J. Cent. South Univ. (2016) 23: 1564-1568
DOI: 10.1007/s11771-016-3209-7
Hydrogenation reaction characteristics and properties of its hydrides for magnetic regenerative material HoCu2
JIN Tao(����)1, WU Meng-xi(������)1, HUANG Jia-le(������)1, TANG Ke(����)1, CHEN Li-xin(������)2
1. Institute of Refrigeration and Cryogenics, State Key Laboratory of Clean Energy Utilization,
Zhejiang University, Hangzhou 310027, China;
2. School of Materials Science and Engineering, Zhejiang University, Hangzhou 310027, China
 Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Abstract: The hydrogenation reaction characteristics and the properties of its hydrides for the magnetic regenerative material HoCu2 (CeCu2-type) of a cryocooler were investigated. The XRD testing reveals that the hydrides of HoCu2 were a mixture of Cu, unknown hydride I, and unknown hydride II. Based on the PCT (pressure-concentration-temperature) curves under different reaction temperatures, the relationships among reaction temperature, equilibrium pressure, and maximum hydrogen absorption capacity were analyzed and discussed. The enthalpy change ��H and entropy change ��S as a result of the whole hydrogenation process were also calculated from the PCT curves. The magnetization and volumetric specific heat capacity of the hydride were also measured by SQUID magnetometer and PPMS, respectively.
1 Introduction
The performance of the regenerator in a cryocooler has a significant impact on the overall efficiency of the cryocooler [1]. One of the frequently used regenerative materials is the metal lead, thanks to its low Debye temperature and relatively large volumetric heat capacity. However, its volumetric heat capacity drops rapidly at the cooling temperatures below 20 K, leading to a significant reduction in the regenerator��s efficiency. As a result, it is difficult for a cryocooler with lead as its regenerative material to cool down to below 10 K. Fortunately, magnetic regenerative materials, such as Er3Ni and ErNi, have been consecutively proposed in the past 30 years as the regenerative materials, which successfully improved the performance of a cryocooler at low temperature thanks to the abnormal peak values of heat capacity induced by magnetic phase transition below 20 K [2-3]. The application of these materials contributed to the breakthroughs of the traditional G-M cryocooler and pulse tube cryocooler in achieving the cooling temperatures as low as liquid helium temperature. Unfortunately, the efficiency of those machines declined dramatically for the cooling temperatures as low as 4 K, since the above-mentioned magnetic regenerative materials have the limit to their lowest transition temperature. To solve this problem, researchers have been looking for novel magnetic materials with lower transition temperatures. Previous studies claimed or verified that the hydrides of some magnetic regenerative material might have higher heat capacity than the corresponding original magnetic regenerative material in some certain temperature range, which indicates the possibility of using the hydride as a new regenerative material [4-7].
The hydrogenation reaction might change the structure and magnetic properties of some rare earth compounds, including HoCu2 [2-3, 8], which then may affect its transition temperature. HoCu2, with a magnetic transition temperature between 6 K and 10 K, has also been commonly used as the magnetic regenerative material at cryogenic temperatures in the past 10 years, thanks to its relatively high heat capacity induced by magnetic phase transition [9-11]. GRATZ et al [12] investigated the magnetization and electrical resistivity properties of HoCu2. WANG et al [5] worked on the crystal structures and the magnetic properties of the crystalline hydride of HoCu2. ZAVALIY et al [13] provided numerous detailed descriptions and explanations of the crystal transition and the structure transformation of HoCu2 hydrogenation. However, the reported studies are not yet comprehensively sufficient in the aspect of reaction characteristics. In this work, we will dig at the hydrogenation reaction characteristics and the structure transformations of HoCu2 compounds. The information about the product types, the crystallinity, the average grain size of the raw material and its hydrides, and the pressure-concentration-temperature (PCT) curves of the reaction have been obtained and presented.
2 Apparatus and experiments
The HoCu2 compound used in the experiments was prepared by the arc melting method in an argon gas atmosphere from the raw material holmium (99.99% in mass fraction) and copper (99.9% in mass fraction). The ingots were turned over and remelted three times to ensure the homogeneity of the sample, and were then cooled in the furnace. The cooled alloy ingots were silver in color and hard in nature. The ingot should be ground into powders with the particle sizes under 0.1 mm in an agate mortar before the hydrogenation reaction. The experimental apparatus for hydrogenation, as shown in Fig. 1, includes reaction chamber, hydrogen reservoir, hydrogen source, vacuum pump, pressure gauge, thermocouple, and computer-based DAQ system. The samples should be firstly activated, and then exposed to high-purity hydrogen gas (10-6 impurity) at different temperatures. In this way, the hydrogen absorption capacity of each sample could be controlled, thus acquiring the PCT curves of the samples at various temperatures. The absorbed hydrogen content is quantitatively determined by pressure difference method based on the equation of state for ideal gas, i.e., by means of monitoring pressure change in the constant volume chamber.
The experimental procedure is as follows: 1) Open valve 2, vacuumize the system for 5 min by the vacuum pump and then close valve 2. 2) Heat the sample to 650 K and then cool it to room temperature (between 280 K and 300 K), repeating three times to get the sample activated. 3) Open valve 1, introduce hydrogen into the reservoir till the pressure p1, then close valve 1. 4) Open valve 4 and record the new pressure p2 (as the deviation is below 1%, we take p1=p2). 5) Wait for about 3 h till the reaction reaches equilibrium, record the equilibrium pressure p3 (p3
2). 6) Close valve 4 and change the hydrogen pressure in the reservoir to p4 (p4>p2). 7) Open valve 4 and the sample will go on absorbing the hydrogen to a new equilibrium pressure p5 (p5
4). 8) Repeat steps 6 and 7 other 18 times to get 20 equilibrium pressures in all. 9) Open valve 3 to empty the hydrogen. If the volume of the hydrogen reservoir is V, the number of moles of the hydrogen atoms absorbed by the sample, ��n, can be calculated from the pressure change:
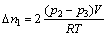 (1)
(1)
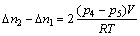 (2)
(2)
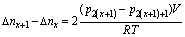 (3)
(3)
where ��nx+1-��nx are the mole numbers of the hydrogen atoms reacting with the compound when the initial pressure is p2(x+1) and the equilibrium pressure is p2(x+1)+1; R is the gas constant; T is the temperature inside the reservoir.
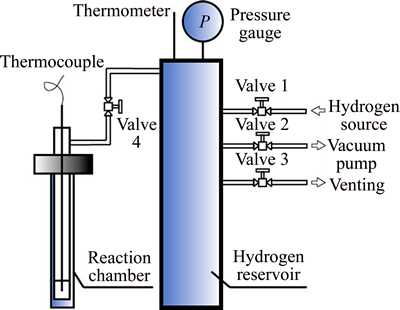
Fig. 1 Schematic diagram of apparatus for hydrogenation of HoCu2
Upon obtaining the equilibrium pressure and the hydrogen absorption content, the PCT curves of the hydrogenation of HoCu2 can be drawn at different reaction temperatures.
The crystal properties HoCu2 and its hydrides are measured by X-ray diffraction (XRD) using Cu-K�� radiation. The XRD spectra are obtained by scanning with a step size 4 (��)/min in the range (diffraction angle) of 20��-80��. The product types, the crystallinity, and the average grain size can be indirectly obtained by analyzing the XRD pattern of the raw material and its hydrides.
The magnetization measurements of the hydrides were carried out using a SQUID magnetometer. The volumetric specific heat capacity values of HoCu2 and its hydrides were measured by the relaxation technique using PPMS.
3 Results and discussions
3.1 HoCu2 and properties of its hydrides
Similar to the intermetallic compounds for hydrogen storage [14], HoCu2 is produced by combining R-type element (Ho) forming stable hydride and M-type element (Cu) forming unstable hydride. Therefore, HoCu2 exhibits the intermediate hydrogenation properties between those of Ho and Cu.
Figure 2 shows the powder XRD patterns of the original compound and its ��hydride��. The results show that all the hydrides are various mixtures. At the three temperatures, the final ��hydrides�� are all the mixture of Cu, unknown hydride I and unknown hydride II. As for the peaks marked with ��, which could not match the PDF database, it means that it is not a known compound. However, the special peaks in the three patterns are in proportion, which indicates that those peaks are from the same compound. On the other hand, as for the peaks marked with ��, it could be either HoH2 or HoH3, because the peaks of the two compounds are almost the same. In conclusion, the hydride II should be some kind of compound made up of Ho and H, but the exact chemical formula cannot be determined. Although the species of the compounds in the three hydrides are the same, their properties are not. By analyzing the XRD patterns, the crystallinities of the original compound and the hydrides are both higher than 95%, namely they both appear in form of crystal. All the hydrogenation products (including unknown hydride I, unknown hydride II and Cu) are cubic crystals. As a result of the decomposition reaction, the average grain size of the hydrides is much smaller than that of the parent alloy, and decreases with the rising reaction temperature. The crystal structure, the crystallinity, and the average grain size of the original compound and the hydrides, obtained from Fig. 2, are shown in Table 1.

Fig. 2 X ray diffraction patterns of HoCu2 hydrides at different reaction temperatures (�ء�Cu; o��HoCu2; �ˡ�Unknown hydride I; ����Unknown hydride II)
According to the experimental results, one single HoCu2 compound can react with at least 3.1 hydrogen atoms (see Table 1). However, if the reaction mentioned above is the unique reaction (which indicates that the maximal hydrogen atoms reacted with one single HoCu2 compound are about 2.7), there must be some hydrogen atoms to hide in the spaces among the HoCu2 atoms in form of solid solution. To simplify, the mixture of Cu, unknown hydride I and unknown hydride II would be expressed into HoCu2Hx.
3.2 Hydrogenation properties
The influence of reaction temperature on the hydrogenation properties of HoCu2 has been investigated. The pressure-concentration-temperature curve (PCT curve) in Fig. 3 presents the thermodynamic characteristics of hydrogen storage alloys, including hydrogen absorption capacity, temperature, and equilibrium pressure in the hydrogenation. Three typical stages can be observed in the PCT curves. At the first stage, the metal initially dissolves some hydrogen atoms as a solid solution (��-phase). As the hydrogen pressure together with the concentration of H in the metal is increased, the interactions between hydrogen atoms become locally important, and the formation of nucleation and the hydride (��-phase) gradually reach at equilibrium. When these two phases coexist, the isotherms appear as flat plateau.
The hydrogen absorption characteristics of HoCu2 are given in Table 2. With the rising reaction temperature,the plateau pressure and the maximum hydrogen absorption capacity drop down. For instance, as the reaction temperature rises from 500 K to 600 K, the plateau pressure drops from 4.8 MPa to 3.0 MPa, and the maximum hydrogen absorption capacity decreases from 1.26% to 1.05%. The plateau slope factors of the PCT curves are also included in Table 2. By controlling the reaction temperature and the equilibrium pressure, we can obtain different phases and different hydrogen contents of hydride, which gives us a useful guide to preparing for our further investigation. The highest operating pressure in the chamber in the three reactions was 7 MPa, and the x in HoCu2Hx is 3.623, 3.149 or 3.103 when the reaction temperature is 500 K, 540 K or 600 K, respectively. The amount of hydrogen atoms absorbed by the alloy decreases as the reaction temperature rises, indicating that the hydrogen absorption process is exothermic. It has also been verified by our experiments. As the hydrogen is charged into the reaction chamber, the temperature would rise by 40-50 K almost immediately.
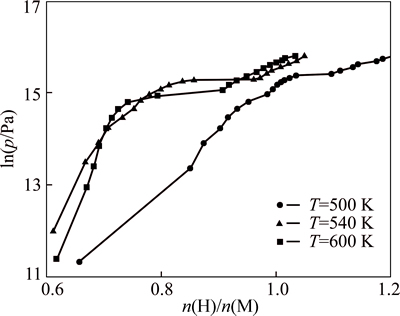
Fig. 3 PCT curves of HoCu2 hydrogenation
Table 1 Crystal structure, crystallinity, and average grain size of original compounds and hydrides
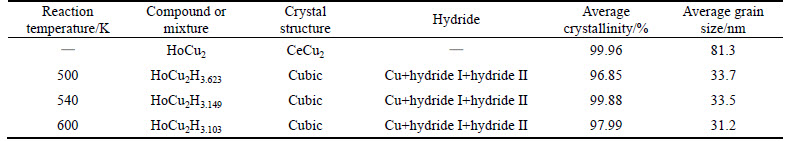
Table 2 Hydrogen absorption characteristics of HoCu2
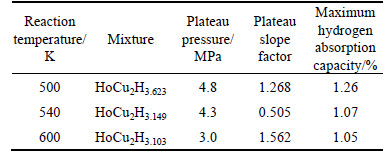
Based on the PCT curves of HoCu2 hydrogenation at different temperatures, the Van��t Hoff plot (logarithm of the equilibrium or plateau pressure against the reciprocal temperature) for hydrogenation is displayed in Fig. 4. The slope of the Van��t Hoff plot can be used to evaluate the whole experimental values of the hydride formation enthalpy ��H. The plateau pressure peq (T) as a function of reaction temperature can be correlated to the enthalpy change ��H and entropy change ��S, by the Van��t Hoff equation [15]:
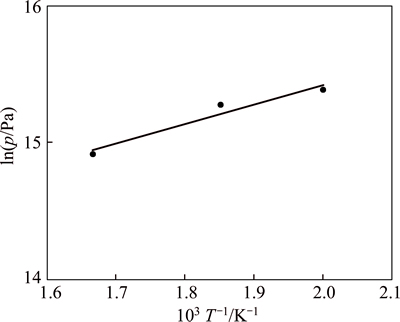
Fig. 4 Van��t Hoff plots of HoCu2
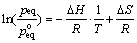 (4)
(4)
According to the Van��t Hoff equation, the enthalpy change ��H and entropy change ��S of the whole hydrogenation processes are -11.90 kJ/mol and 104.40 J/(K��mol), respectively. The enthalpy of reaction is negative, also indicating that the reaction is an exothermic process.
Hydrogen absorption in intermetallic compounds can be characterized by a mean hydrogen capacity of about 1.1 (hydrogen atom per metal atom). As shown in Fig. 4, the amount of hydrogen atoms absorbed by one HoCu2 atom is around 3.2, and the mass content of hydrogen atom is around 1.2%, which is not yet sufficient for a prospective hydrogen storage material. However, HoCu2 has already been used as an excellent magnetic regenerative material [8]. If the hydrides of HoCu2 are also expected to be used as a potential regenerative material for cryocoolers, further work such as the measurement of its physical properties still needs to be done.
3.3 Magnetization and volumetric specific heat capacity
The magnetic property of the hydride of HoCu2 was measured by a SQUID magnetometer, and the results are shown in Fig. 5. With the magnetic field of 0.01 T and the surrounding temperature from 2 K to 100 K, the ferromagnetism of the hydride varied from 2.7 A��m2/kg to almost 0. The magnetization decreases as the temperature rises. The hydride of HoCu2 is still in the state of magnetic material, similar to the parent compound.
The volumetric specific heat capacity values of HoCu2 and HoCu2H3.623 were measured via physical property measurement system (PPMS) as shown in Fig. 6. As the temperature rises from 2 K to 20 K, the volumetric specific heat capacity of HoCu2H3.623varies from 0.049 J/(K��cm3) to 0.226 J/(K��cm3),correspondingly. However, the volumetric specific heat capacity of the hydride is lower than the parent compound, which is out of our expectation.
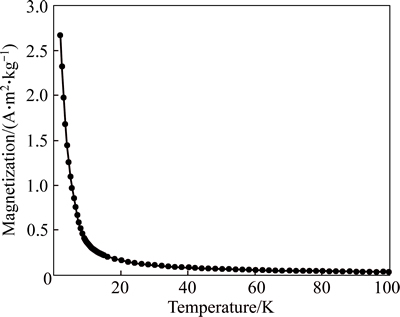
Fig. 5 Temperature dependence of magnetization of HoCu2H3.623 with magnetic field of 0.01 T
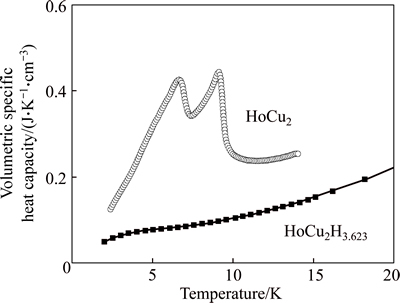
Fig. 6 Volumetric specific heat capacity of HoCu2 and HoCu2H3.623 between 2 K and 20 K
4 Conclusions
1) The crystallinity of the HoCu2 hydrides, with cubic crystal structures, is quite high. The average grain size of the hydrides decreases as the reaction temperature rises.
2) Some hydrogen atoms hide in the spaces among the HoCu2 atoms in form of solid solution.
3) The PCT curves of the hydrogenation of HoCu2 at three different temperatures are presented. The hydrogenation reaction is an exothermic process. Calculated from the PCT curves, the enthalpy change ��H and the entropy change ��S of the hydrogenation processes are -11.90 kJ/mol and 104.40 J/(K��mol), respectively.
4) To be expected as the regenerative materials of a cryocooler, the hydrides of HoCu2 should possess lower transition temperature and higher volumetric specific heat capacity. The reaction characteristics of HoCu2 hydrogenation have already provided a better understanding of the hydride products. Unexpectedly, the measured volumetric specific heat capacity of the hydride is lower than that of the parent compound. Further work is still necessary to explore the relation between the hydrogenation process and the properties of the hydrides.
References
[1] de Waele A T A M. Finite heat-capacity effects in regenerators [J].
Cryogenics, 2012, 52: 1-7.
[2] Hashimoto T, Ogawa M, Li R. Recent advance in magnetic regenerator material [J]. Cryogenics, 1990, 30: 192-198.
[3] JIN Tao, LI Cong-hang, TANG Ke, XU Bin, CHEN Li-xin. Hydrogen absorption characteristics and structural transformations during the hydrogenation process of Er3Ni [J]. Intermetallics, 2013, 32: 162-166.
[4] JIN Tao, LI Cong-hang, TANG Ke, CHEN Li-xin, XU Bin, CHEN Guo-bang. Hydrogenation-induced variation in crystal structure and heat capacity of magnetic regenerative material Er3Ni [J]. Cryogenics, 2011, 51: 214-217.
[5] WANG Dong, LI Yan-li, LONG Yi, YE Rong-chang, CHANG Yong-qin, WAN Fa-rong. Hydrogen induced structural and magnetic transformations in magnetic regenerator materials ErNin (n=1, 2) and HoCu2 [J]. Journal of Magnetism and Magnetic Materials, 2007, 311: 697-701.
[6] ZHANG Yang-huan, ZHAO Dong-liang, LI Bao-wei, MA Zhi-hong, GUO Shi-hai, WANG Xin-lin. Influence of substituting Ni with Co on hydriding and dehydriding kinetics of melt spun nanocrystalline and amorphous Mg2Ni-type alloys [J]. Journal of Central South University, 2011, 18: 303-309.
[7] ZHANG Yang-huan, QI Yan, REN Hui-ping, MA Zhi-hong, GUO Shi-hai, ZHAO Dong-liang. Hydriding and dehydriding kinetics of nanocrystalline and amorphous Mg2Ni1-xMnx (x=0-0.4) alloys prepared by melt spinning [J]. Journal of Central South University, 2011, 18: 985-992.
[8] Wallace W E, Malik S K, Takeshita T, Sankar S G, Gualtieri D M. Magnetic properties of hydrides of the rare earths and rare earth intermetallics [J]. Journal of Applied Physics, 2008, 49: 1486-1491.
[9] Li R, Ogawa M, Hashimoto T. Magnetic intermetallic compounds for cryogenic regenerator [J]. Cryogenics, 1990, 30: 521-526.
[10] LI Song-lin, LIU Yi, CUI Jian-min, YANG Wen-zhi, LI Hao-peng, HE Yi-lun. Synthesis and hydrogen desorption properties of Mg2FeH6 hydrogen storage material by reactive mechanical alloying [J]. Journal of Central South University: Science and Technology, 2008, 39(1): 1-6. (in Chinese)
[11] HUANG Tai-zhong, WU Zhu, YU Xue-bin, CHEN Jin-zhou, XIA Bao-jia, HUANG Tie-sheng, XU Nai-xin. Hydrogen absorption�C desorption behavior of zirconium-substituting Ti�CMn based hydrogen storage alloys [J]. Intermetallics, 2004, 12: 91-96.
[12] Gratz E, Sechovsky V, Sima V, Smetana Z, Str m- Olson J O. Magnetization, electrical resistivity, thermopower, and neutron diffraction in HoCu2 [J]. Physica Status Solidi (b), 1982, 111: 195-201.
m- Olson J O. Magnetization, electrical resistivity, thermopower, and neutron diffraction in HoCu2 [J]. Physica Status Solidi (b), 1982, 111: 195-201.
[13] Zavaliy Y, Cerny R, Verbetsky V N, Denys R V, Riabov A B. Interaction of hydrogen with RECu2 and RE(Cu, Ni)2 intermetallic compounds (RE=Y, Pr, Dy, Ho) [J]. Journal of Alloys and Compounds, 2003, 358: 146-151.
[14] Latroche M. Structural and thermodynamic properties of metallic hydrides used for energy storage [J]. Journal of Physics and Chemistry of Solids, 2004, 65: 517-522.
[15] Ives D J. Chemical Thermodynamics: With special reference to inorganic chemistry [M]. 1st ed. Wisconsin: Macdonald Technical and Scientific, 1971.
(Edited by YANG Hua)
Foundation item: Project(51276154) supported by the National Natural Science Foundation of China; Project(2012010111014) supported by the University Doctoral Subject Special Foundation of China
Received date: 2015-04-17; Accepted date: 2015-09-24
Corresponding author: JIN Tao, PhD; Tel/Fax: +86-571-87953233; E-mail: jintao@zju.edu.cn