��ԭ���������п���Ͽ����ۺϻ����̺�п
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2017���5��
�������ߣ���� ��ѩ�� ��� ������ ���� ���� ����
����ҳ�룺1172 - 1179
�ؼ��ʣ���п���Ͽ�˫��ˮ�����ǣ��ۺ����ã��̣�п������
Key words��manganese-zinc compound ore; hydrogen peroxide; sucrose; comprehensive utilization; manganese; zinc; leaching
ժ Ҫ�����û�ԭ������Ժ�Ǧ�����ĵ�Ʒλ��п���Ͽ�����ۺ����á�ͨ������Mn-Zn-H2Oϵ�Ħ�-pHͼ��������з�����֪���̺�п��pH-2~5.61�����������¿���ͬʱ������ʵ����������˫��ˮ�����Ƕ���ǿ���̺�пͬʱ��������Ч��ԭ��������������Ǧ���������и���������˫��ˮΪ��ԭ���������Ը��Ͽ���л�ԭ����ʱ���̺�п�Ľ����ʷֱ�Ϊ95.88%��99.23%��ͬʱǦ���������е�Ʒλ�ֱ���13.21%��489.36 g/t����������Ϊ��ԭ��ʱ���̺�п�Ľ����ʷֱ�Ϊ98.26%��99.62%��ͬʱǦ���������е�Ʒλ�ֱ���13.92%�� 517.87 g/t��
Abstract: Comprehensive utilization of low grade manganese-zinc compound ore containing lead and silver with a method of reductive acid leaching was studied. According to the ��-pH diagram of Mn-Zn-H2O system, Mn and Zn can be leached simultaneously in the pH range of -2 to 5.61. The results showed that both hydrogen peroxide and sucrose were effective reductants which could intensify the simultaneous leaching of Mn and Zn into leachate as well as enrich Pb and Ag in the residue. 95.88% of Mn and 99.23% of Zn were extracted when the compound ore was leached with hydrogen peroxide in sulfuric acid media, meanwhile the contents of Pb and Ag in the residue were enriched to 13.21% and 489.36 g/t, respectively. When sucrose was used as the reductant, the leaching efficiencies of Mn and Zn separately achieved 98.26% and 99.62%, and contents of Pb and Ag in the residue were as high as 13.92% and 517.87 g/t, respectively.
Trans. Nonferrous Met. Soc. China 27(2017) 1172-1179
Qian LI, Xue-fei RAO, Bin XU, Yong-bin YANG, Ting LIU, Tao JIANG, Long HU
School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China
Received 11 March 2016; accepted 1 September 2016
Abstract: Comprehensive utilization of low grade manganese-zinc compound ore containing lead and silver with a method of reductive acid leaching was studied. According to the ��-pH diagram of Mn-Zn-H2O system, Mn and Zn can be leached simultaneously in the pH range of -2 to 5.61. The results showed that both hydrogen peroxide and sucrose were effective reductants which could intensify the simultaneous leaching of Mn and Zn into leachate as well as enrich Pb and Ag in the residue. 95.88% of Mn and 99.23% of Zn were extracted when the compound ore was leached with hydrogen peroxide in sulfuric acid media, meanwhile the contents of Pb and Ag in the residue were enriched to 13.21% and 489.36 g/t, respectively. When sucrose was used as the reductant, the leaching efficiencies of Mn and Zn separately achieved 98.26% and 99.62%, and contents of Pb and Ag in the residue were as high as 13.92% and 517.87 g/t, respectively.
Key words: manganese-zinc compound ore; hydrogen peroxide; sucrose; comprehensive utilization; manganese; zinc; leaching
1 Introduction
With the continual exploitation of mineral resources, the grades of exploitable mineral resources take on degressive tendence, and the ore compositions of mineral resources become more and more complicated. Therefore, the researches on comprehensive utilization of low grade polymetallic ore have great significance [1].
Over the past few decades, the researches on comprehensive utilization of manganese-iron compound ore containing zinc and lead were mainly focused on pyrometallurgical processes. The most representative traditional pyrometallurgical processes were pellet roasting and blast furnace smelting, in which Zn and Pb were separated from Mn and Fe by volatilization [2]. Although the utilization efficiencies of Fe and Mn by pyrometallurgical processes are relatively high, the recovery rates of Zn and Pb are not satisfactory. On the other hand, the volatilization of Zn can cause the nodulation in blast furnace and block the gas pipeline. Furthermore, these processes have the common disadvantages of high energy consumption, heavy pollution and high investment.
In recent years, many researchers have paid more efforts to hydrometallurgical leaching. However, manganese usually occurs as pyrolusite in low grade ores. Pyrolusite, which is a kind of high valence manganese oxide, needs to be converted to low valence state Mn (II) before leaching because Mn (IV) is insoluble in dilute acid and alkaline media [3,4]. The pyro-reduction processes of pyrolusite mainly used coals as reductant in industries [5-7]. Later on, sulfur and cornstalk instead of coal as reductant also have been explored in laboratory [8,9]. However, all these pyro-reduction processes require more energy consumption and lead to serious environmental pollution and large amount of greenhouse gas emissions [10]. Compared with the pyro-reduction processes, the directly reductive leaching processes, using pyrite, oxalic acid, hydrogen peroxide, glucose and CaS as reductants, have drawn more attention recently because of their low reagent consumption, high efficiency and environmental friendliness [11-14].
All these methods mentioned above were mainly focused on the recovery of Mn. However, hydrometallurgical research on the comprehensive utilization of manganese-zinc compound ore was seldom reported. In this study, the material was a kind of manganese-zinc compound ore containing Pb and Ag. Combining with its characteristics, a method of reductive acid leaching was proposed to utilize the main valuable elements comprehensively. The reduction of pyrolusite as well as the extractions of Mn and Zn can be accomplished in one step leaching by this method. Mn and Zn were dissolved in solution, and Pb and Ag were enriched in the residue because the dissolutions of Pb and Ag were both negligible. The obtained residue containing high Pb and Ag can be mixed with lead concentrate appropriately and then used in lead smelting process [15,16].
2 Experimental
2.1 Material and reagents
The material used in this work was a kind of low grade manganese-zinc compound ore. In order to identify the main chemical composition and valuable elements in this ore, chemical multi-element analysis and chemical phase analysis have been done.
As shown in Table 1, the contents of Mn (27.38%), Zn (11.46%), Pb (4.06%) and Ag (150.53 g/t) in this compound ore are all relatively high, and these metallic elements need to be recovered effectively.
Table 1 Main chemical composition of manganese-zinc compound ore (mass fraction, %)
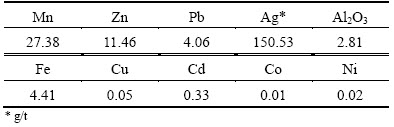
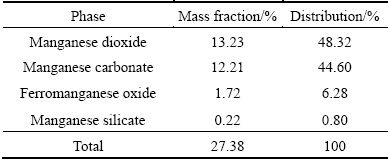
As seen in Table 2, the main phases of Mn are manganese dioxide (MnO2) and manganese carbonate (MnCO3). As shown in Table 3, 68.5% of Zn occurs as zinc oxide, and 29.84% of Zn combined with manganese.
Table 2 Results of chemical phase analysis of Mn in manganese-zinc compound ore
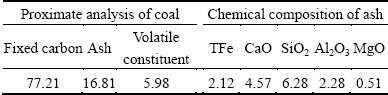
The reagents, such as sulfuric acid, sucrose, hydrogen peroxide and oxalate, used in this study, were all analytically pure. The grade of pyrite was 99%, and the type of the coal was anthracite. Proximate analysis of anthracite coal and chemical composition of ash was shown in Table 4. Deionized water was used throughout all experiments.
Table 3 Results of chemical phase analysis of Zn in manganese-zinc compound ore
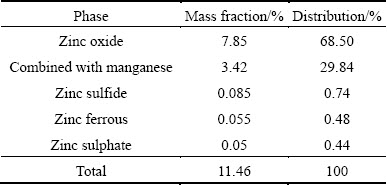
Table 4 Proximate analysis of anthracite coal and chemical composition of ash (mass fraction, %)
2.2 Methods
The raw material was crushed and finely ground to more than 70% finer than 0.074 mm. In each leaching experiment, 20.00 g of ground material and desired quantities of reductant were added to sulfuric acid solution, and the liquid-to-solid ratio was fixed as 5:1. Sulfuric acid was chosen as the lixiviant due to the characters of high boiling point and good stability. Because the hydrogen peroxide is instable and easily decomposed, reductive acid leaching experiments with this reductant were performed under the low leaching temperature of (25��0.5) ��C and the slow agitation speed of 120 r/min in order to reduce its loss caused by decomposition of hydrogen peroxide. There were two methods of adding the hydrogen peroxide into the leaching system: one was that hydrogen peroxide was dropwise added at the beginning of the leaching, the other was that hydrogen peroxide was dropwise added after 30 min of leaching. Both of the two adding methods lasted for 15 min. A faster agitation speed of 400 r/min was applied and different temperatures of 50, 60, 70, 80 and 90 ��C were tested in reductive acid leaching with other reductants, such as pyrite, pulverized coal, oxalate and sucrose. The fluctuation ranges of the leaching temperatures in this work are all within ��0.5 ��C.
The analyses of Mn, Zn, Pb and Ag in solid and leachate were all implemented with an atomic absorption spectrometer (AA-6800, Shimadzu). Phase constitution analysis was employed to determine the distribution percentage of an element for its every phase in research sample. Phase constitutions of Mn and Zn were identified with chemical dissolution method in the Analysis and Test Center of Changsha Research Institute of Mining and Metallurgy, China. Oxygen concentration was tested by dissolved oxygen meter (A336278, Milwaukee).
2.3 Theoretical basis
Based on the data in related literatures [17,18], the thermodynamic equilibrium systems of Mn-H2O and Zn-H2O were analyzed and calculated. The main equations of Mn-H2O and Zn-H2O systems were given in Tables 5 and 6, respectively.
The ��-pH diagrams of the Mn-H2O system and Zn-H2O systems can be drawn by setting p=101.325 kPa, [Mn]=1 mol/L and [Zn]=1 mol/L. Figure 1 shows the superposed ��-pH diagram for the Mn-H2O and Zn-H2O systems under standard conditions. The solid line in Fig. 1 represents the equilibrium line of Mn-H2O system and the dotted line represents the equilibrium line of Zn-H2O system. The area surrounded by thick lines represents the coexistence area of Mn2+ and Zn2+. The existence of the coexistence area in the pH range of -2 to 5.61 indicated that Mn and Zn can be leached simultaneously in acid medium.
It can be seen that the areas of ZnO and Zn2+ are both quite large, and ZnO transforming into Zn2+ only needs to change the value of pH. ��-pH diagram of the Mn-H2O system shows that the higher manganese valence state corresponds to the higher potential value. This implies that the MnO can be transformed into Mn2+ more easily than MnO2, and MnO2 dissolving into Mn2+ decreases the pH value and reduces the potential value. From the above, the leaching of ZnO is much easier than that of MnO2, and the reductant and acid should be added to regulate the potential and pH value so as to maintain Mn2+ and Zn2+ to coexist in solution.
3 Results and discussion
3.1 Effect of reductant type
The selection of efficient and environmentally friendly reductant is the key for the efficient utilization of this manganese-zinc compound ore. A promising reductant should meet the following requirements: 1) high leaching efficiencies of Mn and Zn, 2) fewer impurities brought in the leaching solution, 3) little toxicity and low cost.
In order to select a promising reductant, a series of contrast tests have been done, in which pyrite, pulverized coal, oxalate, sucrose and hydrogen peroxide were considered. The results obtained are shown in Table 7.
Table 5 Main electrode reactions and equilibrium equations in Mn-H2O system
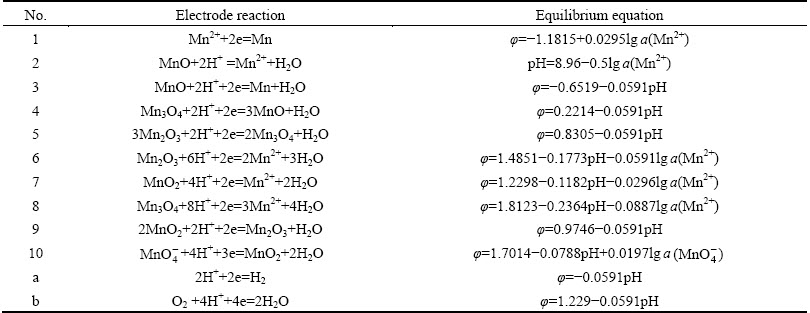
Table 6 Main electrode reactions and equilibrium equations in Zn-H2O system
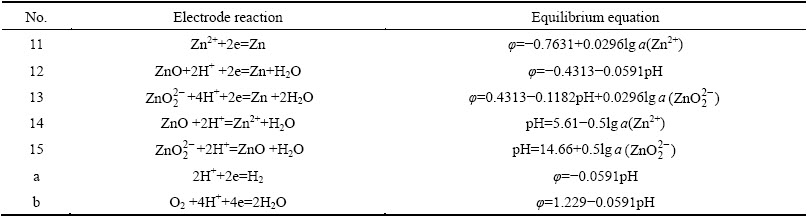
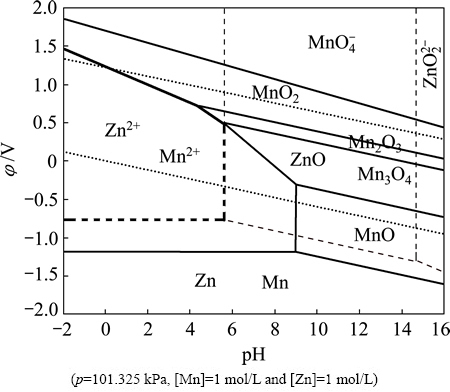
Fig. 1 Superposed ��-pH diagram for Mn-H2O and Zn-H2O systems
Table 7 Results of reductive acid leaching using different reductants
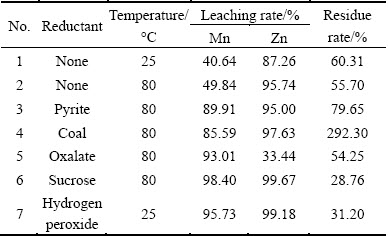
The leaching rates of Mn are less than 50% under the condition of leaching without reductant, and the results are consistent with the phase constitution of Mn, i. e., more than 48% of Mn occurs as MnO2. Besides, the leaching rates of Zn are also lower than those of reductant acid leaching with coal, sucrose and hydrogen peroxide. This implies that a part of Zn is combined with MnO2. It can also be seen from Table 7 that, the leaching rates of Mn and Zn obtained under leaching temperature of 80 ��C are higher than those under 25 ��C. This reveals that the leaching temperature influences the leaching rates of Mn and Zn to a certain degree.
When using pyrite or coal as reductant, the leaching rates of Mn are both improved obviously but still lower than 90%. However, a lot of impurities will be brought in the leaching system with the addition of these reductants [10]. Furthermore, the lead and silver cannot be enriched efficiently due to the high residue rate, especially when coal is used. High dosage of coal also results in high production cost. When oxalate is used as reductant, the leaching rate of Mn is increased to 93.01%. However, the zinc leaching rate is only 33.44% due to the formation of zinc-oxalate precipitates [19].
When sucrose and hydrogen peroxide are used, the leaching rates of Mn and Zn are both more than 95%. Compared with other residue rates shown in Table 7, the residue rates gained by using hydrogen peroxide and sucrose as reductants are relatively low. By combining with the chemical multielement analysis and the chemical phase analysis in Section 2.1, the total contents of manganese and zinc compounds in compound ore were calculated about 69%, and the main chemical phases of Mn and Zn were MnCO3, MnO2 and ZnO which can be dissolved by reductive acid leaching with hydrogen peroxide and sucrose, thus the low residue rates were almost consistent with the corresponding leaching rates shown in Table 7, and a small amount of impurities have been leached. Moreover, the element compositions of the hydrogen peroxide and sucrose are C, H and O, which would be decomposed as CO2 and H2O when used as reductants, so fewer impurities are brought in leaching system [20]. Comprehensively considering, both sucrose and hydrogen peroxide are promising reductants.
3.2 Intensification of Mn and Zn simultaneous leaching with H2O2
Hydrogen peroxide is not only a kind of reductant but also an oxidant. Hence, the ferrous oxide and sulfides such as FeS and ZnS could be oxidized along with the reduction of pyrolusite, without bringing in any impurities [20]. The oxidation of ZnS is beneficial to the increase of Zn leaching rate, and the oxidation of Fe2+ is favorable to the subsequent iron removal in process of lixivium purification [21]. The possible reaction equations are proposed as follows:
MnO2+H2O2+2H+=Mn2++2H2O+O2�� (1)
ZnS+4H2O2=Zn2++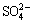 +4H2O (2)
+4H2O (2)
2Fe2++H2O2+2H+=2Fe3++2H2O (3)
In order to find the optimum conditions of reductive acid leaching with hydrogen peroxide as reductant, four main factors, including sulfuric acid concentration, adding method of hydrogen peroxide, hydrogen peroxide dosage and leaching time, were investigated in the following sections.
3.2.1 Effect of sulfuric acid concentration and H2O2 adding method
As shown in Fig. 2, the leaching rates of Mn and Zn with two adding methods are both increased with the increase of sulfuric acid concentration until 2.16 mol/L. Besides, the leaching rates of Zn are higher than those of Mn in the whole abscissa range.
It can also be seen that, at all sulfuric acid concentration levels, the leaching rates of Mn and Zn with adding method II are higher than those with adding method I. The likely reasons for this are as follows: in the initial stage of leaching, large numbers of bubbles were released, which was due to the dissolution of carbonate in sulfuric acid medium, and the bubbles might result in the loss of hydrogen peroxide; when hydrogen peroxide was added after 30 min of leaching, the loss of hydrogen peroxide which was caused by CO2 bubbles can be avoided, for the carbonate was almost reacted with sulfuric acid. Hence, more MnO2 is reduced by adding hydrogen peroxide with method II, and thus more zinc combining with manganese minerals can also be leached.
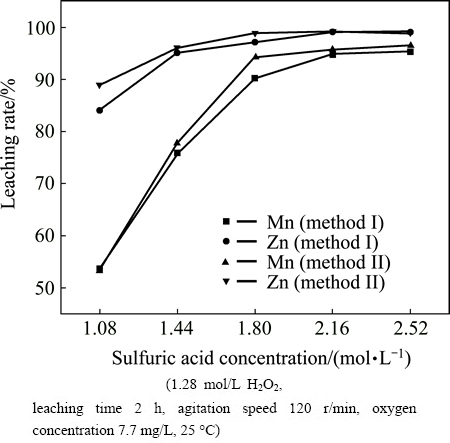
Fig. 2 Effect of sulfuric acid concentration and H2O2 adding method on leaching rates of Mn and Zn
According to the analysis of the superposed ��-pH diagrams for Mn-H2O and Zn-H2O systems in Section 2.3, the leaching of ZnO is much easier than that of MnO2. As shown in Tables 2 and 3, the main chemical phase of Zn is ZnO in this compound ore, meanwhile, MnO2 is the highest percentage chemical phase of Mn. Therefore, the leaching rates of Zn are higher than that of Mn.
3.2.2 Effect of H2O2 dosage and leaching time
The effects of hydrogen peroxide dosage and leaching time on leaching rates of Mn and Zn were studied by varying the hydrogen peroxide dosage from 0 to 1.48 mol/L and leaching time from 1 to 3 h. The results are given in Fig. 3.
Figure 3(a) shows that the hydrogen peroxide dosage plays a critical role in the reductive acid leaching process. Compared with leaching results obtained without hydrogen peroxide, the leaching rate of Mn is rapidly increased from 40.64% to 95.73% when 1.28 mol/L hydrogen peroxide is used, and the leaching rate of Zn is also increased from 87.26% to 99.18%. The result can be explained by the fact that the MnO2 is insoluble in sulfuric acid solution in the absence of a reductant [4]. High valence state Mn(IV) in the form of MnO2 was converted to MnO in the presence of reductant hydrogen peroxide, and MnO could be easily leached in sulfuric acid solution. Moreover, the hydrogen peroxide dosage has a great influence on the reduction degree of MnO2 [14]. Therefore, the more the hydrogen peroxide is used, the higher the manganese leaching rate is. Meanwhile, the increase of leaching rate of Zn may mainly be attributed to the oxidation of ZnS and the exposure of zinc which is wrapped by manganese phases.
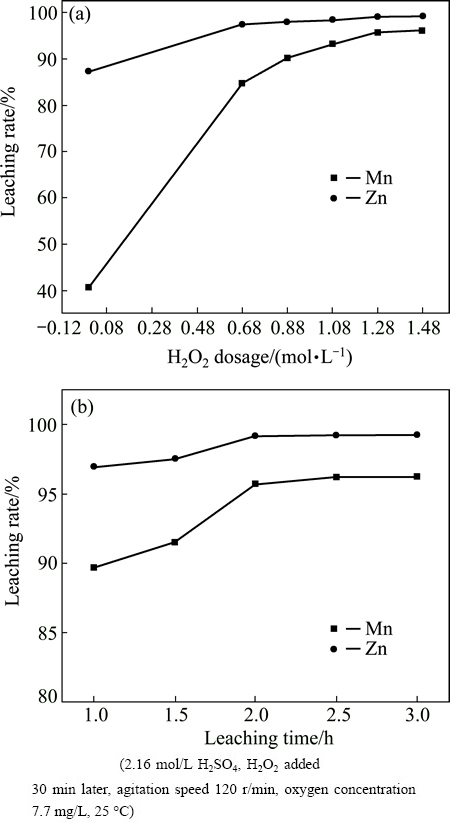
Fig. 3 Effect of H2O2 dosage (a) and leaching time (b) on leaching rates of Mn and Zn
As shown from Fig. 3(b), leaching rates of Zn and Mn give similar trend, and they are both increased with the extension of leaching time until 2 h, and then keep steady. After 1 h of leaching, the leaching rates of Mn and Zn are separately attained 89.70% and 96.94%. This means that most of the Mn and Zn in the compound ore have been leached, especially the components like MnCO3 and ZnO which can be leached easily. As the leaching time is inadequate, there are parts of the Mn and Zn remaining to be dissolved, and this part of Mn may be MnO2 and Zn is combined with it. The chemical phase analysis of Zn in Table 3 shows that more than 29% Zn is combined with Mn and the leaching rates of Mn and Zn shown in Table 7 can support this assumption. Thus, when MnO2 is gradually reduced and leached as the leaching time prolonging, the leaching rate of Zn is increased at the same time.
From the above, optimal reaction conditions are established as follows: 2.16 mol/L sulfuric acid, 1.28 mol/L hydrogen peroxide added after 30 min of leaching, and 2 h leaching time. 95.73% of Mn and 99.18% of Zn are extracted under the optimum conditions, and meanwhile, the contents of Pb and Ag in the residue are 12.99% and 479.67 g/t, respectively.
A scale-up verification experiment has been done with 200.00 g of ground material in 2 L beaker under the optimal conditions described above, 60.47 g leaching residue is obtained, and the chemical multi-element analysis of the residue is shown in Table 8. The leaching rates of Mn and Zn are separately calculated to be 95.88% and 99.23%. The content of Pb in the residue is 13.21% and the content of Ag is 489.36 g/t, and both are enriched more than 3.25 times. Compared with the leaching rates of Mn and Zn, and the contents of Pb and Ag in basic experiment under the same conditions, the results of the scale-up verification experiment have a little difference, and the difference is within the range of permissible error.
Table 8 Main chemical components of reductive acid leaching residue by H2O2 (mass fraction, %)

3.3 Intensification of Mn and Zn simultaneous leaching with sucrose
In this section, reductive acid leaching was carried out in sulfuric acid solution in the presence of sucrose. Although sucrose is a kind of non-reducing sugar, it is used as reductant for it can be hydrolyzed to glucose and fructose in acid medium, and both glucose and fructose are reducing sugar [22]. The leaching process of MnO2 can be described by the following reaction equation:
24MnO2+C12H22O11+48H+=24Mn2++12CO2+35H2O (4)
The effect of sucrose dosage and leaching temperature on leaching rates of Mn and Zn are presented in Fig. 4. From the results shown in Fig. 4(a), it can be observed that the leaching rate of Mn is rapidly increased with the increase of sucrose dosage, and the leaching rate of Zn is also slightly increased. In addition, sucrose dosage of 43.8 mmol/L is adequate for the reductive acid leaching. This indicates that the sucrose is an efficient reductant for pyrolusite, which is in accordance with results in Ref. [23], and the impact trend of sucrose dosage on the leaching rates is similar to that of hydrogen peroxide.
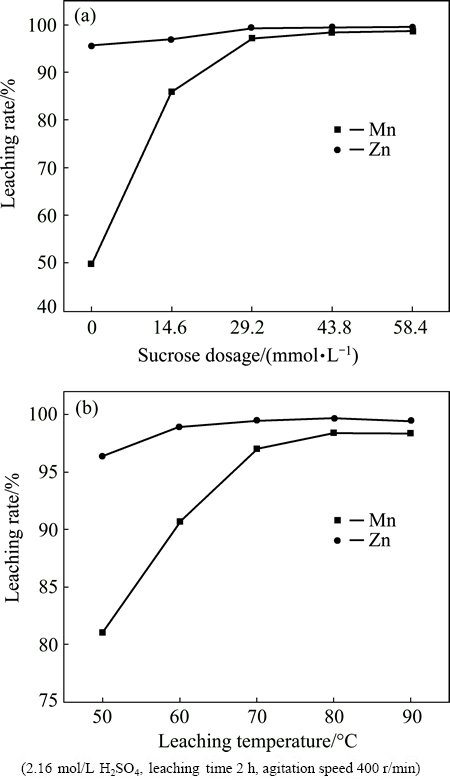
Fig. 4 Effect of sucrose dosage (a) and leaching temperature (b) on leaching rates of Mn and Zn
As indicated in Fig. 4(b), the leaching rates of Mn and Zn are increased with the increase of leaching temperature until they reached the maximum at 80 ��C, and afterward are basically unchanged. The leaching rates of Mn and Zn respectively reached 98.40% and 99.67% under the optimum conditions of sucrose dosage of 43.8 mmol/L and leaching temperature of 80 ��C, and meanwhile, the contents of Pb and Ag in the residue are separately as high as 14.10% and 520.39 g/t, respectively.
A scale-up verification experiment of reductive acid leaching with sucrose has also been done by using 200.00 g of ground material in 2 L beaker under the optimal conditions above. The obtained leaching residue is 58.24 g, and Table 9 shows the chemical multi-element analysis of the residue. The calculated leaching rates of Mn and Zn are 98.26% and 99.62%, respectively. The content of Pb in the residue is enriched to 13.92% and Ag is enriched to 517.87 g/t, and both enrichment ratios are more than 3.42. Compared with the results of its basic experiment under the same conditions, the differences between them are also within the range of permissible error, and the results show a good reproducibility.
Table 9 Main chemical components of reductive acid leaching residue by sucrose (mass fraction, %)

From the results presented above, it can be seen that high leaching rates of Mn and Zn and enrichment ratios of Pb and Ag are achieved by reductive acid leaching with both hydrogen peroxide and sucrose. Reductive acid leaching by using hydrogen peroxide as reductant can be carried out at room temperature, which can obviously reduce the energy consumption. Besides, the Fe2+ can be oxidized by hydrogen peroxide along with the reduction of MnO2 without bringing in any impurities. There still exist some cons, for example, the price of hydrogen peroxide is more expensive and the adding method is inconvenient. Compared with hydrogen peroxide, the reductive acid leaching by using sucrose has the advantages of low dosage, cheap price and convenient use. However, its leaching temperature needs 80 ��C and it will no doubt increase the energy consumption. In summary, both hydrogen peroxide and sucrose are efficient reductant for comprehensive utilization of the manganese-zinc compound ore, and have a very good application prospect in reductive acid leaching.
4 Conclusions
1) Manganese and zinc can be leached simultaneously by using reductant and sulfuric acid to regulate the potential and pH value of leaching system, and the Pb and Ag can be enriched effectively in the residue.
2) Compared with pyrite, coal and oxalate, hydrogen peroxide and sucrose are both effective in intensifying the simultaneous leaching of Mn and Zn.
3) 95.88% of Mn and 99.23% of Zn are extracted by reductive acid leaching using hydrogen peroxide as reductant. Meanwhile, the Pb and Ag in the residue are enriched to 13.21% and 489.36 g/t, respectively.
4) The leaching rates of Mn and Zn by reductive acid leaching with the reductant of sucrose reaches 98.26% and 99.62%, respectively, and the contents of Pb and Ag in the residue are as high as 13.92% and 517.87 g/t, separately.
5) The method proposed in this study has the advantages of mild reaction conditions, high efficiency and little environment pollution.
References
[1] ACHARYA C, KAR R N, SUKLA L B. Studies on reaction mechanism of bioleaching of manganese ore [J]. Minerals Engineering, 2003, 16: 1027-1030.
[2] LI Zhao-jia, WAN Hong-qiang, ZHAO Qiang, NING Shun-ming. New technology of comprehensive utilization of lead and zinc manganese ore [J]. Mining and Metallurgical Engineering, 2001, 31: 85-88. (in Chinese)
[3] HARIPRASAD D, DASH B, GHOSH M K, ANAND S. Leaching of manganese ores using sawdust as a reductant [J]. Minerals Engineering, 2007, 20: 1293-1295.
[4] WU Fang-fang, ZHONG Hong, WANG Shuai, LAI Su-feng. Kinetics of reductive leaching of manganese oxide ore using cellulose as reductant [J]. Journal of Central South University, 2014, 21: 1763-1770.
[5] ERIC R H, BURUCU E. The mechanism and kinetics of the carbothermic reduction of Mamatwan manganese ore fines [J]. Minerals Engineering, 1992, 5: 795-815.
[6] SWAMY Y V, BHOI B, PRAKASH S, RAY H S. Enrichment of the manganese to iron of ferruginous low-grade manganese ores using solid reductant [J]. Minerals and Metallurgical Processing, 1998, 15: 34-37.
[7] ZHANG Yuan-bo, ZHAO Yi, YOU Zhi-xiong, DUAN Dao-xian, LI Guang-hui, JIANG Tao. Manganese extraction from high-iron- content manganese oxide ores by selective reduction roasting-acid leaching process using black charcoal as reductant [J]. Journal of Central South University, 2015, 22: 2515-2520.
[8] CHENG Zhuo, ZHU Guo-cai, ZHAO Yuan. Study in reduction-roast leaching manganese from low-grade manganese dioxide ores using cornstalk as reductant [J]. Hydrometallurgy, 2009, 96(1-2): 176-179.
[9] ZHANG Yuan-bo, YOU Zhi-xiong, LI Guang-hui, JIANG Tao. Manganese extraction by sulfur-based reduction roasting�Cacid leaching from low-grade manganese oxide ores [J]. Hydrometallurgy, 2013, 133: 126-132.
[10] ZHANG Wen-sheng, CHENG Chu-yong. Manganese metallurgy review. Part I: Leaching of ores/secondary materials and recovery of electrolytic/chemical manganese dioxide [J]. Hydrometallurgy, 2007, 89: 137-159.
[11] LI Chang-xin, ZHONG Hong, WANG Shuai, XUE Jian-rong, WU Fang-fang, ZHANG Zhen-yu. Manganese extraction by reduction- acid leaching from low-grade manganese oxide ores using CaS as reductant [J]. Transactions of Nonferrous Metals Society of China, 2015, 25: 1677-1684.
[12] SAHOO R N, NAYAK P K, DAS S C. Leaching of manganese from low grade manganese ore using oxalic acid as reductant in sulfuric acid solution [J]. Hydrometallurgy, 2001, 62: 157�C163.
[13] TIAN Xi-ke, WEN Xiao-xia, YANG Chao, LIANG Yu-jun, PI Zheng-bang, WANG Yan-xin. Reductive leaching of manganese from low-grade manganese dioxide ores using corncob as reductant in sulfuric acid solution [J]. Hydrometallurgy, 2010, 100: 157-160.
[14] NAYL A A, ISMAIL I M, ALY H F. Recovery of pure MnSO4��H2O by reductive leaching of manganese from pyrolusite ore by sulfuric acid and hydrogen peroxide [J]. International Journal of Mineral Processing, 2011, 100: 116-123.
[15] ZHANG Le-ru. Modern lead metallurgy [M]. Changsha: Central South University Press, 2013. (in Chinese)
[16] WEI Wei. The direction and distribution of sliver in the Kivcet collocation zinc leaching residue lead smelting process [D]. Changsha: Central South University, 2012. (in Chinese)
[17] DEAN J A. Lange,s handbook of chemistry [M]. WEI Jun-fa, Transl. Beijing: Science Press, 2003.
[18] CHEN Jia-yong. Hydrometallurgical manual [M]. Beijing: Metallurgical Industry Press, 2005. (in Chinese)
[19] SAYILGAN E, KUKRER T, FERELLA F, AKCIL A, VEGLIO F, KITIS M. Reductive leaching of manganese and zinc from spent alkaline and zinc-carbon batteries in acidic media [J]. Hydrometallurgy, 2009, 97: 73-79.
[20] LIU Ting. Hydrometallurgical extraction and separation of Mn and Zn from manganese-zinc compound ore [D]. Changsha: Central South University, 2015. (in Chinese)
[21] LIU Ming. The separating of iron, zinc, and manganese in mixed sulphate solution by solvent extraction [D]. Changsha: Central South University, 2005. (in Chinese)
[22] LASHEEN T A, EL-HAZEK M N, HELAL A S, EL-NAGAR W. Recovery of manganese using molasses as reductant in nitric acid solution [J]. International Journal of Mineral Processing, 2009, 92:109-114.
[23] BEOLCHINI F, PETRANGELI PAPINI M, TORO L, TRIFONI M,  F. Acid leaching of manganiferous ores by sucrose: Kinetic modelling and related statistical analysis [J]. Minerals Engineering, 2001, 14: 175-184.
F. Acid leaching of manganiferous ores by sucrose: Kinetic modelling and related statistical analysis [J]. Minerals Engineering, 2001, 14: 175-184.
�� 幣���ѩ�ɣ��� ���������� �ã��� �Σ��� ��
���ϴ�ѧ ��Դ�ӹ������﹤��ѧԺ����ɳ 410083
ժ Ҫ�����û�ԭ������Ժ�Ǧ�����ĵ�Ʒλ��п���Ͽ�����ۺ����á�ͨ������Mn-Zn-H2Oϵ�Ħ�-pHͼ��������з�����֪���̺�п��pH-2~5.61�����������¿���ͬʱ������ʵ����������˫��ˮ�����Ƕ���ǿ���̺�пͬʱ��������Ч��ԭ��������������Ǧ���������и���������˫��ˮΪ��ԭ���������Ը��Ͽ���л�ԭ����ʱ���̺�п�Ľ����ʷֱ�Ϊ95.88%��99.23%��ͬʱǦ���������е�Ʒλ�ֱ���13.21%��489.36 g/t����������Ϊ��ԭ��ʱ���̺�п�Ľ����ʷֱ�Ϊ98.26%��99.62%��ͬʱǦ���������е�Ʒλ�ֱ���13.92%�� 517.87 g/t��
�ؼ��ʣ���п���Ͽ�˫��ˮ�����ǣ��ۺ����ã��̣�п������
(Edited by Wei-ping CHEN)
Foundation item: Projects (51574284, 51504293) supported by the National Natural Science Foundation of China; Project (2013IB020) supported by the Science and Technology Program of Yunnan Province, China; Project (CSUZC201606) supported by the Open-End Fund for the Valuable and Precision Instruments of Central South University, China
Corresponding author: Bin XU; Tel: +86-731-88830547; E-mail: xuandy_16@126.com
DOI: 10.1016/S1003-6326(17)60137-7

