Adsorption characteristics of carbon tetrachloride from aqueous solution onto polyacrylonitrile-based activated carbon fiber
来源期刊:中南大学学报(英文版)2010年第5期
论文作者:刘文霞 官宝红 于洁
文章页码:972 - 978
Key words:adsorption; carbon tetrachloride; activated carbon fiber; isotherm; kinetics; breakthrough curve
Abstract: The isotherm, mechanism and kinetics of carbon tetrachloride (CT) adsorption by polyacrylonitrile-based activated carbon fiber (PAN-ACF) were investigated in batch reactors and a continuous flow reactor, and the regeneration of PAN-ACF was also studied. Freundlich and Dubinin-Radushkevich (D-R) adsorption equations can well describe the adsorption isotherm. CT is mainly adsorbed on the exterior surface of PAN-ACF with low boundary layer effect and rate-controlling step of intra-particle diffusion. The adsorption dynamics in the batch reactor well fits with the pseudo-first-order model, and the breakthrough curves in the continuous flow reactor can be well described by the Yoon-Nelson model. The ACF can be recycled through thermal regeneration, whereas the adsorption capacity decreases from 7.87 to 4.98 mg/g after the fourth regeneration. 78%-94% of CT can be removed from the wastewater of a fluorine chemical plant on a pilot scale, which confirms the efficacy of ACF under industrial conditions. The results indicate that PAN-ACF is applicable to CT removal from wastewater.
J. Cent. South Univ. Technol. (2010) 17: 972-978
DOI: 10.1007/s11771-010-0586-1
![]()
LIU Wen-xia(刘文霞), GUAN Bao-hong(官宝红), YU Jie(于洁)
Department of Environmental Engineering, Zhejiang University, Hangzhou 310027, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2010
Abstract: The isotherm, mechanism and kinetics of carbon tetrachloride (CT) adsorption by polyacrylonitrile-based activated carbon fiber (PAN-ACF) were investigated in batch reactors and a continuous flow reactor, and the regeneration of PAN-ACF was also studied. Freundlich and Dubinin-Radushkevich (D-R) adsorption equations can well describe the adsorption isotherm. CT is mainly adsorbed on the exterior surface of PAN-ACF with low boundary layer effect and rate-controlling step of intra-particle diffusion. The adsorption dynamics in the batch reactor well fits with the pseudo-first-order model, and the breakthrough curves in the continuous flow reactor can be well described by the Yoon-Nelson model. The ACF can be recycled through thermal regeneration, whereas the adsorption capacity decreases from 7.87 to 4.98 mg/g after the fourth regeneration. 78%-94% of CT can be removed from the wastewater of a fluorine chemical plant on a pilot scale, which confirms the efficacy of ACF under industrial conditions. The results indicate that PAN-ACF is applicable to CT removal from wastewater.
Key words: adsorption; carbon tetrachloride; activated carbon fiber; isotherm; kinetics; breakthrough curve
1 Introduction
Carbon tetrachloride (CT) has been widely used as solvent, degreaser, and extractant in industry. The improper use and disposal of CT result in the pollution of soil and water. The environmental persistence, carcinogenicity and mutagenicity of CT have made it a serious environmental concern. CT has been listed in the priority pollutants list in many countries and organizations. For example, CT was ranked to be the 43rd in the 2005 CERCLA (Comprehensive Environmental Response, Compensation, and Liability Act, U.S. Environmental Protection Agency) priority list of hazardous substances in view of its known and suspected toxicities. The maximum concentration level of CT is set at 5 and 2 μg/L in safe drinking water act [1] and standards for drinking water quality (GB 5749―2006), respectively. Many kinds of methods were applied to removing CT from water or wastewater, such as reductive dechlorination [2], biodegradation [3], oxidation [4], ultrasonic destruction [5], photo- electrochemical degradation [6], and adsorption [7]. Adsorption is one of the most effective and widely used techniques because of its relatively simple design, easy operation and simple regeneration.
Activated carbon fiber (ACF) has played an important role in adsorption technology over the last few years. Compared with conventional granular activated carbon (GAC) and powder activated carbon (PAC), ACF has the advantages of smaller fiber diameter, which lessens diffusion limitations and favors rapid adsorption and desorption, more concentrated pore size distribution and higher adsorption capacity at low concentration of adsorbate [8]. SHIRATORI et al [9] reported that ACFs were composed of spherical microdomain units with the size of a few nanometers measured by scanning tunneling microscopy. The diffusion distance in ACF is, on average, 100 times smaller than that in GAC, resulting in the higher adsorption rate onto ACF than onto GAC [10]. The surface chemistry composition of ACF significantly influences its adsorption capacities [8]. The two important heteroatoms are oxygen and nitrogen, which affect the surface acidic, basic and hydrophilic properties of ACF [8]. ACF has been extensively and successfully applied to removing a wide range of pollutants from municipal and industrial wastewater as well as drinking water. One of the most important applications is the removal of toxic organic compounds from wastewater, such as diuron [11-12], 2,4-dichlorophenal [13], p-nitroaniline [14], and p- nitrophenol [15]. But there are few reports concerning the adsorption of CT from water or wastewater by ACF. To the best of our knowledge, there is limited research on CT removal from fluorination industrial effluent by ACF.
In this work, polyacrylonitrile-based ACF (PAN-ACF) was used to adsorb CT from aqueous solution. The adsorption isotherm, mechanism, kinetics, and thermal regeneration were evaluated in batch reactors, while a continuous flow reactor was used to study the breakthrough curve. In addition, the feasibility of PAN-ACF for industrial wastewater treatment was explored on a pilot scale.
2 Experimental
2.1 Materials
Commercial PAN-ACF in the form of cloth was used. The specific surface area of the ACF is 1 182.5 m2/g calculated by the BET method. The fresh ACF was washed repeatedly with deionized water, dried in an oven set at 60 ℃ for 2 h and then cut into pieces of 1 cm×1 cm before use.
CT of analytical grade was used to prepare the simulative wastewater. The pH of the solution was adjusted by HCl or NaOH of analytical grade. The industrial wastewater was the effluent from a fluorine chemical plant.
2.2 Procedures
In batch reactors, ACF was added into a 250 mL conical flask and mixed with 250 mL CT solution. The flask was tightly closed with a silicone membrane cap, and then stirred in a thermostatic water shaker at 25 ℃ and a constant rate of 200 r/min. The stirring time was fixed at 30 min. Blank test was measured as described above without PAN-ACF in the conical flask.
Adsorption in the continuous flow reactor was conducted in a fixed column with diameter of 3.0 cm and length of 100 cm. PAN-ACF was loosely filled in the column capped by a sieve plate. A nylon mesh was placed to avoid flow maldistribution and the loss of ACF, and there was a buffer space above the mesh. The solution up flowed in the column by a peristaltic pump. A pilot scale of fixed column (d 0.5 m×1.8 m) was used to treat the effluent from a fluorine chemical plant.
The used ACF was rinsed several times with tap water and then desorbed in a vacuum oven at 120 ℃ for 24 h. After thermal desorption, the ACF was rinsed repeatedly with deionized water till no CT could be detected in the water and then dried in an oven at 60 ℃ for 2 h.
2.3 Analytical methods
At a desired time interval, a solution or wastewater sample of 5.0 mL was withdrawn from the reactor with a 5-mL gastight syringe and then filtered with a 0.22 μm membrane filter. The concentration of CT was measured using GC 9790A (Fuli Analytical Instrument Co. Ltd., Wenling, China) equipped with an electron capture detector and an OV-225 capillary column (30 m× 0.25 mm×0.25 μm) by direct injection of 0.6 μL filtrate. Temperatures of injector, column and detector were fixed at 220, 180 and 230 ℃ correspondingly. Carrier gas was N2 with a flow rate of 2.0 mL/min. The determination was conducted with an external standard method. The amount of CT adsorbed on per unit mass of ACF was calculated from the mass balance equation as:
![]() (1)
(1)
where qe is the equilibrium adsorption capacity (mg/g); C0 is the initial concentration (mg/L); Ce is the equilibrium concentration (mg/L); V is the volume of the solution (L); and m is the mass of PAN-ACF (g).
3 Results and discussion
3.1 Effect of pH and ACF dosage
Fig.1 shows the effect of pH and PAN-ACF dosage on the adsorption. The equilibrium adsorption capacity is higher under neutral condition than under acidic or basic condition, but the discrepancy is small when the ACF dosage is above 2.0 g/L. For ACF dosage from 0 to 2.0 g/L, the removal efficiencies increase distinctly to 77.46% at pH 1.26, 84.58% at pH 6.93, and 75.47% at pH 11.53, respectively. The corresponding equilibrium adsorption capacities are 7.37, 7.40 and 6.24 mg/g. These results indicate that pH slightly affects the adsorption and the influence can be neglected when the adsorbent dosage or adsorption time is enough.
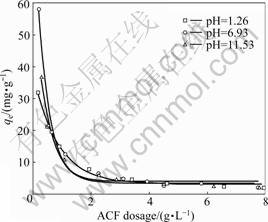
Fig.1 Effect of pH and ACF dosage on adsorption of CT onto polyacrylonitrile-based activated carbon fiber (CT initial concentration: 20 mg/L)
The solution pH is an important parameter for adsorption. The surface binding sites of the adsorbent, aqueous chemistry and the charge of adsorbate are affected by solution pH, which consequently influences the interaction between the adsorbate and adsorbent. The chemical groups on the PAN-ACF surface include acidic groups (such as carboxyl and phenolic hydroxyl), and basic groups (such as amidocyanogen and imino group). The adsorption of p-nitroaniline [14] and pentachlorophenol [10] onto ACF is significantly affected by pH, which may be due to the polarity of these substances. Whereas CT is a nonpolar molecule, and ACF usually possesses a nonpolar surface because of the high temperature condition of its manufacture [16]. Thus, the adsorption driving force is mainly van der Waals force for CT adsorption onto PAN-ACF. Consequently, the influence of pH on the adsorption is slight.
3.2 Adsorption isotherm
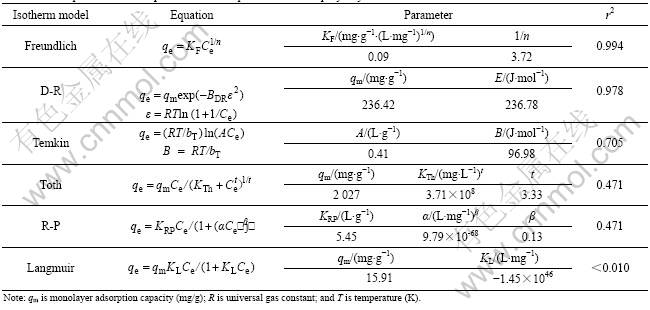
The adsorption isotherm of CT on PAN-ACF, as shown in Fig.2, was obtained under the conditions of initial CT concentration 20 mg/L, ACF dosage 0.80- 8.0 g/L and pH 6.93. The isotherm equations of Freundlich, Dubinin-Radushkevich (D-R) [17], Temkin [18], Toth [19], Redlich-Peterson (R-P) [20] and Langmuir are applied to characterizing the adsorption process. Both of the adsorption constants and the correlation coefficients (r2) for these equations are shown in Table 1.
The values of R2 decrease in the order of Freundlich, D-R, Temkin, Toth, R-P, and Langmuir equation with 0.994, 0.978, 0.705, 0.471, 0.471, and less than 0.010, suggesting that Freundich and D-R equations are better than the other equations in describing the adsorption behavior of CT onto PAN-ACF. Langmuir equation assumes uniform adsorption energies on the adsorbent
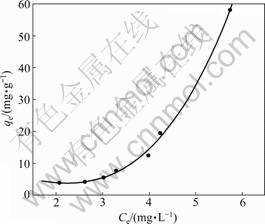
Fig.2 Adsorption isotherm of CT onto PAN-ACF
surface with no transmigration of adsorbate in the plane of the surface, and no interaction among the adsorbed molecules [21]. The contradiction between the results and Langmuir equation indicates that the surface energies of PAN-ACF are not uniform. Whereas the consistence between the results and Freundlich equation reveals that the energy distribution for the adsorption sites is of essentially an exponential type [21]. Thus, some sites are highly energetic and bind CT molecules strongly and the others are much less energetic and bind CT molecules weakly. This implies that the adsorption tends to be of multilayer other than of a homogeneous monolayer. The free energy (E) derived from D-R equation is usually regarded as a measurement for the transfer of adsorbate from the infinity of the solution to the surface of the adsorbent [17]. The magnitude of E gives information about the type of adsorption. If E is smaller than 8 kJ/mol, adsorption type is physisorption
Table 1 Adsorption isotherm equations for adsorption of CT onto polyacrylonitrile-based activated carbon fiber
due to the weak van der Waals forces [22]. E calculated in this experiment is 236.78 J/mol, indicating a physisorption.
3.3 Adsorption mechanism
The intraparticle diffusion model presented as Eq.(2) [23], is applied to revealing the adsorption mechanism. The model presumes that there are at least two regions in the intraparticle diffusion plot. The first, sharper region is the instantaneous adsorption or external surface adsorption. The second region is the gradual adsorption stage in which intraparticle diffusion occurs. The third region may exist in some cases related to the final equilibrium stage.
![]() (2)
(2)
where qt is the adsorption amount at time t (mg/g); Ki is the rate constant (g/(mg・min1/2)); and Ci is the intercept of stage i (mg/g). The increase of C1 indicates the greater boundary layer effect for the instantaneous or external surface adsorption. The larger the C2, the greater the contribution of the surface adsorption in the intraparticle diffusion stage. If the intraparticle diffusion occurs, qt will be linear with t1/2.
Fig.3 shows the plot of qt versus t1/2. The two linear regions in the plot suggest that the process involves surface adsorption and intraparticle diffusion. The values of K1 and C1 are 2.91 g/(mg・min1/2) and -0.08 mg/g respectively, which demonstrates that the adsorption of CT onto PAN-ACF exterior surface is fast and the boundary layer effect is small. The value of C2 is 4.39 mg/g, far away from zero, indicating that surface adsorption plays a significant role in the intraparticle diffusion stage.
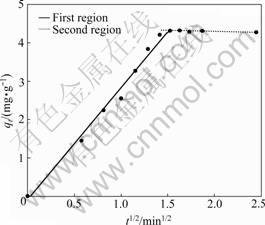
Fig.3 Plot of intraparticle diffusion model for adsorption of CT onto PAN-ACF
Adsorption data are further explored using the model of BOYD et al [24] and REICHENBERG [25] to determine the rate-controlling step of the adsorption.
![]() (3)
(3)
![]() (4)
(4)
when F > 0.85,
![]() (5)
(5)
when F < 0.85,
![]() (6)
(6)
where F is the fractional attainment of equilibrium at time t; Re and B are the mass transfer coefficients (min-1). The linearity of ln(1-F) with t or Bt with t distinguishes whether external or intraparticle mass transport controls the adsorption rate [26]. The parameters are listed in Table 2.
Table 2 Parameters of correlated mass transfer equation for CT adsorption onto polyacrylonitrile-based activated carbon fiber

The intraparticle diffusion is the rate-controlling step because of the higher value of r2 of the intraparticle mass transfer model. The results elucidate that CT is mainly adsorbed onto the external surface of PAN-ACF with the intraparticle diffusion as the rate-controlling step.
3.4 Adsorption kinetics
Fig.4 shows the uptake of CT by PAN-ACF versus adsorption time. The uptake of CT increases sharply in the first 5 min and then reaches equilibrium of 4.22 mg/g. The adsorption equilibrium time of CT onto ACF is
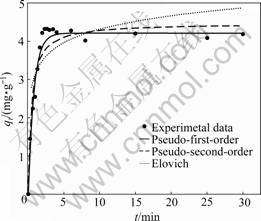
Fig.4 Uptake of CT by PAN-ACF versus adsorption time (CT initial concentration: 10 mg/L; PAN-ACF dosage: 2.0 g/L; pH: 1.29)
much shorter than that of CT onto AC which is over 30 min reported by MILCHERT et al [27]. The rapid adsorption during the first 5 min results from the abundant vacant surface sites that are available for CT. The surface sites are difficult to be occupied after a lapse of time because of the accumulation of CT adsorbed, which leads to the dynamic equilibrium between the desorption and adsorption.
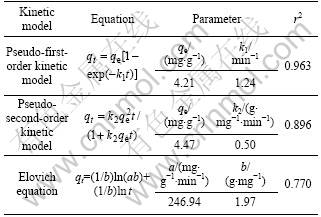
The kinetic models of pseudo-first-order [28], pseudo-second-order [29] and Elovich [30], are applied to fitting the dynamic data. The calculated constants are shown in Table 3. According to correlation coefficient, the pseudo-one-order model appears to be more reasonable for CT adsorption onto PAN-ACF than the other two models. This indicates that the adsorption is a pseudo-first-order reaction. The Elovich model, which is believed to be one of the most useful models for characterizing chemisorption gives the lowest correlation coefficient value of 0.770. Thus, it is most likely that the adsorption is not chemisorption.
Table 3 Adsorption kinetic models for adsorption of CT onto PAN-ACF
3.5 Adsorption in continuous flow reactor
The breakthrough curves in the continuous flow reactor for different flow rates are illustrated in Fig.5. Yoon-Nelson semi-empirical model [31] expressed as Eq.(7) is used to analyze the experimental data. The adsorption capacity is calculated from Eq.(8). The constants calculated are listed in Table 4.
![]() (7)
(7)
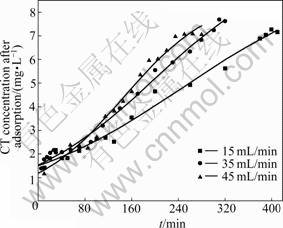
Fig.5 Breakthrough curves of CT adsorption onto PAN-ACF at different flow rates in continuous flow reactor (CT initial concentration: 8.0 mg/L; PAN-ACF mass: 20 g; pH: 1.32)
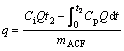 (8)
(8)
where Ci is the initial concentration of CT (mg/L); Cp is the concentration of CT after adsorption (mg/L); t is the adsorption time (min); τ is the time when Cp is half of Ci (min); t2 is the time when Cp is 95% of Ci (min); Q is the flow rate (mL/min); k′ is the flow rate constant (min-1); and mACF is the mass of PAN-ACF filled in the column (g).
The breakthrough curves are steep with slope factors of 0.015 mg/(L・min) at flow rate of 15 mL/min, 0.020 mg/(L・min) at flow rate of 35 mL/min, and 0.025 mg/(L・min) at flow rate of 45 mL/min, suggesting that the mass transfer resistance towards diffusion inside the pores of the column is small. The values of τ calculated from the semiempirical model are 161 min (Q=15 mL/min), 129 min (Q=35 mL/min) and 107 min (Q=45 mL/min), respectively. The values of τ obtained from the experiments are 162, 136 and 114 min in turn. The consistency of τ between the model and the experiment results and the high correlation coefficient indicates that the model can reasonably describe the adsorption in the reactor. Thus, the concentration of CT after adsorption at a given time and the breakthrough time can be calculated from this model.
τ and t2 decrease and k′ increases along with the increase of Q from 15 to 45 mL/min. The adsorption
Table 4 Adsorption parameters in continuous flow reactor at different flow rates

capacity at t2 nearly doubles from 1.10 to 2.01 mg/g when Q increases from 15 to 35 mL/min, and then varies a little at 45 mL/min. The best flow rate in this experiment is 35 mL/min considering the adsorption capacity at t2.
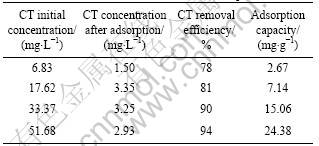
The continuous wastewater treatment was carried out in a pilot scale setup. The wastewater contained 5- 100 mg/L CT and was of pH 1.0-2.0. The dosage of ACF packed was 5.0 kg, the flow rate was 0.5 m3/h, and the hydraulic retention time in the column was fixed at 30 min. The results are listed in Table 5. The removal efficiency and the adsorption capacity increase from 78% to 94% and 2.67 mg/g to 24.38 mg/g when the influent concentration varies from 6.83 to 51.68 mg/L. It proves that PAN-ACF is efficient for removing CT from the industrial wastewater.
Table 5 Pilot experiment results of CT removal by PAN-ACF from industrial wastewater of fluorine chemical plant
3.6 Adsorption onto regenerated PAN-ACF
The uptake of CT by the fresh and regenerated PAN-ACF is shown in Fig.6. The removal efficiencies together with the pseudo-first-order kinetic parameters are listed in Table 6.
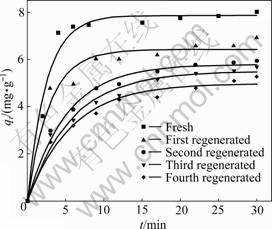
Fig.6 Adsorption of CT onto fresh and regenerated PAN-ACF (initial CT concentration: 18 mg/L; PAN-ACF dosage: 2.0 g/L; pH: 1.29)
Constant k1 decreases from 0.41 through 0.35 and 0.20 to 0.17 min-1 for four times regeneration. The decrease of k1 may be due to the fact that the pore size
Table 6 Removal efficiencies and parameters of pseudo-first- order kinetic model for CT adsorption onto fresh and regenerated PAN-ACF
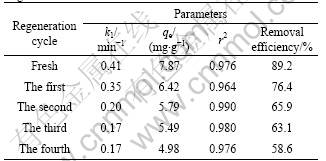
size distribution and surface structure of PAN-ACF are changed during regeneration. The fresh PAN-ACF can adsorb 89.2% of the CT from the solution after 5 min to achieve equilibrium, and the adsorption capacity is 7.87 mg/g. Whereas after the first regeneration, the removal efficiency drops to 76.4%, the equilibrium time prolongs to 10 min, and the adsorption capacity declines to 6.42 mg/g. The results prove that PAN-ACF can be used after thermal regeneration, but the removal efficiency and adsorption capacity for CT decrease along with the increase in regeneration times.
4 Conclusions
(1) The solution pH slightly affects the adsorption due to nonpolar molecule of CT and nonpolarized surface of ACF.
(2) The removal of CT by PAN-ACF from aqueous solution is attributed to the multilayer physisorption, which can be described by Freundlich and D-R equations.
(3) CT is mainly adsorbed onto the exterior surface of PAN-ACF with the rate-controlling step of intraparticle diffusion and the adsorption can be well depicted by the pseudo-first-order kinetic model.
(4) The Yoon-Nelson semiempirical model can well depict the breakthrough curve in the fixed column. The removal efficiencies of CT from industrial wastewater are in the range of 78%-94% in a pilot scale setup, which indicates that PAN-ACF can be applied to practical wastewater treatment.
(5) The ACF can be thermal regenerated, but the removal efficiency and adsorption capacity for CT decrease along with the increase in regeneration times.
References
[1] SITTIG M. Handbook of toxic and hazardous chemicals and carcinogens [M]. 2nd ed. New York: Noyes Publications, 1985: 194.
[2] CHOI J, CHOI K, LEE W. Effects of transition metal and sul?de on the reductive dechlorination of carbon tetrachloride and 1,1,1-trichloroethane by FeS [J]. Journal of Hazardous Materials, 2009, 162(2/3): 1151-1158.
[3] MUN C H, NG W J, HE J Z. Evaluation of biodegradation potential of carbon tetrachloride and chlorophenols under acidogenic condition [J]. Journal of Environmental Engineering, 2008, 134(3): 177-183.
[4] TEEl A L, WATTS R J. Degradation of carbon tetrachloride by modified Fenton’s reagent [J]. Journal of Hazardous Materials, 2002, 94(2): 179-189.
[5] WU Chun-de, LIU Xin-hui, FAN Jin-chun, WANG Lian-sheng. Ultrasonic destruction of chloroform and carbon tetrachloride in aqueous solution [J]. Journal of Environmental Science and Health. Part A: Toxic/Hazardous Substances and Environmental Engineering, 2001, 36(6): 947-955.
[6] OHSAKA T, SHINOZAKI K, TSURUTA K, HIRANO K. Photo-electrochemical degradation of some chlorinated organic compounds on n-TiO2 electrode [J]. Chemosphere, 2008, 73(8): 1279-1283.
[7] HERNANDEZ M A, GONZALEZ A I, ROJAS F, ASOMOZA M, SOLIS S, PORTILLO R. Adsorption of chlorinated compounds (chlorobenzene, chloroform, and carbon tetrachloride) on microporous SiO2, Ag-doped SiO2 and natural and dealuminated clinoptilolites [J]. Industrial and Engineering Chemistry Research, 2007, 46(10): 3373-3381.
[8] SHEN Wen-zhong, WANG Hui, GUAN Ren-gui, LI Zhi-jie. Surface modification of activated carbon fiber and its adsorption for vitamin B1 and folic acid [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2008, 331(3): 263-267.
[9] SHIRATORI N, LEE K J, MIYAWAKI J, HONG S H, MOCHIDA I, AN B, YOKOGAWA K, JANG J, YOON S H. Pore structure analysis of activated carbon fiber by microdomain-based model [J]. Langmuir, 2009, 25(13): 7631-7637.
[10] DIAZ-FLORESP E, LEYVA-RAMOSR, GUERRERO- CORONADOR M, MENDOZA-BARRONJ. Adsorption of pentachlorophenol from aqueous solution onto activated carbon fiber [J]. Industrial and Engineering Chemistry Research, 2006, 45(1): 330-336.
[11] FONTECHA-CAMARA M A, LOPEZ-RAMON M V, ALVAREZ- MERINO M A, MORENO-CASTILLA C. About the endothermic nature of the adsorption of the herbicide diuron from aqueous solutions on activated carbon fiber [J]. Carbon, 2006, 44(11): 2335-2338.
[12] FONTECHA-CAMARA M A, LOPEZ-RAMON M V, ALVAREZ- MERINO M A, MORENO-CASTILLA C. Effect of surface chemistry, solution pH, and ionic strength on the removal of herbicides diuron and amitrole from water by an activated carbon fiber [J]. Langmuir, 2007, 23(3): 1242-1247.
[13] WANG Jian-ping, FENG Hui-min, YU Han-qing. Analysis of adsorption characteristics of 2,4-dichlorophenol from aqueous solutions by activated carbon fiber [J]. Journal of Hazardous Materials, 2007, 144(1/2): 200-207.
[14] LI Kun-quan, ZHENG zheng, FENG Jing-wei, ZHANG Ji-biao, LUO Xing-zhang, ZHAO Guo-hua, HUANG Xing-fa. Adsorption of p-nitroaniline from aqueous solutions onto activated carbon fiber prepared from cotton stalk [J]. Journal of Hazardous Materials, 2009, 166(2/3): 1180-1185.
[15] TANG Deng-yong, ZHENG Zheng, LIN Kui, LUAN Jing-fei, ZHANG Ji-biao. Adsorption of p-nitrophenol from aqueous solutions onto activated carbon fiber [J]. Journal of Hazardous Materials, 2007, 143(1/2): 49-56.
[16] KIM B K, RYU S K, KIM B J, PARK S J. Adsorption behavior of propylamine on activated carbon fiber surfaces as induced by oxygen functional complexes [J]. Journal of Colloid and Interface Science, 2006, 302(2): 695-697.
[17] DUBININ M M. The potential theory of adsorption of gases and vapors for adsorbents with energetically non-uniform surface [J]. Chemical Reviews, 1960, 60(2): 235-241.
[18] TEMKIN M I, PYZHEV V. Kinetics of ammonia synthesis on promoted iron catalyst [J]. Acta Physiochim URSS, 1940, 12: 327-356.
[19] SRIVASTAVA V C, SWAMY M M, MALL I D, PRASAD B, MISHRA I M. Adsorptive removal of phenol by bagasse ?y ash and activated carbon: Equilibrium, kinetics and thermodynamics [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2006, 272(1/2): 89-104.
[20] REDLICH O, PETERSON D L. A useful adsorption isotherm [J]. Journal of Physical Chemistry, 1959, 63(6): 1024.
[21] COONEY D O. Adsorption design for wastewater treatment [M]. London: Lewis Publishers, 1998.
[22] BEKCI Z, SEKI Y, YURDAKOC M K. Equilibrium studies for trimethoprim adsorption on montmorillonite KSF [J]. Journal of Hazardous Materials, 2006, 133(1/2/3): 233-242.
[23] WEBER W J, MORRIS J C. Kinetics of adsorption on carbon from solution [J]. Journal of the Sanitary Engineering Division, 1963, 89(SA2): 31-60.
[24] BOYD G E, ADAMSON A W, MYERS L S. The exchange adsorption of ions from aqueous solutions by organic zeolites. Ⅱ: Kinetics [J]. Journal of the American Chemical Society, 1947, 69(11): 2836-2848.
[25] REICHENBERG D. Properties of ion exchange resins in relation to their structure. Ⅲ: Kinetics of exchange [J]. Journal of the American Chemical Society, 1953, 75(3): 589-597.
[26] KUMAR K V, PORKODI K. Mass transfer, kinetics and equilibrium studies for the biosorption of methylene blue using Paspalum notatum [J]. Journal of Hazardous Materials, 2007, 146(1/2): 214-226.
[27] MILCHERT E, GOC W, PELECH R. Adsorption of CCl4 from aqueous solution on activated carbons [J]. Adsorption Science and Technology, 2000, 18(9): 823-837.
[28] LANGERGREN S, SVENSKA B K. About the theory of so-called adsorption of soluble substances [J]. Kungliga Svenska Veternskapsakademinens Handlingar, 1898, 24(4): 1-39.
[29] HO Y S, MCKAY G. Pseudo-second order model for sorption processes [J]. Process Biochemistry, 1999, 34(5): 451-465.
[30] MCLINTOCK I S. The Elovich equation in chemisorption kinetics [J]. Nature, 1967, 216(5121): 1204-1205.
[31] YOON Y H, NELSON J H. Application of gas adsorption kinetics Ⅰ: A theoretical model for respirator cartridge service life [J]. American Industrial Hygiene Association Journal, 1984, 45(8): 509-516.
Foundation item: Project(2004C33068) supported by the Science and Technology Programs of Zhejiang Province, China; Project(20100933B17) supported by the Social Development and Science Research Program of Hangzhou, China
Received date: 2009-12-28; Accepted date: 2010-04-28
Corresponding author: GUAN Bao-hong, PhD, Associate professor; Tel: +86-571-88273650; E-mail: guanbaohong@zju.edu.cn

