


Trans. Nonferrous Met. Soc. China 22(2012) 1995-1999
Phase structure and electrochemical properties of La1.7+xMg1.3-x(NiCoMn)9.3(x=0-0.4) hydrogen storage alloys
WEI Fan-song1, LI Li2, XIANG Hong-fu1, LI Hui1, WEI Fan-na1
1. School of Materials Science and Engineering,Jiangsu University of Science and Technology, Zhenjiang 212003, China;
2. Environment Monitoring Center of Zhenjiang City, Zhenjiang 212000, China
Received 1 August 2011; accepted 21 December 2011
Abstract: The phase structure and electrochemical properties of La1.7+xMg1.3-x(NiCoMn)9.3(x=0-0.4) alloys were investigated. The XRD analysis reveals that the alloys consist of LaNi5 phase and other phases, such as LaMg2Ni9 phase (PuNi3 structure) and La4MgNi19 phases (Ce5Co19+Pr5Co19 structure, namely A5B19 type). With the increase of the x value, the LaMg2Ni9 phase fades away and La4MgNi19 phases appear, while the abundance of LaNi5 phase firstly increases and then decreases. At the same time, the cell volume of LaNi5 phase and LaMg2Ni9 phase decreases. The electrochemical measurement shows that alloy electrodes could be activated in 4-5 cycles, and with the increase of the x value, the maximum discharge capacity gradually increases from 330.9 mA��h/g (x=0) to 366.8 mA��h/g (x=0.4), but the high-rate dischargeability (HRD) and cyclic stability (S) decrease somewhat (x=0.4, HRD600=82.32%, S100=73.8%). It is found that the HRD is mainly controlled by the electrocatalytic activity on the alloy electrode surface, and the decline of cyclic stability is due to the appearance of A5B19 type phase with larger hydrogen storage capacity, which leads to larger volume expansion and more intercrystalline stress and then easier pulverization during charging/discharging.
Key words: hydrogen storage alloy; A5B19 type; crystal structure; electrochemical property; La-Mg-Ni system
1 Introduction
With the development of portable electronic devices and equipments, the demand for Ni/MH battery with higher capacity is ever growing. Mischmetal-based AB5-type alloys are now widely used as the negative electrode materials for Ni/MH batteries, but their maximum discharge capacity is generally 330 mA��h/g or so which is approaching the theoretical capacity limited by single CaCu5 type structure. It is difficult to meet the demand for increasing energy density of Ni/MH secondary battery [1]. In recent years, a great attention to La-Mg-Ni system alloys has been paid owing to the benefit of low cost and high discharge capacity. KADIR et al [2,3] revealed that RMg2Ni9 (R=rare earth, Ca or Y) alloys still held PuNi3-type rhombohedral structure after hydriding, and their hydrogen storage capacity could reach 1.7%-1.8% (mass fraction) through the appropriate element substitution, which was significantly higher than that of the mischmetal-based AB5-type alloys. Subsequently, CHEN et al [4] found that the discharge capacity of LaCaMg2Ni9 alloy was up to 360 mA��h/g, but the high-rate dischargeability (HRD) and cyclic stability were poor. KOHNO et al [5] investigated the properties of AB3-3.5-type alloy electrode in La-Mg-Ni system, and found that at room temperature, the La0.7Mg0.3Ni2.8Co0.5 alloy had a discharge capacity of 410 mA��h/g which increased by 25% compared with the AB5-type alloy, a better capacity retention rate after 30 cycles and a higher high-rate dischargeability, so it is considered to be a promising candidate for the negative electrode materials for Ni/MH batteries. However, considering the commercial applications, the overall electrochemical properties of such alloys have to be further improved. Researches have indicated that changing the stoichiometry [6,7] and partial substituting the elements of the alloy [8-14] are effective ways to improve the cycle life of alloy, at the same time, the cyclic stability can also be improved significantly by use of different material processing methods such as heat treatment [15,16] and rapid solidification [17,18]. In this work, La1.7+xMg1.3-xNi7.05Co1.98Mn0.27 (x=0-0.4) alloys were prepared by vacuum melting followed with an annealing treatment, and the influence of mass ratio of La to Mg on the phase structure and electrochemical properties of the alloys was investigated in order to improve the overall electrochemical properties of the alloys.
2 Experimental
The alloy samples were prepared by vacuum levitation melting in argon atmosphere and remelted three times to ensure a high homogeneity. The purity of all starting elemental metals was higher than 99.9%. Half the as-cast ingot was annealed under vacuum at 1173 K for 12 h.
The alloy samples thus prepared were ground mechanically into powders below 50 ��m and used for electrochemical tests and XRD analysis. The crystal structure of the alloy samples was determined by XRD analysis using a Rigaku D/max 2500/PC diffractometer with Cu K�� radiation, and the XRD patterns were obtained by the step scan mode with step size of 2��=0.02 ��.
For electrochemical tests, pellet type alloy electrodes (d=10mm) were prepared by cold pressing the mixture of the alloy powder with carbonyl Ni powder in a mass ratio of 1:4. The electrochemical tests were carried out at 298 K in a conventional tri-electrode cell consisting of a working electrode (MH electrode), a sintered Ni(OH)/NiOOH counter electrode and a Hg/HgO reference electrode, and the electrolyte was 6 mol/L KOH solution. The discharge capacity was determined galvanostatically by using an automatic charge/discharge unit (Land). Each electrode was charged at 100 mA/g for 4.5 h, and discharged at 60 mA/g to the cut-off potential of -0.7 V (vs Hg/HgO). The high-rate dischargeability (HRD) defined as Cn/(Cn+C60) was determined by the ratio of the discharge capacity Cn (with n=300 or 600 mA/g, respectively) to the total discharge capacity defined as the sum of Cn and C60, which was the additional capacity measured subsequently at 60 mA/g after Cn was measured. The cycling test was conducted at the charge/discharge current density of 100 mA/g. The cyclic capacity retention rate S100 was defined as S100=C100/Cmax, where C100 was the discharge capacities at the 100th cycles. In evaluating the kinetic properties of the electrode reaction, the linear polarization curves of the electrode were plotted with a Solarton SI 1287 potentiostat by scanning the electrode potential at the rate of 0.1 mV/s from �C5 to 5 mV(versus open circuit potential) at 50% depth of discharge (DOD). The potentiostatic discharge technique was used to evaluate the diffusion coefficient of hydrogen within the alloy bulk. After being fully charged and followed by a 30 min open circuit rest-period, the test electrodes were discharged with 600 mV potential-step for 2000 s on a Sloartron SI1287 potentiostat, and then the experimental data were analyzed by using the CorrWare electrochemical corrosion software.
3 Results and discussion
3.1 Phase structure
Figure 1 shows the XRD patterns of annealed La1.7+xMg1.3-x(NiCoMn)9.3(x=0-0.4) alloys. It can be seen that all the alloys are composed of LaNi5 phase (CaCu5 structure, space group: P6/mmm) and some other phases, such as LaMg2Ni9 phase (PuNi3 structure, space group: R-3m) and La4MgNi19 phases with A5B19 type structure (Ce5Co19+Pr5Co19, space groups are R-3m and P63/mmc respectively). With the increase of the x value, the diffraction peaks of the LaMg2Ni9 phase become lower, and when x=0.4, the peaks of the LaMg2Ni9 phase disappear, while those of La4MgNi19 phases appear. This indicates that the reduction of Mg content in the alloy can promote the increase of La4MgNi19 phases.
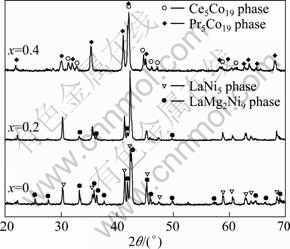
Fig. 1 XRD patterns of annealed La1.7+xMg1.3-x(NiCoMn)9.3 (x=0-0.4) alloys
Figure 2 shows the XRD Rietveld analysis patterns of La2.1Mg0.9Ni7.05Co1.98Mn0.27 alloys, in which the fitting factor (Rp) is 11.6 and goodness of fit (S) is 1.9. The fitting results reveal that the Mg occupies the 4f and 6c positions of La atom in A5B19 type structure, with the same occupation as Mg in PuNi3 structure. The phase composition and lattice parameters of the phases in alloys were calculated by the Rietveld method and are listed in Table 1. It can be seen that as x increases, the abundance of LaMg2Ni9 phase gradually decreases from 61.29% (x=0) to none (x=0.4), but that of LaNi5 phase firstly increases from 38.71% (x=0) to 63.32% (x=0.2) and then decreases to 26.31% (x=0.4). This indicates that the higher mass ratio of La to Mg easily leads to the transformation of LaMg2Ni9 phase into LaNi5 phase and further into La4MgNi19 phase (Ce5Co19+Pr5Co19 type). It can also be seen from Table 1 that the cell volumes of LaMg2Ni9 phase and LaNi5 phase tend down with the increase of x. For example, the volume of LaNi5 phase decreases from 88.6254��10-3 nm3 (x=0) to 88.4473��10-3 nm3 (x=0.4), which may be due to the phase transformation and the variation of their abundance.
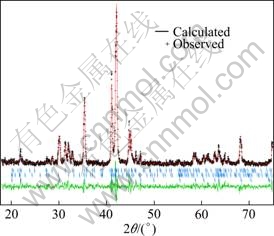
Fig. 2 X-ray diffraction patterns for La2.1Mg0.9Ni7.05Co1.98- Mn0.27 alloy
3.2 Electrochemical properties
3.2.1 Characteristics of discharge platform
Figure 3 shows the electrochemical plateau curves for hydrogen desorption of La1.7+xMg1.3-x(NiCoMn)9.3 (x=0-0.4) alloys. As can be seen from Fig.3, although all the alloys are composed of double phases or multiphases, their discharging plateau curves show a characteristics of single platform, which may be ascribed to their approximate plateau pressures. When x=0.4, the plateau lowers obviously, and referring to the Rietveld analysis in Table 1, the total mass fraction of La4MgNi19 phase (Ce5Co19+Pr5Co19 type structure) with larger cell volume is up to 73.69%, which adds the hydrogen position in the alloy and then leads to a lower platform and a higher capacity.
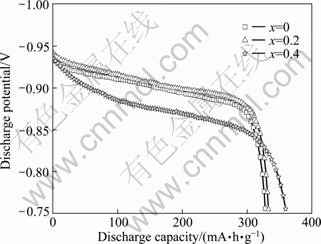
Fig. 3 Electrochemical plateau curves for hydrogen desorption of La1.7+xMg1.3-x(NiCoMn)9.3 (x=0-0.4) alloys at 298 K
3.2.2 Activation performance and discharge capacity
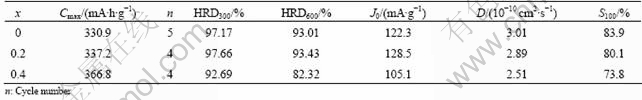
The electrochemical properties of the alloys are summarized in Table 2. It can be seen that alloy electrodes can be activated in 4-5 cycles, and with x increasing, the maximum discharge capacity gradually increases from 330.9 mA��h/g (x=0) to 366.8 mA��h/g (x= 0.4). This means that the higher mass ratio of La to Mg has a little influence on the activation performance, but can significantly enhance the original capacity, which may be related to the higher hydrogen storage capacity of La4MgNi19 phases (about 1.5%).
Table 1 Phase composition and lattice parameters of La1.7+xMg1.3-x(NiCoMn)9.3(x=0-0.4) alloys

Table 2 Electrochemical properties of La1.7+xMg1.3-x(NiCoMn)9.3(x=0-0.4) alloys
3.2.3 High-rate dischargeability
From Table 2, it is also found that the high-rate dischargeabilities of alloys at x=0 and x=0.2 are similar, and when the x value further increases to 0.4, the HRD decreases obviously. At the discharge rate of 300 mA/g, the HRD300 of x=0 or x=0.2 alloy is around 97%, but that of x=0.4 alloy is only 92.69%. When the discharge current increases to 600 mA/g, the HRD600 of x=0 or x=0.2 alloy is up to 93%, but that of x=0.4 alloy obviously decreases to 82.32%. It is known that the high-rate dischargeability of MH electrode is mainly influenced by the electrochemical reaction rate on the alloy surface and the diffusion rate of hydrogen in the bulk of the alloy [19]. To examine the factors of discharge kinetics in alloy electrodes, linear polarization and potential-step experiment were performed on the alloy electrodes, and then exchange current density (J0) and hydrogen diffusion coefficient (D) were calculated respectively according to the obtained curves, as listed in Table 2, and the detailed calculating method can be seen in Ref. [20]. As shown in Table 2, with the increase of the x value, the exchange current density (J0) of alloy electrodes firstly increases and then decreases, but the hydrogen diffusion coefficient (D) continuously decreases from 3.01��10-10 cm2/s (x=0) to 2.51��10-10 cm2/s (x=0.4). Comparing the HRD with the values of J0 and D, it is found that the variation of J0 is consistent with that of HRD values of the alloy electrodes. This indicates that the HRD is mainly controlled by the electrocatalytic activity on the alloy electrode surface.
3.2.4 Cyclic stability
Figure 4 shows the cyclic stability curves of alloy electrodes. From Table 2 and Fig. 4, it can be seen that the alloy with higher x value shows further poor cycling stability. For x=0.4 alloy, although its cycling stability declines obviously, the capacity retention rate (S100) of the alloy after 100 cycles is still up to 73.8%. As seen from the above analysis, there are Ce5Co19+Pr5Co19 type phases with larger hydrogen storage capacity for x=0.4 alloy, which causes larger cell volume expansion and more intercrystalline stress, then resulting in easier pulverization during charge/discharge. It is well accepted that a higher degree of pulverization exposes more surface area to the corrosive electrolyte, then corrodes more and hence has a poor cycling stability.

Fig. 4 Cyclic stability curves of alloy electrodes at 298 K
4 Conclusions
1) The structure analysis reveals that all the alloys are composed of LaNi5 phase (CaCu5 structure) and other phases, such as LaMg2Ni9 phase (PuNi3 structure) and La4MgNi19 phases with A5B19 type structure (Ce5Co19+Pr5Co19). With the increase of the x value, the LaMg2Ni9 phase disappears and La4MgNi19 phase appears, but the abundance of LaNi5 phase increases firstly and then decreases. At the same time, the cell volume of LaNi5 and LaMg2Ni9 decreases.
2) The electrochemical measurement shows that the increase of x value improves the activation and initial capacity, but lowers the high-rate dischargeability somewhat. It is found that the HRD is mainly controlled by the electrocatalytic activity on the alloy electrode surface, and the decline of cyclic stability is due to the appearance of A5B19 type phase with larger hydrogen storage capacity, which leads to larger volume expansion and intercrystalline stress and then easier pulverization during charge/discharge.
3) La2.1Mg0.9(NiCoMn)9.3 alloy shows a high capacity (366.8 mA��h/g), reasonable cyclic stability (S100=73.8%) and good 1 C rate dischargeability (HRD300= 92.69%), which is a promising candidate for a high capacity negative electrode material for Ni/MH batteries.
References
[1] LEI Yong-quan. New energy materials [M]. Tianjin: Tianjin University Press, 2000: 44-45. (in Chinese)
[2] Kadir K, Sakai T, Uehara I. Structural investigation and hydrogen capacity of YMg2Ni9 and (Y0.5Ca0.5)(MgCa)Ni9: New phases in the AB2C9 system isostructural with LaMg2Ni9 [J]. J Alloys Comp, 1999, 287: 264-270.
[3] Kadir K, Sakai T, Uehara I. Structural investigation and hydrogen storage capacity of LaMg2Ni9 and (La0.65Ca0.35)(Mg1.32Ca0.68)Ni9 of the AB2C9 type structure [J]. J Alloys Comp, 2000, 302: 112-117.
[4] Chen J, Kuriyama N, Takeshita H T, Tanaka H, Saka T, Haruta M. Hydrogen storage alloys with PuNi3-type structure as metal hydride electrodes [J]. Electrochem Solid-State Lett, 2000, 3(6): 249-252.
[5] Kohno T, Yoshida H, Kawashima F, Indaba T, Sakai I, Yamamoto M, Kanda M. Hydrogen storage properties of new ternary system alloys: La2MgNi9, La5Mg2Ni23, La3MgNi14 [J]. J Alloys Comp, 2000, 311: L5-L7.
[6] PAN H G, LIU Y F, GAO M X, LEI Y Q, WANG Q D. Study of the structural and electrochemical properties of La0.7Mg0.3(Ni0.85Co0.15)x (x=2.5-5.0) hydrogen storage alloys [J]. J Electrochem Soc A, 2003, 150: 565-570.
[7] LIU Y F, PAN H G, GAO M X, LI R, LEI Y Q. Influence of Ni addition on the structures and electrochemical properties of La0.7Mg0.3Ni2.65+xCo0.75Mn0.1 (x=0-0.5) hydrogen storage alloys [J]. J Alloys Comp, 2005, 389(1-2): 281-289.
[8] Liao B, Lei Y Q, Chen L X, Lu G L, Pan H G, Wang Q D. Effect of the La/Mg ratio on the structure and electrochemical properties of LaxMg3-xNi9 (x=1.6-2.2) hydrogen storage electrode alloys for nickel-metal hydride batteries [J]. J Power Source, 2004, 129: 358-367.
[9] TANG Rui, LIU Li-qin, LIU Yong-ning, YU Guang, ZHU Jie-wu, LIU Xiao-dong. Structure and electrochemical properties of La0.8-xRExMg0.2Ni3.2Co0.6 hydrogen storage alloys [J]. The Chinese Journal of Nonferrous Metals, 2005, 15(7): 1057-1061. (in Chinese)
[10] LIAO Bin, LEI Yong-quan, CHEN Li-xin, L? Guang-lie, PAN Hong-ge, WANG Qi-dong. A study on the structure and electrochemical properties of La2Mg(Ni0.95M0.05)9 (M=Co, Mn, Fe, Al, Cu, Sn) hydrogen storage electrode alloys[J]. Journal of Alloys and Compounds, 2004, 376(1-2): 186-195.
[11] ZHANG X B, SUN D Z, YIN W Y, CHAI Y J, ZHAO M S. Crystallographic and electrochemical characteristics of La0.7Mg0.3Ni3.5-x(Al0.5Mo0.5)x (x=0-0.8) hydrogen storage alloys [J]. J Power Sources, 2006, 154: 290-297.
[12] ZHANG Fa-liang, LUO Yong-chun, SUN Kai, KANG Long, CHEN Jian-hong. A study on the structure and electrochemical properties of La1.5Mg0.5Ni7-xCox (x=0-1.8) hydrogen storage alloys [J]. Functional Materials, 2006, 37(2): 265-268. (in Chinese)
[13] LIU Y F, PAN H G, GAO M X, ZHU Y F, LEI Y Q, WANG Q D. Structures and electrochemical properties of La0.7Mg0.3Ni2.975-xCo0.525Mnx hydrogen storage alloys [J]. J Electrochem Soc A, 2004, 151(3): 374-380.
[14] JIANG Bing-jie, WANG Jing, MU Dao-bin, CHEN Shi, WU Bo-rong, WU Feng. Effect of Al substitution on electrochemical performance of La-Mg-Ni hydrogen storage alloys [J]. The Chinese Journal of Nonferrous Metals, 2008, 18(11): 2036-2043. (in Chinese)
[15] LIU Y F, PAN H G, GAO M X, ZHU Y F, LEI Y Q. Influence of heat treatment on electrochemical characteristics of La0.75Mg0.25Ni2.8Co0.5 hydrogen storage electrode alloy [J]. Transactions of Nonferrous Metals Society of China, 2003, 13: 25-28.
[16] Zhou Zeng-lin, Song Yue-qing, Cui Shun, Lin Chen-guang, GUO Zhi-meng, QU Xuan-hui. Effect of heat treatment on the properties of La-Mg-Ni-system hydrogen storage electrode alloys (II) Hydrogen storage and electrochemical properties [J]. Rare Metal Materials and Engineering, 2008, 37(6): 964-969. (in Chinese)
[17] ZHANG Yang-huan, DONG Xiao-ping, WANG Guo-qing, GUO Shi-hai, REN Jiang-yuan, WANG Xin-lin. Cycling stability of La-Mg-Ni system (PuNi3-type) hydrogen storage alloys prepared by casting and rapid quenching [J]. The Chinese Journal of Nonferrous Metals, 2005, 15(5): 705-710. (in Chinese)
[18] ZHANG Yang-huan, ZHAO Dong-liang, DONG Xiao-ping, QI Yan, GUO Shi-hai, WANG Xin-lin. Effects of rapid quenching on structure and electrochemical characteristics of La0.5Ce0.2Mg0.3Co0.4Ni2.6-xMnx (x=0-0.4) electrode alloys [J]. Transactions of Nonferrous Metals Society of China, 2009, 19(2): 364-371.
[19] Iwakura C, Oura T, Inouse H, Mastsuoka M. Effects of substitution with foreign metals on the crystallographic, thermodynamic and electrochemical properties of AB5-type hydrogen storage alloys [J]. Electrochim Acta, 1996, 41(1): 117-121.
[20] Wei Fan-song, Lei Yong-quan, Chen Li-xin, Ying Tiao, Ge Hong-wei, L? Guang-lie. Influence of material processing on crystallographic and electrochemical properties of cobalt-free LaNi4.95Sn0.3 hydrogen storage alloy [J]. Transactions of Nonferrous Metals Society of China, 2006, 16(3): 527-531.
La1.7+xMg1.3-x(NiCoMn)9.3(x=0~0.4)����Ͻ����ṹ��绯ѧ����
κ����1���� ��2����긣1���� ��1��κ����1
1. ���տƼ���ѧ ���Ͽ�ѧ�빤��ѧԺ���� 212003��
2. ����ʡ���л����������վ���� 212000
ժ Ҫ����La1.7+xMg1.3-x(NiCoMn)9.3(x=0~0.4)����Ͻ���ṹ�͵绯ѧ���ܽ����о����ṹ�����������Ͻ���Ҫ��LaNi5��(CaCu5�ṹ)����������ɣ���LaMg2Ni9�� (PuNi3�ṹ) ��La4MgNi19��(Ce5Co19+Pr5Co19�ṹ)������x�����ӣ�LaMg2Ni9����ʧ������La4MgNi19�࣬��LaNi5��ĺ����������Ӻ��С���Ҿ�������½����绯ѧ�����������Ͻ�缫ֻ��4��5��ѭ�����ɻ������x�����ӣ����ŵ�����������x=0��330.9 mA��h/g���ӵ�x=0.4��366.8 mA��h/g�����߱��ʷŵ�����(HRD)��ѭ���ȶ���(S)�������½�(x=0.4, HRD600=82.32%, S100=73.8% )���о���Ϊ��HRD��Ҫ�ɺϽ�缫����ĵ�����Կ��ƣ���ѭ���ȶ��Ե��½���������x=0.4�Ͻ��г����˾��нϴ���������Ce5Co19��Pr5Co19�ͽṹ�࣬�������������ʺ;���Ӧ������ʹ�Ͻ����������������н��ۻ����¡�
�ؼ��ʣ�����Ͻ�A5B19�ͣ�����ṹ���绯ѧ���ܣ�La-Mg-Niϵ
(Edited by LI Xiang-qun)
Foundation item: Project (2008CL068L) supported by the Natural Science Research Project of Higher Education of Jiangsu Province, China; Project (50901036) supported by the National Natural Science Foundation of China
Corresponding author: WEI Fan-song; Tel: +86-511-85809119; E-mail: zju.wei@yahoo.com.cn
DOI: 10.1016/S1003-6326(11)61419-2