通过KxCrOy便捷水解制备六方和无定型羟基氧化铬
来源期刊:中国有色金属学报(英文版)2020年第5期
论文作者:梁书婷 张红玲 徐红彬
文章页码:1397 - 1405
关键词:水解过程;CrOOH;氧化铬;活性KxCrOy;反应机理
Key words:hydrolysis process; CrOOH; chromium oxide; activated KxCrOy; reaction mechanism
摘 要:系统研究KCrO2和K3CrO4的水解过程和机理。XRD、IR和SEM的表征结果表明,在80 °C的低反应温度和24 h的反应时间下,可以实现水解反应。此外,通过水解单相六方KCrO2,能形成灰绿色六方片状直径为10 μm的α-CrOOH;而通过水解立方K3CrO4,能形成绿色无定形团聚体形态Cr(OH)3溶胶凝胶。通过上述水解反应合成纯相羟基氧化铬是一种非常简便与快速的方法。
Abstract: The hydrolysis process and mechanisms of unique as-prepared KCrO2 and K3CrO4 were systematically investigated. The characterization results of XRD, IR and SEM show that the hydrolysis reaction can be realized at a low reaction temperature of 80 °C and a reaction time of 24 h. Moreover, the greyish-green α-CrOOH with a hexagonal plate-like morphology and a large size of 10 μm is formed via the hydrolysis of the single-phase hexagonal KCrO2, while the green sol-gel of amorphous Cr(OH)3 with a lumpy aggregate morphology is generated through the hydrolysis of a cubic K3CrO4. It is a facile and rapid method to synthesize pure-phase chromium oxyhydroxide via the above hydrolysis.
Trans. Nonferrous Met. Soc. China 30(2020) 1397-1405
Shu-ting LIANG1,2, Hong-ling ZHANG3,4, Hong-bin XU3,4
1. College of Chemistry and Environmental Engineering, Chongqing University of Arts and Sciences, Chongqing 402160, China;
2. Chongqing Key Laboratory of Environmental Materials and Remediation Technologies, Chongqing University of Arts and Sciences, Chongqing 402160, China;
3. National Engineering Laboratory for Hydrometallurgical Cleaner Production Technology, Institute of Process Engineering, Chinese Academy of Science, Beijing 100190, China;
4. Key Laboratory of Green Process and Engineering, Institute of Process Engineering, Chinese Academy of Sciences, Beijing 100190, China
Received 9 June 2019; accepted 30 March 2020
Abstract: The hydrolysis process and mechanisms of unique as-prepared KCrO2 and K3CrO4 were systematically investigated. The characterization results of XRD, IR and SEM show that the hydrolysis reaction can be realized at a low reaction temperature of 80 °C and a reaction time of 24 h. Moreover, the greyish-green α-CrOOH with a hexagonal plate-like morphology and a large size of 10 μm is formed via the hydrolysis of the single-phase hexagonal KCrO2, while the green sol-gel of amorphous Cr(OH)3 with a lumpy aggregate morphology is generated through the hydrolysis of a cubic K3CrO4. It is a facile and rapid method to synthesize pure-phase chromium oxyhydroxide via the above hydrolysis.
Key words: hydrolysis process; CrOOH; chromium oxide; activated KxCrOy; reaction mechanism
1 Introduction
Hydrous chromium oxides (Cr2O3・xH2O) such as chromium hydroxide (Cr(OH)3) and chromium oxyhydroxide (CrOOH) offer a near-ideal combination of distinctive adsorption selectivity and magnetic properties. The remarkable CrOOH and Cr(OH)3 have piqued the interest of many researchers as a consequence of their potential applications as catalysts [1]{Rotter, 2004 #30;Knaak, 1976 #15}, colorants, fluorescent and magnetic bifunctional molecules [2-5]. The chromium oxide (Cr2O3) was also generated from thermal decomposition of CrOOH or Cr(OH)3 [3,6], and it was important in applications such as catalysts [7], functional pigments [8-11], hydrogen storage [12], gas sensors [13], batteries [14] or electrode [15], and metallic ferromagnetic material [16-18].
Nowadays, rhombohedral chromium oxyhydr- oxide (α-CrOOH) has been mostly prepared via the hydrothermal reaction of Cr(NO3)3・9H2O [19-22]. The α-CrOOH with different diameters [23], morphologies [24], adsorption properties [25] and catalytic properties has been prepared through the hydrothermal method. Based on the hydrothermal technique developed [18,19], the α-CrOOH was produced with a size of 10-300 nm. By controlling the synthetic parameters, especially the temperature and pH, the crystallinity of the final products would be greatly influenced [26]. This same hydrothermal reduction of chromate has already been experimentally applied to preparing Cr(OH)3, which was performed in a sealed autoclave to control the atmospheric pressure.
However, in the previous work, the high reaction pressure and temperature, complex process control, and unable monitoring of CrOOH during the crystal-growing process have been widely considered as the weakness. Unfortunately, as one of the crystallographically well-defined KxCrOy compounds, KCrO2 and K3CrO4 were extremely sensitive to moisture and oxygen [27]. They were very difficult to prepare and preserve in the air [28]. In particular, few studies have reported the reactions of KCrO2 and K3CrO4 compounds with water over the past few years.
In this current work, rhombohedral α-CrOOH and amorphous Cr(OH)3 were synthesized via a low-temperature and stress-free process, rather than the traditional preparation method. This alternative techniques could effectively reduce the high reaction temperature and pressure, save the reaction energy consumption, and also optimize the control and monitoring of products in the reaction process. In parallel with this, a new technical concept of hydrolysis of KxCrOy compounds was proposed, and the hydrolysis processes of KCrO2 and K3CrO4 were accidentally discovered, and a series of α-CrOOH and Cr(OH)3 were produced using this hydrolysis reaction. The hydrolysis processes of KCrO2 and K3CrO4 were performed in an inert atmosphere for the sake of preventing their oxidation. The crystal structures, chemical compositions, morphological characteristics, and sizes of hydrolysates (α-CrOOH and Cr(OH)3) were described in more detail. Finally, the formation mechanisms of α-CrOOH and Cr(OH)3 were deeply proposed, respectively.
2 Experimental
99.5% purity potassium chromate (K2CrO4) was purchased from Sinopharm Chemical Reagent Co., Ltd., and used to produce KCrO2 and K3CrO4 [29,30]. 99.99% purity hydrogen gas was purchased from Praxair Inc.. KCrO2 and K3CrO4 were produced via hydrogen reduction, which was reported in previous papers [29,30]. KCrO2 used in this work was obtained via hydrogen reduction of K2CrO4 at 650 °C for 2 h; while K3CrO4 was also obtained via hydrogen reduction of K2CrO4 at 450 °C for 0.5 h. A nickel boat, loaded with 5 g of K2CrO4, was placed into the furnace tube. Hydrogen was introduced into the tube at a flow rate of 0.6 L/min. After the reduction reaction was finished, different reduction products such as KCrO2 and K3CrO4 were added to distilled water immediately, which were maintained constant stirring for more than 24 h at 80 °C. Some greyish- green powdery substances observed in the solution were separated through centrifugation and dried at 100 °C for 12 h.
X-ray diffraction (XRD) patterns were recorded using a Rigaku diffractometer employing Cu Kα radiation. The infrared spectrum was recorded using a Spectro GX FT-IR spectrometer (Perkin-Elmer, USA) in KBr pellets. The morphological characteristic was performed using a JSM-6700F NT scanning electron microscopy (SEM), supplied by Japan. The transmission electron microscopy (TEM), selected area electron diffraction (SAED) and energy dispersive spectroscopy (EDX) were taken with a JEOLJEM 2010 operating at an acceleration voltage of 200 kV.
3 Results and discussion
3.1 Characterization of KCrO2
Using a facile and highly reproducible hydrogen reduction method, KCrO2 was prepared at 650 °C. As shown in Fig. 1(a), the XRD patterns of the synthesized precursor, show that all diffraction peaks can be indexed to the KCrO2 with a rhombohedral crystal structure, indicating that the precursor has a single crystalline phase. The lattice constants of the precursor obtained via the refinement of XRD data were a=0.3022 nm and c=1.7760 nm, which was consistent with the potassium chromate (KCrO2, JCPDS No. 28-0743). According to Ref. [6], KCrO2 was a green powder with a magnetic property, and it was extremely sensitive to moisture and oxygen as well. In the previous work, KCrO2 has been synthesized via an azide route or a nitrate route [26-28], which must be executed at a high temperature and dangerous pressure. Notably, KCrO2 could be prepared by hydrogen reduction of K2CrO4 in this work, which was more pressure-free and clean.
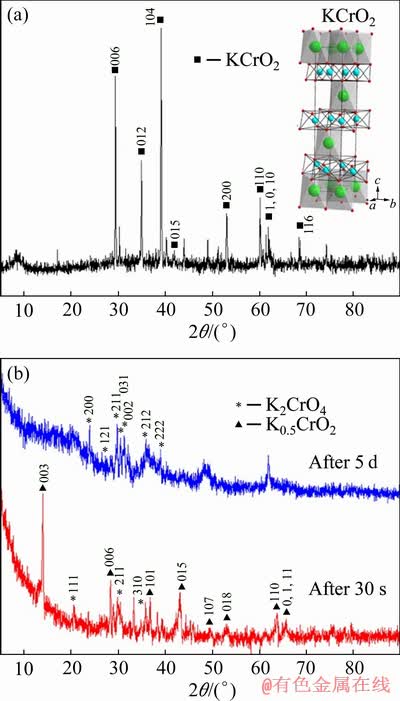
Fig. 1 XRD patterns of KCrO2 and crystal structure of KCrO2 (K (green spheres), Cr (blue spheres) and O (red spheres)) (a) and XRD patterns of oxidation products of KCrO2 in air at room temperature after 30 s and 5 d (b)
The oxidation reaction of KCrO2 occurred naturally at room temperature in air. The behavior of KCrO2 during oxidation with air was discussed. Figure 1(b) shows the XRD results for the oxidation of KCrO2. After exposing in air for 30 s, the KCrO2 was converted to a mixture of K0.5CrO2 and K2CrO4. It has been also reported that KCrO2 was immediately oxidized and yielded a mixture of K0.5CrO2 and K2CrO4 at room temperature in the air [27]. The conversion rate from KCrO2 to K0.5CrO2 was not 100% at room temperature. As illustrated in Fig. 1(b), the reduction product after exposing in air for 30 s contained a mixture of K0.5CrO2 (PDF No.00-28-0745), and K2CrO4 (PDF No. 00-15-0365).
The phase transition from K0.5CrO2 to K2CrO4 was observed as well, and the complete conversion took place after 5 d in air. Therefore, in an atmosphere of air, the most likely process was the conversion of KCrO2 to K0.5CrO2 and then the conversion of K0.5CrO2 to K2CrO4. Hence, it is very important to control the oxygen level at as low a level as possible in the next following hydrolysis process.
3.2 Hydrolysis of KCrO2 to prepare α-CrOOH
Considering the easy oxidation of KCrO2, KCrO2 was cooled naturally to ambient temperature in an inert atmosphere. In the next, KCrO2 (2 g) was immediately added to the 100 mL distilled water for analyzing the hydrolysis process.
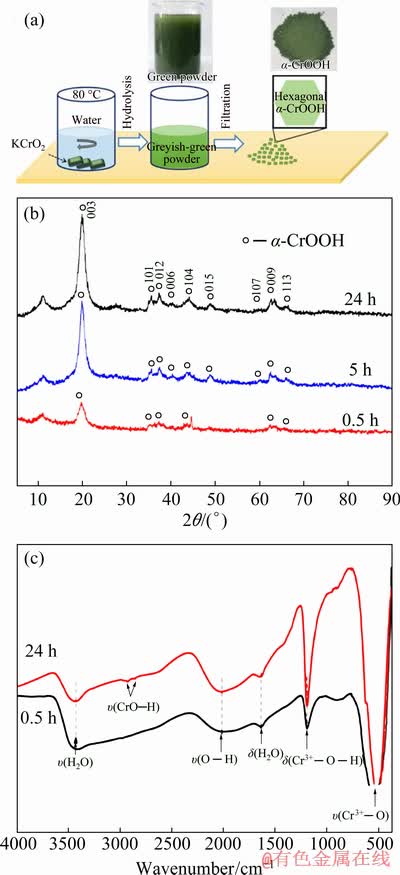
Fig. 2 Schematic diagram of hydrolysis process of KCrO2 and photograph of greyish-green hydrolysis product of KCrO2 (a), XRD patterns of hydrolysis products treated at 80 °C for 0.5, 5, 24 h (b) and FT-IR spectra of hydrolysis products treated for 24 and 0.5 h (c)
An overview of the hydrolysis taken in this work is presented in Fig. 2(a). This material was vigorously stirred at 80 °C for 24 h. After mild hydrolysis of KCrO2, quite a few greyish-green powdered substances were observed in the solution. This remaining solid-green powder was separated by centrifugation and then dried at 100 °C for 12 h. The hydrolysis process of different reduction products would be analyzed.
Different greyish-green powdered materials were formed through the hydrolysis of KCrO2 for different durations of 0.5, 5, and 24 h at 80 °C. As shown in Fig. 2(b), all the patterns observed from hydrolysis product showed the characteristic diffraction peaks of α-CrOOH with a rhombohedral crystal structure (space group of 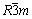 ), and lattice constants of a=0.2984 nm and c=1.34 nm, and all the peaks were in good agreement with the JCPDF No. 09-0331 [23]. From Fig. 2(b), hydrolysis duration could be deduced to have a great influence on the crystallization of α-CrOOH. The XRD peaks of the sample treated for 0.5 h were weak and broad (as shown in red line), indicating that the structure has poor degree of crystallization. Furthermore, hydrolysate after 24 h shows increase in all peak intensities (as shown in black line). The results suggest that the nucleation and growth of α-CrOOH crystallites could be accelerated by a longer hydrolysis duration.
), and lattice constants of a=0.2984 nm and c=1.34 nm, and all the peaks were in good agreement with the JCPDF No. 09-0331 [23]. From Fig. 2(b), hydrolysis duration could be deduced to have a great influence on the crystallization of α-CrOOH. The XRD peaks of the sample treated for 0.5 h were weak and broad (as shown in red line), indicating that the structure has poor degree of crystallization. Furthermore, hydrolysate after 24 h shows increase in all peak intensities (as shown in black line). The results suggest that the nucleation and growth of α-CrOOH crystallites could be accelerated by a longer hydrolysis duration.
The FT-IR absorption spectrum in Fig. 2(c) clearly demonstrated that samples treated for 24 h and 0.5 h showed five bands: 3247, 2017, 1635, 1190 and 599 cm-1. The strong sharp peak at 599 cm-1 corresponded to the Cr3+―O anti- symmetric stretching vibration [23]. Another strong sharp peak at 1190 cm-1 was assigned to Cr3+―O―H bending vibration [31]. A low- intensity band observed at 1635 cm-1 corresponded to the bending modes of non-dissociated water molecules [32]. The appearance of a broadband in the 1700-2100 cm-1 region was assigned to the O―H stretching vibration in the OH group of CrOOH [33]. While the other broadband in the 2800-3500 cm-1 region was assigned to the surface OH stretching groups originated from dissociation chemisorptions of water [34,35]. Table 1 lists the summary of FT-IR bands and their assignments of the hydrolysate. These bands were characteristics of α-CrOOH, which further indicated that the hydrolysis product was composed of α-CrOOH with a rhombohedral crystal structure.
A few representative SEM and TEM images were provided, which were used to determine the final morphology of the product after 24 h of hydrolysis. As shown in Fig. 3(a), the morphology of hydrolysate after treating for 24 h was hexagonal plate-like particles with a diameter of 10 μm.
A more complete set of SAED pattern as well as EDX mapping from the analyzed sample treated for 24 h was available in Figs. 3(b, c). The hexagonal single crystal particle of α-CrOOH gave a well-defined sixfold symmetric electron diffraction pattern, as shown in Fig. 3(b), confirming the existence of the crystalline structure of rhombohedral α-CrOOH. In the meanwhile, the selected area electron diffraction pattern of the hexagonal single crystal particle took along the 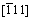 zone axis. As shown in the SAED patterns of Fig. 3(c), diffraction points, which were recorded from the α-CrOOH, could be identified as (101), (110), and (202) planes of the α-CrOOH. The corresponding EDS spectrum (Fig. 3(d)) highlighted the presence of only chromium, oxygen and potassium peaks. In addition, Cu and C could also be observed, which was attributed to the copper TEM sample copper grids and carbon film. In this research, oversize particle (10 μm) was obtained successfully via the hydrolysis technique.
zone axis. As shown in the SAED patterns of Fig. 3(c), diffraction points, which were recorded from the α-CrOOH, could be identified as (101), (110), and (202) planes of the α-CrOOH. The corresponding EDS spectrum (Fig. 3(d)) highlighted the presence of only chromium, oxygen and potassium peaks. In addition, Cu and C could also be observed, which was attributed to the copper TEM sample copper grids and carbon film. In this research, oversize particle (10 μm) was obtained successfully via the hydrolysis technique.
3.3 Characterization of K3CrO4
In the previous work, Cr2O3, KOH, and K2CrO4 were used as the raw materials for the typical route to synthesize K3CrO4, and the reaction was completed at 700 °C for approximately 8 h [24,25]. The dark-green K3CrO4 was also highly hygroscopic and sensitive, and it was spontaneous oxidation at room temperature. Various researchers have suggested that the oxidation state of Cr5+ was rather unstable. In this work, K3CrO4 was prepared through a simple low-temperature hydrogen reduction method. The XRD pattern of the prepared product is presented in Fig. 4(a) for crystal phase identification. It was obvious that the main lines were the diffraction peaks of the crystallized cubic K3CrO4 (lattice constants a=b=c=0.83368 nm, PDF No. 00-31-0994).
Table 1 FT-IR bands and assignments of hydrolysates (cm-1)

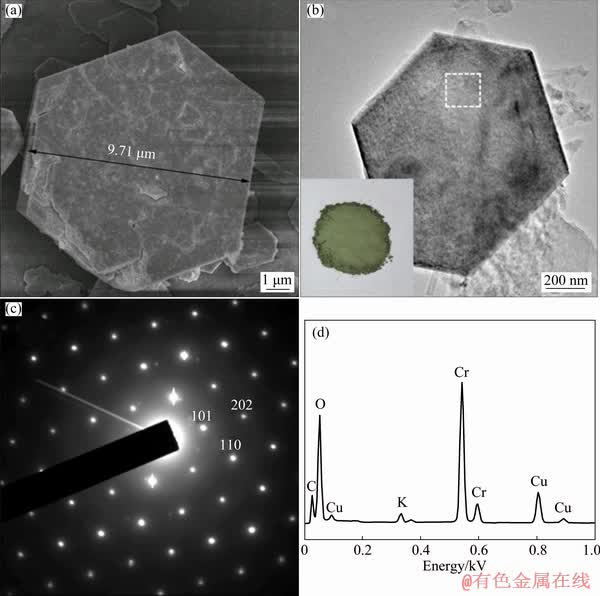
Fig. 3 SEM image (a), TEM image (b), SAED pattern (c) and EDX mapping (d) of hydrolysates (The location of the examined area of SAED pattern and EDX mapping is indicated in (b) with a frame. The region in the inset of (b) shows the photograph of the greyish-green powdered α-CrOOH)
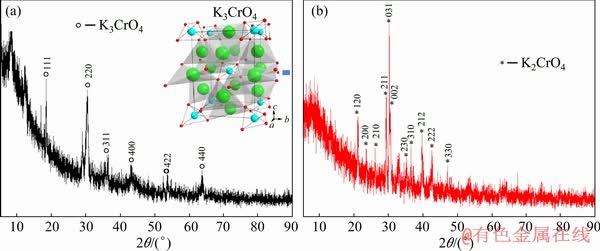
Fig. 4 XRD pattern of K3CrO4 and its crystal structure (K (green spheres), Cr (blue spheres), O (red spheres)) (a) and XRD pattern of oxidation product of K3CrO4 in air for 24 h at room temperature (b)
Figure 4(b) shows the XRD results for the oxidation of K3CrO4 in air at room temperature. It can be seen that the oxidation of K3CrO4 was an easy process in air. It was observed that the diffraction peak for K3CrO4 completely disappeared, whereas the diffraction peak for K2CrO4 appeared. The presence of air significantly promoted the K3CrO4 oxidation. After exposing the K3CrO4 and K2CrO4 system in air for 24 h, only the K2CrO4 remained.
3.4 Hydrolysis of K3CrO4 for preparing Cr(OH)3
The hydrolysis process of K3CrO4 was performed in an inert atmosphere for the sake of preventing its oxidation. After the hydrolysis of K3CrO4, several green flocculent substances were observed in the solution. Interestingly, when the solution was completely placed and heated at 60 °C for 2 h, a type of green sol-gel material was precipitated. The color of the solution was changed from green to yellow, which indicated that Cr6+ was generated. This obtained green gel-like precipitate was centrifuged at 10000 r/min for 10 min and washed three times with distilled water, as shown in Fig. 5(a). Figure 5(b) shows the XRD pattern of the green gel-like precipitate sample prepared by hydrolysis treatment for 0.5 and 24 h, all the hydrolysates presented an amorphous phase, which indicated that it was impossible to directly characterize the hydrolysates in this experiment.
To further understand the main component of the hydrolysis product of K3CrO4, the FT-IR absorption data were evaluated. The FT-IR absorption spectrum was collected and plotted in Fig. 5(c). As observed, the 540 cm-1 broadband corresponded to Cr3+―O lattice vibrations; at 919 cm-1, the peak corresponded to the bending vibration of Cr3+―O―H [31]. While three absorption peaks of Cr(OH)3 at 1377, 1497, and 1634 cm-1 [30], can also be observed in the spectra. The result indicated that the gel-like precipitate contained Cr(OH)3, which was directly generated by the hydrolyzing process of K3CrO4 in this system.
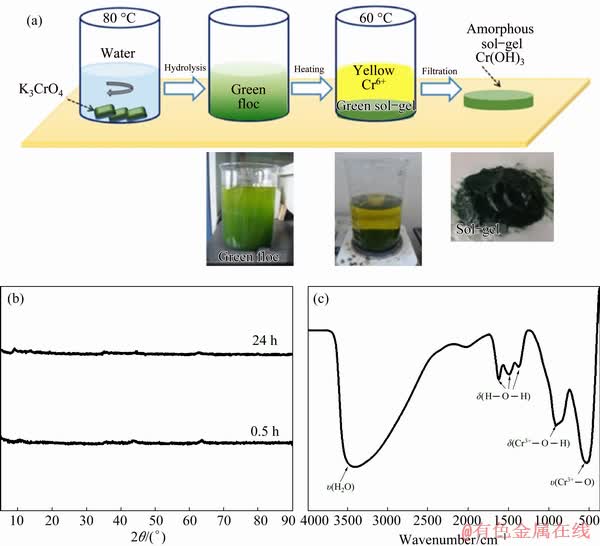
Fig. 5 Schematic diagram of hydrolysis process of K3CrO4 and photos of green flocculent solution and green sol-gel precipitate (a), XRD patterns of hydrolysis products treated at 80 °C for 0.5 and 24 h (b) and FT-IR spectrum of hydrolysates (c)

Fig. 6 TEM image (a), SAED pattern (b) and EDX mapping (c) of hydrolysates (The location of the examined area of SAED pattern and EDX mapping was indicated with a frame. The region in the inset of graph (a) showed the photographs of the Cr(OH)3)
Transmission electron microscopy (TEM) was used to investigate the final morphology and crystallographic properties of the Cr(OH)3 product. TEM analysis revealed that the surface of Cr(OH)3 product was relatively rough, lumpy, and irregular in Fig. 6(a). The SAED spectrum also strongly supported the view that Cr(OH)3 product was amorphous. Energy dispersive spectroscopy (EDS) was performed to further confirm the composition of the prepared product. The corresponding EDS spectrum highlighted the presence of only chromium, oxygen, and potassium peaks. Besides, Cu and C could also be observed, which was attributed to the copper TEM sample copper grids and carbon film.
4 Discussion
This experimental results discussed above confirmed that the formation of unique large hexagonal crystal may be attributed to the presence of KCrO2 with a rhombohedral crystal structure. The results of XRD, FT-IR and SEM analysis were used to explore the hydrolysis mechanism of KCrO2. It could be seen from the XRD and FT-IR results that the peak position of KCrO2 disappeared after hydrolysis, and the peak position of CrOOH appeared. Generally, the formation of α-CrOOH resulted from the decomposition of KCrO2. The possible hydrolysis reaction of the overall process was given as
KCrO2+H2O=α-CrOOH+KOH (1)
Compared with the former hydrothermal method, this novel intrinsic hydrolysis approach has many preponderance for the preparation of α-CrOOH. The hydrolysis treatment is very facile. KCrO2 could thoroughly hydrolyze just though slightly extending hydrolysis duration and increasing the temperature (80 °C, 24 h), which avoided other complicated adjusting processes.
In a recent review, BAMBERGER et al [36] reported that K3CrO4 reacted with water to form hydrosoluble Cr3+, potassium chromate (K2CrO4) and potassium hydroxide (KOH). After boiling for approximately 8 min, the hydrosoluble Cr3+ formed a solid residue of amorphous chromic hydroxide (Cr(OH)3). JOHNSON et al [37] determined that the reaction of K3CrO4 with aqueous acid at 25 °C led to solutions with two-thirds chromium in the hexavalent oxidation state and one-third chromium in the trivalent oxidation state. More recently, by measuring UV-Vis absorption and back-titrated 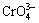 , BAILEY et al [38] have reported that a fused solution of
, BAILEY et al [38] have reported that a fused solution of 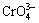 was rapidly oxidized to
was rapidly oxidized to 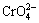 in oxygen, and a solution of
in oxygen, and a solution of 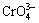 in potassium hydroxide was quickly and disproportionately converted into 2Cr6+ and Cr3+.
in potassium hydroxide was quickly and disproportionately converted into 2Cr6+ and Cr3+.
The experimental results of the hydrolysis of K3CrO4 in this work could be indexed as Cr(OH)3 and K3CrO4, which was consistent with the previous reported result. In air, K3CrO4 reacted with water vapor to form two-thirds of K2CrO4 (Cr6+) and one-third of Cr(OH)3 (Cr3+). As previously discussed, the peak position of K2CrO4 can be obtained from XRD data, which proved the formation of K2CrO4. At the same time, the formation of Cr(OH)3 could be proved from FT-IR data and product morphology. As a result, after exposing K3CrO4 in air, one of the product had a XRD phase of K2CrO4, and Cr(OH)3 was amorphous or had poor crystallinity. The possible hydrolysis reaction of K3CrO4 was
3K3CrO4+4H2O=Cr(OH)3+2K2CrO4+5KOH (2)
5 Conclusions
(1) The obtained α-CrOOH was crystallo- graphically pure, and it was identified with a hexagonal plate-like morphology with a grain size of 10 μm.
(2) The prepared amorphous Cr(OH)3 was identified with lumpy aggregate morphology.
(3) The reaction temperature was only 80 °C and the reaction time was 24 h. In addition, increasing the hydrolysis duration during the crystallization was helpful to increasing the product purity.
(4) This process would be a better alternative for producing Cr(OH)3 and CrOOH compounds due to its easier implementation and higher flexibility.
References
[1] KNAAK J F. γ-CrOOH fluorination catalysts: US Patent, 3992325 [P]. 1976-11-16.
[2] ADBELSADEK M S, BABU S M. A controlled approach for synthesizing CdTe@CrOOH (core-shell) composite nanoparticles [J]. Current Applied Physics, 2011, 11: 926-932.
[3] HUANG Zhong-lin, CHEN Chang-guo, XIE Ji-yun, WANG Zeng-xiang. The evolution of dehydration and thermal decomposition of nanocrystalline and amorphous chromium hydroxide [J]. Journal of Analytical and Applied Pyrolysis, 2016, 118: 225-230.
[4] SU Chang-qing, LI Li-qing, YANG Zhi-hui, CHAI Li-yuan, LIAO qi, SHI Yan, LI Jia-wei. Cr(VI) reduction in chromium-contaminated soil by indigenous microorganisms under aerobic condition [J]. Transactions of Nonferrous Metals Society of China, 2019, 29: 1304-1311.
[5] HU Jin, MENG De-long, LIU Xue-duan, LIANG Yi-li, YIN Hua-qun, LIU Hong-wei. Response of soil fungal community to long-term chromium contamination [J]. Transactions of Nonferrous Metals Society of China, 2018, 28: 1838-1846.
[6] PARDO P, CALATAYUD J M, ALARCON J. Chromium oxide nanoparticles with controlled size prepared from hydrothermal chromium oxyhydroxide precursors [J]. Ceramics International, 2017, 43: 2756-2764.
[7] MAHMOUD H R. Novel mesoporous Gd3+ doped Cr2O3 nanomaterials: Synthesis, characterization, catalytic and antitumor applications [J]. Advanced Powder Technology, 2016, 27: 1446-1452.
[8] SANGEETHA S, BASHA R, SREERAM K J, SANGILIMUTHU S N, UNNI NAIR B. Functional pigments from chromium(III) oxide nanoparticles [J]. Dyes and Pigments, 2012, 94: 548-552.
[9] LIANG Shu-ting, ZHANG Hong-ling, LUO Min-ting, LUO Ke-jun, LI Ping, XU Hong-bin, ZHANG Yi. Colour performance investigation of a Cr2O3 green pigment prepared via the thermal decomposition of CrOOH [J]. Ceramics International, 2014, 40: 4367-4373.
[10] WEI Guang-ye, QU Jing-kui, YU Zhi-hui, LI Yong-li, GUO Qiang, QI Tao. Mineralizer effects on the synthesis of amorphous chromium hydroxide and chromium oxide green pigment using hydrothermal reduction method [J]. Dyes and Pigments, 2015, 113: 487-495.
[11] LI Zhong-fu, DU Yi, CHEN Zhong-tao, SUN Dan-dan, ZHU Chao-feng. Synthesis and characterization of cobalt doped green ceramic pigment from tannery sludge [J]. Ceramics International, 2015, 41: 12693-12699.
[12] POLANSKI M, BYSTRZYCKI J, VARIN R A, PLOCINSKI T, PISAREK M. The effect of chromium (III) oxide (Cr2O3) nanopowder on the microstructure and cyclic hydrogen storage behavior of magnesium hydride (MgH2) [J]. Journal of Alloys and Compounds, 2011, 509: 2386-2391.
[13] SURYAWANSHI D N, PATIL D R, PATIL L A. Fe2O3-activated Cr2O3 thick films as temperature dependent gas sensors [J]. Sensors and Actuators B: Chemical, 2008, 134: 579-584.
[14] ABBAS S M, AHMAD N, ATA UR R, RANA U A, KHAN S U D, HUSSAIN S, NAM K W. High rate capability and long cycle stability of Cr2O3 anode with CNTs for lithium ion batteries [J]. Electrochimica Acta, 2016, 212: 260-269.
[15] MARUTHAPANDIAN V, MURALIDHARAN S, SARASWATHY V. Liquid-free alkaline gel filled reference electrode based on Cr2O3 spheres [J]. Indian Journal of Chemistry A, 2015, 54: 1215-1220.
[16] ZHANG Xiao-yu, CHEN Ya-jie, LI Zhen-ya. Large magnetocaloric effect in chromium dioxide with second-order phase transition [J]. Journal of Physics D: Applied Physics, 2007, 40: 3243-3247.
[17] BELEVTSEV B, DALAKOVA N, OSMOLOWSKY M, BELIAYEV E Y, SELUTIN A. Transport and magnetotransport properties of cold-pressed CrO2 powder, prepared by hydrothermal synthesis [J]. Journal of Alloys and Compounds, 2009, 479: 11-16.
[18] PEI Zhen-zhao, XU Hong-bin, ZHANG Yi. Preparation of CrO2 nanoparticles via oxidation method [J]. Materials Letters, 2012, 76: 205-207.
[19] LAUBENGAYER A W, MCCUNE H W. New crystalline phases in the system chromium(III) oxide-water [J]. Journal of the American Chemical Society, 1952, 74: 2362-2364.
[20] CHRISTENSEN A N. Hydrothermal preparation and magnetic-properties of Alpha-CrOOH, Beta-CrOOH, and Gamma-CrOOH [J]. Acta Chemica Scandinavica A―Physical and Inorganic Chemistry, 1976, 30: 133-136.
[21] CHRISTEN A N. Crystal structure of a new polymorph of CrOOH [J]. Inorganic Chemistry, 1966, 5: 1452-1453.
[22] HAMILTON W C, IBERS J A. Structures of HCrO2 and DCrO2 [J]. Acta Crystallographica, 1963, 16: 1209-1212.
[23] YANG Jing, MARTENS W N, FROST R L. Transition of chromium oxyhydroxide nanomaterials to chromium oxide: A hot-stage Raman spectroscopic study [J]. Journal of Raman Spectroscopy, 2010, 42: 1142-1146.
[24] KITTAKA S, TAHARA T. Thermal decomposition of chromium oxide hydroxide: II. Texture change of α-HCrO2 through thermal decomposition under vacuum [J]. Journal of Colloid and Interface Science, 1986, 112: 252-260.
[25] KITTAKA S, FUJINAGA R, MORISHIGE K, MORIMOTO T. Adsorption of water on the surfaces of α-HCrO2 and Cr2O3 [J]. Journal of Colloid and Interface Science, 1984, 102: 453-461.
[26] YANG Jing, BAKER A, LIU Hong-wei, MARTENS W, FROST R. Size-controllable synthesis of chromium oxyhydroxide nanomaterials using a soft chemical hydrothermal route [J]. Journal of Materials Science, 2010, 45: 6574-6585.
[27] ALI N Z, NUSS J, JANSEN M. A new polymorph of potassium chromate(III), β-KCrO2, and reinvestigation of α-KCrO2 [J]. Journal of Inorganic and General Chemistry, 2013, 639: 241-245.
[28] DELMAS C, DEVALETTE M, FOUASSIER C, HAGENMULLER P. Les phases KXCrO2 (X≤1) [J]. Materials Research Bulletin, 1975, 10: 393-398.
[29] LI Ping, XU Hong-bin, ZHENG Shi-li, ZHANG Yi, LI Zuo-hu, BAI Yu-lan. A green process to prepare chromic oxide green pigment [J]. Environmental Science & Technology, 2008, 42: 7231-7235.
[30] BAI Yu-lan, XU Hong-bin, ZHANG Yi, LI Zuo-hu. Reductive conversion of hexavalent chromium in the preparation of ultra-fine chromia powder [J]. Journal of Physics and Chemistry of Solids, 2006, 67: 2589-2595.
[31] RATNASAMY P, LEONARD A J. Structural evolution of chromia [J]. The Journal of Physical Chemistry, 1972, 76: 1838-1843.
[32] PEI Zhen-zhao, XU Hong-bin, ZHANG Yi. Preparation of Cr2O3 nanoparticles via C2H5OH hydrothermal reduction [J]. Journal of Alloys and Compounds, 2009, 468: L5-L8.
[33] DOUGLASS R M. The crystal structure of HCrO2 [J]. Acta Crystallographica, 1957, 10: 423-427.
[34] YAO Zhi-mao, LI Zuo-hu, ZHANG Yi. Studies on thermal dehydration of hydrated chromic oxide [J]. Journal of Colloid and Interface Science, 2003, 266: 382-387.
[35] SNYDER R G, IBERS J A. O―H―O and O―D―O potential energy curves for chromous acid [J]. The Journal of Chemical Physics, 1962, 36: 1356-1360.
[36] BAMBERGER C E, DONALD M, RICHARDSON D M. Chemical cycle for thermochemical production of hydrogen from water: US Patent, 3927192 [P]. 1975-12-16.
[37] JOHNSON L H, HEPLER L G, BAMBERGER C E, RICHARDSON D M. The enthaply of formation of potassium chromate(V), K3CrO4(c) [J].Canadian Journal of Chemistry, 2011, 56: 446-449.
[38] BAILEY N, SYMONS M C R. Structure and reactivity of the oxyanions of transition metals. Part III. The hypochromate ion [J]. Journal of the Chemical Society, 1957, 1: 203-207.
梁书婷1,2,张红玲3,4,徐红彬3,4
1. 重庆文理学院 化学与环境工程学院,重庆 402160;
2. 重庆文理学院 环境材料与修复技术重庆市重点实验室,重庆 402160;
3. 中国科学院 过程工程研究所 湿法冶金清洁生产技术国家工程实验室,北京 100190;
4. 中国科学院 过程工程研究所 绿色过程与工程重点实验室,北京 100190
摘 要:系统研究KCrO2和K3CrO4的水解过程和机理。XRD、IR和SEM的表征结果表明,在80 °C的低反应温度和24 h的反应时间下,可以实现水解反应。此外,通过水解单相六方KCrO2,能形成灰绿色六方片状直径为10 μm的α-CrOOH;而通过水解立方K3CrO4,能形成绿色无定形团聚体形态Cr(OH)3溶胶凝胶。通过上述水解反应合成纯相羟基氧化铬是一种非常简便与快速的方法。
关键词:水解过程;CrOOH;氧化铬;活性KxCrOy;反应机理
(Edited by Xiang-qun LI)
Foundation item: Project (R2018SCH02) supported by the High-level Talents Foundation of Chongqing University of Art and Sciences, China; Project (P2018CH10) supported by Major Cultivation Program of Chongqing University of Arts and Sciences, China; Project (cstc2019jcyj-msxmX0788) supported by the Natural Science Foundation of Chongqing, China; Project (KJQN201901342) supported by the Science and Technology Research Program of Chongqing Municipal Education Commission, China
Corresponding author: Shu-ting LIANG; Tel: +86-23-61162725; E-mail: stliang@mail.tsinghua.edu.cn
DOI: 10.1016/S1003-6326(20)65305-5

