J. Cent. South Univ. (2012) 19: 2434-2439
DOI: 10.1007/s11771-012-1293-x
Preparation of titanium powders by
calciothermic reduction of titanium dioxide
WAN He-li(�����)1,2,3,4, XU Bao-qiang(�챦ǿ)1,2,3,4, DAI Yong-nian(������)1,2,3,4,
YANG Bin(���)1,2,3,4, LIU Da-chun(����)1,2,3,4, SEN Wei(ɭά)1,2
1. National Engineering Laboratory for Vacuum Metallurgy
(Kunming University of Science and Technology), Kunming 650093, China;
2. State Key Laboratory of Breeding Base of Complex Nonferrous Metal Resources Clear Utilization in
Yunnan Province (Kunming University of Science and Technology), Kunming 650093, China;
3. Key Laboratory for Nonferrous Metals Vacuum Metallurgy of Yunnan Province
(Kunming University of Science and Technology), Kunming 650093, China;
4. Faculty of Metallurgy and Energy Engineering,Kunming University of Science and Technology, Kunming 650093, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2012
Abstract: To develop an effective process for titanium powders production, a calciothermic reduction process of pigment titanium dioxide (w(TiO2)>98%), based on the preform reduction process (PRP), was investigated by means of XRD, SEM and EDS. In this process, the mixture of TiO2 powder and CaCl2 was pressed into pieces as feed preform and was reduced by calcium vapor. Titanium powders was recovered after leaching from the reduced preform with hydrochloric acid and deionized water. The results indicate when the mass ratio of CaCl2 to TiO2 is about 1:4 and at a constant temperature of 1 273 K for 6 h in vacuum furnace, titanium powders with 99.55% purity by EDS analysis and irregular shape (8-15 ��m in particle size) are obtained.
Key words: titanium powders; titanium dioxide; calciothermic reduction; calcium chloride
1 Introduction
Titanium has attracted much attention because of its excellent performance such as high strength, light weight, high-temperature performance, excellent flexibility, extraordinary corrosion-resistance, and biocompatibility characteristics [1]. Its expanding application makes the demand for titanium increase constantly in the 21st century. Currently, the Kroll process [2] is widely used to produce titanium sponge [3-5]. But it has high production cost due to long production process, and slow and batch-type reduction process [6-7].
In order to find a better way to overcome the drawback of the Kroll process, many methods have been developed during past decades, especially the direct reduction processes of titanium oxide such as the FFC process [8-9], MHR process [10], OS process [11-12] and EMR process [13]. However, there are some problems needed to be solved before these processes can be commercialized. Specifically, a large amount of molten salt is required, and the reduction process is slow. Furthermore, the process lacks the flexibility to be scaled up, as they are based on electrochemical reduction. OKABE et al [14] and XU et al [15] investigated a new calcium vapor reduction process for producing fine titanium powders by means of PRP (preform reduction process) as shown in Fig. 1. In this method, the feed preform was fabricated from slurry, by mixing titanium dioxide (TiO2) powder, flux (CaCl2 or CaO) and a collodion solution, then it was placed on the stainless steel plate. The titanium sponge was also placed at the bottom of the vessel for gettering nitrogen gas in the closed system.
In this process, product purity and scalability are easy to control. The contamination from the reaction container or reductant in products can easily be avoided because the feed materials have no physical contact with them. Titanium powders with 99% purity were obtained by this way. Furthermore, the amount of CaCl2 can be reduced, compared to the direct electrochemical reduction processes mentioned above.
In this work, based on the PRP process, a brief process was proposed to reduce TiO2 to metallic Ti, using calcium as a reductant and CaCl2 as the additive, without adding any binder and titanium sponge in the reactor. Reaction vessel was made of high purity graphite crucible instead of stainless steel crucible to ensure reactor working at a high enough temperature. The purpose of this work is to report the above concept in detail and to show some experimental verification.
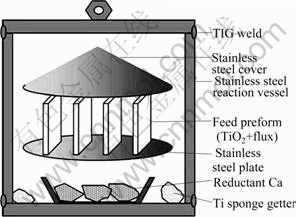
Fig. 1 Schematic illustration of preform reduction process (PRP)
2 Experimental
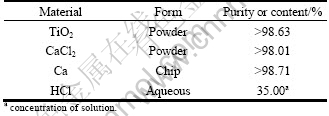
The purity and form of the starting materials used in this work are listed in Table 1. CaCl2 was employed as the additives [12, 14, 16]. The powder mixture of TiO2 and CaCl2 was manually pressed into small pieces at the pressure of 2 MPa (20 mm in diameter and 5.0-7.0 mm in thickness). The pieces were placed in a porous stainless steel vessel, and the calcium was placed at the bottom of a graphite crucible, as shown in Fig. 2. The graphite crucible was heated in the vacuum furnace. In order to reduce TiO2 effectively, excessive calcium was put into the reaction container.
Table 1 Starting materials used in experiments
The experiment included four major steps as follows: 1) The mixed powder was heated at 353-393 K before making pieces in order to remove the water; 2) The mixed powders or preformed pieces of CaCl2 and TiO2 were heated to 1 273 K and held for hours, respectively; 3) The reduction product was dissolved by low content hydrochloric acid (3%-3.5%) and deionized water repeatedly for 4-6 h; 4) The product was dried in a vacuum oven at 373 K, then titanium powders were obtained.
The microstructure and the surface morphology of reduction product were characterized by scanning electron microscopy (SEM: XL30ESEM-TMP, Phillips, Holland). The elemental content of the product was measured by the energy dispersive X-ray analysis (EDS: EDAX, PHOENIXTM, USA) and the phase structure and composition of reduced products were identified by X-ray diffraction (XRD: Japan. D/max-3B).
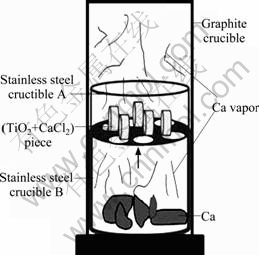
Fig. 2 Schematic illustration of experimental apparatus
3 Results and discussion
3.1 Calcium chloride volatilization
The pieces with different mass ratios of CaCl2 to TiO2 at 1 000 ��C for 6 h were sintered. The results can be seen in Table 2. Scanning electron microscope (SEM) was employed to observe the surface morphology and the size of pores are shown in Fig. 3. When the CaCl2 is not added, the pores are not formed in the sintered products, as shown in Fig. 3(a). It can be seen that the pores are significantly different when proportions of the mixture change. As shown in Fig. 3(b), the smaller the amount of CaCl2 is added (the mass ratios of CaCl2 to TiO2 is less than 1:6), the fewer the pores in volatilization product are, lending to the densification of internal structure. However, when the large amount of CaCl2 is added (the mass ratio of CaCl2 to TiO2 is more than 1:1), the pores become less, which are not uniformly distributed, as shown in Fig. 3(d). When the mass ratio of CaCl2 to TiO2 is controlled at 1:2-1:1.5, more pores are obtained and distributed homogeneously, as shown in Fig. 3(c). The average diameter of pores is 25 ��m, which would increase the contact surface area between calcium vapor and TiO2 particles and help to improve gas-solid reduction process.
Table 2 Different proportions of CaCl2 and TiO2 used in experiments
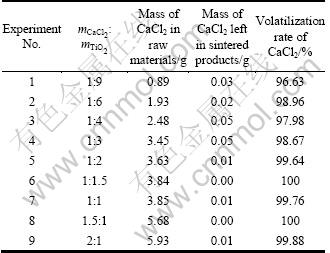
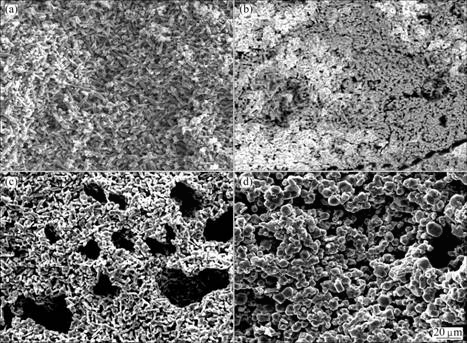
Fig. 3 SEM micrographs of surface with different mass ratios of CaCl2 to TiO2: (a) 0; (b) 1:6; (c) 1:1.5; (d) 1:1
3.2 Effects of additive of CaCl2 on reaction products
Figure 4 shows XRD patterns of products after calciothermic reduction when TiO2 powders, mixed powders of TiO2-CaCl2 and preform pieces of TiO2-CaCl2 are used as the feed materials, respectively. From Fig. 4(a), it can be seen when TiO2 powder is used as the raw material, the product is composed of Ti2O3, TiO, Ti and CaO. It is indicated that TiO2 powders are not completely reduced by calcium vapor because the exothermic reduction process of TiO2 by metallic Ca makes the surface of TiO2 powders sintered, the contact area of sintered TiO2 powder with calcium vapor becomes small, and the oxygen of TiO2 is not removed by calcium effectively.
A graph of the Gibbs free energy changes (��GT) related to the temperature (T) is drawn to clarify these processes (Fig. 5). Because Ti2O3 and TiO exist in the products, TiO2��Ti3O5, Ti3O5��Ti2O3, Ti2O3��TiO, and TiO��Ti are analyzed in these processes:
3TiO2+Ca(g)=Ti3O5+CaO (1)
2Ti3O5+Ca(g)=3Ti2O3+CaO (2)
Ti2O3+Ca(g)=2TiO+CaO (3)
TiO+Ca(g)=Ti+CaO (4)
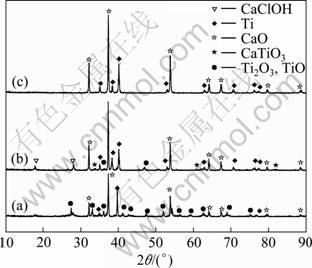
Fig. 4 XRD patterns of reduced products from different raw materials at 1 273 K for 6 h: (a) TiO2 powder; (b) TiO2 and CaCl2 powder; (c) TiO2 and CaCl2 pieces
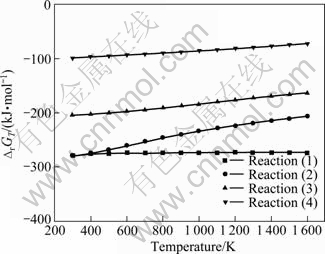
Fig. 5 Relationships between Gibbs free energy changes and temperature
As shown in Fig. 5, these reactions will happen. And it is not conducive to the reduction reaction when the temperature increases. TiO2 would be reduced step by step, and Reaction (4) will be harder than others, which implies that it might be the rate-limiting step on the whole reduction process of titanium dioxide by calcium vapor. In addition, the kinetic conditions, such as reaction activation energy, grain distribution of TiO2 solid, and contact area of reactants, will control this gas-solid reduction process [17].
Figure 4(b) shows XRD pattern of the mixture of powder sample reduced by calcium vapor at 1 273 K for 6 h. The product contains titanium, CaClOH and perovskite calcium titanate (CaTiO3). CaClOH could be resulted from the hydrolysis of CaCl2 after absorbing water as
CaCl2��H2O = CaClOH + HCl�� (5)
CaClOH = CaO + HCl�� (6)
CaO+TiO2= CaTiO3 (7)
When the powder material is reduced, it has less contact area with calcium vapor than pieces materials, which is resulted from the sintering layer on the surface of powders and by-product of CaO on the powder surface preventing calcium vapor from reacting with TiO2, and Ti2O3 and TiO are hardly reduced. Figure 4(c) shows the XRD pattern of the pieces of sample reduced by calcium vapor at 1 273 K for 6 h. The product is almost composed of Ti and CaO after reduction. So the pieces sample is chosen to reduce the effect of sintering in the experiment.
It is reported that by-product of CaO could be dissolved in large amount of molten CaCl2 [18-20], which can improve reduction process. When the mixture of CaCl2 and TiO2 with different mass ratios is used as raw material, the morphology of reduction product is observed by scanning electron microscopy (SEM), as shown in Fig. 6. Seen from Fig. 6(a), the molten surface is formed and the pore in feed is not obvious because of melting of CaCl2 [21]. When the amount of CaCl2 decreases in the raw materials, the molting phenomenon on the surface of samples is relieved, as shown in Figs. 6(b-d). As shown in Fig. 6(e), without CaCl2, accompanying the exothermic reducing reaction, raw materials on reaction surface are sintered seriously, which makes the sample surface more dense, the reaction between Ca and TiO2 can not fully accomplish [1, 22], and the large amount of titanium dioxide exists in reduction products yet.
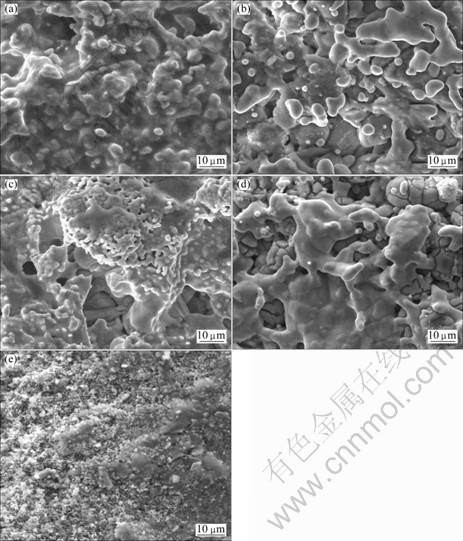
Fig. 6 SEM images of products obtained after reduction (mCa:mTiO2=2:1; reduction temperature, T=1 273 K; reaction time, t=6 h): (a) mCaCl2:mTiO2=1:2; (b) mCaCl2: mTiO2=1:4; (c) mCaCl2:mTiO2=1:6; (d) mCaCl2:mTiO2=1:9; (e) mCaCl2:mTiO2=0
XRD patterns of the products obtained after reduction corresponding to different mass ratios of TiO2 to CaCl2 are shown in Fig. 7. Figure 7(a) shows that CaClOH and Ca(OH)2 should be resulted from the hydrolysis of CaCl2 after absorbing water. When CaCl2 proportion decreases (mCaCl2:mTiO2=1:6), TiO2 is not completely reduced to titanium, as shown in Fig. 7(c). When the mass ratio of CaCl2 to TiO2 is 1:4, as shown in Fig. 7(b), the product does not contain Ti3O5, which effectively improves the purity of the titanium powders.
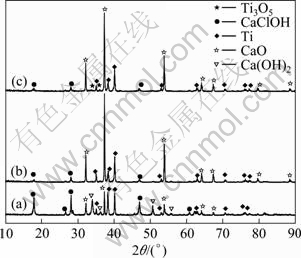
Fig. 7 XRD patterns of products obtained after reduction (mCa:mTiO2=2:1; reduction temperature, T=1 273 K; reaction time, t=6 h): (a) mCaCl2:mTiO2=1:2; (b) mCaCl2: mTiO2=1:4; (c) mCaCl2:mTiO2=1:6
3.3 Effects of reaction time on products
Figure 8 shows XRD patterns of the products obtained after leaching process for different reduction time. In the leaching process, the large number of metal calcium exists in reduction products, and H2 will be produced when calcium is dissolved in the dilute hydrochloric acid which causes the generation of TiH, as shown in Figs. 8(a-c). And CaTiO3 is generated by sintering between CaO and TiO2 [22], which is not dissolved in deionized water and hydrochloric acid. However, after reaction for 8 h, TiC is generated, as shown in Figs. 8(e-f), which should be resulted from the contact of product titanium and carbon graphite crucible.
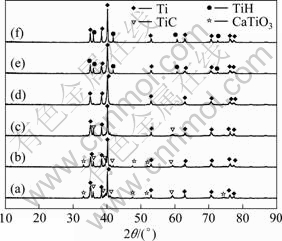
Fig. 8 X-ray diffraction patterns of reduced samples at 1 273 K for different reduction time: (a) 1 h; (b) 2 h; (c) 4 h; (d) 6 h; (e) 8 h; (f) 10 h
When the reaction temperature is 1 273 K and the reaction time is 6 h, titanium powders are obtained, as shown in Fig. 9. The EDS analysis reveals that the compositions of the titanium powders obtained after leaching are 99.55% Ti, 0.17% Ca, and 0.28% Si (mass fraction). Figure 10 shows SEM image of the titanium powder, and an irregular shape is observed clearly. The size of the titanium particles is in the range of 10-15 ��m.
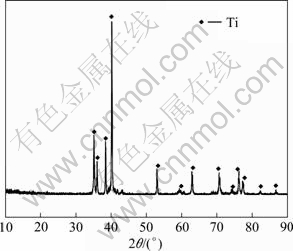
Fig. 9 XRD pattern for titanium powder obtained after leaching

Fig. 10 SEM image of titanium powder obtained after leaching and drying
4 Conclusions
1) Calcium chloride added in feed can help to form a porous bed of raw material, which can increase the contact area of gas-solid reaction between Ca vapor and TiO2 solid.
2) A novel process to prepare the titanium powder from TiO2 is proposed. It is found that the preform of raw materials adding CaCl2 can be more effectively reduced than the powders of raw materials, because it relieves the sintering on the feed surface.
3) The pieces of mCaCl2:mTiO2=1:4 are reduced by calcium vapor at 1 273 K for 6 h, and the pure titanium powders (more than 99.55% purity by EDS) are obtained after leaching with hydrochloric acid and deionized water and dried in vacuum. Titanium powders have an irregular shape and the particle size is 8-15 ��m.
References
[1] CHEN Tai-wu. Analysis and control of high temperature sintering in titanium sponge production by magnesium reduction method [J]. Titanium Industry Progress, 2009, 26(2): 34-36. (in Chinese)
[2] KROLL W J. The production of ductile titanium [J]. Transactions of Electrochemical Society, 1940, 78: 35-47.
[3] DU Ji-hong. Research progress of titanium production technology [J]. Rare Metal Materials and Engineering, 2008, 37(10): 1872-1875.
[4] OGASAWARA T. Progress of the titanium production technology in Japan and future prospects of the field [J]. The Japan Titanium Society, 2005, 53: 103-108.
[5] ZHENG Hai-yan, TORU H. OKABE. Recovery of titanium metal scrap by utilizing chloride wastes [J]. Journal of Alloys and Compounds, 2008, 461: 459-466.
[6] TOMONARI T. Titanium industry��its growing steps and future possibility [R]. Tokyo, Japan: The Japan Titanium Soc, 2000.
[7] SCHWANDTC. ALEXANDER D T L. FRAY D J. The electro-deoxidation of porous titanium dioxide precursors in molten calcium chloride under cathodic potential control [J]. Electrochimica Acta, 2009, 54: 3819-3829.
[8] JIANG K, HU X H, MA M S. Perovskitization-assisted electrochemical reduction of solid TiO2 in molten CaCl2 [J]. Angew Chem Int Ed, 2006, 45: 428-446.
[9] LI Wei, WANG Ying, TANG Ren-heng, XIAO Fang-ming. Reseach development of preparation of titanium by the FFC cambridge process [J]. Materials Research and Application, 2010, 4(4): 555-558. (in Chinese)
[10] MOXSON V S, SENKOV O N, FROES F H. Innovations in titanium powder processing [J]. JOM, 2000, 52(5): 24-26.
[11] ONO K, SUZUKI R O. A new concept for producing Ti sponge: Calciothermic reduction [J]. JOM, 2002, 54(2): 59�C61.
[12] ZHENG Hai-yan, LU Jin-wen, SHEN Feng-man. New technologies on direct production of metallic titanium from TiO2 [J]. The Chinese Journal of Process Engineering, 2009, 9(1): 448�C451. (in Chinese)
[13] OKABE T H, SADOWAY D R. Metallothermic reduction as an electronically mediated reaction [J]. Journal of Materials Research, 1998, 13: 3372-3377.
[14] OKABE T H, TAKASHI O, YOSHITAKA M. Titanium powder production by perform reduction process (PRP) [J]. Journal of Alloy sand Compounds, 2004, 364: 156-163.
[15] XU Bao-qiang, YANG bin, WAN He-li, SEN Wei. Preparing titanium powders by calcium vapor reduction process of titanium dioxide [C]// Proceedings of the 140th Annual Meeting & Exhibition. New York: John Wiley and Sons Incorporation, 2011: 501-508.
[16] OKABE T H, NAKAMURA M, OISHI T, ONUS K. Electrochemical deoxidation of titanium [J]. Metallurgical Transactions B, 1993, 24: 449-455.
[17] HUA Yi-xin. Kinetics of metallurgical process [M]. Beijing: Metallurgy Industry Press, 2004: 84-86. (in Chinese)
[18] OKABE T H, SUZUKI R O, OISHI T, ONO K. Thermodynamic properties of dilute titanium-oxygen solid solution in beta phase [J]. Mat Trans, 1991, 32(5): 485-488.
[19] OSAMU T, TORU H. Okabe. Fundamental study on synthesis and enrichment of titanium subchloride [J]. Journal of Alloys and Compounds, 2008, 457: 376-383.
[20] LIU Mei-feng, LU Shi-gang, KAN Su-rong, LI Guo-xun. Effect of electrolysis voltage on electrochemical reduction of titanium oxide to titanium in molten calcium chloride [J]. Rare Metals, 2007, 26(6): 547-551.
[21] GUO Shi-ju. Powder sintering theory [M]. Beijing: Metallurgy Industry Press, 1998: 11-21. (in Chinese)
[22] DU Ji-hong, XI Zheng-ping, LI Qing-yu, XU Qian, TANG Yong, LI Zheng-xian. Effect of TiO2 cathode performance on preparation of Ti by electro-deoxidation [J]. Trans Nonferrous Met Soc China, 2007, 17: 514-521.
(Edited by YANG Bing)
Foundation item: Project(51004058) supported by the National Natural Science Foundation of China; Project(2011FB039) supported by the Natural Science Foundation of Yunnan Province, China
Received date: 2011-08-30; Accepted date: 2011-12-03
Corresponding author: XU Bao-qiang, Associate Professor; Tel: +86-871-5161583; E-mial: kmxbq@126.com