Preparation and characterization of high salts polymer electrolyte based on poly(lithium acrylate)
TANG Ai-dong(�ư���), HUANG Ke-long(�ƿ���), PAN Chun-yue(�˴�Ծ), LU Cui-hong(¬���)
(School of Chemistry and Chemical Engineering, Central South University, Changsha 410083,China)
Abstract: Novel polymer electrolytes were prepared by highly mixing poly(lithium acrylate)(PPALi) with eutectic lithium salts of lithium acetate and lithium nitrate. Poly(lithium acrylate) was preparaed by inverse emulsion polymerization from crylic acid and LiOH. Phase transition temperatures were measured for all the eutectic lithium of binary system samples as a function of the concentration of Li(CH3COO), and the mixtures exhibit the lowest phase transition temperatures of (448��2)K at about 50% (mass fraction) Li(CH3COO). Thermogravimetry(TG) and X-ray diffraction(XRD) analysis indicate the formation of a novel polymer-salt complex. The highest conductivity (approximately 4.97��10-5S��cm-1) is found at room temperature with the electrolyte composition of eutectic mixture of about 80% (mass fraction), poly(lithium acrylate)20% under quickly cooling condition, which is 150% higher than that under natural cooling condition.
Key words: polymer electrolyte; thermal stability; ionic conductivity CLC number: O631.3
Document code: A
1 INTRODUCTION
Worldwide research and efforts are currently underway to fabricate all-solid-state, rechargeable Li and Li-ion batteries utilizing Li+-conductive polymer electrolytes[1]. Solid polymer electrolytes possesses many advantages including high ionic conductivity, high specific surface energy, solvent free, wide electrochemical stability windows, light and easy processability[2]. Apart from polyethylene oxide (PEO)[3], poly(vinyl alcohol)(PAV)[4], poly(tetramethylene glycol)(PTG)[5], poly(vinyl difluoride)(PVDF)[6], poly(trimethylene carbonate(PTMC)[7] and poly (acrylonitrile) (PAN)[8], poly(lithium acrylate) (PPALi) has also been used as the polymer host material. In most solid polymer electrolytes, the polymer host is doped with inorganic salts and one or more plasticizers in order to improve the conductivity. The conductivity is highly related to the species of polymer and additive salts. The main purpose of the present work is to discuss the effects of the extremely salt-riched concentration and different cooling mode on ionic conductivity of polymer electrolyte, and also to determine the structure of novel polymer-in-salt mixture based on poly(lithium acrylate). Although a certain amount of general information is available on the thermal behavior of the nitrate and acetate[9], we have also re-measured these salts for the sake of completeness in order to make comparisons with the other investigated substances measured under the same experimental conditions.
2 EXPERIMENTAL
2.1 Preparation of binary lithium salts sample
All reagents used in this experiment were analytical grade. The Li(CH3COO) and LiNO3 were dried to constant mass at 150��, and stored over silica gel in desiccator before used. The binary lithium salts were prepared by mixing appropriate amounts of Li(CH3COO) and LiNO3, the obtained binary lithium salts were milled in an agate mortar to form fine powders. The samples were done in quartz crucible to melt (no chemical reaction between the quartz and lithium salts was observed) and stored over silica gel in desiccator until used for further experiments.
2.2 Preparation of poly(lithium acrylate)(PLA)
50mL acrylic acid monopolymer solution (0.02mol/mL) was slowly added to 100mL LiOH solution, stirring at 40�� for 30min, the solution was then heated at 70�� for 1h. After adding 100mL NH4S2O4 (0.1mol/mL) and methyl alcohol, the precipitate, poly(lithium acrylate)(PLA) was obtained by filtered and washed three times by methyl alcohol, the product was dried in vacuum at 90�� for 48h.
2.3 Preparation of polymer electrolyte
Samples of lithium nitrate and lithium acetate were dried at 90�� for 4h before used. After thoroughly mixing PLA, LiNO3, LiOAc in the desired proportions at proper temperature, polymer electrolyte films were prepared by quick cooling in liquid nitrogen or natural cooling with oven to ambient temperature, then stored over silica gel in desiccators for use.
2.4 Thermal analysis
Thermal analysis of the polymer electrolyte film and lithium salts were carried out using a WRT-3P Micro-Thermalbalance and SCRKZ220-5 Differential Thermal Analysis (DTA) apparatus in a quiescent air atmosphere at heating rate of 8��/min, with ��-Al2O3 used as a reference material. Time, temperature and temperature differences were recorded simultaneously during the experiment.
2.5 Conductivity measurement
The total ionic conductivities at ambient temperature were obtained in a sandwich format cell (INOX/electrolyte/INOX) by electrochemical impedance spectroscopy using an electrochemical working station (CHI660A, made in china) in 0.01Hz-100kHz.
2.6 X-ray diffraction (XRD)
The structure of the materials were characterized using the X-ray power diffraction (SIMENS-D500 XRD) using Cu K�� irradiation (��=1.5406�@) at 50kV at room temperature. The data were collected at rate of 0.5(��)/min in the diffraction angle range of 8�� to 47��.
3 RESULTS AND DISCUSSION
3.1 FTIR studies
The IR spectra of acrylate lithium and poly(lithium acrylate) are shown in Fig.1.
The vibrational bands at 3405cm-1 and 1440cm-1 in Fig.1(a) were assigned to C��H asymmetric stretching and bending vibrations of acrylate lithium[10] , respectively. The frequencies 1645 and 996cm-1 were due to C-C stretching and bending vibrations of acrylate lithium in Fig.1(a). The peaks at 1572, 1278 and 845cm-1 may be assigned to stretching vibrations of C-O, C��O and symmetrical stretching of C��O��C of acrylate lithium and poly(lithium acrylate). The absorption peak at 640cm-1 in Fig.1(b) may be attributed to the amorphous poly(lithium acrylate). The spectra shows that there is no absorption bands of C-C at 1645 and 996cm-1, indicating no existence of C-C in PPALi.

Fig.1 IR spectra of acrylate lithium(a) and poly(lithium acrylate)(b)
3.2 DTA analysis and phase diagram of binary Li(CH3COO)-LiNO3 system
Based on DTA curves for all the compositions during the heating process, the phase diagram of Li (CH3COO)- LiNO3 system is shown in Fig.2. The change of phase state is answer for the law of binary simple eutectic mixture. Li (CH3COO) and LiNO3 formed the eutectic mixture at about 50% Li (CH3COO) with the melting temperature of (175��2)��. The melting point reduces about 77-112�� compared with the melting point of single composition.

Fig.2 Phase diagram of Li (CH3COO)-LiNO3 system
The conductivities at room temperature for various Li(CH3COO) concentration of binary system are shown in Fig.3. The lowest conductivity of LiNO3-Li (CH3COO) mixtures is observed when the content of Li(CH3COO) increases to 50%.

Fig.3 Plot of lg�� versus concentration of Li(CH3COO) at room temperature
3.3 TG analysis of ��CH2CHOOLi��n, polymer electrolyte and lithium salts
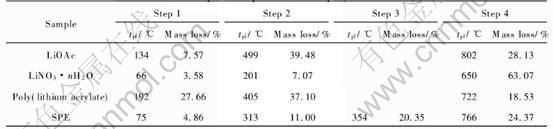
Thermal characterization of polymer electrolyte and various salts are shown in Table 1. The sample of ��CH2CHOOLi��n starts to lose mass below 200��. This initial mass loss results from residual solvent evaporation. The second mass loss happens around 405��. This may be due to the decomposition of sample. The third mass loss begins around 722��, which can be attributed to the decomposition of the residual.
The TG data of poly(lithium acrylate)/salts complex shows that mass loss of 4.86% exists below 100��, which may be due to the absorbed moisture. The polymer electrolyte decomposes around 313�� with mass loss of 11%, and around 354�� just 20.35%. The final mass loss of 24.37% around 766�� results from decomposition of the residual polymer electrolyte.
The anhydrous lithium acetate shows a theoretical mass loss of 39.24%, expected in the temperature from 358��. Corresponding to decomposition of Li(CH3COO), the result in in excellent agreement with the experimental value of 39.48%. At 802��, decomposition with the generation of Li2CO3 takes place. A mass loss of 28.46% is expected (observed mass loss, 28.13%). The thermolysis of LiNO3��nH2O is rather simple. Water is given off from 65�� to 201�� (experimental mass loss 10.57%, calculated for loss of 0.45 mol water, 10.50%). A mass loss of 63.00% is expected (mass loss observed, 63.07%). The results of thermal behavior of Li(CH3COO)-LiNO3, PLA and SPE show that polymer/salts complex was formed after poly(lithium acrylate) dissolving in lithium salts because of no single thermal characterization of raw material (Li(CH3COO)-, LiNO3 and PLA).
The presence of some extent of water content was characterized by TG of about 1%-5%, even after careful drying procedures in the range of the salt studied. This water content could give an additional contribution to the dissolution and thus increases the conductivities. So, polymer electrolytes can be obtained by dissolving the appropriate polymers and lithium salts in solvent, then evaporating the solvent and drying of the samples at high vacuum.
3.4 XRD
Fig.4 shows XRD patterns of polymer/salts complexes under different cooling conditions.

Fig.4 X-ray diffraction spectrum of complex with poly(lithium acrylate) (20)-[Li(CH3COO)-LiNO3] (80) under natural cooling(a) and quick cooling(b)
Table 1 Thermal decomposition parameters of polymer electrolyte and salts
There are peaks corresponding to the pure Li(CH3COO) and LiNO3 appeared in the complex which indicates the incomplete dissolution of high concentration salt in poly(lithium acrylate) matrix.
There was a decrease in the relative intensity of the peaks under quickly cooling condition. This result can be interpreted by considering the Hodge et al��s Criterion that establishes a correlation between the height of the peak and the degree of crystallinity[11]. The X-ray diffraction analysis reveals that the quickly cooling sample consisting of [Li(CH3COO) (40)-LiNO3 (40) ]-poly(lithium acrylate) (20) shows less crystallinity compared to the natural cooling sample[10].
3.5 Ionic conductivity
The typical impedance plot (Z�� and Z��) for the electrolyte composition LiNO3(40)-LiCH3COO (40)-poly(lithium acrylate)(20) is shown in Fig.5. In the impedance response behaviour, the disappearance of the high frequency semicircular portion leads to a conclusion that the current carriers are ions and this leads one to further conclude that the total conductivity is mainly the result of ion conduction[5]. The conductivity was calculated according to the value of R in the complex impedance polt and the known area and thickness of the SPE film. The room temperature conductivity of the quickly cooled sample is 4.97��10-5S��cm-1, which is 150% higher than that obtained under natural cooling condition[10].
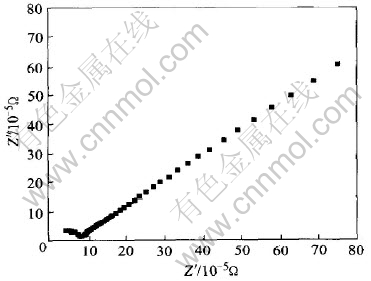
Fig.5 Impedance diagram of LiNO3(40)-LiCH3COO(40)-poly(lithium acrylate)(20) polymer complex at room temperature under quick cooling condition
4 CONCLUSIONS
The phase diagram of binary Li(CH3COO)-LiNO3 system is suggested, there is a eutectic at (175��2)�� and with 50% Li(CH3COO), so the highest conductivity (approximately 4.97��10-5 S��cm-1) is found at room temperature with LiNO3(40)-LiCH3COO (40)-poly(lithium acrylate)(20) polymer complex under quick cooling condition, which is 150% higher than that obtained under natural cooling condition.
REFERENCES
[1]Alamgir M, Abraham K M. Room temperature rechargeable polymer electrolyte batteries[J]. J Power Sources, 1995, 54(11): 40-45.
[2]Ramesh S, Arof K. Structural,thermal and electrochemical cell characteristics of poly(vinyl chloride)-based polymer electrolytes[J]. J of Power Source, 2001, 99(1-2): 41-47.
[3]Hayamizu K, Aihara Y, Araietal S. Diffusion, conductivity and DSC studies of a polymer gel composed of cross-linked PEO/��-BL and LiBF4[J]. Solid State Ionics, 1998, 107(1-2): 1-12.
[4]Rhoo H J, Kim H T, Park J K, et al. Time- and temperature- dependent changes of the in situ conductivity of polyaniline and polyindoline [J]. Electrochimica Acta, 1997, 40(2): 213-219.
[5]Furtado A C, Silva G G, Pimenta A M, et al. Conductivities, thermal properties and Raman studies of poly(tetramethylene glycol) based polymer electrolytes[J]. Electrochimica Acta, 1998, 43(10-11): 1477-1480.
[6]Core F, Appetecchi G B, Slane S, et al. Impedance and lithium-7NMR studies of polymer electrolytes based on PVDF[J]. Solid State Ionics, 1996, 86-88: 307-312.
[7]Michael J S, Maria M S, Sandra C, et al. Preparation and characterization of a lithium ion conducting electrolyte based on poly(trimethylene carbonate)[J]. Solid State Ionics, 2001, 140(3-4): 345-351.
[8]Hlaoyoki A, Kochi S. Electrochemical properties of polymer electrolytes based on PAN[J]. Industry Materials, 1999, 47(2): 56-59.(in Japanese)
[9]Tobon-Zapata E G, Eerrer F G, Etcheverry B S, et al. Thermal behaviour of pharmacologically active lithium compounds[J]. J of Thermal Analysis and Calorimetry, 2000, 61: 29-35.
[10]Tang A D, Pan C Y, Zhao X N, et al. Preparation and characterization of new solid electrolyte LiNO3-LiOOCCH3/poly(lithium acrylate)[J]. Acta Polymerica Sinica, 2002, 5: 636-639.
[11]Hodge R M, Edward G H, Simon G P. Water absorption and states of water in semicrystalline poly(vinyl alcohol) films [J]. Polymer, 1996, 37(8): 1371-1376.
(Edited by LONG Huai-zhong)
Received date: 2003-10-08; Accepted date: 2004-12-24
Correspondence:PAN Chun-yue, Professor; +86-731-8876099; E-mail: panchunque@sina.com