
Effects of microwave pretreatment on zinc extraction from spent catalyst saturated with zinc acetate
ZHANG Ze-biao(�����)1, 2, ZHANG Zheng-yong(������)1, 2, NIU Hao(ţ ��)1, 2, PENG Jin-hui(�����)1, 2,
ZHANG Li-bo(������)1, 2, QU Wen-wen(������)2, PAN Hui-juan(�˻۾�)1, 2
1. Faculty of Metallurgical and Energy Engineering, Kunming University of Science and Technology,Kunming 650093, China;
2. Key Laboratory of Unconventional Metallurgy, Ministry of Education,Kunming University of Science and Technology, Kunming 650093, China
Received 6 July 2009; accepted 30 December 2009
_____________________________________________________________________________________________________
Abstract: A new technology for microwave pretreatment of spent catalyst on Zn extraction by HCl leaching was proposed and the temperature-change curve of spent catalyst under microwave irradiation was measured. The influence of microwave pretreatment temperature and microwave irradiation time on zinc extraction was investigated and the mechanism of microwave pretreatment for spent catalyst was analyzed. The results show that microwave pretreatment can greatly enhance the leaching rate of Zn. The Zn extraction reaches 96.58% under the conditions of microwave pretreatment temperature of 950 �� and the microwave irradiation time of 12 min. The blocked pores of spent catalyst can be opened through microwave pretreatment, increasing the contact area of leaching reagent and zinc.
Key words: microwave pretreatment; spent catalyst; leaching rate; zinc
_____________________________________________________________________________________________________
1 Introduction
Activated carbon saturated with zinc acetate is used as catalyst during the process of vinyl acetate production by the acetylene method. Fresh catalysts would deactivate after being used in a period of time, and these catalysts lose their catalytic property and become wastes [1]. They are usually deposited and stacked around the factories which cause great pollution to our environment. So far, several kinds of methods have been utilized to regenerate spent catalyst, such as washing by organic solvents, leaching with inorganic acid and ultrasonic irradiation methods[2-5]. But even under the optimum conditions, the leaching rate of zinc was less than 95%. The cause of the low extraction rate of above-mentioned method was due to the fact that zinc acetate on the catalyst could react with by-product and formed a resin-like polymer. Those materials would block and fill the pores of carbon, so that the contact areas between leaching agent and spent catalyst were largely diminished. This makes further extraction processing of spent catalyst difficult and costly. Therefore, how to increase the specific area of leaching agent and zinc acetate becomes the bottleneck which restricts the improvement of zinc extractive rate.
Microwave heating has gradually been utilized in extractive metallurgy in these years[6-8]. The main advantage of using microwave heating is that the shorter treatment time, which in many cases represents a reduction in the energy consumption as well. Recently, some studies focus on the application of microwaves for improving size reduction and mineral liberation[9-11]. After being pretreated by microwave heating, the leaching performance and leaching rate of raw materials can be improved effectively in the leaching processes [12-15]. This attribution to an effective mineral breakage and liberation improvement approach which has been observed to be very promising is the application of microwave irradiation[16].
Based on the advantage of microwave pretreatment mentioned above. In this work, a new technology for microwave pretreatment of spent catalyst of zinc in hydrochloric acid solution was developed, in order to find out the optimum condition for microwave pretreatment. The effect of microwave pretreatment on zinc leaching rate of spent catalyst saturated with zinc acetate was investigated systematically, in order to optimize the optimum conditions of microwave pretreatment, and make use of advantages of microwave heating highly efficient, selective, integral, uniform to decompose the resin-like polymer in the spent catalyst and open up the blocked pores. The mechanism of microwave pretreatment to spent catalyst was also studied, trying to lay a theoretical foundation preliminarily and providing necessary parameters for the industrial utilization of spent catalyst.
2 Experimental
2.1 Materials and equipment
Spent catalyst was obtained from a chemical plant in Yunnan Province, China, which contained 26% zinc acetate. And the main compositions of the spent catalyst are presented in Table 1. Before the experiment, the materials should be dried to constant mass at 110 ��, and then reserved in the dryer.
Table 1 Main compositions of spent catalyst (mass fraction, %)

Microwave pretreatment was carried out adopting the self-made microwave heating equipment which has a temperature measurement system, under the condition of output microwave power of 750 W at 2 450 MHz. The scheme of the microwave pretreatment experimental apparatus is shown in Fig.1.
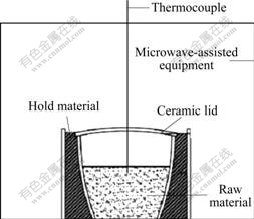
Fig.1 Scheme of microwave pretreatment experimental apparatus
2.2 Experimental method
100 g pretreated spent catalyst was weighed and exposed to the self-made microwave heating device, considering the different effects of pretreatment temperatures and holding times. The leaching condition chosen in this work was as follows: the leaching agent 30 g/L hydrochloric acid solution; the stirred time 60 min; the ratio of liquid to solid 4?1; and the temperature of microwave irradiation 950 ��. The zinc extraction was determined by analyzing zinc concentration using EDTA complexometric titration method. The X-ray diffraction (XRD, BRUKER D8 ADVANCE, Germany) measurement was used for XRD analysis using Cu K�� radiation. Scanning electron microscope (SEM, XL30ESEM-TMP, Philips) was used for the observation of the microscopic crystallographic structure.
3 Results and discussion
3.1 Temperature curve of spent catalyst by microwave heating
The temperature curve of spent catalyst by microwave heating is shown in Fig.2.
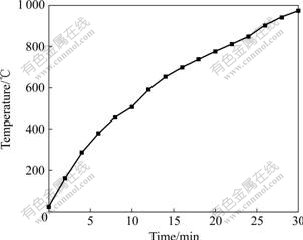
Fig.2 Temperature curve of spent catalyst by microwave heating
It could be seen that the temperature of samples increased rapidly with increasing microwave irradiation time, and after 30 min heating, the temperature almost reached 1 000 ��, showing that the spent catalyst was microwave absorbing material and its temperature could be improved rapidly by microwave irradiation.
3.2 Effect of different pretreated temperatures on leaching rate
The influence of different microwave pretreatment temperatures on the leaching rate was investigated at 650, 750, 850, 950 and 1 050 ��. When the heating temperature of the spent catalyst reached the pre-setting point, the sample was held for 6 min, and then was taken out from the device for cooling to room temperature for leaching test. The effect of different pretreatment temperatures on the leaching rate is shown in Fig.3.
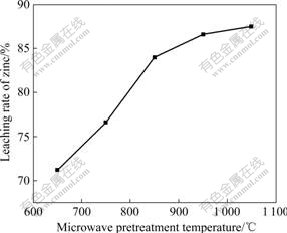
Fig.3 Relationship between microwave pretreatment temperature and leaching rate of zinc
It was indicated that the increase of microwave pretreatment temperature had a great effect on the leaching rate of zinc. When the pretreatment temperature was increased from 650 to 950 ��, the leaching rate of zinc increased from 71.2% to 86.9%. When the pretreatment temperature was 1 050 ��, the leaching rate increased slightly and the leaching rate was 87.5%. For the adsorption capacity of activated carbon contained in the spent catalyst was powerful, part of zinc was still adsorbed in the pores of activated carbon and it was hardly to be desorbed completely. Otherwise, the zinc oxide discomposed from the zinc acetate was reduced to zinc by carbon and evaporated, causing zinc loss and giving rise to a lower zinc leaching rate. In order to decrease evaporation of zinc, the microwave pretreatment temperature should not exceed 950 ��. So, the microwave pretreatment temperature was chosen to be 950 ��.
3.3 Effect of different holding times on leaching rate
The spent catalyst was pretreated at 950 �� by microwave irradiation, then the holding time was changed to test the leaching rate of the samples. The relationship between holding time and leaching rate of zinc is shown in Fig.4.
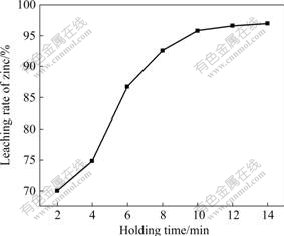
Fig.4 Relationship between holding time and leaching rate of zinc
It could be seen that the leaching rate increased with increasing holding time. When the holding time was 12 min, the leaching rate reached 96.58%, and when holding time was 14 min, the leaching rate was 96.95%, indicating that the leaching rate only increased slightly after 12 min of holding, which showed that 12 min of holding time for microwave pretreatment was enough for decomposition and opening the blocked pores of activated carbon. On the contrary, if the holding time was prolonged, the internal pores of the pretreated samples might collapse; and the interior structure of the spent catalyst might be burnt off, giving rise to a lower adsorption capacity of regenerated activated carbon. This is mainly due to the fact that, when the heating rate of microwave irradiation was higher, the spent catalyst could absorb microwave energy rapidly in a short time. The heat transfers from internal to external, therefore every portions of the spent catalyst were uniformly heated, resulting in more destruction to the zinc acetate and organic compounds which absorbed in the spent catalyst, so the blocked pores of the spent catalyst were opened in a short time.
3.4 Comparison of leaching test
Comparison of the results between the spent catalyst being pretreated at 950 �� for 12 min and the spent catalyst untreated by microware irradiation was made. It was shown that the leaching rate of the pretreated sample was 96.58%, while that of the untreated sample was 42.35%. This indicated that microwave pretreatment could play an important role in the improvement of spent catalyst leaching rate. This was mainly because microwave pretreatment was rapid. So, the blocked pores of the spent catalyst could be opened rapidly, then the zinc compound was exposed onto the surface of the activated carbon. The contact area between the leaching agent and the spent catalyst were increased, which was helpful for leaching agent entering into the pore canals, finally improving the leaching rate of zinc effectively.
4 Analysis of mechanism on microwave pretreatment process
XRD pattern and SEM image of the spent catalyst saturated with zinc acetate are shown in Fig.5 and Fig.6, respectively.
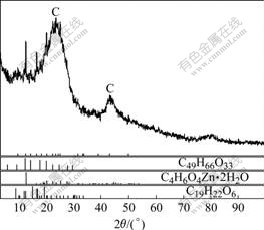
Fig.5 XRD pattern of spent catalyst

Fig.6 SEM image of spent catalyst
From Fig.6, it could be found that the main cause of catalyst deactivation was that the pores of activated carbon were blocked by the zinc acetate and organic compounds. XRD pattern indicated that the spent catalyst was mainly composed of zinc acetate and activated carbon, when the spent catalyst was heated, due to the different microwave absorption characteristics. The microwave absorption property of activated carbon was better than that of zinc acetate, resulting in different heating rates of microwave irradiation. Different parts of activated carbon were heated to different temperatures subsequently, giving rise to temperature gradient in the spent catalyst and causing the thermal stress between the heated activated carbon and zinc acetate.
When this thermal stress increased to a certain degree, cracks in the contact face turned out. In this situation, it was easier for the zinc compound to be separated from the pores of activated carbon, and it was easier to expose the zinc compound to the surface of activated carbon, which increased the effective contact area between leaching agent and spent catalyst.
When the microwave pretreated temperature reached a certain value, zinc acetate was decomposed into zinc oxide gradually. The XRD pattern of the spent catalyst pretreated at 950 �� by microwave irradiation for 12 min is shown in Fig.7.

Fig.7 XRD pattern of sample by microwave pretreatment
It could be seen that the diffraction peak of activated carbon (002) appeared at 2��=26?. This indicated that, as the temperature increased, the zinc acetate was gradually decomposed and the residual organic compound also transformed into volatile species, subsequently the pores of activated carbon were opened, so that the absorbing capacity of activated carbon gradually recovered. It was found from the SEM image in Fig.8 that, as the pretreatment temperature increased, the zinc oxide was reduced by the carbon, and part of the zinc vapor was pressed from internal to the external of the carbon, which made the pores of activated carbon become richer, resulting in a smaller crystal particles of zinc oxide and a larger contact area between zinc and leaching agent. So, the leaching rate of microwave pretreated samples was improved.

Fig.8 SEM image of sample by microwave pretreatment
5 Conclusions
Microwave heating has the advantages of internal and volumetric heating, which eliminates the cold centers of the conventional surface heating. The experimental results prove that microwave pretreatment can improve the leaching rate of zinc. XRD and SEM were employed to analyze the mechanism in microwave pretreatment process, and some interesting conclusions are summarized as follows:
1) The temperature-change curve of spent catalyst is measured under microwave irradiation, and the result indicates that the temperature of spent catalyst can be increased rapidly by microwave irradiation. The results of comparison of the leaching test show that microwave pretreatment has resulted in a great enhancement of the leaching of zinc.
2) The optimum conditions obtained are as follows: the microwave pretreatment temperature of 950 �� and the microwave irradiation time of 12 min. The rate of extraction reaches 96.58% under above-mentioned conditions.
3) Activated carbon and zinc acetate contained in the spent catalyst have different microwave absorption characteristics. This results in local temperature gradient and causes zinc compound to separate from pores of activated carbon, exposing the zinc compound to the surface of activated carbon. So, the blocked pores of activated carbon are opened, and the leaching agent can react with zinc easily.
References
[1] CHEN Chen, LIN Xing-yi, CHEN Xiao-hui, ZHENG Qi. Advances in the catalysts for synthesis of vinyl acetate from acetylene and acetic acid[J]. Industrial Catalysis, 2003, 11(11): 7-12. (in Chinese)
[2] MARAFI M, STANISLAUS A. Options and processes for spent catalyst handling and utilization[J]. Journal of Hazardous Materials, 2003, 101(2): 123-132.
[3] DABEK L. Sorption of zinc ions from aqueous solutions on regenerated activated carbons[J]. Journal of Hazardous Materials, 2003, 101(2): 191-201.
[4] LI W, PENG J H, ZHANG L B, ZHANG Z B, LI L, ZHANG S M, GUO S H. Pilot-scale extraction of zinc from the spent catalyst of vinyl acetate synthesis by microwave irradiation[J]. Hydrometallurgy, 2008, 92(1/2): 79-85.
[5] BIAO Ci-guang, ZHAO Yi-yun, FENG Ruo, ZHU Chang-pin. Recovery of active charcoal from waste catalyst via ultrasonic wave[J]. Journal of Nanjing University: Natural Sciences Edition, 1995, 31(2): 242-247. (in Chinese)
[6] AL-HARAHSHEN M, KINGMAN S M. Microwave leaching�CA review[J]. Hydrometallurgy, 2004, 73(3): 189-203.
[7] HAQUE K E. Microwave energy for mineral treatment processes�CA brief review[J]. International Journal of Mineral Processing, 1999, 57(1): 1-24.
[8] NANTHAKUMAR B, PICKLES C A, KELEBEK S. Microwave pretreatment of a double refractory gold ore[J]. Minerals Engineering, 2007, 20(11): 1109-1119.
[9] AMANKWAH R K, PICKLES C A. Microwave roasting of a carbonaceous sulphidic gold concentrate[J]. Minerals Engineering, 2009, 22(13): 1095-1101.
[10] LANGOVA ?, LE?KO J, MATYSEK D. Selective leaching of zinc from zinc ferrite with hydrochloric acid[J]. Hydrometallurgy, 2009, 95(3/4): 179-182.
[11] SCOTT G, BRADSHAW S M, EKSTEEN J J. The effect of microwave pretreatment on the liberation of a copper carbonatite ore after milling[J]. Hydrometallurgy, 2008, 85(4): 121�C128.
[12] AL-HARAHSHEN M, KINGMAN S M. The influence of microwaves on the leaching of sphalerite in ferric chloride[J]. Chemical Engineering and Processing: Process Intensification, 2007, 46(10): 883-888.
[13] KRISHNAN K H, MOHANTY D B, SHARMA K D. The effect of microwave irradiations on the leaching of zinc from bulk sulphide concentrates produced from Rampura�CAgucha tailings [J]. Hydrometallurgy, 2007, 89(3/4): 332-336.
[14] JONGES D A, LELYVELD T P, MAVROFIDIS S D, KINGMAN S W, MILES N J. Microwave heating applications in environmental engineering�CA review[J]. Resources, Conservation and Recycling, 2002, 34(2): 75-90.
[15] ZHU S D, WU Y X, YU Z N, WANG C W, YU F Q, JIN S W, DING Y G, CHI R A, LIAO J T, ZHANG Y. Comparison of three microwave/chemical pretreatment processes for enzymatic hydrolysis of rice straw[J]. Biosystems Engineering, 2006, 93(3): 279�C283.
[16] OLUBAMBI P A. In?uence of microwave pretreatment on the bioleaching behaviour of low-grade complex sulphide ores[J]. Hydrometallurgy, 2009, 95(1/2): 159-165.
____________________________
Foundation item: Project(2008DFA91500) supported by the China International Science and Technology Cooperation Program; Project(2006GH01) supported by the International Collaboration Project of Yunnan Provincial Science and Technology Department; Project(14051157) supported by Yunnan Provincial Natural Science Foundation
Corresponding author: PENG Jin-hui; Tel: +86-871-5191046; E-mail: jhpeng@kmust.edu.cn
(Edited by YANG You-ping)