
Effects of Al partial substitution for Ni on properties of LaNi5-xAlx
CAO Da-li(曹大力)1, CHENG Hong-hui(程宏辉)2, MA Lei(马 雷)1, CHEN De-min(陈德敏)2,
L? Man-qi(吕曼祺)2, YANG Ke(杨 柯)2
1. School of Materials Science and Engineering, Shenyang Institute of Chemical Technology,Shenyang 110141, China;
2. Institute of Metal Research, Chinese Academy of Sciences, Shenyang 110016, China
Received 15 July 2007; accepted 10 September 2007
Abstract: The hydrogen storage properties of four LaNi5-xAlx (x = 0.25, 0.50, 0.75, 1.00) pseudobinary alloys were systematically studied. The characteristics of microstructure before and after hydrogenation, activation, kinetics and thermodynamics properties, as well the anti-combustibility properties of the four pseudobinary alloys were investigated. The results reveal that the alloys have excellent activation properties and kinetics properties. X-ray diffraction analysis of crystal lattice of the alloys show that the crystal structures of alloys do not change with the addition of Al in the range of 0≤x≤1, but the lattice constants slightly increase. It is found by measuring thermodynamic properties of the alloy-H2 systems that with increasing x value the equilibrium pressure, hydrogen-absorbing ability and hysteresis decrease, whereas the absolute value of the enthalpy increases. It is also found that the hydrogen absorbing velocity of the alloys decreases with increasing x value.
Key words: hydrogen storage; LaNi5-xAlx alloy; kinetics property; thermodynamics property; crystal lattice parameter
1 Introduction
The hydrogen storage properties of some metals and alloys have been widely investigated. Among these materials, the LaNi5 alloy exhibits excellent hydrogen storage characteristics such as high volumetric storage density, easy activation, moderate kinetics properties etc. However, LaNi5 alloy has unsatifactory properties such as the storage capacity decreasing considerably over cyclic absorption/desorption process, easy pulverization, easy combustion after pulverization, plateau pressure too high and so on[1].
Cyclic life, pulverization, moderate plateau pressures of LaNi5 alloy can be modulated by substituting different elemental species into the La or Ni lattice sites[2-6]. Absorption/ desorption cycling effects have been reported for different systems including LaNiSny (y = 0.1, 0.2) [7], LaNiAl and Ni by Mn, Zn,Cr, Fe, Co, Cu or even by Al and Li[5-8] to improve the hydrogen storage capacity.
Much attention has been particularly focused on LaNixAl5-x compounds[7-10] for their application to energy storage and the construction of nickel-metal hydride (Ni/MH) battery negative electrodes. It was found that the hydrogen sorption properties were strongly altered when nickel was partially replaced by aluminum in the LaNi5 compound[11-12]. LaNixAl5-x is considered to be a very useful material for H absorption because of its low plateau pressure and resistance to impurities in hydrogen gas[7-12]. It was also found that the partial replacement Ni in LaNi5 alloy by a small amount of Al resulted in a prominent increase in the cycle life time without causing much decrease in hydrogen absorption capacity and minimizes corrosion attack of the hydride electrode[7-10]. But partial substitution amount of Al for Ni is below 0.5.
In this work, for the purpose of application, partial substitution amount of Al for Ni in LaNi5 alloys was selected from 0.25 to 1, hydrogen storage properties of four LaNi5-xAlx (x = 0.25, 0.50, 0.75, 1.00) alloys were systematically studied.
2 Experimental
The LaNi5-xAlx alloy was prepared in a high-frequency induction melting furnace of stoichiometric amounts of the pure metals (lanthanum with a purity of 99.50%, nickel 99.95%, aluminum 99.70%, in mass fraction) using argon atmosphere protection, the ingots were turned over and remelted twice for homogeneity. The ingot was then annealed at 1 323 K for 8 h in argon atmosphere in a sealed quartz tube. The alloy was a single phase with hexagonal CaCu5 structure examined by X-ray diffractometry(XRD). In order to investigate the hydrogen storage properties, four samples of LaNi5-xAlx (x=0.25, 0.50, 0.75, 1.00) about 1 g were placed individually in cells made of stainless steel. These sample cells were then connected with testing apparatus, whose volume was calibrated precisely. This testing apparatus was set up with actuator-driven valves, needle valves. Pressure transducers and thermocouple were attached to the system for accurate pressure and temperature measurement. Water bath and oil bath were used in order to keep a constant testing temperature.
High-purity hydrogen (H2 99.999%, O2 (Ar) 1×10-6, N2 2×10-6, CO 0.2×10-6, CO2 0.4×10-6, CH4 0.2×10-6 and H2O 2.6×10-6) was used for activation and absorption/desorption cycle test. PC isotherms for absorption/desorption as well as absorption kinetics after initial activation.
The samples, after initial activation and absorption/desorption cycles, were characterized by XRD to observe possible changes in the phases. The pulverization size distribution of the particles were investigated with a Malvern Mastersizer mirco laser particle sizer.
3 Results and discussion
3.1 Phase structure of alloys
The XRD patterns of LaNi5-xAlx (x = 0.25, 0.50, 0.75, 1.00) hydrogen storage alloys are shown in Fig.1. It can be seen that the alloys with different Al contents have similar diffraction patterns and they have corresponded to single phase material with the CaCu5 structure, no peak of secondary phase or residual starting materials was observed, which implies that a single structure forms in these alloys. However, the diffraction peaks shift to smaller angles with increasing Al content, which implies a increasing in the lattice parameters[5].
Lattice parameters (a and c) together with the calculated unit cell volumes for the samples, are presented in Table 1. The lattice parameters and the calculated unit cell volumes indicates that partial substitution of Ni for Al increases slightly the unit cell dimensions. It should also be noted that these parameters are a little increasing with the increase of x, the ratio of a to c decreases with the increase of x. The function of cell volume(V) and x is described as V=3.26x+86.67±0.15. This linear relationship means that the cell volume increase with x increasing. This is attributed to that the atomic radius of Al (0.143 nm) is somewhat larger than that of Ni (0.125 nm).
Table 1 Unit cell volume and lattice constant of LaNi5-xAlx
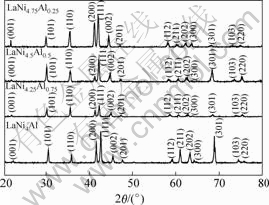
Fig.1 XRD patterns of LaNiAl series alloys
3.2 Thermodynamic properties
All of the LaNi5-xAlx (x = 0.25, 0.50, 0.75, 1.00) alloys can be easily activated to their maximum adsorption hydrogen capacity within 3-4cycles, and all of alloys have excellent activation properties, activation procedures have hardly change with the addition of Al in the range of 0≤x(Al)≤1.
Fig.2 shows the absorption hydrogen PC isotherms of the LaNi5-xAlx alloy after activation, it can be found that the adsorption hydrogen concentration decreases and plateau equilibrium pressure significantly decreases with increasing Al content. The molecular formula of saturated alloy hydrids at 353 K are respectively: LaNi4.75Al0.25H5.7, LaNi4.5Al0.5H5.3, LaNi4.25Al0.75H4.2, LaNi4AlH3.6, the function between the plateau pressure and Al content(x) is lnPeq= [(-3 308-2 018x)/T]+12.4. After LaNi5-xAlx absorb hydrogen, hydrogen atoms accommodate in crystal lattice interstice (especially in tetrahedral interstice) as interstitial atoms. The larger the crystal lattice interstice is, the easier the hydrogen atoms enter into interstice. That the cell volume increases with increasing x content leads to the increase of the accommodation interstitial sizes for hydrogen atoms and consequently the decreasing of dissociation plateau pressures, which is responsible for stability of the hydrides.

Fig.2 Hydriding p-C-T curves of LaNiAl series alloys: (a) LaNi4.75Al0.25; (b) LaNi4.5Al0.5; (c) LaNi4.25Al0.75; (d) LaNi4Al
The enthalpy and the entropy of hydride formation can be determined from the temperature dependence of the equilibrium pressures of hydrogen absorption isotherms by using the van’t Hoff plots in Fig.3. We can see that the enthalpy and the entropy of hydride formation increase with content x increasing in Table 2.
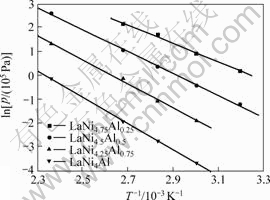
Fig.3 van’t Hoff plotes of LaNiAl series alloys with hydrogen absorption
Table 2 Change of enthalpy and entropy of LaNi5-xAlx series alloys after hydrogen absorption

The heat of hydride formation is the most important parameter characterizing the alloy as a proper hydrogen absorber for various specific applications. HONG[12] has stated that the heat of alloy hydride formation should be between -40 and -15 kJ/mol. In case it is more positive than -15 kJ/mol, the alloy hydride is not stable enough for charging the MH electrode at room temperature. On the other hand, the hydrides that are too stable make discharging too difficult.
3.3 Kinetics properties
The absorption kinetics curves of LaNiAl series alloys after 25 circles at 353 K and 1 020 kPa (initial pressure of storage cell) are shown in Fig.4. From these kinetics curves, it can be seen that all of LaNiAl alloys can absorb 90% of its maximum hydrogen capacity in 90 s, and the absorption hydrogen rate decreases with increasing x, that is in order from low to high: LaNi4Al, LaNi4.25Al0.75, LaNi4.5Al0.5, LaNi4.75Al0.25.Ni in LaNi5-xAlx can catalyze the reaction of alloys and H, so absorption rate of alloys are very rapidly.
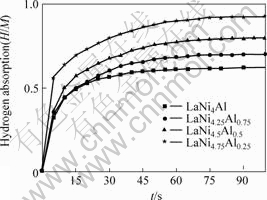
Fig.4 Hydriding kinetics curves of LaNiAl series alloys
More cracks and fissures formed both on the surface and inside of the samples with decreasing the Al content, which could shorten the hydrogen atoms diffusion distance and provide more fresh surfaces for hydrogen atoms association, improve the absorption kinetics. So the absorption hydrogen rate decreases with increasing x.
3.4 Pulverized properties
It was found that the sample pulverized in the activation due to the volume expansion from hydrogen absorption. The particle diameter distribution plots of LaNiAl series alloys after 25 circles are shown in Fig.5. From the plots, it can be seen that the particle diameter of LaNi5 focus on about 10 ?m, LaNi4.75Al0.25 focus on about 15 ?m, laNi4.25Al0.75 focus on about 20 ?m, LaNi4Al focus on about 30 ?m. It was found that the sample pulverized after 25 circles decreases with increasing x. According to GIIA[10], Al accumulates on grain boundaries and in association with segregated La,forms a porous oxide layer which protects the material against further corrosion and oxidation. SUZUKI[3] subjected LaNi5 and LaNi4.7Al0.3 up to 2000 absorption/desorption cycles at 293 K in super-high purity (99.999 99%) hydrogen, found that the amount of Ni precipitation increased with increasing the number of cycles and Al substitution could improve the cycle stability of LaNi5.
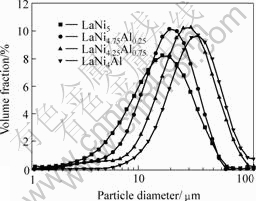
Fig.5 Plots of particle diameter vs volume fraction
4 Conclusions
1) The crystal structures of alloys do not change with the addition of Al in the range of 0≤x≤1, but the lattice constants slightly increase, cell volume slightly increases too, and a/c value slightly decreases. A function between cell volume(V) of alloys LaNi5-xAlx and x is V=3.26x+86.67±0.15.
2) LaNi5-xAlx alloys have excellent activation properties and activation procedures have hardly change with the addition of Al in the range of 0≤x≤1;
3) The hydrogen-absorbing ability and the hysteresis decrease, but the absolute value of the enthalpy increases with the increase of x; the function between the plateau pressure and Al content (x) is ln peq = [(-3 308 - 2 018x)/T]+12.4. In addition, the desorption entropy increases with Al additive, while the desorption enthalpy and Gibbs free energy increase with Al contents which diminish the maximum absorbing hydrogen content.
4) The hydrogen absorbing velocity of the all alloys is very fast, and it slightly decreases with increasing x value.
5) The anti-pulverization after 25 circles is better with the increase of Al content.
References
[1] MUELLER W M, BLACKLEDGE J P, LIBOWITZ G G. Metal hydrides[M]. New York: Academic Press, 1968.
[2] MENDELSOHN M H, GRUEN D M. LaNi5-xAlx is a versatile alloy system for metal hydride applications[J]. Nature, 1977, 269(9): 45-47.
[3] LIU Y F, PAN H G, GAO M X, ZHU Y F, LEI Y Q, WANG Q D. The effect of Mn substitution for Ni on the structural and electrochemical properties of LaxMg0.7Ni2.55-x Co 0.45Mn0.3 hydrogen storage electrode alloys[J]. Int J Hydrogen Energy, 2004, 29(3): 297-302.
[4] LIU Y F, PAN H G, GAO M X, ZHU Y F, LEI Y Q, WAN G. Electrochemical studies on La0.7Mg0.3Ni3.4-xCo0.6Mnx metal hydride electrode alloys[J]. Mater Chem Phys, 2004, 84(1): 171-178.
[5] LIU Y F, PAN H G, GAO M X, LI R, LEI Y Q. Effect of Co content on the structural and electrochemical properties of the La0.7Mg0.3Ni 3.4-xMnxCo0.1 hydride alloys: I. The structure and hydrogen storage[J]. J Alloys Compd, 2004(376): 296-302.
[6] LIU Y F, PAN H G, GAO M X, LI R, LEI Y Q. Effect of Co content on the structural and electrochemical properties of La0.7 Mg0.3Ni3.4-x? MnxCo0.1 the hydride alloys: II. Electrochemical Properties[J]. J Alloys Compd, 2004(376): 304-310.
[7] ZHANG W, CIMATO J, GOUDY A J. The hydriding and dehydriding kinetics of some LaNi5-xAlx alloys[J]. J Alloys Compod, 1993(201): 175-180.
[8] FENG F, HAN J W, GENG M M, NORTHWOOD D O. Hydrogen desorption kinetics of LaNi4.7Al0.3 metal hydride electrode using potentiostatic measurements [J]. Solar Energy Mater Solar Cells, 2000, 62(1/2): 51-61.
[9] WANG X L, SUDA S. Effect of Al-substitution on hydriding reaction rates of LaNi5-xAlx[J]. J Alloys Compod, 1993(202): 5-10.
[10] KIYONORI S, KAZUHIRO I. Effect of hydrogen absorption/desorption cycling on hydrogen storage performance ofLaNi5 and LaNi4.7Al0.3[J]. Materials Transactions JIM, 2000, 40(5): 581-589.
[11] CHENG H H, YANG H G, LI S L. Effect of hydrogen absorption/desorption cycling on hydrogen storage performance ofLaNi4.25Al0.75[J]. J Alloys Compod, 2006(425): 321-328.
[12] GIZA K, IWASIECZKO W, PAVLYUK V V. Hydrogen absorption and corrosion resistance of LaNi4.8Al0.2 and LaNi4.8Al0.1 Li0.1 alloys[J]. J Alloys Compod, 2007(429): 352-357.
(Edited by YANG Hua)
Foundation item: Project (50276063) supported by the National Natural Science Foundation of China
Corresponding author: CAO Da-li, Tel: +86-24-83680245; E-mail: caodali2008@126.com