Trans. Nonferrous Met. Soc. China 23(2013) 3063-3067
Selective depression of galena and chalcopyrite by O,O-bis(2,3-dihydroxypropyl) dithiophosphate
Zheng-jie PIAO1, De-zhou WEI1, Zhi-lin LIU1,2, Wen-gang LIU1, Shu-ling GAO1, Ming-yang LI1
1. College of Resources and Civil Engineering, Northeastern University, Shenyang 110004, China;
2. Faculty of Resource and Environmental Engineering, Jiangxi University of Science and Technology, Ganzhou 341000, China
Received 28 August 2012; accepted 7 November 2012
Abstract: A novel synthesized reagent, O,O-bis(2,3-dihydroxypropyl) dithiophosphate (DHDTP), was investigated as depressant on the depression of chalcopyrite and galena, when ammonium dibutyl dithiophosphate (DDTP) was used as the collector in flotation tests. Zeta potential and adsorption measurement were performed to study the interaction between depressant and minerals. The flotation tests of two minerals show that DHDTP has slight depression on chalcopyrite in the whole pH range and strong depression on galena in the pH range of 6-10. When DHDTP dosage is increased, the recovery of galena decreases rapidly, while that of the chalcopyrite decreases slightly. The satisfied separation results of artificially mixed samples are that the copper grade and recovery rates of concentrate are 24.08% and 81%, respectively, when the pH is 6 with 278 mg/L DHDTP. Zeta potential and adsorption measurements show that DHDTP has more strongly adsorption capacity to galena than chalcopyrite.
Key words: O,O-bis(2,3-dihydroxypropyl) dithiophosphate; flotation; chalcopyrite; galena; depressant
1 Introduction
Inorganic compounds have been mainly used as depressants in the flotation separation of complex sulfide ores for a long time, but there are many problems in the application, such as the high dosage, unfriendliness to the environment and poor selectivity. Compared with inorganic depressants, organic depressants have advantages of better selectivity and environmentally friendliness, and moreover they can be synthesized according to the design. In recent years, scholars mainly focused on the study of organic depressants. HUANG et al [1] clarified that chitosan, a natural polymer extracted from crustacean shells, selectively depressed chalcopyrite while galena was floated by xanthate. Chitosan-metal ions adsorption test, TOF-SIMS and X-ray photoelectron spectroscopy (XPS) were employed to study the interaction of chitosan with chalcopyrite and galena. TOF-SIMS measurements indicated that when chalcopyrite and galena were together in the suspension, chitosan hardly adsorbed on galena but adsorbed on chalcopyrite easily.
According to the synergistic effect of carboxymethyl cellulose (CMC), silicate sodium and sodium sulfite, DONG et al [2] found that mixture of three reagents could well inhibit galena, and chalcopyrite was floated. BULATOVIC et al [3,4] reported that RB-SO2-starch could separate chalcopyrite from galena by depressing galena well. LIU et al [5] showed that chalcopyrite was depressed slightly by organic depressant ferrochrome lignin (FCLS), while galena was depressed strongly. IR spectrum analysis indicated that FCLS adsorbed strongly on the surface of galena while the absorption on chalcopyrite was comparatively weak. CHEN et al [6] pointed out that a new organic depressant ASC could depress galena well, while chalcopyrite was floated by butyl xanthate. QIN et al [7] revealed that sodium pyrophosphate (SPH) separated chalcopyrite from galena at acidic condition with O-isopropyl-N-ethyl thionocarbamate (IPETC) as collector. The infrared spectral analysis indicated chemical adsorption between SPH and galena. VALDIVIESO et al [8], DRZYMALA et al [9] and LIU et al [10] indicated that dextrin selectively depressed galena while chalcopyrite was floated with xanthate as the collector. WEI [11] screened out a new organic depressant PPD, and testified that PPD depressed galena but activated chalcopyrite. It was thought that PPD anion could react with Pb2+ to produce hydrophilic compound, which covered on the surface of galena and hindered the collector to adsorb on the surface of galena. The interaction was chemisorption.
The separation of chalcopyrite from galena is difficult. In this work a new organic depressant, O,O-bis (2,3-dihydroxypropyl) dithiophosphate (DHDTP) was studied and its adsorption effects on two minerals were also examined at different pulp pH using ammonium dibutyl dithiophosphate (DDTP) as the collector, and the depression mechanism by measurements of Zeta potential and adsorption isotherms measurements was discussed.
2 Experimental
2.1 Materials
Pure mineral sample of chalcopyrite was obtained from Jiujiang, Jiangxi Province in China, and galena was obtained from Mongolia. Chemical composition analysis of chalcopyrite gave the following chemical composition: 32.9% Cu, 29.1% Fe, 33.2% S, 4.8% others; chemical composition of galena was 86% Pb, 13.3% S, 0.7% others.
2.2 Reagents
Ammonium dibutyl dithiophosphate (DDTP) was used as collector, and terpenic oil was used as frother. They were supplied by Tieling Flotation Reagents Factory, Liaoning, China. O,O�Cbis(2,3-dihydroxy propyl) dithiophosphate (DHDTP) synthesized in our lab was used as depressant. Solutions of HCl and NaOH were employed to adjust the pH of the system.
2.3 Flotation tests
The flotation tests were carried out in a microflotation cell with a 35 mL effective volume. The amount of samples used in each experiment was 2 g, and they were ultrasonically washed for 5 min to remove any possible oxides on the mineral surface. The washing solution of samples was decanted, and the remaining part was floated for at least 3 min. The flotation recovery(R) was calculated as follows: R=[m1/(m1+m2)]��100%, where m1 and m2 are the masses of the floated and unfloated products respectively.
2.4 Zeta potential measurement
Zeta potential was measured by means of Nano-ZS90 apparatus (Made in England). The pure minerals were ground to be smaller than 5 ��m in the agate mortar. A small amount of mineral powder was added to a beaker with 50 mL distilled water, then the mineral was cleaned using ultrasonic generator for 5 min to remove the oxidation film. At last, the reagents were added to the solution, and the suspension was stirred for 3 min with a magnetic stirring apparatus. When the pH of suspension was regulated with HCl or NaOH solution to a desired value, the suspension was ready for the measurement.
2.5 Adsorption isotherms measurement
The mineral samples were transferred to a glass conditioning vessel. Then, a known concentration of reagent solution was introduced into the vessel and continuously stirred until adsorption equilibrium was reached [12]. At last, the mineral suspension was centrifuged for 8 min at 5 000 r/min and the supernatant was sucked out for measuring reagent concentration by spectrophotometric method [13]. It was assumed that the amount of reagent depleted from the solution had been adsorbed totally onto the mineral surface. The amount of reagent absorbed on mineral was calculated from the initial and residual concentrations of reagent.
3 Results and discussion
3.1 Flotation of pure minerals
The recovery of chalcopyrite by the collector DDTP (6.5 mg/L) with or without DHDTP depressant (278 mg/L) as a function of pH is shown in Fig. 1. The results demonstrate that chalcopyrite has a good floatability without DHDTP depressant. The recovery of chalcopyrite decreases slightly in the presence of DHDTP, and the depression effect of DHDTP gets weak with increasing of pH.
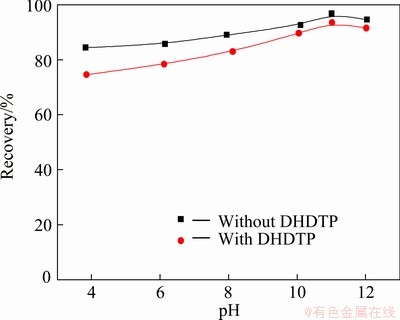
Fig. 1 Effects of pH on flotation of chalcopyrite by collector DDTP (6.5 mg/L) with or without DHDTP depressant (278 mg/L)
The effects of pH on flotation of galena by the collector DDTP (6.5 mg/L) with or without DHDTP as depressant (278 mg/L) are described as a function of pH in Fig. 2. It is shown that the recoveries of galena are basically above 80% in the range of pH 4-10; however, the recovery of galena is dropped dramatically to about 55%, when pH 11. In the presence of DHDTP depressant, the recoveries of galena are below 10% in the range of pH 6-10, but the recoveries of galena are above 25% in the strong acidic or alkaline conditions. This is mainly attributed to the competition mechanism between DDTP and DHDTP on the mineral surface. Hydrophilic groups of DHDTP are adsorbed on the surfaces of galena to form a hydrophilic film and hinder the absorption of DDTP, thus galena is depressed in the flotation test [14,15].
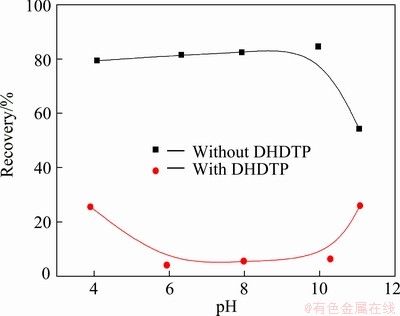
Fig. 2 Effects of pH on flotation of galena by collector DDTP (6.5 mg/L) with or without DHDTP depressant (278 mg/L)
The effects of concentrations of DHDTP on flotation of chalcopyrite and galena by collector DDTP (6.5 mg/L) are considered, and an optimal dosage at pH 6 is found by flotation test. As seen from Fig. 3, the recoveries of chalcopyrite decrease slightly with the increase of the DHDTP concentration, but the recoveries of galena decrease more rapidly with increasing DHDTP dosage. When the concentration is 278 mg/L, the recoveries of chalcopyrite and galena are about 85% and 5%, respectively.
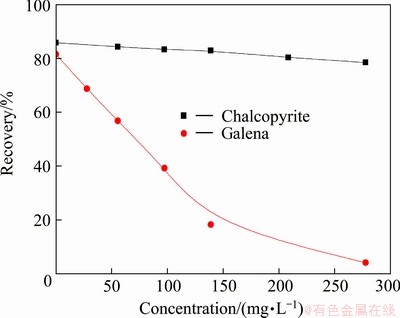
Fig. 3 Effects of concentration of DHDTP on flotation of chalcopyrite and galena by collector DDTP (6.5 mg/L) at pH 6
3.2 Flotation of artificially mixed minerals
Pure minerals flotation results show that DHDTP has slight depression to chalcopyrite and strong depression to galena. Artificially mixed samples are adopted to demonstrate that it is a good depressant to separate chalcopyrite from galena.
The separation efficiency of chalcopyrite by DHDTP depressant (278 mg/L) with the collector DDTP (6.5 mg/L) as a function of pH is shown in Fig. 4. The copper grade and recovery of concentrate are 24.08% and 81% respectively when pH is 6, and the copper grade and recovery of concentrate decrease with pH increasing or decreasing.
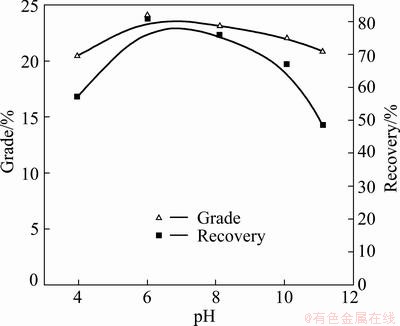
Fig. 4 Effects of pH on separation of chalcopyrite by DHDTP depressant (278 mg/L) with collector DDTP (6.5 mg/L)
The effects of DHDTP dosage on separation efficiency of chalcopyrite by collector DDTP (6.5 mg/L) are considered, and an optimal dosage at pH 6 is found by flotation test. It can be seen from Fig. 5 that the copper grade of concentrate increases with DHDTP dosage increasing, while the copper recovery of concentrate decreases. The appropriate concentration of DHDTP is 278 mg/L, and chalcopyrite can be well separated from galena.
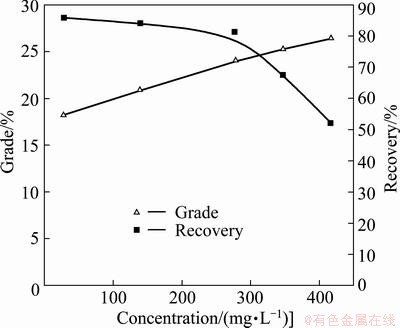
Fig. 5 Effects of concentrations of DHDTP on separation of chalcopyrite by collector DDTP (6.5 mg/L) at pH 6
3.3 Zeta potential measurement
The effects of DHDTP on Zeta potential of chalcopyrite and galena are shown in Figs. 6 and 7 respectively. It is revealed that the adsorption of anionic depressant DHDTP increases negative charges of galena more than chalcopyrite, namely the flotation selectivity between chalcopyrite and galena is good with DHDTP as depressant. The adsorption of DHDTP strongly increases the negative potential of mineral, so the potential is almost negative in the whole range of pH. The potential of mineral becomes negative in the presence of DHDTP at a low pH, revealing the electrostatic attraction between DHDTP and minerals. When the potential of galena is negative without DHDTP, the addition of DHDTP still clearly increases the negative potential, indicating the existence of other types of interaction between DHDTP and galena, such as hydrogen bonding and chemisorption.
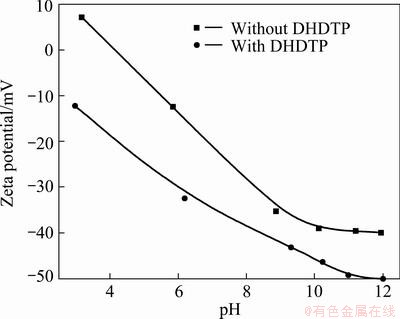
Fig. 6 Zeta potential of chalcopyrite as function of pH
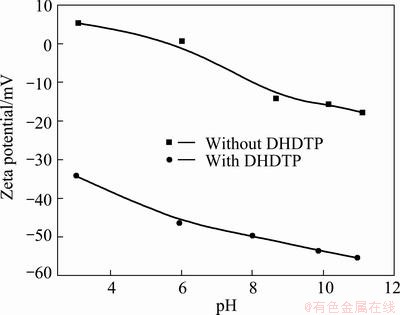
Fig. 7 Zeta potential of galena as function of pH
3.4 Adsorption isotherms measurement
The adsorption amount may be related with the depression performance, as it is found that the more the adsorption, the more the depression. The adsorption capacities of DHDTP on chalcopyrite and galena with different concentrations of depressant at pH 6 are shown in Fig. 8. It is found that increasing adsorption amount with galena is faster compared with chalcopyrite, which is consistent with the result of flotation test (Fig. 3). The adsorption dosage of galena grows nearly linearly, which may be due to the chemisorption between DHDTP and galena [16,17]. While the adsorption dosage of chalcopyrite grows slowly, the adsorption equilibrium can be approached with a large amount of DHDTP.
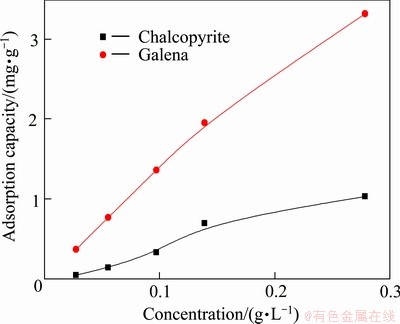
Fig. 8 Adsorption capacity of DHDTP on chalcopyrite and galena as function of DHDTP concentration (pH=6)
Adsorption capacities of DHDTP on chalcopyrite and galena as a function of pH are illustrated in Fig. 9. It is shown that the adsorption amount of DHDTP on galena is larger than that on chalcopyrite in the broad pH range. The dosage of DHDTP on chalcopyrite is getting less with the increase of pH, which is also consistent with the results of flotation test (Fig. 1). The adsorption capacity of DHDTP on galena as a function of the pH is similar to the parabolic profile, and the dosage of DHDTP at pH=11 is less than that at pH=4. This trend is also in agreement with the flotation test (Fig. 2).
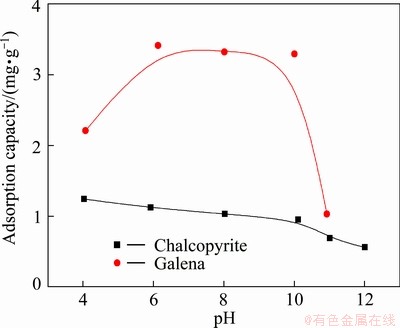
Fig. 9 Adsorption capacity of DHDTP on chalcopyrite and galena as function of pH
4 Conclusions
1) O,O-bis (2,3-dihydroxypropyl) dithiophosphate (DHDTP) depresses chalcopyrite slightly and galena strongly with ammonium dibutyl dithiophosphate (DDTP) as the collector in the flotation of pure minerals. Galena is depressed by DHDTP in the range of pH 6-10. The satisfied results of artificially mixed samples are that the copper grade and recovery of concentrate are 24.08% and 81% respectively when DHDTP dosage is 278 mg/L and pH is 6.
2) Zeta potential experiments indicate that DHDTP absorbs galena more efficiently than chalcopyrite, which is due to the electrostatic attraction between DHDTP and minerals.
3) The adsorption of DHDTP on chalcopyrite and galena illustrates that there may be chemisorption between DHDTP and galena.
References
[1] HUANG Peng, CAO Ming-li, LIU Qi. Using chitosan as a selective depressant in the differential flotation of Cu-Pb sulfides [J]. International Journal of Mineral Processing, 2012, 106-109: 8-15.
[2] DONG Yan-fei. Study on the electrochemistry mechanism of flotation separating copper-lead sulphide minerals [D]. Changsha: Central South University, 2011. (in Chinese)
[3] BULATOVIC S. Flotation process: United States, US4246096A [P]. 1981-01-20.
[4] BULATOVIC S, WYSLOUZIL D M. Selection and evaluation of different depressants systems for flotation of complex sulphide ores [J]. Minerals Engineering, 1995, 8(1): 63-76.
[5] LIU Run-qing, SUN Wei, HU Yue-hua. Study on organic depressant FCLS for separation of chalcopyrite and galena [J]. Mining and Metallurgical Engineering, 2009, 29(3): 29-32. (in Chinese)
[6] CHEN Jian-hua, FENG Qi-ming, LU Yi-ping. Research on a new organic depressant ASC for separation chalcopyrite and galena [J]. Conservation and Utilization of Mineral Resources, 2000(5): 39-42. (in Chinese)
[7] QIN Wen-qing, WEI Qian, JIAO Fen, LI Ning, WANG Pei-pei, KE Li-fang. Effect of sodium pyrophosphate on the flotation separation of chalcopyrite from galena [J]. International Journal of Mining Science and Technology, 2012, 22(3): 345-349.
[8] VALDIVIESO A L, LOPEZ A A S, SONG S, MARTINEZ H A G, ALMADA S L. Dextrin as a regulator for the selective flotation of chalcopyrite, galena and pyrite [J]. Canadian Metallurgical Quarterly, 2007, 46(3): 301-309.
[9] DRZYMALA J, KAPUSNIAK J, TORNASIK P. Removal of lead minerals from copper industrial flotation concentrates by xanthate flotation in the presence of dextrin [J]. International Journal of Mineral Processing, 2003, 70(1-4): 147-155.
[10] LIU Qi, ZHANG Ya-hui. Effect of calcium ions and citric acid on the flotation separation of chalcopyrite from galena using dextrin [J]. Minerals Engineering, 2000, 13(13): 1405-1416.
[11] WEI Ming-an. Fundamental research on flotation separation of chalcopyrite and galena [D]. Shenyang: Northeastern University, 2008. (in Chinese)
[12] MORRIS G E. The adsorption characteristics of polymeric depressants at the talc-water interface [D]. Adelaide: University of South Australia, 1996.
[13] DUBIOS M, GILLES K A, HAMILTON J K, REBERS P A, SMITH F. Colorimetric method for determination of sugars and related substances [J]. Analytical Chemistry, 1956, 28(3): 350-356.
[14] HE Ming-fei, QIN Wen-qing, LI Wei-zhong, ZENG Ke. Pyrite depression in marmatite flotation by sodium glycerine-xanthate [J]. Transactions of Nonferrous Metals Society of China, 2011, 21(5): 1161-1165.
[15] WANG Dian-zuo. Molecular design of reagents for mineral and metallurgical processing [M]. Changsha: Central South University of Technology Press, 1996. (in Chinese)
[16] NATARAJAN R, SHARMA J, NIRDOSH I. Adsorption of N-hydrocinnamoyl-N-phenyl-hydroxylamine on pure minerals [J]. Journal of the International Adsorption, 2010, 16(6): 541-548.
[17] XIONG Dao-ling. The synthesis of xanthate orgnic depressant and its depressing mechanisms in zinc-iron sulfide minerals [D]. Changsha: Central South University, 2006. (in Chinese).
O,O-��(2,3-���ǻ�����)���������Ի�ͭ��ͷ�Ǧ���ѡ��������
������1��κ����1��������1,2�����ĸ�1��������1��������1
1. ������ѧ ��Դ����ľ����ѧԺ������ 110004��
2. ����������ѧ ��Դ�뻷��ѧԺ������ 341000
ժ Ҫ���Զ�狀�ҩΪ���ռ�������һ���������Ƽ�O,O-��(2,3-���ǻ�����)���������(DHDTP)�Ի�ͭ��ͷ�Ǧ����и�ѡ��ͨ������λ���������IJ��ԣ�̽�����Ƽ�DHDTP����������û�������ѡ������������������pH��Χ�ڣ����Ƽ�DHDTP�Ի�ͭ����������ú�������pH 6~10��Χ�ڣ����Ƽ�DHDTP�Է�Ǧ���к�ǿ���������á��������Ƽ�DHDTP���������࣬��Ǧ��Ļ�����Ѹ�ٽ��ͣ�����ͭ��Ļ�����ֻ��С���ȵĽ��͡���DHDTP����278 mg/L����pH 6�������£��˹���Ͽ�ѡ�ó��ľ�����ͭ��Ʒλ�ͻ����ʽϺã��ֱ�Ϊ24.08%��81%��ͨ������λ���������IJ��Խ����֪��DHDTP�ڷ�Ǧ���ϵ������������Ը��ڻ�ͭ��ġ�
�ؼ��ʣ�O,O-��(2,3-���ǻ�����)����������ѡ����ͭ��Ǧ�����Ƽ�
(Edited by Hua YANG)
Foundation item: Project (2008BAB34B01) supported by the National Key Technology R&D Program of China
Corresponding author: De-zhou WEI; Tel: +86-24-83673863; E-mail: dzwei@mail.neu.edu.cn
DOI: 10.1016/S1003-6326(13)62834-4