Synthesis and characterization of red-emission PPV copolymers containing fluorenone unit
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2010���2��
�������ߣ���ӦƼ �˴�Ծ ���� ���� ��Ծ�� �ܿƳ� �ܵt ������
����ҳ�룺269 - 276
Key words��conjugated polymers; fluorenone; photophysical properties; red-emission polymers; electron affinity
Abstract: Two soluble copolymers of fluorenone and dioctoxylbenzene (PFN) or anthracene (PFNAn) were synthesized through Heck polymerization, and were characterized by gel permeation chromatography (GPC), FT-IR, 1H-NMR, elemental analysis and thermogravimetric analysis. The polymers possess good solubility in common organic solvents and high thermal stability with the onset decomposition temperature at higher than 410 ��. The photophysical properties of the polymers were investigated in both solutions and spin-coated films. Cyclic voltammetry results revealed that the copolymers possess higher electron affinity and reversible reduction/re-oxidation processes. Their electroluminescent properties were further investigated. PFN and PFNAn show stable and saturated red light emission with high thermal stability and high electron injection ability. This type of conjugated polymers may be promising for the applications as electron acceptors in polymer photovoltaic cells and electron transporting materials.
������Ϣ��the National Natural Science Foundation of China
J. Cent. South Univ. Technol. (2010) 17: 269-276
DOI: 10.1007/s11771-010-0041-3 ![]()
ZOU Ying-ping(��ӦƼ)1, PAN Chun-yue(�˴�Ծ)1 , ZHANG Zhen-hua(����)1, LIU Bo(����)1,
HE Yue-hui(��Ծ��)2, ZHOU Ke-chao(�ܿƳ�)2, ZHOU Yi(�ܵt)3, LI Yong-fang(������)3
1. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China;
2. State Key Laboratory of Powder Metallurgy, Central South University, Changsha 410083, China;
3. Key Laboratory of Organic Solids, Institute of Chemistry, Chinese Academy of Sciences,
Beijing 100190, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2010
Abstract: Two soluble copolymers of fluorenone and dioctoxylbenzene (PFN) or anthracene (PFNAn) were synthesized through Heck polymerization, and were characterized by gel permeation chromatography (GPC), FT-IR, 1H-NMR, elemental analysis and thermogravimetric analysis. The polymers possess good solubility in common organic solvents and high thermal stability with the onset decomposition temperature at higher than 410 ��. The photophysical properties of the polymers were investigated in both solutions and spin-coated films. Cyclic voltammetry results revealed that the copolymers possess higher electron affinity and reversible reduction/re-oxidation processes. Their electroluminescent properties were further investigated. PFN and PFNAn show stable and saturated red light emission with high thermal stability and high electron injection ability. This type of conjugated polymers may be promising for the applications as electron acceptors in polymer photovoltaic cells and electron transporting materials.
Key words: conjugated polymers; fluorenone; photophysical properties; red-emission polymers; electron affinity
1 Introduction
The field of polymer optoelectronics has witnessed a great deal of progress since the discovery of the conducting properties of polyacetylene in 1977 [1]. Conjugated polymers can be used as the active materials in optoelectronic devices such as organic light emitting diodes (OLEDs), thin film transistors (TFTs), photovoltaic cells and ion detection [2-14]. Red-, green-, and blue-emitting organic and polymer materials have attracted much attention in the last two decades since the reports of Kodak group [15] and the Cambridge group [16] on OLED and polymer light-emitting diode (PLED).
Recently, great efforts have been devoted to charge-transfer (CT) type polymers containing an electron-accepting unit and an electron-donating unit [17]. It is expected that the polymers or oligomers with the CT structure are useful in optic-electronic devices [18-19] such as PLEDs [20], photovoltaic cells [21-23], frequency doublers and high speed modulators, because the conjugated polymers with such a CT structure or p-stacked structure usually show good properties such as low band gap values and good nonlinear optical properties[24].
For the further development of the CT type polymers, studies on the polymers with various electron- accepting and electron-donating units should shed some light on the relationship between the chemical structures and chemical properties of the polymers. Among the electron accepting units, fluorenone unit is very attractive for good electron-injection ability [25]. Combination of poly(p-phenylenevinylene) (PPV) and fluorenone unit to a polymer main chain may obtain the polymers with both hole transfer ability and electron injection ability; such polymers may have some special applications in optic-electrical fields. Although there have been many reports about PPV derivatives, no reports about the CT type fluorenone-containing alternating PPV copolymers were found in literatures.
Herein, we introduced dioctoxyloxylbenzene, anthracene moiety which is known to have such unique properties of suppressing this excimer/aggregate emission [26] and fluorenone unit into the backbone to obtain two copolymers (PFN, PFNAn), respectively. The polymers possess high thermal stabilities, high electron affinity, which are potential as electron transporting materials (ETM) and hole-blockers, and also as n-type materials for photovoltaic applications [27-28]. In the contribution, we presented the optical, thermal, electro- chemical, electroluminescence properties of the polymers. Interestingly, to our best knowledge, this is seldom for the polymers containing fluorenone unit in the main chain to emit red light. The above results are firmly believed to a better understanding of the relationship between the chemical structures and properties of the new polymers.
2 Experimental
2.1 Materials
Pd(AcO)2 and P(o-tri-o-tolylphosphine)3 were obtained from Aldrich Chemical Co. Inc. (USA) without further purification. N, N-Dimethyl-formamide (DMF) and triethylamine (TEA) were dried over Na/benzophenone ketyl and freshly distilled prior to use. The other materials were common commercial level and used as received.
2.2 Measurements
1H NMR spectra were recorded using a Bruker DMX-300 spectrometer, and chemical shifts were recorded. Elemental analysis was carried out on a Flash EA 1112 elemental analyzer. MALDI-TOF mass spectrometry was performed on Bruker Biflex MALDI-TOF. Fourier transform infrared (FT-IR) spectra were recorded on a BIO FTS-135 spectrometer by dispersing samples in KBr disks. The relative molecular masses and polydispersities of polymers were determined by gel permeation chromatography (GPC) analysis relative to polystyrene calibration (Waters 515 HPLC pump, a Waters 2414 differential refractometer, and three Waters Styragel columns (HT2, HT3, and HT4)) using THF as eluent at a flow rate of 1.0 mL/min at 35 ��. Thermogravimetric analysis (TGA) was conducted on a Perkin-Elmer 7 thermogravimetric analyzer at a heating rate of 20 ��/min under a nitrogen atmosphere. The UV-Vis spectra were recorded on a Hitachi UV-3010 spectrometer. The photoluminescence (PL) and electroluminescence (EL) spectra were obtained with a Hitachi F-4500 fluorescence spectrophotometer.
The cyclic voltammograms were recorded with a computer controlled Zahner IM6e electrochemical workstation (Germany) using polymer films on Pt plate as the working electrode, Pt wire as the counter electrode and Ag/Ag+ (0.1 mol/L) as the reference electrode.
The current density�Cvoltage (J-V) and luminance- voltage (L-V) characteristics of the light-emitting devices were recorded with a computer-controlled Keithley (USA) 236 source measure unit and a Keithley 2000 Multimeter coupled with a Si photomultiplier tube. All the measurements of device were performed under ambient atmosphere at room temperature.
2.3 Synthesis of monomers and polymers
Synthetic route of the monomers and polymers are shown in Scheme 1.
(1) Synthesis of 2, 5-dioctoxy benzene (compound 1)
To 100 mL dimethyl sulfone, potassium hydroxide (28 g, 0.5 mol) was added with violently stirring for 0.5 h under nitrogen. 16.53 g hydroquinone and 1.7 g cetyl trimethyl ammonium bromide (CTAB) as the phase transfer catalyst were added while stirring, then n-octyl bromide (83 g, 0.43 mol) was added dropwisely to the mixture. The reaction proceeded for 7 h. After the mixture was poured into plenty of distilled water, there was light-yellow solid in the upper layer. The crude product was recrystallized by isopropyl alcohol after filtration, which was washed with distilled water, 10% sodium hydroxide aqueous solution, and then dried under vacuum. The white sheet-like product was afforded. Yield: 93%, melting point (mp): 44.5-45.2 ��.
FT-IR (KBr, cm-1): 1 045 (Ar��O��), 933 (C��O��C).
1H NMR (CDCl3): chemical shift ��=6.81 (s, 4H), 3.88-3.91 (t, 4H), 1.72-1.77 (m, 8H), 1.28-1.43 (t, 16H), 0.88-0.90 (t, 6H).
(2) Synthesis of 1, 4-bis(bromomethyl)-2,5- octyloxylbenzene (compound 2) [29-30]
A mixture of 2, 5-dioctoxy benzene (15.03 g, 45 mmol), paraformaldehyde (4.7 g, 157 mmol), sodium bromide (15.8 g, 0.153 mol), glacial acetic acid (85 mL) was added in a flask under stirring for 1 h, then concentrated sulfuric acid (8.3 mL, 148 mmol) and isochoric glacial acetic acid were dropped into the flask within 1 h. The system was stirred at room temperature for 1 h. After that it was heated to 60 �� for 7 h. After being cooled to the room temperature, the mixture was poured into 500 mL of distilled water, filtered, washed with water. The crude product was recrystallized from petroleum ether and dried under vacuum to give white powder with 78.1% yield, and mp of 82-83 ��.
FT-IR (KBr, cm-1): 1 665 (��, CH2), 1 042 (Ar�� O��), 931 (C��O��C), 865 (��, ��CH2��).
1H NMR (CDCl3): ��=7.00 (s, 2H, Ar��H), 4.52 (s, 4H), 3.97-4.00 (t, 4H), 1.80-1.83 (m, 4H), 1.49-1.54 (m, 4H), 1.32-1.35 (m, 16H), 0.87-0.91 (t, 6H).
(3) Synthesis of 2, 5-dioctoxyl-1, 4-xylene-bis (triphenyl phosphonium bromine) (compound 3) [31-32]
A mixture of compound 2 (2.6 g, 5 mmol), triphenyl phosphine (3.93 g, 15 mmol) and 10 mL of freshly distilled DMF was heated to 95 ��, and the reaction proceeded under the temperature while stirring for 24 h. The resulting mixture was poured into diethyl ether. The white solid was filtered and washed with diethyl ether repeatedly to remove the by-products, dried under vacuum to afford a white powder. Yield: 91%, mp�� 200 ��.
FT-IR (KBr, cm-1): 535(P��Br).
1H NMR (CDCl3): ��=7.92-7.69 (m, 30H), 7.11 (s, 2H), 5.41-5.38 (d, 4H), 3.18-3.15 (t, 4H), 2.05 (m, 6H), 1.30-1.11 (m, 18H), 0.85-0.83 (t, 6H).
(4) Synthesis of 2,5-dioctyloxy-1,4-divinyl-benzene (compound 4)
A mixture of compound 3 (1.93 g, 1.85 mmol), 27 mL of methylene chloride and 7.2 mL of formalin solution was added into 100 mL flask, then 20% sodium hydroxide aqueous solution was slowly dropped into the flask at 0 �� for 1 h. The reaction proceeded at 25 �� for 24 h. Pouring the reaction mixture into 50 mL of water, the mixture was extracted by methylene chloride, the organic layer was collected, dried with anhydrous MgSO4, and the solvent was removed through rotary evaporator. The crude product was recrystallized twice from 20% ethanol to give 0.71 g of white crystal. Yield: 54%, mp: 42-43 ��.
FT-IR (KBr, cm-1): 962 (trans��HC=CH2).
1H NMR (CDCl3): ��=7.11 (s, 2H), 5.75-5.71 (d, 4H), 5.26-5.24 (d, 2H), 3.99-3.96 (m, 4H), 1.82-1.76 (m, 4H), 1.57-1.31 (m, 20H), 0.91-0.89 (t, 6H).
(5) Synthesis of 2, 7-dibromoflurenone (compound 5)
CrO3 (2.3 g, 23 mmol) was added to a suspension of 2,7-dibromofluorene (3 g, 9.27 mmol) in 45 mL of acetic anhydride under stirring at room temperature. The reaction mixture was stirred for 4 h, and then put into a 100 mL of 2% HCl solution. The suspension was filtered and washed with cold water, and the product was recrystallized from iso-propanol to provide 3 g of the title product as yellow solid with the yield of 95% and mp of 203-204 ��.
Mass spectrometry (MS): 338.
1H NMR (CDCl3): ��=7.76 (d, 2H), 7.63 (q, 2H), 7.61 (dd, 2H), 7.37 (d, 2H).
(6) Synthesis of 9, 9��-dibromoanthracene (compound 6)
After bromine (8.95 g, 60 mmol) was slowly dropped into the mixture of anthracene (5 g, 28 mmol) in 150 mL glacial acetic acid within 0.5 h, yellow solid appeared. The reaction stirred overnight, after filtration, washed for several times with water, bright-yellow solid was obtained with a yield of 88%.
MS: 324.
1H NMR (CDCl3): ��=8.61-8.50 (d, 4H), 7.65-7.50 (m, 4H).
(7) Synthesis of PFN
The polymer was prepared by the Heck coupling reaction from compounds 5 and 4. A mixture of compounds 5 (338 mg, 1 mmol), 4 (386 mg, 1 mmol), Pd(OAc)2 (9 mg, 0.04 mmol), tri-o-tolylphosphine (71 mg, 0.23 mmol), and 1 mL triethylamine in 15 mL of DMF was stirred at 140 �� for 24 h under inert atmosphere. After being cooled to room temperature, the reaction mixture was poured into 50 mL of methanol and then filtered through a Soxhlet thimble, which was then subjected to Soxhlet extraction with methanol, hexane, and CHCl3. The polymer was recovered from the CHCl3 fraction by rotary evaporation. The target product was dried thoroughly in vacuum for 1 d to give a dark red solid with a yield of 71%.
1H NMR (CDCl3): ��=7.8 (s, 1H), 7.66-7.42 (br, 6H), 7.20-7.11 (br, 5H), 4.08 (br, 4H), 1.91-1.03 (br, 20 H), 0.89 (s, 6H).
Analytics calculated (Anal. Calcd). For (C39H42O3)n: C 83.87, H 8.42; Found: C 82.15, H 8.32.
GPC (THF): Mw: 12 037, Mn: 9 120.
UV-Vis(CHCl3): ��max=450 nm
FL(CHCl3): ��max=644 nm.
(8) Synthesis of PFNAn
A mixture of compounds 5 ( 85 mg, 0.25 mmol), 6 (252 mg, 0.75 mmol), 4 (386 mg,1 mmol), Pd(OAc)2 (9 mg, 0.04 mmol), tri-o-tolylphosphine (71 mg, 0.23 mmol), and 1 mL triethylamine in 15 mL of DMF was stirred at 140 �� for 24 h under inert atmosphere. After being cooled to room temperature, the reaction mixture was poured into 50 mL of methanol and filtered through a Soxhlet thimble, which was then subjected to Soxhlet extraction with methanol, hexane, and CHCl3. The polymer was recovered from the CHCl3 fraction by rotary evaporation. The final polymer was dried thoroughly in vacuum for 1 d to give a dark red solid with a yield of 68%.
1H NMR (CDCl3): ��=8.36-8.31 (br, 3H), 7.88 (br, 2H), 7.36-7.34 (m, 7H), 7.21-7.10 (br, 1.5H), 4.00-3.93 (br, 4H), 1.86-1.20 (br, 20H), 0.88 ( br, 6H).
Analytics calculated (Anal. Calcd). For (C37.75H46.5O2.25)n: C 84.60, H 8.68; Found: C 83.10, H, 8.38.
GPC(THF): Mw: 14 570, Mn: 8 650.
UV-Vis(CHCl3): ��max=430 nm
FL(CHCl3): ��max=531 nm, 630 nm (shoulder).
3 Results and discussion
3.1 Synthesis and characterization
The polymers were synthesized by a Heck cross-coupling reaction as shown in Scheme 1. 1H NMR results confirmed the structure of the polymers.

Scheme 1 Synthetic route of PFN and PFNAn
The thermal stability of the polymers was evaluated by thermogravimetric analysis (TGA), as shown in Fig.1. As reflected by the results of TGA, the onset degradation temperature was 411 �� for PFN and 410 �� for PFNAn, in a nitrogen atmosphere with a heating rate of 10 ��/min. Hence, the thermal stability of the polymers is enough for the applications in optoelectronic devices.
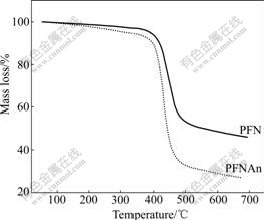
Fig.1 TGA of PFN and PFNAn at heating rate of 10 ��/min
3.2 Optical properties
The photophysical properties of PFN and PFNAn were measured by UV-Vis and fluorescence spectroscopy in chloroform solutions and thin films on quartz plates. The results are shown in Fig.2. PFN and PFNAn in chloroform solution exhibited the maximum peaks at about 420 and 430 nm, respectively, which were corresponding to �ШC��* transition. The shoulder peak at about 300 nm was mostly attributed to the absorption of alkyloxyl moiety. PFN and PFNAn emitted red and green light in dilute chloroform solutions, respectively. As shown in Fig.2(a), the photoluminescence (PL) spectra of PFN were regular and symmetrical, and the maximum emission of PFN in dilute chloroform solution was observed at about 644 nm, while PPFNAn exhibited the maximum emission at 527 nm, and a shoulder peak at 625 nm, which is due to the intermolecular charge transfer state.
Fig.2(b) shows the optical absorption and PL spectra of PFN and PFNAn in solid state. The absorption spectra of the polymers in solid state were a little red-shifted and similar to those in chloroform solutions, and the maximum absorption of PFN and PFNAn corresponding to �ШC��* transition is 428 and 445 nm, respectively. The onset positions of the UV-Vis absorption band of PFN and PFNAn appeared at longer wavelength position than those of the copolymers in solutions. Optical band gaps (Eg) determined from the absorption edge of the solid-state spectra of PFN and PFNAn were found to be 1.94 and 1.98 eV. The PL spectra of PFN in solid state were almost identical to those in dilute chloroform solution, and the maximum emission peak of PFN was observed at about 633 nm. Intrestingly, compared to PL spectra in the solutions, the thin film PL spectra of the polymer hypsochromic shift, which suggests that the sterical hindreded polymers are highly twisted, which maybe concerns with the state of aggregates. The reason needs to be investigated further. For PFNAn, the solution showed two peaks at about 527 and 625 nm, while in the film state, it exhibited a maximum peak at 620 nm, which may be the complete energy transfer from the unit of anthracene to the unit of fluorenone. The optical and photoluminescence properties of the polymers are summarized in Table 1.
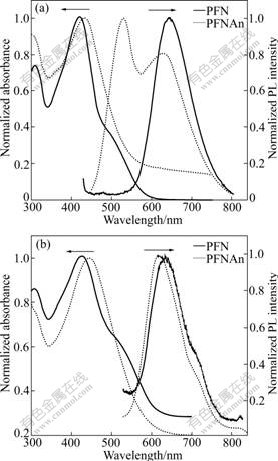
Fig.2 UV-Vis absorption and PL spectra of PFN and PFNAn: (a) In chloroform solution; (b) In film on quartz plate
Table 1 Optical and photoluminescence properties of polymers

3.3 Electrochemical properties
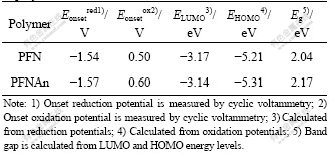
In order to gain information on the charge injection, cyclic voltammetry (CV) was employed to estimate the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) energy levels of the polymers [33]. Fig.3 shows the oxidation of the polymers started at 0.50 V for PFN and 0.60 V for PFNAn (vs Ag/Ag+), indicating that the oxidation potentials of these copolymers positively shifted in comparison with those of MEH-PPV (0.41 V) and P3HT (0.05 V) measured under the same conditions. On sweeping the polymers cathodically, polymers exhibit reversible n-doping/dedoping process, the onset potentials of the n-doping process for PFN and PFNAn occurred at -1.54 and -1.57 V, respectively. The reduction potentials of these copolymers also positively shifted in comparison with those of MEH-PPV (-2.01 V) and P3HT (-2.16 V), which suggests that the copolymers possess higher electron affinity. Then, we can get the HOMO and LUMO energy levels of the copolymers according to the equations [34]: EHOMO=-e(Eonox+4.71) (eV); ELUMO=-e(Eonred+4.71) (eV). The HOMO energy levels of PFN and PFNAn were estimated to be -5.21 and -5.31 eV, and the LUMO energy levels of PFN and PFNAn were estimated to be -3.17 and -3.14 eV, respectively.
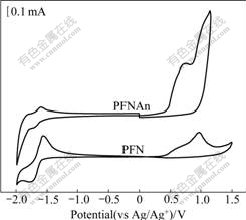
Fig.3 Cyclic voltammograms of PFN and PFNAn films on platinum plates in acetonitrile solution of 0.1 mol/L Bu4NPF6 (Bu=butyl) at sweep rate of 50 mV/s
From the onset potentials of the oxidation and reduction processes, the band gaps of the PFN and PFNAn were estimated to be 2.04 and 2.17 eV, respectively. The electrochemical bandgap EgEC is somewhat higher than the corresponding optical bandgap Egopt, but within the range of error (0.2-0.5 eV). The electrochemical data of the copolymers are summarized in Table 2. It is obvious that the nature of the fluorenone unit can help to regulate the LUMO and HOMO energy levels of the materials to move downwards.
Table 2 Electrochemical potentials and energy levels of copolymers
3.4 Electroluminescence properties of PLED devices
In order to investigate the electroluminescent properties of the polymers, the preliminary single-layer polymer light-emitting diodes (PLEDs) with the device structure of ITO/PEDOT/polymer/Ca/Al were fabricated and characterized. It was observed that PFN and PFNAn emitted stable red and saturated red light, respectively. As shown in the spectra, PFN and PFNAn gave an emission maximum located at 640 and 621 nm. Compared to those of PL, the EL spectra of PFN and PFNAn exhibited the similar emission, as shown in Fig.4, which indicates that the same excitation mechanisms are involved in both cases.
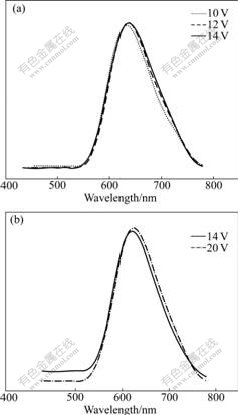
Fig.4 EL spectra of ITO/PEDOT /polymer/Ca/Al devices: (a) PFN; (b) PFNAn
Fig.5 shows current density�Cvoltage (J-V) and luminance�Cvoltage (L-V) characteristics of the single- layer PLED devices. The current density increased exponentially with increasing forward bias voltage, which was a typical diode characteristic. The turn-on voltage of the PLEDs based on PFN and PFNAn was approximately 8.5 and 11.0 V, respectively. The maximum luminance of single-layer PLEDs based on PFN and PFNAn was about 174 cd/m2 at 17.2 V, and 98 cd/m2 at 17.0 V, respectively. As shown in Fig.6, the maximum EL efficiency of the devices based on PFN and PFNAn was 0.067 and 0.01 cd/A, respectively. Lower EL efficiency and higher turn-on voltage were observed for PFNAn. The phenomena can be attributed to the mismatch in energy levels with that of the PEDOT hole transporting material(HOMO level 5.0 eV) and with the work function of the Ca cathode (about 2.9 eV). Further improvement in device performance can be expected through the optimization of device configuration, as well as the optimization of the effective conjugation length of the polymers. The optimization of device configurations is in progress.
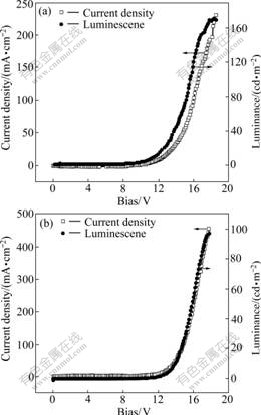
Fig.5 Current density�Cvoltage (J-V) and luminance�Cvoltage (L-V) characteristics of ITO/PEDOT /polymer/Ca/Al devices of PFN (a) and PFNAn (b)

Fig.6 EL current efficiency curves of ITO/PEDOT/polymer/Ca/ Al devices
4 Conclusions
(1) The polymers possess good solubility in common organic solvents and high thermal stability with the onset decomposition temperature at higher than 410��.
(2) The maximum absorptions of PFN and PFNAn films are at 428 and 445 nm, and the maximum PL emissions of PFN and PFNAn films are at about 633 and 620 nm, respectively.
(3) Cyclic voltammetry results reveal that the copolymers possess higher electron affinity and reversible reduction re-oxidation processes. Their electroluminescent spectra demonstrate that PFN and PFNAn emit saturated red light with the EL emission maximum located at 640 and 621 nm, respectively.
(4) Excellent thermal and chemical stability, relatively high electron affinity could also result in these materials having potential applications in ETM in OLEDs and electron acceptors in photovoltaic cells.
References
[1] SKOTHEIM T A, ELSENBAUMER R L, REYNOLDS J R. Hand book of conducting polymers [M]. 2nd ed. New York: Marcel Dekker, 1998: 921.
[2] CHEN Y, ARAKI Y, DOYLE J, STREVENS A, ITO O, BLAU W J. Synthesis, characterization and optoelectronic properties of novel PFO/PPV copolymer [J]. Chem Mater, 2005, 17: 1661-1666.
[3] WANG Ping, CHAI Chun-peng, YANG Qian, WANG Fu-zhi, SHEN Zhi-hao, GUO Hai-qing, CHEN Xiao-fang, FAN Xing-he, ZOU De-chun, ZHOU Qi-feng. Synthesis and characterization of bipolar copolymers containing oxadiazole and carbazole pendant groups and their application to electroluminescent devices [J]. J Polym Chem Part A: Polym Chem, 2008, 46(16): 5452-5460.
[4] PENG Qiang, LI Ming-jun, TANG Xing-hua, LU Shi-qiang, PENG Jun-biao, CAO Yong. New polyphenylene- and polyphenyl- enevinylene-based copolymers containing triarylpyrazoline units in the main chains [J]. J Polym Chem Part A: Polym Chem, 2007, 45(9): 1632-1640.
[5] ZOU Ying-ping, ZHOU Yi, WU Guang-long, LI Yong-fang, PAN Chun-yue. Electroluminescent fluorene-based alternating polymers bearing triarylamine or carbazole moieties in the main chain: Synthesis and properties [J]. J App Polym Sci, 2009, 111(2): 978-987.
[6] ZOU Ying-ping, TAN Song-ting, YUAN Zhu-liang, YU Zong-qiang. Synthesis, photo- and electroluminescence properties of a PPV derivative with di(ethylene oxide) segment in the backone [J] . J Mater Sci, 2005, 40(13): 3569-3571.
[7] TAN Song-ting, ZOU Ying-ping, ZHU Wei-guo, JIANG Chang-Yun. Synthesis, photo- and electroluminescence properties of asymmetrical alternating copoly (p-phenylenevinylene) derivatives [J]. Opt Mater, 2006, 28(8/9): 1108-1114.
[8] ZOU Ying-ping, TAN Song-ting, YUAN Zhu-liang, YU Zong-qiang. A novel green light emitting material containing both hole and electron transporting units [J]. J Mater Sci, 2005, 40(13): 3561-3563.
[9] ZOU Y P, DAVID G, R?DA B A, AHMED N, TAO Y, MARIO L. A high-mobility low-bandgap poly(2,7-carbazole) derivative for photovoltaic applications [J]. Macromolecules, 2009, 42(8): 2891- 2894.
[10] ZOU Ying-ping, WU Wei-ping, SANG Guang-yi, YANG Yi, LIU Yun-qi, LI Yong-fang. Polythiophene derivative with phenothiazine- vinylene conjugated side chain: Synthesis and its application in field-effect transistors [J]. Macromolecules, 2007, 40(20): 7231-7237.
[11] ZOU Ying-ping, SANG Guang-yi, WU Wei-ping, LIU Yun-qi, LI Yong-fang. A polythiophene derivative with octyloxyl triphenylamine-vinylene conjugated side chain: Synthesis and its applications in field-effect transistor and polymer solar cell [J]. Synth Met, 2009, 159: 182-187.
[12] LI Yong-fang, ZOU Ying-ping. Conjugated polymer photovoltaic materials with broad absorption band and high charge carrier mobility [J]. Adv Mater, 2008, 20: 2952-2958.
[13] ZOU Ying-ping, WAN Mei-xiu, SANG Guang-yi, YE Ming-fu, LI Yong-fang. An alternative copolymer of carbazole and thieno[3,4b]-pyrazine: Synthesis and mercury detection [J]. Adv Funct Mater, 2008, 18: 2724-2732.
[14] ZOU Ying-ping, SANG Guang-yi, WAN Mei-xiu, TAN Song-ting, LI Yong-fang. Synthesis, electrochemical and photovoltaic properties of multi-armed polythiophenes with triphenylamine trivinylene as conjugated linker [J]. Macromol Chem Phys, 2008, 209: 1454-1462.
[15] TANG C W, VANSIYKE S A. Organic electroluminescent diodes [J]. Appl Phys Lett, 1987, 51(12): 913-915.
[16] BURROUGHES J H, BRADLEY D D C, BROWM A R, MARKS R N, MACKAY K, FRIEND R H, BURN P L, HOLMES A B. Light-emitting diodes based on conjugated polymers [J]. Nature, 1990, 347(11): 539-541.
[17] BARRY C, THOMPSON L, MADRIGA G, MAURICIOR P, TAESIK K, KIRKA S. Donor-acceptor copolymers for red- and near-infrared-emitting polymer light-emitting diodes [J]. Polym Sci Part A: Polym Chem, 2005, 43(7): 1417-1431.
[18] ZOU Ying-ping, HOU Jian-hui, YANG Chun-he, LI Yong-fang. A novel n-type conjugated polymer DOCN-PPV: Synthesis, optical and electrochemical properties [J]. Macromolecules, 2006, 39(26): 8889-8891.
[19] XIA Yang-jun, DENG Xian-yu, WANG Li, LI Xian-zhen, ZHU Xu-hui, CAO Yong. An extremely narrow-band-gap conjugated polymer with heterocyclic backbone and its use in optoelectronic devices[J]. Macromol Rapid Commun, 2006, 27(15): 1260-1264.
[20] HUANG Jian, XU Yi-she, HOU Qiong, YANG Wei, YUAN Ming, CAO Yong. Novel red electroluminescent polymers derived from carbazole and 3,6-bis(2-thienyl)-1,2,7-benzothiadiazole macromol [J]. Rapid Commun, 2002, 23(12): 709-712.
[21] COLLADET K, FOURIER S, CLEIJ T J, LUTSEN L, GELAN J. Low band gap donor-acceptor conjugated polymers toward organic solar cells applications [J]. Macromolecules, 2007, 40(1): 65-72.
[22] BLOUIN N, MICHAUD A, LECLERC M. A low-bandgap poly(2,7-carbazole) derivative for use in high-performance solar cells [J]. Adv Mater, 2007, 19: 2295-2300.
[23] ZOU Ying-ping, SANG Guang-yi, ZHOU Er-jun, LI Yong-fang. Copolymers of thiophene and cyano-substituted phenylene-facile tuning of electronic energy levels of conjugated polymers and their photovoltaic application [J]. Macromol Chem Phys, 2008, 209: 431-438.
[24] ZHAN Xiao-wei, LIU Yun-qi, ZHU Dao-ben, HUANG Wen-tao, GONG Qi-huang. Femtosecond third-order optical nonlinearity of conjugated polymers consisting of fluorene and tetraphenyldiaminobiphenyl units:Structure-property relationships [J]. Phys Chem B, 2002, 106(8): 1884-1888.
[25] BRUN M, DEMADRILE R, RANNOU P, PRON A, TRAVERS J P, GREVIN B. Multiscale scanning tunneling microscopy study of self-assembly phenomena in two-dimensional polycrystals of ��-conjugated polymers: The case of regioregular poly(dioctylbithiophene-alt-fluorenone) [J]. Adv Mater, 2004, 16: 2087-2092.
[26] KLAERNER G, DAVEY M H, CHEN W D, SCOTT J C, MILLER R D. Colorfast blue-light-emitting random copolymers derived from di-n-hexylfluorene and anthracene [J]. Adv Mater, 1998, 10(13): 993-997.
[27] VEENSTTA S C, VERHEES W J H, KROON J M, KOETSE M M, SWEELSEEN J. Photovoltaic properties of a conjugated polymer blend of MDMO-PPV and PCNEPV [J]. Chem Mater, 2004, 16(12): 2503-2508.
[28] FANG Qiang, JIANG Biao, XU Bing, WANG Wan-jun, YU Feng, WU Xia-qin. Preparation and properties of new soluble ��-conjugated polymers containing a fumaronitrile unit in the main chain [J]. Macromol Rapid Commun, 2004, 25(15): 1429-1431.
[29] ELDO J, ARUNKUMAR E, AJIAYAGHOSH A. Fluorescent bispyrroles: New building blocks for novel ��-conjugated polymers [J]. Tetra Lett, 2000, 41(32): 6241-6244.
[30] ANNA H, DAVID H, GEORGE M. Electron transfer in bis-porphyrin donor-acceptor compounds with polyphenylene spacers shows a weak distance dependence [J]. J Am Chem Soc, 1992, 114(15): 6227-6238.
[31] JUNK H K, LEE J K, KANG M S, KIM S W, KIM J J, PARK S Y. Synthesis and properties of poly(p-phenylenevinylene-co-sulfonylene) for a blue light-emitting diode [J]. Polym Bull, 1999, 43(1): 13-20.
[32] LIAO L, DING L M, KARASZ F E, PANG Y. Poly[(2- alkoxy-5-methyl-1,3-phenylene vinylene)-alt-(phenylene vinylene)] derivatives with different contents of cis- and trans-olefins: The effect of the olefin bond geometry and conjugation length on luminescence [J]. J Polym Sci Part A: Polym Chem, 2004, 42(2): 303-316.
[33] LI Y F, CAO Y, GAO J, WANG D L, YU G, HEEGER A J. Electrochemical properties of luminescent polymers and polymer light-emitting electrochemical cells [J]. Synth Met, 1999, 99(3): 243-248.
[34] SUN Qing-jiang, WANG Hai-qiao, YANG Chun-he, LI Yong-Fang. Synthesis and electroluminescence of novel copolymers containing crown ether spacers [J]. J Mater Chem, 2003, 13(4): 800-806.
Foundation item: Projects(50803074, 50633050) supported by the National Natural Science Foundation of China; Project supported by the Opening Fund of State Key Laboratory of Powder Metallurgy and Start-up Fund of Central South University, China
Received date: 2009-04-21; Accepted date: 2009-07-09
Corresponding author: ZOU Ying-ping, PhD; Tel: +86-731-88836961; E-mail: zyp2008@iccas.ac.cn
(Edited by YANG You-ping)

