�����������ͭ��п�������������
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2014���11��
�������ߣ�Vesna T. CONI Mirjana M. RAJI VUJASINOVI Vlastimir K. TRUJI Vladimir B. CVETKOVSKI
����ҳ�룺3688 - 3695
�ؼ��ʣ����������������������Ⱦ���Acidithiobacillus ferrooxidans
Key words��bioleaching; polymetallic sulphide concentrate; mesophiles; Acidithiobacillus ferrooxidans
ժ Ҫ���������»�Ͼ�Acidithiobacillus ferrooxidans, Acidithiobacillus thiooxidans�� Leptospirillum ferrooxidans�Ե�Ʒλ����Cu-Zn-Pb-Fe-Ag-Au�������������������Ӧ���н�������������þ���Ϊ������ά��Bor����ͭ���������Һ��ɸѡ��һ�������������Ӫ��ҺΪpH 1.6��9KӪ��Һ��87%�Ŀ������ȴ���10 ��m�����ܶ�Ϊ8%(w/v)���ڲ��������£�п��ͭ�����Ľ����ʷֱ�ﵽ89%��83%��68%������ѧ��������������������Spencer-Topleyģ��������ֲܾ���Ӧ��ɢ���ơ�
Abstract: Bioleaching of low-grade complex Cu-Zn-Pb-Fe-Ag-Au sulphide concentrate (of Majdanpek ore body, RTB Bor, Serbia) was carried out in an aerated bioleach reactor in the presence of mesophilic mixed bacterial culture of Acidithiobacillus ferrooxidans, Acidithiobacillus thiooxidans, and Leptospirillum ferrooxidans. A mesophilic acidophiles culture was isolated from the acidic solution of the underground copper mine of Bor, Serbia. The nutrient medium was 9K at pH 1.6. 87% of the particles were <10 ��m in size, with a pulp density of 8% (w/v). Bioleaching efficiencies of 89% for zinc, 83% for copper, and 68% for iron can be achieved in the examined conditions. Kinetic analysis shows that the change in leaching corresponds to the Spencer-Topley kinetic model for diffusion-controlled topochemical reactions.
Trans. Nonferrous Met. Soc. China 24(2014) 3688-3695
Vesna T.  1, Mirjana M.
1, Mirjana M.  2, Vlastimir K.
2, Vlastimir K.  1, Vladimir B. CVETKOVSKI1
1, Vladimir B. CVETKOVSKI1
1. Laboratory for Bioleaching and Solvent Extraction, Mining and Metallurgy Institute Bor, Bor 19210, Serbia
2. Technical Faculty in Bor, University of Belgrade, Bor 19210, Serbia
Received 22 January 2014; accepted 20 May 2014
Abstract: Bioleaching of low-grade complex Cu-Zn-Pb-Fe-Ag-Au sulphide concentrate (of Majdanpek ore body, RTB Bor, Serbia) was carried out in an aerated bioleach reactor in the presence of mesophilic mixed bacterial culture of Acidithiobacillus ferrooxidans, Acidithiobacillus thiooxidans, and Leptospirillum ferrooxidans. A mesophilic acidophiles culture was isolated from the acidic solution of the underground copper mine of Bor, Serbia. The nutrient medium was 9K at pH 1.6. 87% of the particles were <10 ��m in size, with a pulp density of 8% (w/v). Bioleaching efficiencies of 89% for zinc, 83% for copper, and 68% for iron can be achieved in the examined conditions. Kinetic analysis shows that the change in leaching corresponds to the Spencer-Topley kinetic model for diffusion-controlled topochemical reactions.
Key words: bioleaching; polymetallic sulphide concentrate; mesophiles; Acidithiobacillus ferrooxidans
1 Introduction
Bio-hydrometallurgical processing of complex low-grade non-ferrous metal concentrates has an important role in contemporary research on the possibility of achieving higher overall recoveries of metal values from such mineral resources [1-5]. High grade non-ferrous and noble metal ores are being exhausted, creating a need for the development of new technologies that could meet growing technological, economic, and ecological demands with regard to the available resources and capacities [6-10]. Chemical analyses of such resources in copper mines in Bor, Serbia, indicate that the presence of Zn, Cu, Pb, Ag, and Au is most common. The processing of complex raw materials with an average copper content of 2% in the concentrate using existing pyrometallurgical technology is not economically satisfactory. The processing of polymetallic concentrates by bioleaching can have commercial benefits compared to the conventional pyrometallurgical processing method. Compared to hydrometallurgy, zinc concentrate bioleaching has the advantage that it does not require roasting, a sulphuric acid plant, or washing of gaseous effluents, energy consumption and cost are low, and flow process is short [1,11].
Acidithiobacillus (A.) ferrooxidans, one of the most important bioleaching microorganisms, bases its action on direct and indirect mechanisms. The first one refers to sulphur and sulphide oxidation by the bacteria attached to the particle surface. The latter is related to the oxidation of sulphur compounds by ferric ions resulted from the bacterial oxidation of ferrous ions. Although it is not easy to define the predominance of one or the other mechanism, the importance of cell attachment to the particles in order to carry out biooxidation is widely accepted [4,12].
Detailed study of A. ferrooxidans, an iron and sulphur oxidizing acidophile, showed that it grows much faster in the presence of ferrous ions than another mesophilic iron oxidizing chemolitotroph, Leptospirillum ferrooxidans. Besides that, some researchers proved that A. ferrooxidans grows best with a higher concentration of ferrous ions [13].
However, L. ferrooxidans is highly tolerant to acidic environments, and may enhance the regeneration of Fe3+ ions in acidic environments. That is why L. ferrooxidans is as important as A. ferrooxidans in the indirect leaching mechanism (over ferric ions). Furthermore, in some environments, L. ferrooxidans is to be found to a greater extent than A. ferrooxidans and it seems to be the dominant bacterium in industrial continuous-flow biooxidation tanks [14]. Some researchers have shown that the indigenous microorganisms which can be found in mineral raw materials are more efficient than many of the consortia created in laboratories [15,16].
Polymetallic sulphide concentrates of Majdanpek ore body in RTB Bor Complex contain 8%-27% Zn, 2%-4% Cu, 4%-14% Pb (mass fraction), 170-500 g/t Ag, and 3.7-700 g/t Au. When using the existing pyrometallurgical technology of the copper smelter in Bor to process them, the formation of their oxides and thus the loss of these metals could not be avoided. This indicates that it is possible to use Pb-Zn sulphide concentrates only as a supplement to the feed for smelting, in order to correct the feed composition to make it suitable for further processing, and not as an independent concentrate.
The aim of this work is to present an experimental research on the bio-hydrometallurgical processing of complex sulphide concentrates in the pilot laboratory plant for bio-hydrometallurgical copper production of the Mining and Metallurgy Institute Bor, Serbia, in order to define the optimum process conditions for maximum utilization of useful components.
2 Materials and methods
2.1 Concentrates characterization
Polymetallic sulphide concentrate was taken from the ore deposit of Majdanpek ore body (Serbia). The sample contained 27.2% Zn, 3.7% Cu, 18.4% Fe, 4.6% Pb (mass fraction), 3.5 g/t Au, and 178.9 g/t Ag. From the results of chemical and mineralogical analysis of the concentrate, it can be seen that the treated sample is polymetallic Zn, Cu, Fe, Pb, Ag, and Au sulphide concentrate. Mineralogical analysis of the concentrate and bioleach residue was performed using the X-ray diffraction (XRD) Explorer model analyser (GNR) with Cu Ka (0.154 nm). The operating conditions are 40 kV and 30 mA. The mineralogical XRD pattern is presented in Fig. 1, where it can be seen that the concentrate mostly consists of sphalerite (ZnS), chalcopyrite (CuFeS2), galena (PbS), and pyrite (FeS2). The concentrate was milled and 87% of particles were <10 ��m in size.

Fig. 1 XRD pattern for tested polymetallic sulphide concentrate
2.2 Microbiological culture
Mesophilic mixed bacterial culture of A. ferrooxidans, A. thiooxidans, and Leptospirillum ferrooxidans was isolated from underground mine water of the Bor Copper Mine in Serbia. The mesophilic mixed culture is presented in Fig. 2. Bacterial growth was observed on an Exacta Optech binocular microscope with 1000 times magnification. A sample of mine water was taken from the ore body ��Tilva  ��.
��.

Fig. 2 Optical micrograph of mixed mesophilic culture with 1000 times magnification
2.3 Microbiological culture characterization
Characterization of microbiological species presented in RTB Bor resources (mine waters) was conducted using quantitative polymerase chain reaction (Q-PCR) analysis-based molecular equipment, and determination of their number was done within the EU FP6 BioMinE project by Bioclear (the Netherlands), applying the Q-PCR and T-RFLP methods [17]. The natural microbial population in RTB Bor mine waters is listed in Table 1, from which it can be seen that the relative presence of cultures which exist in natural mine habitats is not large.
Table 1 Quantification of microbial population using Q-PCR

2.4 Analytical determinations
Ferrous and ferric ions concentrations were determined by titration with permanganate. This chemical analysis reliability is ��5%. The value of oxidation�Creduction potential (ORP) was measured by Pt electrode in relation to a Ag/AgCl reference electrode (mV (vs Ag/AgCl)). The acidity of the solution was measured by combined pH electrode. Zinc, copper, and iron in solution were measured every seven days by absorption spectrometry (Perkin-Elmer 403) at the Mining and Metallurgy Institute Bor, Serbia. Expanded uncertainties of concentrations are ��8.8%, ��7.5% and ��10% for zinc, copper, and iron, respectively. XRD analysis of the pulverized samples was conducted before and after the leaching process.
3 Results and discussion
The results of the dissolution of sulphides, including sphalerite, chalcopyrite, galena, and pyrite, by mixed mesophilic culture bioleaching were presented in this work. The analysis was done by applying the Q-PCR and T-RFLP methods [17]. For the treated sulphides, extended leaching time is required to achieve a high extraction degree using A. ferrooxidans, A. thiooxidans, and Leptospirillum ferrooxidans bacteria, which are active at temperatures in the range of 30-40 ��C. These sulphides are much more easily dissolved by extremely thermophilic bacteria compared to mesophilic and moderately thermophilic bacteria. However, extremely thermophilic bacteria show higher sensitivity to solids content as well as to the particles granulation of the dissolved material. Thermophiles are intolerant even to the small concentration of silver. If the concentrate contains silver, it will have a negative impact on the thermophiles�� growth [18]. Leaching by moderately thermophilic and extremely thermophilic bacteria requires higher energy consumption and corrosion- resistant equipment [19]. The chemical and mineralogical characterization of polymetallic concentrate was discussed in this work. For the test performed in the bioleaching reactor, a mixed mesophilic culture was used. The efficiency of extraction of Zn, Cu, and Fe was monitored simultaneously with the pulp ORP measurement, and the kinetics of the leaching process of all three metals was also examined.
3.1 Bacterial culture and nutrient
The culture isolated from acidic solution from underground exploitation of the Bor copper mine (Serbia) is mesophilic iron-sulphur oxidizing bacterial culture. Development of the culture was achieved in the testing laboratory of the Mining and Metallurgy Institute Bor, Serbia [20,21]. Development of mesophiles was achieved in 9K nutrient medium. The mineral salts of the 9K nutrient medium, produced by Lachner, are of p.a. quality and have the following composition: (NH4)2SO4 (3.0 g/L), KCl (0.1 g/L), K2HPO4 (0.5 g/L), MgSO4��7H2O (0.5 g/L), Ca(NO3)2��4H2O (0.01 g/L), FeSO4��7H2O (9 g/L). The acidity of the solution was adjusted to pH=1.8 with 1 mol/L H2SO4. The bacterial concentration at the end of the incubation period was within the limit of 108 cell/mL, which corresponds to the literature data [22].
3.2 Culture adaptation
The bacterial culture prepared by isolation and growth was adapted for oxidation of polymetallic Zn-Cu-Pb-Fe-Ag-Au sulphide concentrate by processing in the 2 L volume glass reactor with magnetic stirring at 150 r/min and a temperature of 30 ��C in the presence of (0.15%) CO2 enriched air. The total solution volume was 1 L. The solution contained 5% inoculum with nutrient medium and 5 g/L polymetallic concentrate for culture adaptation. The initial pH value was adjusted to pH 1.6. The used 1 mol/L H2SO4 (produced by Molar) was of p.a. quality. The process was monitored by daily measurement of the pH value and ORP. Complete oxidation of ferrous ions was achieved in a period of 6 d. The value of ORP in that period gradually increased from 380 to 550 mV. In the period from 6 to 8 d, the ORP value remained at the maximum value of 550 mV.
3.3 Minerals characterization
Modal analysis of the tested sample (Table 2) shows that the content of basic sulphide minerals was 40.5% sphalerite, 10.6% chalcopyrite, 5.3% galena, and 32% pyrite. The chemical composition of the concentrate is presented in Table 3.
Table 2 Mineralogical components of concentrate

Table 3 Chemical composition of concentrate

3.4 Particle size analysis
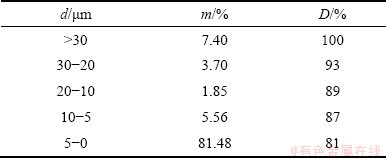
The concentrate was milled in the laboratory of the technical faculty in Bor in Siebtechnik vibrating ring mill. The concentrate was milled to 87% of particles <10 ��m in size. The particle size of the concentrate after milling is presented in Table 4, where m indicates the mass fraction of the appropriate fraction and D is the cumulative undersize. The two finest fractions were used in the experiment.
Table 4 Particle size analysis of concentrate
3.5 Laboratory bioleaching test
After adaptation by adding fresh quantities of polymetallic concentrate of 75 g/L, the total solids content of the pulp was 8% (w/v). The value of the redox potential ORP dropped to an initial value of 380 mV. The test was continued under the same conditions as during the culture adaptation in a 2 L glass reactor with a solution volume of 1 L at 150 r/min and a temperature of 30 ��C. Air enriched with CO2 (0.15%) was introduced into the reactor through an air diffuser. The acidity of the solution was corrected to pH 1.6 by adding 1 mol/L of sulphuric acid. Distilled water was added into the reactor to compensate for the evaporation of the process water. The potential versus the Ag/AgCl electrode was gradually increased from 380 to 550 mV, as presented in Fig. 3. The change in the ORP value was followed by the increase in the Zn, Cu, and Fe concentrations in the solution. After 40 d of mesophilic leaching, the extraction efficiency remained at the same level. In that period, by leaching of 80 g/L of concentrate, the final concentrations of 19.56 g/L Zn, 2.4 g/L Cu, and 10.45 g/L Fe were achieved. Chalcopyrite was dissolved at ORP values higher than 510 mV, while pyrite oxidation was only important when the ORP was higher than 450 mV, which is in agreement with the findings of some other researchers [23].

Fig. 3 Graph illustration of oxidation-reduction potential (ORP) during bioleaching test
The following reactions describe the direct and indirect oxidation mechanisms of Zn-Cu-Pb-Fe sulphides. In the direct mechanism, metal sulphides can be oxidized by mesophilic microorganisms into soluble sulphates according to reactions (1) to (4):
ZnS+1/2O2+H2SO4 ZnSO4+H2O+S (1)
ZnSO4+H2O+S (1)
CuFeS2+O2+2H2SO4 CuSO4+FeSO4+2H2O+2S (2)
CuSO4+FeSO4+2H2O+2S (2)
2FeS2+7O2+2H2O 2FeSO4+2H2SO4 (3)
2FeSO4+2H2SO4 (3)
PbS+1/2O2+H2SO4 PbSO4+H2O+S (4)
PbSO4+H2O+S (4)
In the indirect mechanism of sulphide minerals oxidation, the sulphides oxidation by ferric ions proceeds according to the following reaction:
(Zn, Cu, Fe, Pb)S+Fe2(SO4)3��(Zn, Cu, Fe, Pb)SO4+2FeSO4+S0 (5)
According to the presented mechanism, the dissolution of metals takes place by a cyclic process between reactions (5) and (6).
4FeSO4+2H2SO4+O2 2Fe2(SO4)3+2H2O (6)
2Fe2(SO4)3+2H2O (6)
By formation of H+ ions during sulphur biooxidation, according to reaction (7)
2S0+3O2+2H2O 2H2SO4 (7)
2H2SO4 (7)
the solubility and total recovery are increased.
In the sulphide areas, sulphuric acid is generated by indirect and direct bacterial attacks. By sulphuric acid generation, the acid balance is improved, which creates equilibrium in the system. Also, the reaction is exothermic, so it can be directed to increase the temperature, which improves the kinetics [24].
Distortion of the sulphide mineral crystal lattice occurs because of the much higher ionic radius of the sulphate ions. Thus, microbes gain access to new unreacted places [25].
3.6 Mineralogical analysis of solid residue
After the bioleaching process, the mass fraction of insoluble residue was 30% of the initial value. By bioleaching of polymetallic concentrate, Zn and Cu in the solution are converted into the sulphates, which can be further extracted by different extractants [26]. At the same time, Pb, Au, and Ag remain insoluble in the bioleach residue. By the application of the PLINT process, it is possible to obtain Au, Ag, and Pb from the bioleach residue [27]. XRD analysis of the solid residue, presented in Fig. 4, showed that the galena structure is converted into lead sulphate, which also dominates in the secondary beudantite, whereby it remains in the leach residue together with Ag and Au. Based on the mineralogical analysis of the concentrate and bioleach residue, it could be observed that the destruction of the sample crystal lattice occurred.
The fact that the leaching rate obtained by bioleaching with mixed cultures is higher than that obtained by leaching with pure culture indicates that the influence of mineralogical composition is also important. Relevant information on ore mineralogical composition helps in understanding the interpretation of the microbial dissolution process [19]. Knowledge about the mineralogical composition of minerals in the ore is very important for understanding the biological processes of microbial dissolution, allowing for optimization of the process and processing method. The purpose for testing the mineralogical composition of the concentrate is to optimize the process parameters of bioleaching of complex sulphide concentrates using mixed mesophilic culture of A. ferrooxidans, A. thiooxidans, and Leptospirillum ferrooxidans with reference to the leaching of zinc and copper. The processing parameters of bioleaching processes are particle size, stirring speed, dissolution time, inoculum volume, pulp density, and pH value of the process.

Fig. 4 XRD pattern for bioleach residue
The minerals supergene zone contains Zn, Cu, Fe, and Pb. This zone and the adjacent rocks also contain quartz minerals with relicts of present ore minerals including galena. Members of the solid solution series beudantite-segnitite and lead jarosite represent a small part of a large group of natural compounds with alunite type structure. This alunite structure may also contain quartz. Under the influence of sulphuric acid, the decomposition of bedrock alunite occurs, whereby the beudantite can be extracted from galena, while quartz from such bedrock remains in the form of bioleach slurry.
Bioleaching experiments were performed in a glass reactor by using the mixed culture, which shows a higher leaching than the pure cultures [28]. In the incubation process in 9K nutrient solution, the number of bacteria reaches a value of 108 cell/mL. The acidity of the solution was adjusted to pH 1.6. The acidic condition is the bottleneck for the chemical regeneration of ferric ions from ferrous ions. Therefore, it is necessary to use the iron oxidizing bacteria A. ferrooxidans [3]. The ratio of ferric to ferrous ions in the bioleaching process is presented in Fig. 5. If the acidity of the solution was lower, there would be a concern about jarosite appearing, which would precipitate copper in the slurry and further reduce the recovery from leaching [2,5].

Fig. 5 Iron concentration released into solution with change of ��(Fe3+)/��(Fe2+) ratio and ��(Fe2+), ��(Fe3+) and ��tot(Fe) concentrations during bioleaching of concentrate by using mesophilic mixed culture at 30 ��C
At the beginning of fresh sulphide minerals bioleaching, the ORP potential, ��h, is very low, which corresponds to the highest concentration of ferrous iron in the leaching solution. This should not be interpreted as an ineffective situation. In fact, under these conditions, the reactor can show great bacterial activity and a high sulphide leaching rate. When the ORP potential, Eh, begins to increase, the concentrations of zinc, copper, and iron in the solution also increase. Under these conditions, the maximum increase in the ratio of ��(Fe3+)/��(Fe2+) over time also occurs. When the maximum value of the ORP potential, 550 mV, is reached, the highest content of metal leaching is also reached (Fig. 2), which has also been proven by some other researchers [29]. The change in the ratio of ��(Fe3+)/��(Fe2+) is shown in Table 5. It can be seen that the maximum value of the ions ratio reaches 3.5.
Table 5 Change in ferrous and ferric ions concentration as well as ��(Fe3+)/��(Fe2+) ratio at different time

The solid phase content in the pulp was chosen to be 8% (w/v) because the density of the pulp must ensure the flow of CO2 gas [4]. The experiment was conducted with particle size <10 ��m. By reducing the particle size, the degree of leaching increases because the number of cells attached to the mineral increases. A particle size below 6.4 ��m could have a negative impact on the microbial leaching performance [30]. If the particle size is additionally lowered, the oxygen supply would be reduced. For a larger particle size, the number of microbial attachments, and therefore the cells�� ability to grow and develop, would be lower. Less metal would be exposed to leaching, which would reduce the contact between bacteria and minerals. Also, there would be a non-uniform distribution of sulphur, which is required for microbial growth [19].
The final values of zinc, copper, and iron recovery achieved by bioleaching are 89%, 83%, and 68%, respectively. Sphalerite (ZnS) has the lowest electrochemical potential and is easy to be oxidized. Chalcopyrite (CuFeS2) has higher electrochemical potential and is more difficult to be oxidized, while oxidation of pyrite (FeS2) is the most difficult, including bacterial oxidation [31].
3.7 Analysis of bioleaching kinetics
The fractions of zinc, copper, and iron extraction during the process of bioleaching as a function of time are presented in Fig. 6. One can see that the maximum metal leaching is achieved for a period of 40 d. When the bioleaching time exceeds 40 d, the degree of leaching does not increase any further. Since the particles are of nearly spherical shape with three equal geometric coordinates, they have the largest surface change while the process is occurring (assuming that the reaction rate is the same in all directions). Geometrical changes in this reaction type were studied by SPENCER and TOPLEY [32] using samples in which the particle radius changed linearly with time.
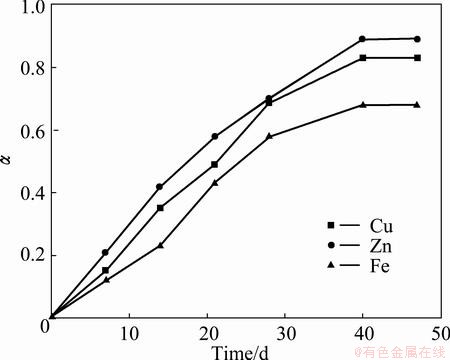
Fig. 6 Dependence of ��-fraction of reacted Zn, Cu and Fe metals on time in 1 L solution at 30 ��C with 8% pulp density
The usual criterion for the application of each kinetic equation in the kinetic process parameters calculation is its linearization in an appropriate coordinate system using the experimental results ��= f(��).
Namely, to determine the kinetic model, the leaching efficiencies ��=f(��) have been introduced in equations corresponding to different possible reaction mechanisms. The best linearization was achieved by using the Spencer-Topley equation [33,34]:
1-(1-a)1/2=kt (8)
where �� is the mass fraction of metals extracted; k is the specific rate constant (s-1); t is the reaction time (d).
The reaction rate is controlled by surface contact of the reactant with the product, indicating that the rate is controlled by geometry topochemical reaction.
A graphical representation of the experimental data in the coordinate system which corresponds to the Spencer-Topley equation is shown in Fig. 7.

Fig. 7 Relationship beteen 1-(1-��)1/2 and time for complex concentrate bioleaching according to data in Fig. 6
Good linearization exists for all three investigated metals. In all three cases, straight lines start from the origin of the coordinate system. The slopes of these lines correspond to the rate constants of the corresponding reactions (Table 6).
Table 6 Reaction rate constants for Cu, Zn, and Fe leaching at 30 ��C

The last measured values of a for all three metals (the last points on the curves presented in Fig. 6) are not taken into account during the linearization because saturation occurs in 40 d. By plotting the linearization it is possible to assume that the bioleaching follows the proposed Spencer-Topley diffusion model.
The future investigation will be done with isolated culture of Leptospirillum ferrooxidans and A. thiooxidans without A. ferrooxidans at 40 ��C. L. ferrooxidans has a higher affinity for Fe2+ than A. ferrooxidans. L. ferrooxidans can grow at stronger acidity levels and higher temperatures, whereby the percentage of the leached metals should be higher and the period of leaching time should be shorter [1,35-37]. But, it should be mentioned that the efficient microbial adaptation is usually a long-term process.
4 Conclusions
1) Mixed mesophilic bacterial culture of A. ferrooxidans, A. Thiooxidans, and Leptospirillum ferrooxidans is proved to be effective for leaching of a polymetallic sulphide concentrate from the Majdanpek ore body (RTB Bor, Serbia). Increasing pyrite content along with minimization of the pH leaching value minimizes jarosite precipitation.
2) Mesophilic mixed bacterial cultures can be used for bioleaching of polymetallic concentrate, whereby the efficiencies of extraction are 89% for Zn, 83% for Cu, and 68% for Fe.
3) The process of polymetallic sulphide concentrates bioleaching follows the Spencer-Topley equation for a topochemical reaction on particles in which all three dimensions are equally developed.
Acknowledgements
This work arose from the results of a project approved and funded by the Ministry of Education, Science and Technology Development of the Republic of Serbia (Project Nos. TR 34004 and TR 34024).
The authors would like to thank Inez DINKLA from Bioclear, the Netherlands, for characterization and number determination of microbiological species presented in mine waters from RTB Bor resources, which was done within the EU FP6 BioMinE project by Bioclear, the Netherlands (European project contract NMP2-CT-2005-500329-1).
References
[1] GIAVENO A, LAVALLE L, CHIACCHIARINI P, DONATI E, Bioleaching of zinc from low-grade complex sulphide ores in an airlift by isolated Leptospirillum ferrooxidans [J]. Hydrometallurgy, 2007, 89(1-2): 117-126.
[2] GERICKE M, MULLER H H, van STADEN P J, PINCHES A. Development of a tank bioleaching process for the treatment of complex Cu-polymetallic concentrates [J]. Hydrometallurgy, 2008, 94(1-4): 23-28.
[3] PATEL B C, TIPRE D R, DAVE S R. Optimization of copper and zinc extractions from polymetallic bulk concentrate and ferric iron bioregeneration under metallic stress, [J]. Hydrometallurgy, 2012, 117-118(1): 18-23.
[4] RAWLINGS D E, JOHNSON D B. The microbiology of biomining: Development and optimization of mineral-oxidizing microbial consortia [J]. Microbiology, 2007, 153(2): 315-324.
[5] QIN Wen-qing, LI Wei-zhong, LAN Zhuo-yue, QIU Guan-zhou. Simulated small-scale pilot plant heap leaching of low-grade oxide zinc ore with integrated selective extraction of zinc [J]. Minerals Engineering, 2007, 20(7): 694-700.
[6] TIPRE D R, VORA S B, DAVE S R. Medium optimization for bioleaching of metals from Indian bulk polymetallic concentrate [J]. Indian Journal of Biotechnology, 2004, 3(1): 86-91.
[7] DAREZERESHKI E, SCHAFFIE M, LOTFALIAN M, SEIEDBAGHERY S A, RANJBAR M. Use of mesophilic and thermophilic bacteria for the improvement of copper extraction from a low-grade ore [J]. International Journal of Minerals, Metallurgy and Materials, 2011, 18(6): 138-143.
[8] BRIERLEY J A. A perspective on developments in biohydrometallurgy [J]. Hydrometallurgy, 2008, 94(1-4): 2-7.
[9] GERICKE M, NEALE J W, van STADEN P J. A Mintek perspective of the past 25 years in minerals bioleaching [J]. The Journal of the Southern African Institute of Mining and Metallurgy, 2009, 109(1): 567-585.
[10] GERICKE M,  V,
V,  D, CVETKOVSKI V. New treatment of flotation concentrates from RTB copper mine [J] Rudarski Radovi, 2008(2): 89-106.
D, CVETKOVSKI V. New treatment of flotation concentrates from RTB copper mine [J] Rudarski Radovi, 2008(2): 89-106.
[11] WANG Jun, ZHAO Hong-bo, ZHUANG Tian, QIN Wen-qing, ZHU Shan, QIU Guan-zhou. Bioleaching of Pb-Zn-Sn chalcopyrite concentrate in tank bioreactor and microbial community succession analysis [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(12): 3758-3762.
[12] GONZALEZ R, GENTINA J C, ACEVEDO F. Attachment behaviour of Thiobacillus ferrooxidans cells to refractory gold concentrate particles [J]. Biotechnology Letters, 1999, 21(8): 715-718.
[13] HAGHSHENAS D F, BONAKDARPOUR B, KESHAVARZ E A, NASERNEJAD B. Optimization of physicochemical parameters for bioleaching of sphalerite by Acidithiobacillus ferrooxidans using shaking bioreactors [J]. Hydrometallurgy, 2012, 111(1): 22-28.
[14] CORAM N J, RAWLINGS D E. Molecular relationship between two groups of the genus leptospirillum and finding that leptospirillum ferriphilum sp. nov. dominates South African the commercial biooxidation tanks that operate at 40 ��C [J]. Appl Environ Microbial, 2002, 68(2): 838-845.
[15] ASMAH R, BOSORMPEM K, OSEI Y, RODRIGUES F, ADDY M, CLEMENT C, WILSON M. Isolation and characterization of mineral oxidizing bacteria from the Obuasi gold mining site, Ghana [C]//Proceedings of International Biohydrometallurgy Symposium. Madrid Spain: Elsevier, 1999: 657-661.
[16] KEELING S E, PALMER M L, CARACATSANIS F C, JOHNSON J A, WATLING H R. Leaching of chalcopyrite and sphaleriteusing bacteria enriched from a spent chalcocite heap [J]. Minerals Engineering, 2005, 18(13-14): 1289-1296.
[17] DINKLA I, GONZALEZ-CONTRERAS P, SEKHARGAHAN C, WEIJMA J, BUISMAN C, HENSSEN M, SANDSTROM A. Quantifying microorganisms during biooxidation of arsenite and bioleaching of zinc sulfide [J]. Minerals Engineering, 2013, 48: 25-30.
[18] NEALE J W, GERICKE M, RAMCHARAN K. The application of bioleaching to base metal sulfides in southern Africa: Prospects and opportunities [C]//Proceedings of the 6th Southern African Base Metals Conference. Cape Toun: SAIMM, 2011: 367-388.
[19] OLUBAMBI P A, NDLOVU S, POTGIETER J H, BORODE J O. Role of ore mineralogy in optimizing conditions for bioleaching low-grade complex sulphide ores [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(5): 1234-1246.
[20]  V, CVETKOVSKI V,
V, CVETKOVSKI V,  M. Pilot plant for biohydrometallurgical production of copper [J]. Chemical Industry, 2009, 63(1): 51-57.
M. Pilot plant for biohydrometallurgical production of copper [J]. Chemical Industry, 2009, 63(1): 51-57.
[21]  V, CVETKOVSKI V,
V, CVETKOVSKI V,  M. Enrichment of mesophilic acidophiles from the underground copper mine Bor [J]. Chemical Industry, 2009, 63(1): 47-50.
M. Enrichment of mesophilic acidophiles from the underground copper mine Bor [J]. Chemical Industry, 2009, 63(1): 47-50.
[22] KARAVAIKO G, ROSSI G, AGATE A, GROUDEV S, AVAKYAN Z, Biogeotechnology of metals, manual [M]. Moscow: Centre for International Projects GKNT Moscow Publishers, 1988: 10-11.
[23] BOON M, HEIJNEN J J, HANSFORD G. The mechanism and kinetics of bioleaching sulphide minerals [J]. Miner Process Extr Metall Rev, 1998, 19(1): 107-115.
[24] ROBERTSON S W, VERCUIL A, van STADEN P J, CRAVEN P A. Bacterial heap leaching approach for the treatment of low grade primary copper sulphide ore [C]//Proceedings of the Third Southern African Conference on Base Metals. Johannesburg, Elasevier, 2005: 471-484.
[25]  Application tiobacteria isolated from tailings and waste water in Bor for leaching of Bor tailings [C]//Proceedings of Mining and Metallurgy Faculty and Copper Institute Bor. Bor, 1978: 157.
Application tiobacteria isolated from tailings and waste water in Bor for leaching of Bor tailings [C]//Proceedings of Mining and Metallurgy Faculty and Copper Institute Bor. Bor, 1978: 157.
[26] CHENG Y, GUO Z, LIU X, YIN H, QIU G, PAN F, LIU H. The bioleaching feasibility for Pb/Zn smelting slag and community characteristics of indigenous moderate-thermophilic bacteria [J]. Bioresource Technology, 2009, 100(10): 2737-2740.
[27]  LOZANO J I. Silver, gold and lead recovery from bioleaching residues using the PLINT process [J]. Minerals Engineering, 2002, 15(11): 877-878.
LOZANO J I. Silver, gold and lead recovery from bioleaching residues using the PLINT process [J]. Minerals Engineering, 2002, 15(11): 877-878.
[28] AKCIL A, CIFTCI H, DEVECI H. Role and contribution of pure and mixed cultures of mesophiles in bioleaching of a pyritic chalcopyrite concentrate [J]. Minerals Engineering, 2007, 20(3): 310-318.
[29] HALINEN A K, RAHUNEN N, KAKSONEN A, PUHAKKA J A. Heap bioleaching of a complex sulfide ore. Part I: Effect of pH on metal extraction and microbial composition in pH controlled columns [J]. Hydrometallurgy, 2009, 98(1-2): 92-100.
[30] NEMATI M, LOWENADLER J, HARRISON S. Particle size effects in bioleaching of pyrite by acidophilic thermophile Sulfolobus metallicus (BC) [J]. Applied Microbiology and Biotechnology, 2000, 53(2): 173-179.
[31] VARDANYAN N S, VARDANYAN A K. Selective extraction of metals from zinc concentrate by association of chemolithotrophic bacteria [J]. Applied Biochemistry and Microbiology, 2011, 47(5): 515-519.
[32] SOHN H, WADSWORTH M E. Rate process of extractive metallurgy [M]. New York: Plenum, 1979.
[33] TATHAVADKAR V D, ANTONY M P. The soda-ash roasting of chromite minerals: Kinetics considerations [J]. Metallurgical and Materials Transactions B, 2001, 32(4): 593-602.
[34] LAHIRI A, JHA A. Kinetics and reaction mechanism of soda ash roasting of ilmenite ore for the extraction of titanium dioxide [J]. Metallurgical and Materials Transactions B, 2007, 38(6): 939-948.
[35] QIU M Q, XIONG S Y, ZHANG W M, WANG G X. A comparison of bioleaching of chalcopyrite using pure culture or a mixed culture [J]. Minerals Engineering, 2005, 18(9): 987-990.
[36] WANG Jun, QIU Guan-zhou, QIN Wen-qing, ZHANG Yan-sheng. Microbial leaching of marmatite by Acidithiobacillus ferrooxidans and Acidithiobacillus thiooxidans [J]. Transactions of Nonferrous Metals Society of China, 2006, 16(4): 937-942.
[37] WANG Jun, QIN Wen-qing, ZHANG Yan-sheng, YANG Cong-ren, ZHANG Jian-wen, NAI Shao-shi, SHANG He, QIU Guan-zhou. Bacterial leaching of chalcopyrite and bornite with native bioleaching microorganism [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(6): 1468-1472.
Vesna T.  1, Mirjana M.
1, Mirjana M.  2, Vlastimir K.
2, Vlastimir K.  1, Vladimir B. CVETKOVSKI1
1, Vladimir B. CVETKOVSKI1
1. Laboratory for Bioleaching and Solvent Extraction, Mining and Metallurgy Institute Bor, Bor 19210, Serbia
2. Technical Faculty in Bor, University of Belgrade, Bor 19210, Serbia
ժ Ҫ���������»�Ͼ�Acidithiobacillus ferrooxidans, Acidithiobacillus thiooxidans�� Leptospirillum ferrooxidans�Ե�Ʒλ����Cu-Zn-Pb-Fe-Ag-Au�������������������Ӧ���н�������������þ���Ϊ������ά��Bor����ͭ���������Һ��ɸѡ��һ�������������Ӫ��ҺΪpH 1.6��9KӪ��Һ��87%�Ŀ������ȴ���10 ��m�����ܶ�Ϊ8%(w/v)���ڲ��������£�п��ͭ�����Ľ����ʷֱ�ﵽ89%��83%��68%������ѧ��������������������Spencer-Topleyģ��������ֲܾ���Ӧ��ɢ���ơ�
�ؼ��ʣ����������������������Ⱦ���Acidithiobacillus ferrooxidans
(Edited by Yun-bin HE)
Corresponding author: Vesna T.  ; Tel: +38-1611618224; E-mail: vesna.conic@irmbor.co.rs
; Tel: +38-1611618224; E-mail: vesna.conic@irmbor.co.rs
DOI: 10.1016/S1003-6326(14)63516-0

