�Խ���������Ϊԭ�ϸĽ�ȼ�շ��ϳɦ�-LiAlO2
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2018���9��
�������ߣ�Daniel CRUZ H. B. ORTIZ-OLIVEROS L. M. PEREZ-DIAZ R. M. FLORES-ESPINOSA G. ROSANO-ORTEGA
����ҳ�룺1793 - 1800
�ؼ��ʣ����أ������ȼ�գ�����ﮣ��մ�
Key words��urea; oxide; combustion; lithium aluminate; ceramic
ժ Ҫ�����һ�ֺϳɦ�-LiAlO2���������������Ľ�ȼ�շ�ֱ�Ӻϳɦ�-LiAlO2��������������Լķ�Ӧ��ϵ�У�ԭ��Ϊ�������Ի�������Al2O3��LiOH��ȼ��Ϊ���ء�����1:1��1.5:1��2:1�ķǻ�ѧ����Li/AlĦ���ȣ���900��1000 ��C�·�Ӧ5 min���Ʊ�LiAlO2����������֯�ͽṹ���б���������Li/AlĦ���ȶԲ�����ò�ߦ����߷����²����ȶ��Ե�Ӱ�졣������������÷���ľ���ṹΪg-LiAlO2��a-LiAlO2����ȡ����Li/AlĦ���ȡ���ˣ��ø÷������Գɹ��ϳ�ש״��������״�Ͳ�״g-LiAlO2���������κκ����������÷��ս�����������õ���g-LiAlO2���ֽ⣬ֻ�γ�������Li2CO3���ɴ˿���ȷ�������ջᵼ�¹̽ᣬ������밵���Ч��ȡ�����֤������ȼ�շ������ߴ���g-LiAlO2����Ҫ������ǰ���塣
Abstract: An alternative solution for the direct formation of g-LiAlO2 was presented by a modified combustion method, to apply it to rather simple systems, utilizing non-oxidizer compounds such as Al2O3 and LiOH, and urea as fuel. LiAlO2 was prepared via non-stoichiometric 1:1, 1.5:1 and 2:1 of Li/Al molar ratios at 900 and 1000 ��C for 5 min. Textural and structural characterization of ��-LiAlO2 was performed. Also, the effect of different Li/Al molar ratios on material morphology and its stability before high gamma radiation gradients was evaluated. The results showed that the crystal structures of the obtained powders were ��-LiAlO2 and a-LiAlO2, depending on the Li/Al molar ratio. The results obtained demonstrate that g-LiAlO2 microbricks, polyhedral and laminar shapes can be successfully synthesized with the proposed method and without any subsequent process. Additionally, gamma irradiation showed that the g-LiAlO2 obtained does not decompose, forming only small amounts of Li2CO3. It can be established that the irradiation produces consolidation, which is not favourable for an efficient extraction of tritium. Finally, it could be demonstrated that nitrate precursors are not necessary in the combustion method to produce ��-LiAlO2 with high purity.
Trans. Nonferrous Met. Soc. China 28(2018) 1793-1800
Daniel CRUZ1, H. B. ORTIZ-OLIVEROS2, L. M.  1, R. M. FLORES-ESPINOSA2, G. ROSANO-ORTEGA3
1, R. M. FLORES-ESPINOSA2, G. ROSANO-ORTEGA3
1. Facultad de Ingenieria Quimica, Benemerita Universidad Autonoma de Puebla, 14 Sur y San Claudio, Ciudad Universitaria, C.P. 72570, Cd. Puebla, Mexico;
2. Instituto Nacional de Investigaciones Nucleares, Direccion de Investigacion Tecnologica, Apartado Postal 18-1027, 11801 Mexico, DF, Mexico;
3. Universidad Popular Autonoma del Estado de Puebla, 21 Sur no. 1103, Colonia Santiago, C.P. 72410, Puebla, Mexico
Received 2 November 2017; accepted 10 May 2018
Abstract: An alternative solution for the direct formation of y-LiAlO2 was presented by a modified combustion method, to apply it to rather simple systems, utilizing non-oxidizer compounds such as Al2O3 and LiOH, and urea as fuel. LiAlO2 was prepared via non-stoichiometric 1:1, 1.5:1 and 2:1 of Li/Al molar ratios at 900 and 1000 ��C for 5 min. Textural and structural characterization of ��-LiAlO2 was performed. Also, the effect of different Li/Al molar ratios on material morphology and its stability before high gamma radiation gradients was evaluated. The results showed that the crystal structures of the obtained powders were ��-LiAlO2 and ��-LiAlO2, depending on the Li/Al molar ratio. The results obtained demonstrate that y-LiAlO2 microbricks, polyhedral and laminar shapes can be successfully synthesized with the proposed method and without any subsequent process. Additionally, gamma irradiation showed that the y-LiAlO2 obtained does not decompose, forming only small amounts of Li2CO3. It can be established that the irradiation produces consolidation, which is not favourable for an efficient extraction of tritium. Finally, it could be demonstrated that nitrate precursors are not necessary in the combustion method to produce ��-LiAlO2 with high purity.
Key words: urea; oxide; combustion; lithium aluminate; ceramic
1 Introduction
It is well documented that economic development and consumption of energy resources in the last 200 years have caused the deterioration of environment, as evidenced by phenomena such as the degradation of water resources, deforestation, loss of biodiversity and global warming due to the emission of greenhouse gases [1,2]. Global warming is an alarming symptom that shows the unsustainability of the form of energy production and consumption [3-5]. Total energy consumption at the end of 2014 was 12.928 million tons of oil equivalent barrels [6]. According to the International Energy Agency, in the same year around 32190 million tons of CO2 were emitted [7].
In this context, nuclear energy has a fundamental role to meet the growing energy demand, as it is considered by many countries as an energy base of operation, contributing to the reduction of greenhouse gases [5,8].
Currently nuclear energy production was carried out by nuclear fission, having the disadvantage of generating a significant amount of radioactive waste. Projects with the International Thermonuclear Experimental Reactor, work on the development of nuclear fusion reactors, as well as large-scale and carbon-free source [9,10]. Fission reactors use lithium ceramics such as tritium breeders. A variety of breeding blanket concepts have been considered, ranging from more conservative concepts to higher-risk higher-payoff concepts for future fusion reactors. The major candidates for breeding materials consist of liquid breeders, mainly liquid metals although recently some attention has been given to FLiBe, and lithium ceramic breeders. Candidate breeder materials are mainly lithium oxide (Li2O), lithium orthosilicate (Li4SiO4), lithium metatitanate (Li2TiO3), lithium metazirconate (Li2ZrO3) and lithium aluminate (LiAlO2) [11-13]. LiAlO2 is one of the most promising ceramics for excellent chemical, structural and mechanical stability against intense fields of thermal and gamma radiation, as well as mixed fields (gamma-neutron) [14].
Additionally, it is used in polymeric composites, matrix of molten carbonate fuel cells (MCFC), electrolyte tiles in MCFC, among other applications such as electrical pumped ultraviolet-blue, light emitting diodes, laser, biosensors and photon detectors, due to its thermal conductivity [15-18].
The lithium aluminate crystal structure exhibits four allotropic forms depending on thermodynamic states and chemical environmental conditions, namely, ��-LiAlO2 (hexagonal), b-LiAlO2 (orthorhombic), y-LiAlO2 (tetragonal) and d-LiAlO2 (tetragonal) [11,19]. Among all polymorph modifications of LiAlO2, y-LiAlO2 is the most stable one, so that at elevated temperature (above 750 ��C, and present lattice parameters a=(5.1687�� 0.0005)  and c=(6.2679��0.0006)
and c=(6.2679��0.0006)  [18].
[18].
The ��-LiAlO2 has been obtained by several conventional powder synthesis techniques, including solid-state synthesis, co-precipitation and sol-gel process [20-23]. Although these methods have advantages, elevated temperatures and long heat treatment time are required to ensure the formation of pure y-LiAlO2. The situation is even more complicated due to many sophisticated techniques and time- consuming procedures that are required, which are not cost effective and represent obstacles to reproducibility and reliability of the final powder [15,24-26].
Combustion synthesis is an innovative and promising powder preparation method, it is a very rapid chemical process based on the principle of explosive decomposition of nitrate reagents and fuel mixtures, using the instantaneous heat generated by the chemical reaction between the desired metal nitrates (the oxidizing agent) and a reducing agent, which is called as the fuel [27-30]. The advantages of this method have been demonstrated as it needs little heating to start a rapid reaction and it permits considerable savings of time and energy compared with other techniques [31,32]. The fuel (glycine, urea, alanine, carbohidrazide) produces two different effects: first, it produces complexes with metal cations, which increases their solubility and prevents selective precipitation when water is evaporated; and second, it serves as fuel for the combustion reaction [33-36]. It was proved that combustion synthesis allowed the direct formation of the desired oxide compound with and without any subsequent calcination. For instance, starting from a stoichiometric mixture of lithium nitrate/aluminium as starting materials and urea/carbohidrazide as fuels, IANOS et al [29] and LI et al [33] reported the formation of pure y-LiAlO2 by the combustion synthesis only after thermal treatment at 900 and at 450 ��C, respectively.
In this work, the aim is to present an alternative solution for the direct formation of y-LiAlO2 by a modified combustion method, to apply it to rather simple systems, utilizing non-oxidizers compounds such as aluminium oxide (Al2O3), lithium hydroxide (LiOH) and, urea as fuel.
2 Experimental
2.1 Synthesis of ��-LiAlO2
��-LiAlO2 powder was prepared, as starting materials, analytical grade reagents without further purification: aluminium oxide (Al2O3 powder, J. T. Baker, 99.0%), lithium hydroxide (LiOH powder, Merck, 98.0%) and urea (CH4N2O powder, Merck, 99.5%). The required mass of each reactant was calculated from the desired mass of the products, according to the chemical reactions. In this work, the main parameters studied were: lithium hydroxide molar ratio and calcination temperature (900-1000 ��C). Different molar ratios of LiOH to Al2O3 (1:1, 1.5:1 and 2:1) were prepared and were placed in distilled water with urea at a molar ratio of 3:1 [37,38]. The mixture was dried at 60 ��C until most of the water was evaporated resulting in a humid integrated solid; this mixture was transferred into 30 mL crucible which was introduced during 5 min into a High Temperature Muffle Furnace Barnstead Thermolyne Corporation at different heat treatment temperatures (900-1000 ��C).
Samples were referred in this work as their LiOH:Al2O3:CH4N2O molar ratios. For example, sample (1:1:3) is the name given to the sample prepared with lithium hydroxide, aluminium oxide and urea with the corresponding LiOH:Al2O3:CH4N2O molar ratio of 1:1:3 at the study temperatures (900 ��C or 1000 ��C).
2.2 Textural and structural characterization of ��-LiAlO2
Powder XRD patterns were recorded on a Siemens D-5000 diffractometer using Cu K��1 radiation. The relative percentages of the various compounds present in the pellets were estimated semi-quantitatively from the total area under the most intense diffraction peak for each phase identified, with the simplifying assumption this was proportional to the volume fraction of each compound. In this way, the amounts of the crystalline compounds present in the samples were obtained within an estimated experimental error of 5% [39,40].
Since no internal standard was introduced, the X-ray absorption for each compound was assumed to be the same. Standard deviations of the measured samples were around 3.0%. A Philips XL-30 scanning electron microscopy (SEM), equipped with an energy dispersive X-ray spectroscopy (EDS) detector was used to study morphology of the samples. Powders were covered with gold to prevent the lack of conductivity. The thermogravimetric analyses (TGA) were performed with a heating rate of 10 ��C/min up to 800 ��C, flowing N2 (TGA 51, Thermogravimetric Analyzer, T.A. Instruments) to determine the mass loss at high temperatures.
2.3 Gamma irradiation of ��-LiAlO2
The gamma irradiation of y-LiAlO2 synthesized by the proposed method was performed in order to determine structural changes due to gamma radiation. Exposure to gamma radiation was performed in an industrial irradiator JS-6500 equipped with a 60Co source of 28971.56 TBq (1 July 2016). The solid was placed in aluminium containers (in duplicate) and subjected to a dose of 12000 kGy, applying a calculated dose rate of 14.09 kGy/h (8 September 2016).
3 Results and discussion
3.1 Synthesis and structural characterization of ��-LiAlO2
Synthesis of lithium aluminate ceramics by a modified combustion method was investigated using insoluble metal oxide. Most samples obtained utilizing different LiOH:Al2O3:CH4N2O molar ratios and different heating temperatures were white opaque powders.
Figure 1 compares the diffraction patterns of samples with n(LiOH):n(Al2O3):n(CH4N2O)=1:1:3, 1.5:1:3, 2:1:3 heat treated in a muffle furnace at 900 and 1000 ��C. It was found in sample (1:1:3)900 that y-LiAlO2 is a main compound (72%), but ��-LiAlO2 and Al2O3 were also present (14% in both samples) showing that the reaction was not completed under these working conditions (Fig. 1(a) and Fig. 2(a)). Although LiAlO2 exists in three forms, only �� and g phases were observed, it can be explained due to that �� phase form only exists at high pressures [41].

Fig. 1 XRD patterns of samples prepared by modified combustion method

Fig. 2 Percentage of products obtained by modified combustion method as function of temperature and lithium concentration according to XRD
In this work, the synthesis of LiAlO2 was carried out to atmospheric pressure. The low-temperature �� tetragonal structure transforms to the high-temperature g structure irreversibly by sintering the �� phase at 1350 ��C [40,42]; however, in this work samples were heated in the presence of urea (fuel) at 900 ��C. Although combustion temperature was not measured directly, we can predict that the combustion of the urea reached a temperature of at least 1200 ��C, to carry out the transformation of �� tetragonal structure to g structure of high-temperature. In the present work, the maximum combustion temperature during reaction was inferred due to the fact that:
1) The Li2O-Al2O3 phase diagram reported by NAKAGAWA et al [43] allows us to predict that the combustion of the urea reached a temperature of at least 1200 ��C, to carry out the transformation of �� tetragonal structure to g structure of high-temperature.
2) CHICK et al [34] have reported that fuel combustion can be achieved at one peak flame temperature of about 1450 ��C. Then, following reactions will occur:
LiOH+Al2O3+CH4N2O ��-LiAlO2
��-LiAlO2 ��-LiAlO2+H2O+CO2+N2 (1)
��-LiAlO2+H2O+CO2+N2 (1)
Heat due to combustion urea:
��-LiAlO2+CO2��Li2CO3+Al2O3 (2)
LiOH+CO2��Li2CO3+H2O (3)
3) Finally, ZU et al [44] have reported that the combustion process can be divided into three stages: a slowly heating zone (Stage I), a rapidly reaction zone (Stage II), and a cooling zone (Stage III). In Stage I, the oxidation reaction is essentially absent at the beginning due to the low temperature. As the sample is heated, the temperature continues to increase (about 1397 ��C). The obtained high heating rate indicates that the combustion reaction is violent with a large amount of heat released.
The presence of Al2O3 can be attributed to incomplete reaction with LiOH, probably due to Li sublimation at 900 ��C [40,41]. Although the XRD analyses did not show the presence of Li2CO3, the thermal analyses demonstrated that materials contained some minor impurities. Note that the lower limit detection by XRD technique depends on the scattering properties of the components of the materials.
In order to obtain y-LiAlO2, LiOH molar ratio was increased to 1.5, to compensate Li2O sublimation (Fig. 1(b)). Under these conditions in sample (1.5:1:3)900, it was observed that Al2O3 decreased from 14% to 4% probably because it was able to react with the LiOH in excess. The main product found was y-LiAlO2 (76%); however, ��-LiAlO2 increased up to 20% (Fig. 2(a)). High concentration Li reacts with Al2O3 to produce ��-LiAlO2 first, and subsequently, due to the combustion of urea, it produces y-LiAlO2 (Reaction 1). BECKERMAN et al [45] reported that y-LiAlO2 is produced by ��-LiAlO2 at temperatures between 650 and 1350 ��C. In order to obtain pure y-LiAlO2, LiOH molar ratio was increased from 1.5 to 2. Figure 1(c) shows that y-LiAlO2 increased considerably (88%) and ��-LiAlO2 decreased (8%); however, Al2O3 was still present (4%), probably due to decomposition of ��-LiAlO2 to Al2O3 according to reaction 2. It is well known that �� and b phases of LiAlO2 are unstable at high temperatures and are transformed into y-LiAlO2 when heat treated at temperatures of 650-1350 ��C for 4 h [45]. Therefore, in order to eliminate the ��-phase, samples were heat treated at 1000 ��C.
Increase of the muffle furnace temperature to 1000 ��C caused a considerable change in the percentage of the powders. Figures 1(d), (e) and (f) compare the diffraction patterns of samples with mole ratios of 1:1:3, 1.5:1:3 and 2:1:3 heat treated at 1000 ��C. y-LiAlO2 (Fig. 1(d)) was found as main compound (80%) in sample (1:1:3)1000, but ��-LiAlO2 and Al2O3 were also present (7% and 13% respectively, Fig. 2(b)) showing that the reaction is not completed under these working conditions. As at 1000 ��C Li2O sublimates in high proportion, LiOH molar ratio was increased to 1.5 to compensate Li2O sublimation. Under these conditions Al2O3 was present in 6% (Fig. 1(e) and Fig. 2(b)). The main product found was y-LiAlO2 (86%), and ��-LiAlO2 was still present in the sample (8%). By increasing the LiOH molar ratio to 2 (Fig. 1(f)), the only product obtained was y-LiAlO2 (100%) showing that molar ratio (2:1:3)1000 was the best condition to obtain pure y-LiAlO2 (Fig. 2(b)). Although Li2CO3 was not observed in XRD, TG analysis showed a mass loss attributed to carbonates present in the sample. Comparing the products obtained by the modified combustion method and the conventional combustion method [33], it was found that the conventional combustion method produces nitrogen contaminants. While in this work, due to the use of metal oxide instead metal nitrate, it was not observed contamination due to nitrate compounds. LI et al [33] have reported that y-LiAlO2 could be synthesized using lithium nitrate/aluminium nitrate as starting materials and urea as fuel by the combustion process. In this study, the pure y-LiAlO2 could be easily synthesized using lithium oxide/aluminium oxide and urea as fuel by the similar combustion process.
Representative scanning electron microscope (SEM) images of the reaction products are shown in Fig. 3 (1:1:3, 1.5:1:3 and 2:1:3 samples heat-treated at 900 ��C). As shown in Fig. 3(a), the product obtained by the modified combustion method with (1:1:3)900 molar ratio clearly reveals a brick-like morphology with one cross-section rectangular; hence they could be called ����microbricks����. Length of microbricks was 2-3.5 ��m. In this case, growth of bricks is not template-directed, or surfactant assisted, because no additional template or surfactants were introduced into the reaction. X-ray diffraction patterns in Fig. 1(a) showed that microbricks are composed mainly of crystalline tetragonal ��-LiAlO2 structures. Previous work reported the synthesis of Li2TiO3 by the modified combustion method, where reaction between LiOH and TiO2 depends on the lithium incorporation, diffusion and reaction into the TiO2 particles, in the present work, reaction between lithium and Al2O3 should be taking a similar mechanism as in the case of titanium [46]. Increasing LiOH concentration from 1 to 1.5 (Fig. 3(b)), polygonal particles were observed between 0.4 and 4.5 mm and agglomerates of about 5 mm. Finally, (2:1:3)900 sample (Fig. 3(c)) showed smaller particles of about 0.5-2 mm with laminar shapes ranging between which form considerably bigger agglomerates than the previous ones, of about 25 mm. The formation of the different structures can be attributed to the lithium volatilization during the calcination process. Due to easy volatilization of Li element, the excess Li element existing in the powder easily migrates to the surface of particles and turns into liquid phase. The local existence of liquid phase is conducive to the formation of new LiAlO2 particles at the site and also encourages the formation of networks between the particles [47,48].

Fig. 3 SEM images of LiAlO2 synthesized at 900 ��C with different molar ratios
Figure 4 shows micrographs of 1:1:3, 1.5:1:3 and 2:1:3 samples heat-treated at 1000 ��C. The morphology is denser than that discussed previously, mainly due to the sinterization effect produced by the high temperature. The particle size in sample (1:1:3)1000, ranges between 0.4 and 2 mm (Fig. 4(a)), with agglomerates of about 5 mm. (1.5:1:3)1000 sample (Fig. 4(b)) shows particles between 0.4 and 4.1 mm. Finally, (2:1:3)1000 sample (Fig. 4(c)) shows particles between 1 and 2 mm with a laminar shape and agglomerates considerably bigger ones (about 20 mm). Therefore, the morphology of the resulting y-LiAlO2 crystals also seems to depend upon the lithium concentration and temperature. It is well known that one of the major parameters that determine the phase formation of the ceramics is the Li/metal molar ratio and temperature [25,46]. In this study, the effects of Li/metal molar ratio and temperature on the characteristics of the synthesized powder were investigated in a precursor solution consisting of urea as a fuel and a mixed oxide of lithium and aluminium. Finally, morphology has been reported which affected product parameters such as surface reaction on the grain, diffusion through pores of sintered particles and diffusion through the boundary layer formed on the particle surface must be considered, and therefore tritium production can be altered [49].

Fig. 4 SEM images of LiAlO2 synthesized at 1000 ��C with different molar ratios
Figure 5 shows the thermogravimetric curves of samples with molar ratios (1:1:3)900 and (1.5:1:3) 900 (Figs. 5(a) and (b)), no significant mass loss was observed (<1%). The mass loss observed throughout the analysis can be attributed to the evaporation of Li element due to its low melting temperature [48].
Samples (2:1:3)900 and (2:1:3)1000, presented total mass loss of about 10% (Fig. 5(c)). The first mass loss of about 3%, in both samples, occurs at 25-250 ��C. This process is associated to the dehydration of the samples, which were hydrated after the combustion during the cooling by two processes [14]; one at T<573 K (physic- sorption) and second one at T>573 K (chemisorption). At temperatures between 580 and 650 ��C, a mass increase of ~1% could be observed, probably due to one of the following two processes: 1) chemical reactions with the components of the purge gas, N2, in the present case, forming non-volatile or less volatile compounds, and 2) physical transformations, as adsorption of gaseous products in the sample. Finally, the second mass loss of about 5%, between 600 and 800 ��C, is attributed to decarbonation process. The presence of carbonates in samples can be attributed to capturing process of CO2 over lithium aluminate [50].
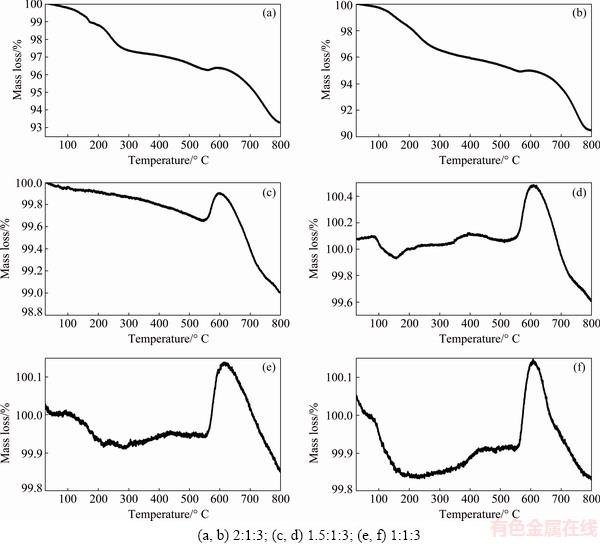
Fig. 5 TGA analyses of samples with different molar ratios prepared by modified combustion method at 900 (a, c, e) and 1000 ��C (b, d, f)
3.2 Gamma irradiation of ��-LiAlO2
The results obtained from the gamma irradiation of y-LiAlO2 showed that the material synthesized changed from white to grey colour as a consequence of gamma dose received. The XRD spectra obtained (not shown) of the y-LiAlO2 allowed to observe that the y-LiAlO2 decreases from 97% to 89% with the formation of Al2O3 and Li2CO3. Li2CO3 formation could be explained by the reaction of excess carbon and lithium present in the sample. Figure 6 shows the micrograph of y-LiAlO2 irradiated. It is observed that the gamma radiation causes changes in the morphology and particle size of the material. The radiation caused the formation of agglomerates of polyhedral particles of a size of approximately 170 mm.

Fig. 6 SEM image of y-LiAlO2 irradiated at 12000 kGy
However, y-LiAlO2 does not decompose, forming only small amounts of Li2CO3. It can be established that the irradiation produces consolidation, which is not favourable for an efficient extraction of tritium. The extraction efficiency is increased by decreasing the particle size.
4 Conclusions
1) The present study demonstrates that y-LiAlO2 microbricks, polyhedral and laminar shapes can be successfully synthesized from Al2O3 and LiOH by simple modified combustion method in 5 min without any subsequent process. A critical effect of Li/Al molar ratio and synthesis temperature was clearly demonstrated on the morphology of the products, at 900 ��C, n(Li)/n(Al)= 1:1 ratio gives microbricks, whereas n(Li)/n(Al)=2:1 ratio gives laminar shapes. However, at 1000 ��C microbricks structures were not observed, instead polyhedral structures were observed in (1:1:3)1000 and (1.5:1:3)1000 samples. Sample (2:1:3)1000 presented laminar shapes. ��-LiAlO2 was obtained with a yield of 100%.
2) The complexion effect of the fuel with Al2O3 was not possible; however, it was demonstrated that insoluble metal oxides can substitute the metal nitrates to produce LiAlO2 by a modified combustion method with high purity.
3) It can be established that the irradiation produces consolidation, which is not favourable for an efficient extraction of tritium. Finally, the modified combustion process described for the synthesis of y-LiAlO2 seems to have a great potential in the preparation of ceramic materials.
Acknowledgments
This work was supported by National Institute of Nuclear Research (ININ), Autonomous University of Puebla (BUAP) and co-financed by National Council of Science and Technology (CONACYT, Mexico). Furthermore, the authors thank to M. Espinosa, L. Banos, C. Rodr��guez an L. Carapia for the analysis.
References
[1] HOUGHTON R A, SKOLE D L. 1 Carbon [C]//The earth as transformed by human action. Global and regional changes in the biosphere over the past 300 years. New York, United States of America: University Press, 1995: 393-421.
[2] SALVATI L, SATERIANO A, ZITTI M. Long-term land cover changes and climate variations��A country-scale approach for a new policy target [J]. Land Use Policy, 2013, 30: 401-407.
[3] KHANDEKAR M L, MURTY T S, CHITTIBABU P. The global warming debate: A review of the state of science [J]. Pure and Applied Geophysics, 2005, 162: 1557-1586.
[4] LIOR N. Sustainable energy development: The present (2009) situation and possible paths to the future [J]. Energy, 2010, 35: 3976-3994.
[5] SAITO S. Role of nuclear energy to a future society of shortage of energy resources and global warming [J]. Journal of Nuclear Materials, 2010, 398: 1-9.
[6] BRITISH PETROLEUM. BP Statistical review of world energy 2015 [M]. London: BP, 2016.
[7] International Energy Agency. World energy outlook [M]. Paris, France: IEA, 2015.
[8] LOVERING J, YIP A, NORDHAUS T. Historical construction costs of global nuclear power reactors [J]. Energy Policy, 2016, 91: 371-382.
[9] CHARPIN J, BOTTER F, BRIEC M, RASNEUR B, ROTH E, ROUX N. Investigation of y lithium aluminate as tritium breeding material for a fusion reactor blanket [J]. Fusion Engineering and Design, 1989, 8: 407-413.
[10] JOHNSON C E. Tritium behavior in lithium ceramics [J]. Journal of Nuclear Materials, 1999, 270: 212-220.
[11] MA Sheng-gui, GAO Tao, LI Shi-chang, MA Xi-jun, SHEN Yan-hong. Theoretical investigations on the ��-LiAlO2 properties via first-principles calculation [J]. Fusion Engineering and Design, 2016, 113: 324-330.
[12] COLLING B R, MONK S D. Development of fusion blanket technology for the DEMO reactor [J]. Applied Radiation and Isotopes, 2012, 70: 1370-1372.
[13] KHORRAMIE S A, BAGHCHESARA M A, GOHARI D P. Fabrication of aluminum matrix reinforced with Al2ZrO3 nano particulates synthesized by sol-gel auto-combustion method [J]. Transactions of Nonferrous Metals Society of China, 2013, 23: 1556-1562.
[14] KAWAMURA Y, NISHIKAWA M, TANAKA K, MATSUMOTO H. Adsorption characteristics of water vapor on gamma-lithium aluminate [J]. Journal of Nuclear Science and Technology, 1992, 29: 436-444.
[15] KIM S D, HYUN S H, LIM T H, HONG S A. Effective fabrication method of rod-shaped ��-LiAlO2 particles for molten carbonate fuel cell matrices [J]. Journal of Power Sources, 2004, 137: 24-29.
[16] ANTOLINI E. The stability of LiAlO2 powders and electrolyte matrices in molten carbonate fuel cell environment [J]. Ceramics International, 2013, 39: 3463-3478.
[17] MA Sheng-gui, SHEN Yan-hong, GAO Tao, CHEN Pi-heng. First-principles calculation of the structural, electronic, dynamical and thermodynamic properties of ��-LiAlO2 [J]. International Journal of Hydrogen Energy, 2015, 40: 3762-3770.
[18] TSAI S C, CHEN H C, HUANG J C, CHANG C M, CHOU M C. Size and orientation effect on the mechanical properties of LiAlO2 single crystal [J]. Materials Science and Engineering: A, 2016, 677: 302-306.
[19] LI X, KOBAYASHI T, ZHANG F, KIMOTO K, SEKINE T. A new high-pressure phase of LiAlO2 [J]. Journal of Solid State Chemistry, 2004, 177: 1939-1943.
[20] OKSUZOMER F, SKOC S N, BOZ I, GURKAYNAK M A. Effect of solvents on the preparation of lithium aluminate by sol�Cgel method [J]. Materials Research Bulletin, 2004, 39: 715-724.
[21] KHOMANE R B, AGRAWAL A, KULKARNI B D. Synthesis and characterization of lithium aluminate nanoparticles [J]. Materials Letters, 2007, 61: 4540-4544.
[22] KHARLAMOVA O A, MITROFANOVA R P, ISUPOV V P. Mechanochemical synthesis of fine-particle ��-LiAlO2 [J]. Inorganic Materials, 2007, 43: 645-650.
[23] NASCIMENTO A C, QUEIROZ M E, MOHALLEM D S. Synthesis of high purity ��-lithium aluminate by freeze drying [J]. Advanced Materials Letters, 2016, 7: 267-270.
[24] NAYAK M, KUTTY T R N, JAYARAMANB V, PERIASWAMY G. Preparation of the layered double hydroxide (LDH) LiAl2(OH)7��2H2O, by gel to crystallite conversion and a hydrothermal method, and its conversion to lithium aluminates [J]. Journal of Materials Chemistry, 1997, 7: 2131-2137.
[25] RIBEIRO R A, SILVA G, MOHALLEM N D S. The influences of heat treatment on the structural properties of lithium aluminates [J]. Journal of Physics and Chemistry of Solids, 2001, 62: 857-864.
[26] KWON S W, PARK S B. Effect of precursors on the morphology of lithium aluminate prepared by hydrothermal treatment [J]. Journal of Materials Science, 2000, 35: 1973-1978.
[27] RAMANATHAN S, KAKADE M B, ROY S K, KUTTY K K. Processing and characterization of combustion synthesized YAG powders [J]. Ceramics International, 2003, 29: 477-484.
[28] IANOS R, LAZAU I, PACURARIU C, BARVINSCHI P. Peculiarities of CaO��6Al2O3 formation by using low-temperature combustion synthesis [J]. European Journal of Inorganic Chemistry, 2008: 925-931.
[29] IANOS R, LAZAU I, PACURARIU C. Metal nitrate/fuel mixture reactivity and its influence on the solution combustion synthesis of c-LiAlO2 [J]. Journal of Thermal Analysis and Calorimetry, 2009, 97: 209-214.
[30] CHENG J, ZOU Xiao-ping, LI F, ZHANG Hong-dan, REN Peng-fei. Synthesis of bamboo-like carbon nanotubes by ethanol catalytic combustion technique [J]. Transactions of Nonferrous Metals Society of China, 2006, 16: 435-437.
[31] CRUZ D, BULBULIAN S. Synthesis of Li4SiO4 by a modified combustion method [J]. Journal of the American Ceramic Society, 2005, 88: 1720-1724.
[32] INGLE J T, GAWANDE A B, SONEKAR R P, OMANWAR S K, WANG Y, ZHAO L. Combustion synthesis and optical properties of Oxy-borate phosphors YCa4O(BO3)3:RE3+ (RE=Eu3+, Tb3+) under UV, VUV excitation [J]. Journal of Alloys and Compounds, 2014, 585: 633-636.
[33] LI F, HU K, LI J, ZHANG D, CHEN G. Combustion synthesis of ��-lithium aluminate by using various fuels [J]. Journal of Nuclear Materials, 2000, 300: 82-88.
[34] CHICK L A, PEDERSON L R, MAUPIN G D, BATES J L, THOMAS L E, EXARHOS G J. Glycine-nitrate combustion synthesis of oxide ceramic powders [J]. Materials Letters, 1990, 10: 6-12.
[35] STELLA K C, NESARAJ A S. Effect of fuels on the combustion synthesis of NiAl2O4 spinel particles [J]. Iranian Journal of Materials Science and Engineering, 2010, 7: 36-44.
[36] LI Y, LIU M. Gas sensing properties of Y-doped ZnO nanosheets synthesized via combustion method [J]. Transactions of Nonferrous Metals Society of China, 2015, 25: 2247-2252.
[37] CRUZ D, BULBULIAN S. Synthesis of lithium silicate tritium breeder powders by a modified combustion method [J]. Journal of Nuclear Materials, 2003, 312: 262-265.
[38] CRUZ D, BULBULIAN S, LIMA E, PFEIFFER H. Kinetic analysis of the thermal stability of lithium silicates (Li4SiO4 and Li2SiO3) [J]. Journal of Solid State Chemistry, 2006, 179: 909-916.
[39] JIMENEZ-BECERRIL J, BOSCH P, BULBULIAN S. Synthesis and characterization of ��-LiAlO2 [J]. Journal of Nuclear Materials, 1991, 185: 304-307.
[40] PFEIFFER H, KNOWLES K M. Reaction mechanisms and kinetics of the synthesis and decomposition of lithium metazirconate through solid-state reaction [J]. Journal of the European Ceramic Society, 2004, 24: 2433-2443.
[41] COOK L P, PLANTE E R. Phase diagram of the system Li2O-Al2O3 [J]. Ceramic Transactions, 1992, 27: 193-222.
[42] LU C H, WEI-CHENG L. Reaction mechanism and kinetics analysis of lithium nickel oxide during solid-state reaction [J]. Journal of Materials Chemistry, 2000, 10: 1403-1407.
[43] NAKAGAWA K, OHZU H, AKASAKA Y, TOMIMATSU N. Allotropic phase transformation of lithium aluminate in MCFC electrolyte plates [J]. Denki Kagaku, 1997, 65: 231-235.
[44] ZU B, LI F, SUN Y, WANG Q, WU Y, ZHU Z. The effects of additives on the combustion characteristics of aluminum powder in steam [J]. RSC Advances, 2017, 7: 5725-5732.
[45] BECKERMAN S J, FORD R B, NEMETH M T. Conversion of gamma lithium aluminate to lithium aluminium carbonate hydroxide hydrate [J]. Powder Diffraction, 1996, 11: 312-317.
[46] CRUZ D, PFEIFFER H, BULBULIAN S. Synthesis of Li2MO3 (M=Ti or Zr) by the combustion method [J]. Solid State Sciences, 2006, 8: 470-475.
[47] ZHAU H, LIU H, LI H, ZHENG L, HU C, ZHANG X, LI Q, YANG J. Hydrothermal-assisted sintering strategy towards Porous- and hollow-structured LiNb3O8 anode material [J]. Nanoscale Research Letters, 2017, 12: 463.
[48] ZHAU H, LIU H, LI H, ZHENG L, HU C, ZHANG X, LI Q, YANG J. The effects of Li/Nb ratio on the preparation and photocatalytic performance of Li-Nb-O compounds [J]. Nanoscale Research Letters, 2017, 12: 496.
[49] CARRERA L M,  J, BASURTO R, ARENAS J, LOPEZ B E, BULBULIAN S, BOSCH P. Tritium recovery from nanostructured LiAlO2 [J]. Journal of Nuclear Materials, 2001, 299: 242-249.
J, BASURTO R, ARENAS J, LOPEZ B E, BULBULIAN S, BOSCH P. Tritium recovery from nanostructured LiAlO2 [J]. Journal of Nuclear Materials, 2001, 299: 242-249.
[50] KORAKE P V, GAIKWAD A G. Capture of carbon dioxide over porous solid adsorbents lithium silicate, lithium aluminate and magnesium aluminate at pre-combustion temperatures [J]. Frontiers of Chemical Science and Engineering, 2011, 5: 215-226.
Daniel CRUZ1, H. B. ORTIZ-OLIVEROS2, L. M.  1, R. M. FLORES-ESPINOSA2, G. ROSANO-ORTEGA3
1, R. M. FLORES-ESPINOSA2, G. ROSANO-ORTEGA3
1. Facultad de Ingenieria Quimica, Benemerita Universidad Autonoma de Puebla, 14 Sur y San Claudio, Ciudad Universitaria, C.P. 72570, Cd. Puebla, Mexico;
2. Instituto Nacional de Investigaciones Nucleares, Direccion de Investigacion Tecnologica, Apartado Postal 18-1027, 11801 Mexico, DF, Mexico;
3. Universidad Popular Autonoma del Estado de Puebla, 21 Sur no. 1103, Colonia Santiago, C.P. 72410, Puebla, Mexico
ժ Ҫ�����һ�ֺϳɦ�-LiAlO2���������������Ľ�ȼ�շ�ֱ�Ӻϳɦ�-LiAlO2��������������Լķ�Ӧ��ϵ�У�ԭ��Ϊ�������Ի�������Al2O3��LiOH��ȼ��Ϊ���ء�����1:1��1.5:1��2:1�ķǻ�ѧ����Li/AlĦ���ȣ���900��1000 ��C�·�Ӧ5 min���Ʊ�LiAlO2����������֯�ͽṹ���б���������Li/AlĦ���ȶԲ�����ò�ߦ����߷����²����ȶ��Ե�Ӱ�졣������������÷���ľ���ṹΪy-LiAlO2�ͦ�-LiAlO2����ȡ����Li/AlĦ���ȡ���ˣ��ø÷������Գɹ��ϳ�ש״��������״�Ͳ�״y-LiAlO2���������κκ����������÷��ս�����������õ���y-LiAlO2���ֽ⣬ֻ�γ�������Li2CO3���ɴ˿���ȷ�������ջᵼ�¹̽ᣬ������밵���Ч��ȡ�����֤������ȼ�շ������ߴ���y-LiAlO2����Ҫ������ǰ���塣
�ؼ��ʣ����أ������ȼ�գ�����ﮣ��մ�
(Edited by Xiang-qun LI)
Corresponding author: H. B. ORTIZ-OLIVEROS; Tel: +52-5553297200-13460; E-mail: huemantzin.ortiz@inin.gob.mx
DOI: 10.1016/S1003-6326(18)64823-X

