
J. Cent. South Univ. (2018) 25: 1076-1083
DOI: https://doi.org/10.1007/s11771-018-3807-7
Effects of ceramsite on methane and hydrogen sulphide productions from macroalgae biomass
SUN Meng-ting(������)1, 2, FU Shan-fei(���Ʒ�)1, 2, HE Shuai(��˧)1, 2,FAN Xiao-lei(������)1, GUO Rong-bo(���ٲ�)1
1. Shandong Industrial Engineering Laboratory of Biogas Production & Utilization, Key Laboratory of Biofuels, Shandong Provincial Key Laboratory of Synthetic Biology, Qingdao Institute of Bioenergy and Bioprocess Technology, Chinese Academy of Sciences, Qingdao 266101, China;
2. University of Chinese Academy of Sciences, Beijing 100049, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2018
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2018
Abstract: The easy acidification and high hydrogen sulfide (H2S) production during anaerobic digestion of macroalgae limited its application in biomethane production. In order to investigate the effects of ceramsite on methane and H2S productions during anaerobic digestion of macroalgae, batch experiments of Macrocystis pyrifera were carried out. Four groups named C0, C1, C2 and C3 added with 0, 1.5, 3.0 and 4.5 g/gsubstrate of ceramsite, respectively, were studied and compared. The highest cumulative methane yield of 286.3 mL/gsubstrate is obtained in C2, which is 40.11% higher than that of C0. The cumulative H2S yields of C1, C2 and C3 are 32.67%, 44.66% and 53.21% lower than that of C0, respectively. Results indicate that ceramsite addition permits higher methane yields, shorter lag-phase time and lower H2S yields during anaerobic digestion of Macrocystis pyrifera.
Key words: anaerobic digestion; macroalgae; ceramsite; methane production; hydrogen sulfide
Cite this article as: SUN Meng-ting, FU Shan-fei, HE Shuai, FAN Xiao-lei, GUO Rong-bo. Effects of ceramsite on methane and hydrogen sulphide productions from macroalgae biomass [J]. Journal of Central South University, 2018, 25(5): 1076�C1083. DOI: https://doi.org/10.1007/s11771-018-3807-7.
1 Introduction
Energy crisis is a global issue for sustainable development of economy [1, 2]. The marine macroalgae is a promising alternative feedstock for generating biofuels and biomaterials because of its major advantages, including high growth rate, energy-efficient and without competition with food crops for cultivated land [3�C5]. Methane production through anaerobic digestion (AD) is a feasible solution to utilize marine macroalgae [6]. A few studies obtained decent methane yields from macroalgae via AD processes. JARD et al [7] conducted batch and semi-continuous AD of Palmaria palmate and Saccharina latissima, and results showed that Palmaria palmata permitted a better methane yield than Saccharina latissima either in batch (279.0 mL/gsubstrate vs 210.0 mL/gsubstrate) or in semi-continuous reactor (320.0 mL/gsubstrate vs 270.0 mL/gsubstrate). FAN et al [8] obtained a methane yield of (217.1��2.4) mL/gsubstrate from AD of Macrocystis pyrifera with marine sediments as inocula in seawater system. During the AD of Nizimuddinia zanardini macroalgae, YAZDANI et al [9] obtained biogas yield of 170 to 200 m3/t from dried algae biomass by hot water pretreatment.Methane production from macroalgae biomass needs to overcome feedstock-related obstacles. Due to the high carbohydrate content and low lipid level of macroalgae, the hydrolysis rate of macroalgae is fast, which benefits the release of soluble organics and VFA accumulation. However, acidification happens easily in the AD process of macroalgae, which possibly inhibits methanogenesis process [10�C12].
Another restrictive factor of macroalgae AD process is the high yield of hydrogen sulfide (H2S). Existing literatures show that the volatile sulfur compounds (VSCs), including H2S and methyl mercaptan (MM), could be generated from sulfurated biomass under anaerobic condition [13, 14]. Since macroalgae biomass is a kind of sulfer-rich substrate, high H2S yields emerge in AD processes [11, 14]. High concentration of H2S (��0.10%) might have an inhibitory effect on methanogenesis and restrain methane production and AD efficiency of macroalgae [15�C17]. In addition, the produced H2S could corrode biogas production facilities [18, 19]. Therefore, H2S yield decrease is an important aspect during AD of macroalgae.
Present researches point out that porous material addition in AD system might be an attractive choice to prevent process from acidification and contribute to system stability. MILAN et al [20] pointed out that the natural zeolite reduced the values of TVFA/Alk ratio in AD system while increasing pH value and relieving acidification. In addition, natural zeolite had a capacity for microbial immobilization, which eventually improved methane yield. WATANABE et al [21] reported the addition of cedar charcoal during co-digestion of sewage sludge and crude glycerol obtained a 1.6 time higher methane yield. Cedar charcoal could immobilize microbial community. It also improved VFAs degradation efficiency and made pH value more stable and appropriate for methane production.
In addition, porous material was also used in H2S removal in several studies. Zeolite was used for absorbing H2S from biogas in virtue of its unique structure, uniform pores and channels, high selectivity and compatibility towards polar compounds [22]. It has been also applied to zeolite/TiO2 photocatalytic reactor for in-situ H2S removal from AD system due to its absorption capacity and harmlessness to AD process [18].
Ceramsite is another economical porous material with similar structure and absorption potential compared to zeolite. Although ceramsite has been utilized for absorbing H2S in mixed gas [23, 24], literatures about its effects on AD of macroalgae are deficient so far. In this study, impacts of ceramsite addition on the methane and H2S productions from anaerobic digestion of macroalgae were investigated, which could provide a feasible approach for improving methane yield and restraining H2S yield in AD system of M. pyrifera.
2 Methods
2.1 Substrate, inoculum and ceramsite
The substrate used in this study was M. pyrifera biomass, which was gathered from Zhangzi Island, Dalian, China and sun-dried for storage. Before use, M. pyrifera biomass was milled to a size of about 2.0 mm. The total solid (TS) of substrate was 90.42% and the volatile solid (VS) was 61.77% (based on TS). The C, N, H and S in macroalgae were 24.45%, 1.20%, 3.84% and 1.00%, respectively. Proportions of protein, carbohydrate and lipid were 6.01%, 45.12% and 8.30% (based on TS), respectively.
Inoculum used in this study was granule sludge obtained from Tsingtao Brewery Co., Ltd.; TS and VS of inoculum were 2.73% and 72.06% (based on TS), respectively.
The ceramsite used in this study was purchased from Tsingtao Zhiyuanlvke Co., Ltd., which was russet spherical particle with diameter of (4.0��0.5) mm and mass of (0.225��0.034) g. The major component of ceramsite was SiO2, and it was also rich in Ca2+, Al3+ and Na+.
2.2 Biogas fermentation process
Batch anaerobic digestions of M. pyrifera biomass were carried out in triplicate in 250 mL bottles. 3.0 g of M. pyrifera biomass and 45.83 mL of granule sludge were mixed in bottles. Four set-ups named C0, C1, C2, C3 were added with 0, 4.5, 9.0, 13.5 g (0, 1.5, 3.0, 4.5 g/gsubstrate) of ceramsite, respectively. Deionized water was added in each vial to reach a total working volume of 160 mL. Each bottle was flushed with N2 for 10 min to replace air and closed with rubber stoppers. Digesters were incubated at 37 ��C with constant shaking at 140 r/min. Methane and H2S concentrations were tested periodically.
2.3 Analytical methods
TS and VS contents were analyzed according to the standard method [25]. Elemental analysis was conducted by Vario EL III elemental analyzer. Carbohydrate and lipid contents in macroalgae were tested by phenol sulfuric acid method [26]. The protein content was determined based on the total elemental nitrogen measurement using the conversion factor 6.35 [27]. Biogas volume was measured by water replacement method. Methane and VFA concentrations were measured by gas chromatograph (GC-2014, SHIMADZU, Japan) as described previously [28]. H2S concentrations were measured by gas chromatograph (GC-2014, SHIMADZU, Japan) using helium as carrier gas. The pH values were measured using a pH meter (B-212, HORIBA). Component measurement of ceramsite was accomplished by the wide angle X-ray diffraction (Bruker D8 Advance) at 40 kV and 40 mA in the scanning angle 2�� of 5��C60�� at a scanning speed of 1.0��/min.
The methane-producing characteristics were also fitted by the Modified Gomperz equation, which was written as [29]
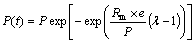
where P(t) is the cumulative total methane yield (mL/gsubstrate); P is the total methane production potential (mL/gsubstrate); Rm is the maximum methane production rate (mL/(gsubstrate��d�C1)); �� is the lag-phase time (d); and t is the elapsed time (d).
3 Results and discussion
3.1 Methane yield during AD process of Macrocystis pyrifera
The cumulative methane yields during AD processes of M. pyrifera are depicted in Figure 1. Results show that the cumulative methane yield increased apparently with ceramsite addition. The cumulative methane yields of C1, C2 and C3 are 283.4, 286.3 and 263.1 mL/gsubstrate,respectively, which are 38.72%, 40.11% and 28.78% higher than that of C0 (204.3 mL/gsubstrate), respectively.
The modified Gomperz equation fitted results are shown in Table 1. R2 values of C0, C1, C2 and C3 are 0.972, 0.987, 0.983 and 0.979, respectively, which reflect that the modified Gomperz equation fitted results could well explain the experimental data. With ceramsite addition, the lag-phase time (��) becomes shorter and the maximum methane production rate (Rm) is higher compared to the control (C0). The lag-phase times of C1, C2 and C3 are all 0 d, which are much shorter than that of C0 (8.285 d). Additionally, the maximum methane production rates of C1, C2 and C3 are 14.4, 10.3 and 9.4 mL/gsubstrate, respectively, which are higher compared to C0 (5.7 mL/gsubstrate).
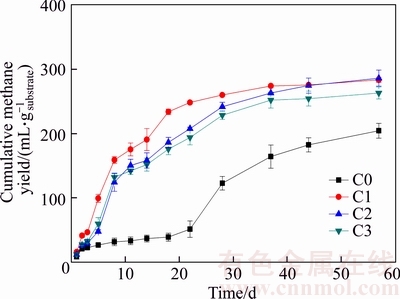
Figure 1 Methane yields during AD processes of Macrocystis pyrifera (C0 stands for 0 g/gsubstrate addition of ceramsite with C1 for 1.5 g/gsubstrate; C2 stands for 3.0 g/gsubstrate; C3 stands for 4.5 g/gsubstrate)
Table 1 Modified Gomperz equation fitted with methane production data

Apparently, compared to C0, the C1, C2 and C3 permit higher cumulative methane yields and faster methane productions, which indicates that ceramsite addition could improve methane production of macroalgae biomass.
3.2 Hydrogen sulfide yield during AD process of Macrocystis pyrifera
The cumulative H2S yields of AD processes of M. pyrifera are depicted in Figure 2. The digesters with ceramsite obtain lower cumulative H2S yields compared to the control. The cumulative H2S yields of C1, C2 and C3 are 3.7, 3.1 and 2.6 mL/gsubstrate, respectively, which are 32.67%, 44.66% and 53.21% lower than that of C0 (5.5 mL/gsubstrate),respectively.

Figure 2 Hydrogen sulfide yields during AD processes of Macrocystis pyrifera (C0 stands for 0 g/gsubstrate addition of ceramsite, C1 for 1.5 g/gsubstrate, C2 for 3.0 g/gsubstrate and C3 for 4.5 g/gsubstrate)
The cumulative H2S yield is restrained with ceramsite addition, and decreases with increased amount of ceramsite (Figure 2), which reveals that ceramsite performs well in reducing H2S yield. COSOLI et al [22] conducted the Grand Canonical Monte Carlo (GCMC) simulations to study H2S removal from biogas streams by different zeolites, and quantum mechanics (QM) molecular simulations were performed to obtain structures and partial charges of sorbates, and results demonstrated that the absorption ability of zeolite was owed to electrostatic interaction between the polar molecule H2S and the framework in zeolite. Ceramsite is a kind of porous material and the main compositions are SiO2, (Na,Ca)Al(Si,Al)3O8, Na(Si3Al)O8 and SiS2. Apparently, ceramsite is rich in Si4+, Ca2+, Al3+ and Na+, and these cations possibly consist of the framework in ceramsite, thus the electrostatic interaction exists between polar molecule H2S and framework. In this work, electrostatic attraction from framework of ceramsite absorbs H2S molecule; consequently, the H2S yield was reduced. The H2S toxicity for methanogenesis causes inhibitory effect on methane production because the H2S molecule is able to penetrate the methanogenesis cell membrane, interfere with disulfide bridges between polypeptide chains and obstruct coenzyme activities [15, 30]. Therefore, the declined H2S yield might favor methanogenesis. Compared to C0, digesters with ceramsite start methane production quicker and obtain higher methane yields (Figure 1). It speculates that the declined H2S yield caused by absorption from ceramsite framework could be one of the reasons for methane production improvement.
3.3 pH value and VFAs variations during AD process of Macrocystis pyrifera
The pH values during AD processes of M. pyrifera are depicted in Figure 3. The digesters with ceramsite permit more lenitive pH decreases than C0. The pH value of C0 declines from 8.0 to 7.3 during the 1st day, and drops to 5.8 on the 5th day, then it maintains approximately 5.6 until the 18th day. Eventually, it returns to 7.9 on the 28th day and remains stable subsequently. Comparably, the pH values of C1, C2 and C3 decline from 8.0 to 7.5 on the 1st day, and maintain approximately 7.5 until the 5th day, then return to 8.2 on the 8th day and remain stable subsequently. C0 possesses a noticeable pH drop (approximately 5.6) which lasts for 14 d (from the 5th day to the 18th day), while C1, C2 and C3 show slight pH decreases (approximately 7.5) which exist for about 4 d (from the 1st day to the 5th day).
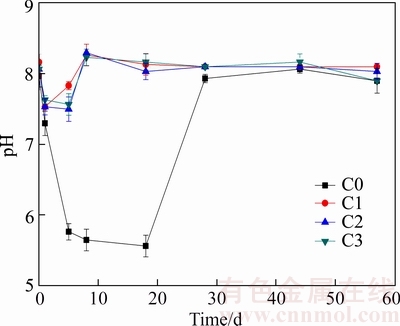
Figure 3 pH variations during AD processes of Macrocystis pyrifera (C0 stands for 0 g/gsubstrate addition of ceramsite, C1 for 1.5 g/gsubstrate, C2 for 3.0 g/gsubstrate and C3 for 4.5 g/gsubstrate)
In the groups of C1, C2 and C3, the pH decreases turn to slighter compared to C0 (Figure 3), which infers that ceramsite addition make pH value more stable. The XRD analysis points out that ceramsite is mainly composed of Si4+, Ca2+, Al3+ and Na+. It suggests that several kinds of cations in crystal structure, including Si4+ and Al3+, could be easily replaced by H+, then the unit cells need to absorb equivalent cations to reach the charge balance [31]. In this work, when ceramsite is added in AD system, Si4+, Ca2+, Al3+ and Na+ in unit cells of ceramsite are replaced by H+ from liquid slurry. Afterwards, the unit cells become electronegative, then further absorb equivalent cations from liquid to reach the charge balance. Consequently, H+ concentration of liquid slurry declines and the pH value goes up. It implies that the ceramsite addition could contribute to elevating pH value and relieving acidification in AD system. The pH value of 6.8�C7.2 is the optimal range for methane production process and the pH value of 6.5�C8.0 is proper for methane production, whereas the pH value below 6.5 could restrain methane productivity [32�C34]. Apparently, the pH values of C1, C2 and C3 (7.5�C8.2) are more stable and appropriate for methanogenesis than that of C0 (5.6�C8.0). It suggests that the increasing and stable pH value in AD system, which is benefitted from ceramsite addition, favors methane production.
The concentrations of TVFA and VFAs during AD processes of M. pyrifera are shown in Figure 4. The results indicate that the digesters with ceramsite possess slighter VFAs accumulation compared to C0. The TVFA concentrations during AD processes of M. pyrifera are depicted in Figure 4(a). The TVFA concentration of C0 reaches peak value of 3.26 g/L on the 8th day and reduces to approximately 0.60 g/L on the 44th day, then levels off. Comparably, the TVFA concentrations of C1, C2 and C3 increase and reach the peak values of 1.76, 2.75 and 2.91 g/L on the 5th day, respectively, then reduce to 0.60 g/L and maintain stable after the 28th day. The concentrations of VFAs during M. pyrifera AD process are shown in Figures 4(b)�C(d). The acetic acid concentration of C0 increases to the maximum value of 2.00 g/L on the 8th day and decreases slowly to 0.08 g/L on the 44th day. Relatively, the acetic acid concentration in C1, C2 and C3 increase to the maximum values of 0.72, 1.50 and 1.67 g/L on the 5th day, respectively, and decline soon to approximately 0.08 g/L on the 28th day (Figure 4(b)). The propionic acid concentration of C0 keeps in a high level after the 8th day and obtains a maximum value of 0.33 g/L, whereas the propionic acid concentrations of C1,C2 and C3 rise dramatically to the maximum values of 0.25, 0.32 and 0.31 g/L on the 5th day, respectively, then recover to low levels (approximately 0.08 g/L) after the 28th day (Figure 4(c)). The butyric acid concentration increases to the maximum value of 0.60 g/L on the 8th day, and decreases slowly until the 44th day and then keep stable. Comparably, in C1, C2 and C3, the butyric acid concentrations increase dramatically to the maximum values of 0.45, 0.56 and 0.55 g/L on the 5th day, respectively, and decline soon to approximately 0.18 g/L on the 28th day (Figure 4(d)).

Figure 4 TVFA and VFAs concentrations during AD processes of Macrocystis pyrifera
The excessive VFAs accumulation inhibits the activity of methanogenesis [35, 36]. It was recommended that the methane productivity was restrained when the TVFA concentration above 20.00 g/L occurred [35]. DOGAN et al [36] verified the inhibition of VFAs on methane production from granular sludge in a upflow anaerobic sludge blanket (UASB) reactor, and argued that the concentrations of 4.00 g/L, 2.00 g/L and 6.00 g/L were the beginning of acetic acid, propionic acid and butyric acid inhibition, respectively. Entirely, the TVFA, acetic acid, propionic acid and butyric acid concentrations in AD processes of macroalgae are below inhibitory values, which implies that VFAs inhibition fails to occur in all groups. However, with ceramsite addition, the TVFA and VFAs concentrations of C1, C2 and C3 increase to lower peak values and recover to balanced levels faster than C0. Apparently, digesters with ceramsite permit slighter VFAs accumulation and acidification compared to C0 (Figure 4), which manifests that ceramsite is rewarding to keep VFAs dynamic balance. BABLE et al [35] mentioned that the VFAs stayed in a dynamic balance in AD system since VFAs were generated by hydrolysis/ acidogenesis and consumed by methanogenesis subsequently. The ceramsite addition causes reduction of H2S yield and increase of pH value in AD system. These two effects favor methanogenesis and methane production, then lead to a result that the VFAs generated from biomass degradation in hydrolysis/acidogenesis stages are converted to CH4 soon, which relieves VFAs accumulation. Accordingly, with ceramsite addition, the reductions of VFAs concentrations are not the main influential factors on improvement of methane production; however, ceramsite addition could benefit VFAs dynamic balance, which provides a feasible solution to prevent AD system from excessive VFAs accumulation and protect methanogenesis from VFAs inhibition.
It is worth mentioning that, several porous materials, including zeolite and charcoal, have been demonstrated to be capable of immobilizing methanogenesis and improving methane yield during AD process [37, 38]. WATANABE et al [21] placed cedar charcoal in a continuous mixed digester co-digesting sewage sludge and crude glycerol, obtained a 1.6 time higher methane yield compared to control, and microbial attachment to the charcoal pores was unveiled through the scanning electron microscope (SEM); while examination of microbial community revealed a high abundance of methanogenesis. The ceramsite used in the macroalgae AD system is a kind of analogous porous material with zeolite and charcoal, and it might possibly have a potential to immobilize microorganism on its porous. Therefore, ceramsite possibly immobilizes microbial community in macroalgae AD system, which might be another positive factor for methane production improvement.
4 Conclusions
1) Ceramsite addition is demonstrated to perform successfully in methane yield improvement and H2S yield reduction during anaerobic fermentation of M. pyrifera.
2) The highest cumulative methane yield of 286.3 mL/gsubstrate is obtained with the ceramsite addition of 3.0 g/gsubstrate, which is 40.11% higher than that of control.
3) With ceramsite addition of 4.5 g/gsubstrate, the cumulative H2S yield is reduced by 53.21% compared to control. The H2S yield decrease could relieve H2S toxicity to methanogenesis and further benefit the methane production.
4) Ceramsite possibly immobilizes methanogenesis which also favors methane productivity.
5) Additionally, ceramsite is beneficial to VFAs dynamic balance during AD process which provides a alternative solution to cope with the acidification.
References
[1] GOYAL H B, SEAL D, SAXENA R C. Bio-fuels from thermochemical conversion of renewable resources: A review [J]. Renewable and Sustainable Energy Reviews, 2008, 12(2): 504�C517.
[2] DEMIRBAS A, DEMIRBAS M F. Importance of algae oil as a source of biodiesel [J]. Energy Conversion and Management, 2011, 52(1): 163�C170.
[3] KONDA N V S N M, SINGH S, SIMMONS B A, KLEIN- MARCUSCHAMER D. An investigation on economic feasibility of macroalgae as a potential feedstock for biorefineries [J]. Bioenergy Research, 2015, 8(3): 1046�C1056.
[4] GHPSH S, KLASS D L, CHYNOWETH D P. Bioconversion of macrocystis pyrifera to methane [J]. Journal of Chemical Technology and Biotechnology, 1981, 31(12): 791�C807.
[5] CHEN Hui-hui, ZHOU Dong, LUO Gang, ZHANG Shi-cheng, CHEN Jian-min. Macroalgae for biofuels production: Progress and perspectives [J]. Renewable and Sustainable Energy Reviews, 2015, 47: 427�C437.
[6] DEBOWSKI M, ZIELINSKI M, GRALA A, DUDEK M. Algae biomass as an alternative substrate in biogas production technologies��Review [J]. Renewable and Sustainable Energy Reviews, 2013, 27: 596�C604.
[7] JARD G, JACKOWIAK D, CARRERE H, DELGENES J P, TORRIJOS M, STEYER J P, DUMAS C. Batch and semi-continuous anaerobic digestion of Palmaria palmata: Comparison with Saccharina latissima and inhibition studies [J]. Chemical Engineering Journal, 2012, 209: 513�C519.
[8] FAN Xiao-lei, GUN Rong-bo, YUAN Xian-zheng, QIU Yan-ling, YANG Zhi-man, WANG Fei, SUN Meng-ting, ZHAO Xiao-xian. Biogas production from Macrocystis pyrifera biomass in seawater system [J]. Bioresource Technology, 2015, 197: 339�C347.
[9] YAZDANI P, ZAMANI A, KARIMI K, TAHERZADEH M J. Characterization of Nizimuddinia zanardini macroalgae biomass composition and its potential for biofuel production [J]. Bioresource Technology, 2015, 176: 196�C202.
[10] BRUHN A, DAHL J, NIELSEN H B, NIKOLAISEN L, RASMUSSEN M B, MARKAGER S, OLESEN B, ARIAS C, JENSEN P D. Bioenergy potential of Ulva lactuca: Biomass yield, methane production and combustion [J]. Bioresource Technology, 2011, 102(3): 2595�C2604.
[11] MONTINGELLIA M E , TEDESCO S, OLABI A G. Biogas production from algal biomass: A review [J]. Renewable and Sustainable Energy Reviews, 2015, 43: 961�C972.
[12] GHADIRYANFAR M, ROSENTRATER K A, KEYHANI A, OMID M. A review of macroalgae production, with potential applications in biofuels and bioenergy [J]. Renewable and Sustainable Energy Reviews, 2016, 4: 473�C481.
[13] HIGGINS M J, CHEN Y C, YAROSZ D P, MURTHY S N, MAAS N A, GLINDEMANN D, NOVAK J T. Cycling of volatile organic sulfur compounds in anaerobically digested biosolids and its implications for odors [J]. Water Environment Research, 2006, 78(3): 243�C252.
[14] DU Wei-wei, PARKER W J. Behavior of volatile sulfur compounds in mesophilic anaerobic digestion [J]. Water Environment Federation, 2009, 12(1): 3963�C3979.
[15] BELLE A J, LANSING S, MULBRY W, WEIL R R. Methane and hydrogen sulfide production during co-digestion of forage radish and dairy manure [J]. Biomass and Bioenergy, 2015, 80: 44�C51.
[16] ABBOTT T, ESKICIOGLU C. Effects of metal salt addition on odor and process stability during the anaerobic digestion of municipal waste sludge [J]. Waste Management, 2015, 46: 449�C458.
[17] CHEN Ye, CHENG J J, CREAMER K S. Inhibition of anaerobic digestion process: A review [J]. Bioresource Technology, 2008, 99(10): 4044�C4064.
[18] HAO Xiao-yan, HOU Guang-ying, ZHENG Pei-xue, LIU Ru-tao, LIU Chun-guang. H2S in-situ removal from biogas using a tubular zeolite/TiO2 photocatalytic reactor and the improvement on methane production [J]. Chemical Engineering Journal, 2016, 294: 105�C110.
[19] JIA Li-juan, NING Ping, WANG Xiang-yu, QU Guang-fei, XIONG Xiang-feng. Dynamics of liquid-phase catalytic oxidation of hydrogen sulfide removal in rural biogas [J]. Journal of Central South University, 2014, 21(7): 2843- 2847.
[20] MILAN Z, SANCHEZ E, WEILAND P, BORJA R, MARTIN A, ILANGOVAN K. Influence of different natural zeolite concentrations on the anaerobic digestion of piggery waste [J]. Bioresource Technology, 2001, 80(1): 37�C43.
[21] WATANABE R, TADA C, BABA Y, FUKUDA Y, NAKAI Y. Enhancing methane production during the anaerobic digestion of crude glycerol using Japanese cedar charcoal [J]. Bioresource Technology, 2013, 150: 387�C392.
[22] COSOLI P, FERRONE M, PRICL S, FERMEGLIA M. Hydrogen sulphide removal from biogas by zeolite adsorption, Part I. GCMC molecular simulations [J]. Chemical Engineering Journal, 2008, 145(1): 86�C92.
[23] CHEN Yi-qing, FAN Zhi-dong, MA Li-xia, YIN Juan, LUO Man, CAI Wang-feng. Performance of three pilot-scale immobilized-cell biotrickling filters for removal of hydrogen sulfide from a contaminated air steam [J]. Saudi Journal of Biological Sciences, 2014, 21(5): 450�C456.
[24] JIANG An-xi, ZHAO Yu-xin, XU Gui-qin, LI Li, MA Li. Evolution of air biofilter for H2S and NH3 biodegradation [J]. Journal of Science of Heilongjiang University, 2003, 20(1): 92�C95. (in Chinese)
[25] YUAN Xian-zheng, SHI Xiao-shuang, ZHANG Pei-song, WEI Yue-li, GUO Rong-bo, WANG Li-sheng. Anaerobic biohydrogen production from wheat stalk by mixed microflora: Kinetic model and particle size influence [J]. Bioresource Technology, 2011, 102(19): 9007�C9012.
[26] DUBOIS M, GILLES K A, HAMILTON JK, REBERS P A, Smith F. Colorimetric method for determination of sugars and related substances [J]. Analytical Chemistry, 1956, 28(3): 350�C356.
[27] SAFI C, CHARTON M, PIGNOLET O, SILVESTRE F, VACA-GARCIA C, PONTALIER P Y. Influence of microalgae cell wall characteristics on protein extractability and determination of nitrogen-to-protein conversion factors [J]. J Appl Phycol, 2013, 25(2): 523�C529.
[28] HE Shuai, FAN Xiao-lei, KATUKURI N R, YUAN Xian-zheng, WANG Fei, GUO Rong-bo. Enhanced methane production from microalgal biomass by anaerobic bio-pretreatment [J]. Bioresource Technology, 2016, 204: 145�C151.
[29] HUILINIR C, QUINTRIQUEO A, ANTILEO C, MONTALVO S. Methane production from secondary paper and pulp sludge: Effect of natural zeolite and modeling [J]. Chemical Engineering Journal, 2014, 257: 131�C137.
[30] GUERRERO L, MONTALVO S, HUILINIR C, CAMPOS J L, BARAHONA A, BORJA R. Advances in the biological removal of sulphides from aqueous phase in anaerobic processes: A review [J]. Environmental Reviews, 2016, 24(1): 84�C100.
[31] BORJA R, BANKS C J. Semicontinuous anaerobic digestion of soft drink wastewater in immobilised cell bioreactors [J]. Biotechnology Letters, 1993, 15(7): 767�C772.
[32] ZHAI Ning-ning, ZHANG Tong, YIN Dong-xue, YANG Gai-he, WANG Xiao-jiao, REN Guang-xin, FENG Yong-zhong. Effect of initial pH on anaerobic co-digestion of kitchen waste and cow manure [J]. Waste Management, 2015, 38: 126�C131.
[33] ZHANG Tong, MAO Chun-lan, ZHAI Ning-ning, WANG Xiao-jiao, YANG Gai-he. Influence of initial pH on thermophilic anaerobic co-digestion of swine manure and maize stalk [J]. Waste Management, 2015, 35: 119�C126.
[34] KIM M, GOMEC C Y, AHN Y, SPEECE R E. Hydrolysis and acidogenesis of particulate organic material in mesophilic and Thermophilic anaerobic digestion [J]. Environmental technology, 2003, 24(9): 1183�C1190.
[35] BABEL S, FUKUSHI K, SITANRASSAMEE B. Effect of acid speciation on solid waste liquefaction in an anaerobic acid digester [J]. Water Research, 2004, 38(9): 2417�C2423.
[36] DOGAN T, INCE O, OZ N A, INCE B K. Inhibition of volatile fatty acid production in granular sludge from a UASB reactor [J]. Journal of Environmental Science and Health, 2005, 40(3): 633�C644.
[37] ROMERO-GUIZA M S, VILA J, MATA-ALVAREZ J, CHIMENOS J M, ASTALS S. The role of additives on anaerobic digestion: A review [J]. Renewable and Sustainable Energy Reviews, 2016, 58: 1486�C1499.
[38] MONTALVO S, GUERRERO L, BORJA R, SANCHEZ E, MILAN Z, CORTES I, ANGELES DE LA LA RUBIA M. Application of natural zeolites in anaerobic digestion processes: A review [J]. Applied Clay Science, 2012, 58: 125�C133.
(Edited by YANG Hua)
���ĵ���
�����Ծ����������ͼ�������������Ӱ��
ժҪ���������������̴������ữ����������ߵ����⣬������������������Ƽ��鷽���Ӧ�á�Ϊ���о������Ծ����������͵ļ��������������Ӱ�죬��չ���Ծ���Macrocystis pyrifera������Ϊ��������η������顣ʵ���4�飬C0��C1��C2��C3���ֱ�������0��1.5��3.0��4.5 g/gsubstrate�����������У�C2��õ�����ߵļ����ۻ�������Ϊ286.3 mL/gsubstrate����C0�������40.11%��C1��C2��C3��������ۻ������ֱ��C0������32.67%��44.66%��53.21%�������ʾ�����������ľ��巢����ϵ���������ߡ����������̡�����������͡�
�ؼ��ʣ��������ͣ����壻���������飻����
Foundation item: Project(2014BAC31B01) supported by the National Science & Technology Support during the 12th Five-Year Plan Period, China; Projects(2015GSF117016, 2015GSF115037) supported by the Key Research & Development Project of Shandong Province, China
Received date: 2016-11-20; Accepted date: 2017-02-23
Corresponding author: GUO Rong-bo, PhD; Tel: +86�C532�C58782862; E-mail: guorb@qibebt.ac.cn; ORCID: 0000-0003-0697-7771