of duration.
Key words: Bacillus subtilis; biosurfactant; surfactant; emulsification activity; emulsion stability; hydrophobicity
1 Introduction
Biosurfactants are biologically surface-active agents produced by various microorganisms, such as bacteria, yeast and fungi [1-2]. On the basis of the relative molecular mass, biosurfactants are generally classified into two groups: (1) low-molecular mass biosurfactants, such as glycolipids, lipopeptides and phospholipids; and (2) high-molecular mass biosurfactants, such as emuslans, alasan, liposan, polysaccharides and protein complexes [3-5]. In recent years, much attention has been paid to biosurfactants because of their various advantages, such as low toxicity, good biodegradability, good environmental compatibility, high foaming, high selectivity, specific activity at extreme temperatures, pH and salinity, and the ability to be synthesized from renewable feed stocks [6-8]. With environmental compatibility becoming an increasingly important factor in the selection of industrial chemicals, the use of biosurfactants in environmental applications, such as bioremediation and the dispersion of oil spills, increases. In addition, biosurfactants have other uses in the petroleum industry, such as enhanced oil recovery and transportation of crude oil. Other possible application fields are in the food, cosmetic and pharmaceutical industries. In these industries, most biosurfactants are used as emulsifiers [9].
B. subtilis, which can produce many kinds of lipopeptide biosurfactants including surfactins, iturins and fengycins, is one of the well-known bacterium species [10-12]. The acidic precipitation and organic solvent extraction method is usually used to isolate the lipopeptide surfactants produced by B. subtilis from fermentation medium. However, the rest of the precipitate after extraction was not studied in more depth formerly.
Therefore, in this work, our attention was paid to the rest of the acidic precipitate after extraction by chloroform and methanol. Fortunately, a new biosurfactant was found, which might provide a way to find some useful biosurfactants from the materials that did not receive enough attention. The preliminary properties of the biosurfactant including the composition, critical micelle concentration (CMC) and emulsification ability were also studied.
2 Experimental
2.1 Materials and microorganisms
Bovine serum albumin was obtained from Shanghai Sangon Biological Engineering Technology and Services Co., Ltd., China. Other chemicals were of analytical grade.
B. subtilis CCTCC AB93108 was obtained from the China Center for Type Culture Collection (Wuhan, China). The microorganism was maintained at 4 ℃ on peptone agar slants, and transferred to fresh agar slants every 30 d to maintain viability.
2.2 Cultivation conditions
The basal medium used for the growth of biosurfactant-producing bacteria was the modified basic mineral salt medium as described by KIM et al [9] (g/L): glucose, 40; NH4NO3, 6.8; K2HPO4・3H2O, 13.8; NaH2PO4・2H2O, 1.95; MgSO4・7H2O, 0.5; MnSO4・H2O, 0.036; yeast extract, 0.5; pH 7.0. The inoculum of B. subtilis was prepared by transferring cells grown on an agar slant to 250 mL Erlenmeyer flasks containing 50 mL nutrient medium (g/L): peptone, 5.0; beef powder, 3.0; NaCl, 5.0; pH 7.0. For incubation, a culture grown in nutrient medium at 30 ℃ and 200 r/min for 24 h was added into the batch shake flasks to a final concentration of 2% (volume fraction). Then the cultivation was carried out in 1 000 mL Erlenmeyer flasks containing 200 mL basal medium at orbital shaking (200 r/min) and 30 ℃ for 72 h.
2.3 Biomass determination
The growth of bacteria was monitored by measuring the optical density of the culture at 600 nm after serial dilution (OD600) on a UV2552 spectrophotometer (Shimadzu, Japan).
2.4 Surface tension measurement
The measurement of the surface tension was carried out on the cell-free cultures or the isolated biosurfactant solutions on a Kruss K100 surface tensiometer (Hamberg, Germany) using the Du Noüy Ring method. The cell-free cultures were obtained by centrifuging the cultures at 10 000 r/min for 15 min. The biosurfactant solutions with different concentrations were obtained by dissolving the isolated biosurfactant in distilled water. The CMC was determined by measuring the surface tension of the biosurfactant solutions up to a constant value. At the CMC, sudden changes in surface tension and detergency were observed [13]. The CMC value was determined as mass per unit volume of biosurfactant (g/L).
2.5 Isolation of biosurfactant
Bacterial cells were removed from surfactant- containing medium by centrifugation (10 000 r/min, 15 min). The supernatant was subjected to acid precipitation by adding 6 mol/L HCl to achieve a final pH 2.0 and a precipitate was formed at 4 ℃ overnight [12]. The precipitate obtained was pelleted at 10 000 r/min for 15 min, redissolved in distilled water, adjusted to pH 7.0 by 1 mol/L NaOH and finally freeze-dried. Then, the dried material was extracted with organic solvent. The dried material was placed into the organic solvent at a ratio of 1:1 (ratio of dried material mass to organic solvent volume) to form a mixture. The organic solvent was made up of chloroform and methanol (65:15, volume ratio). Then, the mixture was filtered by 0.45 μm filter. The filter residual was collected and extracted with the organic solvent again as before. The extractions were repeated three times for a complete extraction. At last, the crude biosurfactant was obtained when the residual on the filter was air-dried under natural conditions.
2.6 Biosurfactant composition
Protein in the isolated biosurfactant was estimated by the Coomassie blue dye-binding method using bovine serum albumin as the standard [14]. Polysaccharide was measured by the phenol-sulfuric acid method using glucose as the standard [15]. To determine the lipid content, 0.2 g sample was blended with 6.0 mL chloroform/methanol mixture (2:1, volume ratio) and agitated acutely. Solvent phase was recovered by centrifuging at 10 000 r/min for 15 min. The extraction process was carried out three times. The whole solvent was collected, evaporated and dried under vacuum. The lipid content was determined by gravimetric estimation [12].
2.7 Emulsification capacity
The emulsification capacity was determined according to the method described by COOPER and GOLDENBERG [16] with slight modification. 2 mL of the cell-free culture after 60 h fermentation or the isolated biosurfactant solution was mixed with 2 mL of the hydrophobic compound (such as benzene, xylene, n-pentane, n-nonane, gasoline and diesel oil) in a screw-cap test tube. The concentration of biosurfactant in the aqueous solutions was 1.64 g/L. The mixture was homogenized by vortex at room temperature for 2 min. Measurements were made 24 h later. The emulsification index (E24) was calculated by dividing the measured height of the emulsion layer by the mixture’s total height and multiplying 100. Other two parameters were calculated following the method described by KATARZYNA et al [17]. Parameter A was described as the percentage of the hydrocarbon phase that was emulsified. Parameter B, the content of hydrocarbon in the emulsion, was calculated by dividing the volume of hydrocarbon in the emulsion phase by the total volume of the emulsion.
E24=(a/b)×100% (1)
A=(1-c/d)×100% (2)
B=[(d-c)/a]×100% (3)
where a is the height of the emulsion layer; b is the total height of the mixture; c is the height of the hydrocarbon un-emulsified; and d is the initial height of the hydrocarbon. All the above assays were performed in triplicate.
2.8 Effects of temperature, pH and time on emulsification capacity
Stability studies were performed using the cell-free culture obtained by centrifuging the cultures at 10 000 r/min for 15 min when the cultivation was maintained after 60 h. The thermal stability studies were carried out. 2 mL of the culture free of cells (pH 5.8-6.2) was maintained at 4, 40, 80 or 100 ℃ for 15 min, separately, and then kept at room temperature. At last E24 was measured. To study the pH stability of the cell-free culture activity, the culture was adjusted to different pH values (2-12) with 1 mol/L NaOH or HCl solution. Then, E24 was measured at room temperature. The effect of time on the emulsification capacity of the culture free of cells (pH 5.8-6.2) was determined as well. E24 was measured at the 1st, 3rd and 7th day when the mixtures were maintained at room temperature, respectively.
3 Results and discussion
3.1 Growth kinetics and biosurfactant production
The growth phases of B. subtilis CCTCC AB93108 cultivated in the fermentation medium are shown in Fig.1.
(1) The exponential phase (0-24 h). After inoculation cells grow at an exponential rate. The biomass increases obviously with increasing concentration of the biosurfactant in the medium.
(2) The stable phase (24-60 h). The cell optical density maintains a stable level, whereas the concentration of the biosurfactant still increases.
(3) The declining phase. The optical density and the concentration of biosurfactant both decrease. The similar phenomenon occurred when surfactin was produced by B. subtilis under Mn2+-free condition [18].
During the preliminary experiments, the bio- surfactant was successfully isolated. Acidic precipitation
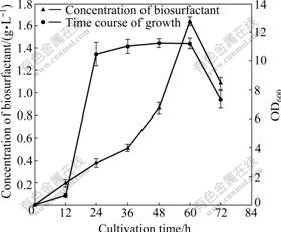
Fig.1 Time course of growth and concentration of biosurfactant by B. subtilis CCTCC AB93108 in fermentation medium (Error bars indicate standard deviations from means on basis of three replicate experiments)
was used to isolate many kinds of biosurfactants [12, 19-20]. For the biosurfactant produced by B. subtilis CCTCC AB93108, precipitation by HCl solution assured high recovery rate and low breakdown of the emulsification activity. The extraction by the organic solvent made up of chloroform and methanol was attempted to remove the lipopeptide biosurfactants (e.g., surfactins, bacilysins, and iturins) produced by B. subtilis [11-12, 21]. The whole process was described in details in section 2. The extracted precipitate might be thrown away in the former experiments. But in this process, it received much attention. The biosurfactant remained on the filter is brown floccule. The maximum concentration of the biosurfactant was 1.64 g/L when the bacterium was cultivated for 60 h (Fig.1).
3.2 Composition and CMC value
B. subtilis, which can produce many kinds of lipopeptide biosurfactants, is one of the well-known bacteria species [10-12]. The biosurfactant synthesized by the bacterium after cultivation for 60 h is used for chemical analysis. The examined agent is a polymeric material consisting of protein (45.3%-45.9%) and polysaccharide (8.6%-9.2%); no lipids are detected; and the left composition may be inorganic salts. This is a powerful evidence to prove that the mixture contains a new kind of biosurfactant, which is different from lipopeptides produced by B. subtilis before. The CMC of the biosurfactant is about 2.96 g/L. The surface tension of the aqueous solution reduces to 45.9 mN/m (Fig.2). This is an other powerful evidence to prove that the found material contains a new biosurfactant. Until now no information is reported that the biosurfactants produced by B. subtilis have similar surface tension value.

Fig.2 Surface tension vs concentration of isolated biosurfactant produced by B. subtilis CCTCC AB93108
3.3 Properties of emulsions
For practical purpose, it is important to distinguish an efficient surfactant from an effective surfactant. Efficiency is measured by the surfactant concentration required to produce a significant reduction in the surface tension of water, whereas, effectiveness is measured as the minimum value to which the surface tension can be reduced. Therefore, important properties of a potent surfactant include its ability to reduce the surface tension of an aqueous solution, and its low CMC [22]. However, if the surfactant is used as an emulsifier, its activity depends on its affinity for hydrocarbon substrates, which involves a direct interaction with the hydrocarbon itself rather than an effect on the surface tension of the medium [13]. The emulsification activity of some surfactants was also studied formerly. When the cell-free culture contains the surfactant produced by Curvularia lunata emulsified kerosene, parameter A, which was detected with the same method reported in section 2, was 86.4% as the maximum value [17]. Alasan produced by Acinetobacter radioresistens was able to emulsify long-chain aliphatics, but emulsified short-chain hydrocarbons (e.g., pentane) and aromatic compounds (e.g., benzene and xylene) poorly or not at all [23].
The emulsification activity of the isolated biosurfactant on different hydrophobic substrates is shown in Fig.3. The biosurfactant concentration is 1.64 g/L. For the six kinds of hydrocarbons studied in this work, the biosurfactant has the highest emulsification activity on benzene (E24, 70.0%). The second high emulsification activity is on xylene, and the lowest is on n-nonane (E24, 60.0%). Parameter B always achieves high efficiencies in this work. The lowest is 71.4% for benzene while the highest is 83.3% for n-nontane. That is to say, the volume of the hydrocarbons
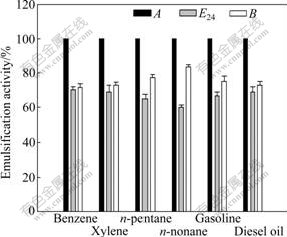
Fig.3 Emulsification activity (A, E24 or B) of various hydrocarbons by biosurfactant produced by B. subtilis CCTCC AB93108 (Isolated biosurfactant is dissolved in distilled water at concentration of 1.64 g/L; error bars indicate standard deviations from means on basis of three replicate experiments)
is 2.5-5.0 times of water in the emulsion layer. Parameter A, maintains 100% in the six instances. The results indicate that the hydrocarbons, such as benzene, xylene, n-pentane, n-nonane, gasoline and diesel oil, are all emulsified completely by the isolated biosurfactant at the specific concentration. This implies that the biosurfactant can emulsify not only aromatic compounds but also short- and long-chain hydrocarbons.
An important property of surfactants is their effectiveness over a wide range of temperature, time, and pH [13, 24]. These environmental factors affect the activity of the surface-active compounds in different ways. Benzene and xylene are chosen as the substrates because the isolated biosurfactant has the highest emulsification activity on them. The effects of thermal treatment (4, 40, 80 and 100 ℃) on the activity of the biosurfactant are shown in Fig.4(a). The emulsification index changes a little when the surfactant emulsifies both benzene and xylene. After 7 d storage, the stability of the biosurfactant still changed little (Fig.4(b)). E24 is stable during the whole observed periods. The stable emulsions extend to 30 d (data not shown) and may be longer. The results suggest that the emulsification activity of the biosurfactant is stable over a wide range of temperature (4-100 ℃) and a long period of duration. The effects of pH (2-12) on the emulsification activity of the cell-free culture are also studied when the surfactant emulsifies benzene and xylene (Fig.4(c)). The highest emulsification activity appears at pH 6; at pH 8, the emulsification activity is still at high levels; however, it is weak or ineffective at both pH 2 and pH 12. Though benzene and xylene are both aromatic hydrocarbons with one phenyl, there are some different complexions about emulsification on them when pH changes. At pH 4, the emulsification activity is strong on benzene, but weak on xylene. However, at pH 10, the status is just reversed. Generally, the extremely high or low pH transforms more surface-active species into less active emulsifier in this work. That is just contrary to the activity of surfactant described by SARUBBO et al [25], whereas the emulsi-

Fig.4 Effects of temperatures (a), time (b) and pH (c) on emulsification activity (E24) of biosurfactant produced by B. subtilis CCTCC AB93108 when surfactant emulsifies benzene and xylene (Error bars indicate standard deviations from means on basis of three replicate experiments)
fication activity is stable at pH 3-11 for surfactin produced by B. subtilis [12].
4 Conclusions
(1) A new biosurfactant produced by B. subtilis CCTCC AB93108 is found. It is different from the lipopeptide biosurfactants produced by B. subtilis formerly. The maximum concentration of the biosurfactant reaches 1.64 g/L when the strains grow on the water-soluble medium for 60 h.
(2) The biosurfactant is a complex of protein and polysaccharide without lipids. It reduces surface tension of distilled water to 45.9 mN/m at the CMC of 2.96 g/L.
(3) The biosurfactant causes the formation of stable emulsions on aromatic and aliphatic hydrocarbons, such as benzene, xylene, n-pentane, n-nonane, gasoline, and diesel oil. The emulsification activity is stable over a wide range of temperature (4-100 ℃) and a long period of duration.
References
[1] BANAT I M, MAKKAR R S, CAMEOTRA S S. Potential commercial application of microbial surfactants [J]. Applied Microbiology and Biotechnology, 2002, 53: 495-508.
[2] ZHONG Hua, ZENG Guang-ming, YUAN Xing-zhong, FU Hai-yan, HUANG Guo-he, REN Fang-yi. Adsorption of dirhamnolipid on four microorganisms and the effect on cell surface hydrophobicity [J]. Applied Microbiology and Biotechnology, 2007, 77: 447-455.
[3] DESAI J D, BANAT I M. Microbial production of surfactants and their commercial potential [J]. Microbiology and Molecular Biology Reviews, 1997, 61: 47-64.
[4] RON E Z, ROSENBERG E. Natural roles of biosurfactant [J]. Environmental Microbiology, 2001, 3: 229-236.
[5] ROSENBERG E, RON E Z. High- and low-molecular-mass microbial surfactants [J]. Applied Microbiology and Biotechnology, 1999, 52: 154-162.
[6] JOSHI S, BHARUCHA C, JHA S, YADAV S, NERURKAR A, DESAI A J. Biosurfactant production using molasses and whey under thermophilic conditions [J]. Bioresource Technology, 2008, 99: 195-199.
[7] XIA Wen-bin, LI Xin, GAO Hui, HUANG Bao-rong, ZHANG Hui-zhi, LIU Yun-guo, ZENG Guang-ming, FAN Ting. Influence factors analysis of removing heavy metals from multiple metal-contaminated soils with different extractants [J]. Journal of Central South University of Technology, 2009, 16: 108-111.
[8] TANG Lin, ZENG Guang-ming, SHEN Guo-li, LI Yuan-ping, ZHANG Yi, HUANG Dan-lian. Rapid detection of picloram in agricultural field samples using a disposable immunomembrane- based electrochemical sensor [J]. Environmental Science and Technology, 2008, 42: 1207-1212.
[9] KIM H S, YOON B D, LEE C H, SUH H H, OH H M, KATSURAGI T, TAN Y. Production and properties of a lipopeptide biosurfactant from Bacillus subtilis C9 [J]. Journal of Fermentation and Bioengineering, 1997, 84: 41-46.
[10] DAS K, MUKHERJEE A K. Comparison of lipopeptide biosurfactants production by Bacillus subtilis strains in submerged and solid state fermentation systems using a cheap carbon source: Some industrial applications of biosurfactants [J]. Process Biochemistry, 2007, 42: 1191-1199.
[11] TSUGE K, AKIYAMA T, SHODA M. Cloning, sequencing, and characterization of the iturin A operon [J]. Journal of Bacteriology, 2001, 183: 6265-6273.
[12] MAKKAR R S, CAMEOTRA S S. Production of biosurfactant at mesophilic and thermophilic conditions by a strain of Bacillus subtilis [J]. Journal of Industrial Microbiology and Biotechnology, 1998, 20: 48-52.
[13] RUFINO R D, SARUBBO L A, CAMPOS-TAKAKI G M. Enhancement of stability of biosurfactant produced by Candida lipolytica using industrial residue as substrate [J]. World Journal of Microbiology and Biotechnology, 2007, 23: 729-734.
[14] CIRIGLIANO M C, CARMAN G M. Isolation of a bioemulsifier from Candida lipolytica [J]. Applied and Environmental Microbiology, 1984, 48: 747-750.
[15] DUBOIS M, GILLES K A, HAMILTON J K, REBERS P A, SMITH F. Colorimetric method for determination of sugars and related substances [J]. Analytical Chemistry, 1956, 28: 350-356.
[16] COOPER D G, GOLDENBERG B G. Surface-active agents from two Bacillus species [J]. Applied and Environmental Microbiology, 1987, 53: 224-229.
[17] KATARZYNA P, ANITA K, JERZY D. Emulsifier production by steroid transforming filamentous fungus Curvularia lunata: Growth and product characterization [J]. Journal of Biotechnology, 2002, 92: 287-294.
[18] WEI Y H, LAI C C, CHANG J S. Using Taguchi experimental design methods to optimize trace element composition for enhanced surfactin production by Bacillus subtilis ATCC 21332 [J]. Process Biochemistry, 2007, 42: 40-45.
[19] MULLIGAN C N, YONG R N, GIBBS B F, JAMES S, BENNETT H P J, MORIKAWA M, HIRATA Y, IMANAKA T. Metal removal from contaminated soil and sediments by the biosurfactant surfactin [J]. Environmental Science and Technology, 1999, 33: 3812-3820.
[20] MORIKAWA M, HIRATA Y, IMANAKA T. A study on the structure-function relation of lipopeptide biosurfactants [J]. Biochimica et Biophysica Acta, 2000, 1488: 211-218.
[21] YAZGAN A, ?ZCENGIA G, MARAHIEL M A. Tn10 insertional mutations of Bacillus subtilis that block the biosynthesis of bacilysin [J]. Biochimica et Biophysica Acta, 2001, 1518: 87-94.
[22] LEE S C, LEE S J, KIM S H, PARK I H, LEE Y S, CHUNG S Y, CHOI Y L. Characterization of new biosurfactant produced by Klebsiella sp. Y6-1 isolated from waste soybean oil [J]. Bioresource Technology, 2008, 99: 2288-2292.
[23] NAVONVENEZIA S, ZOSIM Z, GOTTLIEB A, LEGMANN R, CARMELI S, RON E Z, ROSENBERG E. Alasan, a new bioemulsifier from Acinetobacter radioresistens [J]. Applied and Environmental Microbiology, 1995, 61: 3240-3244.
[24] HU Xing, LIU Hui, ZOU Guo-lin. Iron(Ⅱ) tetrasulfophthalocyanine mimetic enzymatic synthesis of conducting polyaniline in micellar system [J]. Journal of Central South University of Technology, 2009, 16: 743-748.
[25] SARUBBO L A, FARIAS C B B, CAMPOS-TAKAKI G M. Co-utilization of Canola oil and glucose on the production of a surfactant by Candida lipolytica [J]. Current Microbiology, 2007, 54: 68-73.
Foundation item: Project(IRT0719) supported by the Program for Changjiang Scholars and Innovative Research Team in University of China; Project (2004AA649370) supported by the National High-Tech Research and Development Program of China; Projects(50425927, 50225926) supported by the Natural Foundation for Distinguished Young Scholars; Project(2007F3102) supported by Technology Foundation for Creative Young Scholars of Fujian Province, China
Received date: 2009-06-23; Accepted date: 2009-10-08
Corresponding author: ZENG Guang-ming, PhD, Professor; Tel: +86-731-88822754; Fax: +86-731-88823701; E-mail: zgming@hnu.cn
(Edited by CHEN Wei-ping)