Influence of hydrolysis in sulfate process on titania pigment producing
来源期刊:中国有色金属学报(英文版)2009年增刊第3期
论文作者:田从学 杜剑桥 陈新红 马维平 罗志强 程晓哲 胡鸿飞 刘代俊
文章页码:829 - 833
Key words:titanyl sulfate; hydrolysis; F value; pre-adding water; heating rate
Abstract: Using industrial titanyl sulfate solution as feedstock, titania white pigment was prepared by self-generated seeded thermal hydrolysis method. The influence of some factors (such as F value, volume ratio of pre-adding water to TiOSO4, heating rate and pH of pre-adding water) on the structure and pigment properties of the as-prepared slurry metatitanic acid and titania samples were studied. The samples were characterized by XRD, particle size distribution, SEM and pigment properties test. The results show that all samples show anatase phase, and the calcined samples prepared under suitable conditions are with narrow particle size distribution and good pigment properties. All these influencing factors have great impacts on the hydrolysis velocity, nucleation rate, crystal growth rate, particle size distribution, crystal structure and pigment properties. The suitable F value is 1.73-1.93, the volume ratio of pre-adding water and TiOSO4 solution is 0.28:1, the optimum heating rate is 0.94-1.33 ℃/min and pH of the pre-adding water should be at near-neutral range.
基金信息:Pangang Group

TIAN Cong-xue(田从学)1, 2, DU Jian-qiao(杜剑桥)1, CHEN Xin-hong(陈新红)1, MA Wei-ping(马维平)1,
LUO Zhi-qiang(罗志强)1, CHENG Xiao-zhe(程晓哲)1, HU Hong-fei(胡鸿飞)1, LIU Dai-jun(刘代俊)2
1. Panzhihua Iron and Steel Research Institute, Pangang Group, Panzhihua 617000, China;
2. School of Chemical Engineering, Sichuan University, Chengdu 610065, China
Received 10 August 2009; accepted 15 September 2009
Abstract: Using industrial titanyl sulfate solution as feedstock, titania white pigment was prepared by self-generated seeded thermal hydrolysis method. The influence of some factors (such as F value, volume ratio of pre-adding water to TiOSO4, heating rate and pH of pre-adding water) on the structure and pigment properties of the as-prepared slurry metatitanic acid and titania samples were studied. The samples were characterized by XRD, particle size distribution, SEM and pigment properties test. The results show that all samples show anatase phase, and the calcined samples prepared under suitable conditions are with narrow particle size distribution and good pigment properties. All these influencing factors have great impacts on the hydrolysis velocity, nucleation rate, crystal growth rate, particle size distribution, crystal structure and pigment properties. The suitable F value is 1.73-1.93, the volume ratio of pre-adding water and TiOSO4 solution is 0.28?1, the optimum heating rate is 0.94-1.33 ℃/min and pH of the pre-adding water should be at near-neutral range.
Key words: titanyl sulfate; hydrolysis; F value; pre-adding water; heating rate
1 Introduction
Titanium dioxide (TiO2) is used in a wide range of products, ranging from paints to foodstuffs. Due to its high opacity, small crystal size, and inertness, it is the best choice of white pigment. The commercial production of TiO2 follows one of two routes, sulfate process and chloride Process. There is abundant of ilmenite in Panzhihua. However, due to its high content of calcium and magnesium, only the sulfate process can be adopted to produce white pigment. Hydrolysis of industrial titanyl sulfate solution (TiOSO4 solution, black liquor) is the essential step of the process. And the particle morphology, size and size distribution of the hydrolyzed product, namely the hydrated titania or metatitanic acid, determine the quality of titania at some extent[1]. The hydrolysis of TiOSO4 solution has been widely studied, such as the formation mechanism[2-3], the precipitation procedure[4-5], the growth of TiO2 hydrate[6] and the thermal hydrolysis kinetics[7], and all the reports reached the point that the hydrolysis process undergoes three steps, i.e. grain formation of crystallization, nuclei growth and hydrated titania precipitation, hydrated titania aggregation and composition changes of the precipitation. SATHYAMOORTHY et al[8] investigated the particle formation process of anatase and the mechanisms behind particle formation during anatase precipitation from seeded concentrated titanyl sulfate solution. Recently, HIDALGO et al[9] optimized thermal hydrolysis of TiOSO4 to prepare highly photoactive supported TiO2. The hydrogen peroxide solution was also used at low temperature to prepare anatase/rutile dual layer deposition[10], and other methods such as detonation method[11], homogeneous hydrolysis[12] and composite templating method[13] were used to prepare functional titania. GRZMIL et al[14-16] investigated the influences of admixtures, TiOSO4 and free sulphuric acid concentrations in the starting solution, as well as various kinds and amounts of titanium dioxide nuclei addition on the hydrolysis process and the products. All these reports focused on the particle size and its distribution, as well as the structure of the crystal and precipitation, but without further investigation of its pigment properties and no detail information of the precipitation. The particle size and distribution, crystal structure, even the precipitation process are the heart of pigment production and determine the final pigment quality and profits. In this paper, some hydrolysis factors which influence the structure and pigment properties in self-generated seeded hydrolysis process were studied.
2 Experimental
Industrial titanyl sulfate (TiOSO4) solution was taken from the sulfate process of ilmenite (Titanium Dioxide Factory of Pangang Group, PRC), and the composition was as follows: TiO2 of 231.07 g/L, F= m(effective H2SO4)/m(TiO2)=1.73, m(Fe)/m(TiO2)=0.26, Ti3+ of 2.01 g/L. The influencing factors, such as F value, volume ratio of pre-adding water to titanyl sulfate solution, heating rate from the end of feeding to the first boiling point and pH of the pre-adding water were investigated.
A typical procedure of preparing metatitanic acid is as follows. The industrial titanyl sulfate solution and pre-adding water (volume ratio 4?1) were warmed up to (96±1) ℃, respectively, and then the heated TiOSO4 solution was dropped into the pre-adding water at a constant speed in 17 min. After feeding off, the mixture solution was heated to the boiling point (called the first boiling point) by controlling the heating rate. When the solution turned into grey, heating and stirring were stopped immediately. After 30 min, heating and stirring were turned on again, then the solution was heated to boiling point (called the second boiling point) in another few minutes, and the mixture gradually changed into milky white. After some time from the second boiling point, distilled water was dropped into the system to increase the hydrolysis ratio of titanium and maintain the hydrolysis slurry concentration at some degree. The hydrolysis process was over after 3 h from the beginning of the second boiling point, then the slurry was filtered and washed using diluted sulfate solution and distilled water, and metatitanic acid was obtained. The as-prepared metatitanic acid was then treated by phosphate and potassium salts, and so on. The salt treated slurry was dried and calcined to prepare titania pigments.
The as-prepared slurry, metatitanic acid and titania were characterized by the following measurements. X-ray diffraction (XRD, D/max-rC) was used to investigate the crystal phase composition. Particle size distribution test was carried out on a Malvern particle size analyzer (Zetasizer Nano ZS90). Scanning electron microscopy (SEM) studies were carried out on a JEOL electron microscope (JSM-7100F, 15.0 kV). The white pigment properties (achromic ability and whiteness) were also tested.
3 Results and discussion
3.1 Influences of F value on structure and pigment properties
F value was changed by adding concentrated sulfate acid or sodium carbonate, adjusted to 1.53, 1.63, 1.73, 1.83, 1.93 and 2.03. The obtained metatitanic acids were denoted as AM1, AM2, AM3, AM4, AM5, AM6 and titania samples were denoted as AA1, AA2, AA3, AA4, AA5, AA6, respectively. The XRD patterns of the as-prepared metatitanic acid are shown in Fig.1.
All the XRD patterns of the samples are clearly consistent with anatase (JCPDS 21-1272), indicating the presence of anatase nanocrystalline in the metatitanic acid. As the F value increases, the intensity of the XRD peaks increases, suggesting that the crystalline of anatase is increased. Hydrolysis of hydrous titanium ion is a rapid reaction, but can be inhibited by acidity. Higher acidity may reduce the nucleation rate, and accelerate the crystal growth rate, leading to larger microcrystal, then aggregate smaller colloidal particle. The colloidal particle with high surface energy then aggregates into primary agglomerate (also called primary particle), and the formed agglomerate re-aggregate into the aggregate precipitation at last during the hydrolysis. The particle size and pigment properties of titania influenced by F value are shown in Table 1.
All the achromic ability of the calcined samples and the whiteness of the products are higher than those of PTA120 (the first grade anatase TiO2 pigment) when the F value higher than 1.73. Average particle size of titania with F value of 1.73 is the smallest. It can be seen that the suitable particle size is one of the key factors which can affect pigment properties. Sample AA3 shows the best achromic ability. While with the increase of F value, more sulfate ion may be absorbed into the precipitation, which may result in the forming of steric hindrance during calcinations to hinder the crystal growth, and then

Fig.1 XRD patterns of metatitanic acid influenced by F value
Table 1 Particle size and pigment properties of titania influenced by F value
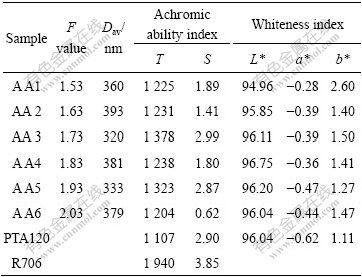
Note: Dav is the average particle size; T is tinting carbon strength; S is scattering carbon strength; L* is the lightness; a* is color value from blue to red; b* is color value from green to yellow.
cause larger particle forming. While with lower F value, low ion strength of the hydrolysis system causes the increase of effective concentration of TiO2+, resulting in secondary nucleation and wide particle size distribution, worsening the pigment properties. The suitable F value is from 1.73 to 1.93 in the experiments.
3.2 Influences of volume ratio of pre-adding water to TiOSO4 on structure and pigment properties
The volume ratios of pre-adding water to TiOSO4 solution are 0.20, 0.23, 0.25, 0.28, 0.30. The obtained metatitanic acid samples are denoted as BM1, BM2, BM3, BM4, BM5 and prepared titania samples are denoted as BA1, BA2, BA3, BA4, BA5, respectively. The XRD patterns of the as-prepared metatitanic acid and the calcined BA4 are shown in Fig.2 and Fig.3.
All the XRD patterns of the samples in Fig.2 are clearly consistent with anatase (JCPDS 21-1272), indicating the presence of anatase nanocrystalline in the metatitanic acid. The calcined sample BA4 shows entire
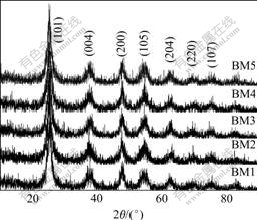
Fig.2 XRD patterns of metatitanic acid with different volume ratios of pre-adding water to TiOSO4
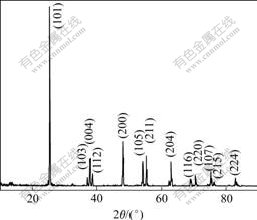
Fig.3 XRD pattern of calcined titania (BA4)
anantase phase with high crystalline (Fig.3). After calcination, the peaks become acuter and narrower, indicating the rapid crystal growth, and high temperature heat treatment accelerating crystal growth and decreasing the lattice strain and crystal defects.
Table 2 lists the particle size and pigment properties of the as-prepared samples. All the achromic ability of the calcined samples are higher than that of PTA120, and the whiteness of the products is higher than or close to that of PTA120. The volume ratio of pre-adding water to TiOSO4 is 0.28. The SEM image of sample BA4 is shown in Fig.4.
Table 2 Particle size and pigment properties of titania samples
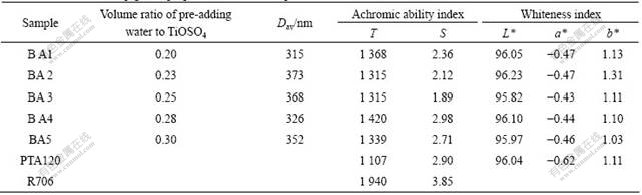

Fig.4 SEM image of sample BA4
The size of anatase titania particle arranges from 80 nm to 350 nm (Fig.4) and the average particle size is 326 nm. The smaller particle may congregates together to form the larger one.
It can be also observed that the dissolving-clear time increases as the ratio increases when the first added TiOSO4 solution causes the system milky white. The hydrolized Ti dissolves into the solution to form nuclei, the amount of nuclei increases with the increase of water content. The ratio affects the hydrolizing rate, the number of nuclei, the particle size distribution and crystal structure of titania. Because the hydrolysis of TiOSO4 solution are the essential step of sulfate process, and the number, activity and quality of the nuclei is the essential parameters in the hydrolysis process, suitable pre-adding water ratio is convenient to produce suitable nuclei and control the hydrolysis rate, resulting in better pigment properties. In order to obtain higher pigment property, the volume ratio of pre-adding water to TiOSO4 solution should be selected at 0.28?1.
3.3 Influences of heating rate on structure and pigment properties
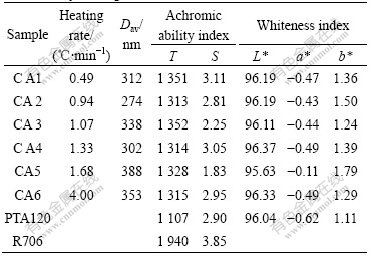
The heating rates are at 0.49, 0.94, 1.07, 1.33, 1.68, and 4.00 ℃/min. The prepared titania samples are denoted as CA1, CA2, CA3, CA4, CA5, CA6, respectively. Table 3 lists the particle size and pigment properties of the as-prepared samples.
Heating rate influences the hydrolysis rate of TiOSO4, namely the nucleation rate and crystal growth rate, as well as the aggregating rate. The Rietveld refined results show that when the heating rate is 1.07 ℃/min, the particle has suitable crystal size and the lattice strain is the smallest. All the samples with the heating rate range of 0.94-1.33 ℃/min have the narrowest particle size distribution. The achromic ability (T) of the samples is higher than 1 300, and their scattering carbon strength is close to that of PTA120, showing good pigment properties except sample CA5. Slow heating rate may induce slow crystal growth and hydrolysis rate, although with better crystal structure, easily inducing secondary nucleation due to its high supersaturation. While higher heating rate may increase crystal defects as hydrolysis rate is accelerated, also the collision frequency, resulting in wide particle size distribution. So, the heating rate should be kept at 0.94-1.33 ℃/min. And this is also consistent with other researchers’ reports[8].
Table 3 Particle size and pigment properties of titania influenced by heating rate
3.4 Influences of pH of pre-adding water on structure and pigment properties
The pH values of pre-adding water adjusted by sulfate acid and sodium hydroxide are 0, 1.0, 2.0, 4.0, 5.0, 10.0. The prepared titania samples are denoted as DA1, DA2, DA3, DA4, DA5, DA6, respectively. Table 4 lists the particle size and pigment properties of the as-prepared samples.
Table 4 Particle size and pigment properties of samples influenced by pH of pre-adding water
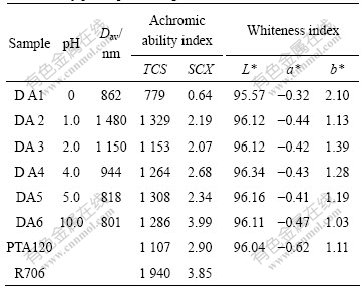
The pH of pre-adding water also influences the hydrolysis rate of TiOSO4, namely the nucleation rate and crystal growth rate, as well as the aggregating rate, just like the pre-adding water ratio. The average particle size of metatitanic acid increases firstly and then decrease. Higher pH brings the hydrolysis system with higher acidity, which can hinder the hydrolysis rate, forming less nuclei, resulting in smaller precipitation particle. With the pH rising, more nuclei formed at the beginning and lower acidity of the system will raise the hydrolysis velocity, leading to the precipitation particle size increasing. When pH is higher than 1.0, the precipitation particle size gradually decreases for that secondary nucleation is aggravated. All the pigment properties of the samples except DA1 are higher than those of PTA120. In order to obtain better particle size distribution and higher quality white pigment, the pH of pre-adding water should be kept at near-neutral range.
4 Conclusions
1) The factors, such as F value, pre-adding water volume ratio, heating rate and pH of pre-adding water, have great influences on the structure and pigment properties of the as-prepared samples produced by self-generated seeded hydrolysis process.
2) All the samples show anatase phase, and the calcined ones are with narrow particle size distribution and good pigment properties.
3) All these factors have great impacts on the hydrolysis velocity, nucleation rate, crystal growth rate, as well as particle size distribution, crystal structure and pigment properties of the samples.
4) The suitable condition are as follows: F value is 1.73-1.93; volume ratio of pre-adding water to TiOSO4 solution is 0.28?1; heating rate is 0.94-1.33 ℃/min; pH of the pre-adding water is near-neutral range.
References
[1] TANG Zhen-ning. Production and environmental treatment of titanium dioxide [M]. Beijing: Chem Ind Press, 2000. (in Chinese)
[2] ZHAO J H, WANG Z C, WANG L W. The preparation and mechanism studies of porous titania [J]. Materials Chemistry and Physics, 2000, 63: 9-12.
[3] RICHARDS R G, DUNCAN J F. Hydrolysis of titanium (Ⅳ) sulphate-solutions: Solution equilibria, kinetics and mechanism [J]. New Zealand Journal of Science, 1976, 19(2): 179-183.
[4] HIXSON W, RALPHE C. Hydrolysis of titanyl sulfate solutions [J]. Industrial and Engineering Chemistry, 1945, 37(7): 678-684.
[5] ZHAO Ming. Precipitation of hydrated titanium dioxide [J]. Coating Industry, 1982(3): 5-8. (in Chinese)
[6] BECKER H, KLEIN E, RECHMANN H. The growth of TiO2 hydrate during hydrolysis of Ti(SO4)2 solutions [J]. Chemical Engineering Journal, 1964, 70: 779-787.
[7] SANTACESATIA E. Kinetics of titanium dioxide precipition by thermal hydrolysis [J]. Journal of Colloid and Interface Science, 1986, 111(1): 45-53.
[8] SATHYAMOORTHY S, MOGGRIDGE G D, HOUNSLOW M J. Particle formation during anatase precipitation of seeded titanyl sulfate solution [J]. Crystal Growth & Design, 2001, 1(2): 123-129.
[9] HIDALGO M C, BAHNEMANN D. Highly photoactive supported TiO2 prepared by thermal hydrolysis of TiOSO4: Optimisation of the method and comparison with other synthetic routes [J]. Applied Catalysis B―Environmental, 2005, 61(3/4): 259-266.
[10] XIAO F, TSURU K, HAYAKAWA S, OSAKA A. Anatase/rutile dual layer deposition due to hydrolysis of titanium oxysulfate with hydrogen peroxide solution at low temperature [J]. Journal of Materials Science, 2007, 42(15): 6339-6346.
[11] QU Y D, LI X J, LI R Y, YAN H H, OUYANG X, WANG X H. Preparation and characterization of the TiO2 ultrafine particles by detonation method [J]. Materials Research Bulletin, 2008, 43(1): 97-103.
[12] HOUSKOVA V, STENGL V, BAKARDJIEVA S,MURAFA N, TYRPEKL V. Photocatalytic properties of Ru-doped titania prepared by homogeneous hydrolysis [J]. Central European Journal of Chemistry, 2009, 7(2): 259-266.
[13] TIAN Cong-xue, ZHANG Zhao. Synthesis of ordered mesoporous TiO2 from industrial titanyl sulfate solution and its formation mechanism [J]. Journal of Inorganic Materials, 2009, 24(2): 225-228. (in Chinese)
[14] GRZMIL B, GRELA D, KIC B, PODSIADLY M. The influence of admixtures on the course of hydrolysis of titanyl sulfate [J]. Polish Journal of Chemical Technology, 2008, 10(3): 4-12.
[15] GRZMIL B U, GRELA D, KIC B. Hydrolysis of titanium sulphate compounds [J]. Chemical Papers, 2008, 62(1): 18-25.
[16] GRZMIL B, GRELA D, KIC B. Formation of hydrated titanium dioxide from seeded titanyl sulphate solution [J]. Chemical Papers, 2009, 63(2): 217-225.
(Edited by ZHAO Jun)
Foundation item: Project (YK08T02) supported by Pangang Group
Corresponding author: TIAN Cong-xue; Tel: +86-812-3380578; E-mail: tcx7311@163.com

