
Differences of grain-refining effect of Sc and Ti additions in aluminum by empirical electron theory analysis
YE Yi-cong(叶益聪)1, HE Liang-ju(何良菊)2, LI Pei-jie(李培杰)1
1. National Center of Novel Materials for International Research, Department of Mechanical Engineering,
Tsinghua University, Beijing 100084, China;
2. School of Aerospace, Tsinghua University, Beijing 100084, China
Received 21 November 2008; accepted 30 June 2009
Abstract: The differences of grain-refining effect between Sc and Ti additions in aluminum, which cannot be substantially explained by traditional theories, were carefully studied. The empirical electron theory (EET) of solids and molecules was employed to calculate the valence electron structures (VES) of Al3Ti and Al3Sc. The conclusions can be drawn that, in the two alloys Al-Ti and Al-Sc, the different valence electron structures of Al3Ti and Al3Sc and the consequent differences of growth habit of the two particles, and the different interfacial electron density between particles and matrix fundamentally lead to the differences of grain-refining effect between Sc and Ti additions on aluminum and make Sr the better grain-refiner of aluminum.
Key words: grain refining; empirical electron theory (EET); valence electron structure (VES); Al3Sc; Al3Ti
1 Introduction
It has long been established that the formation of a fine scale, equiaxed grain structure is desirable in Al-castings because it can improve the mechanical properties, reduce hot tearing, increase feeding to eliminate shrinkage porosity, and give a more uniform distribution of second phases[1]. The usual method of controlling the cast grain structure of aluminum is the deliberate addition of master alloys containing melt inoculants, the most common of which is based on the Al-Ti system[2-3]. However, nowadays, researchers from different countries have paid much attention to scandium (Sc) additions in aluminum, which can perform a better grain-refining effect[4-8].
Ti and Sc, which are in the adjacent locations of periodic system of elements, are both effective refiners of aluminum and aluminum alloys. A large number of primary particles act as effective nucleation sites during the subsequent solidification[4, 9-10]. Much of the existing literature has demonstrated that Sc is so far found to be the most effective grain refiner of aluminum, which, however, to our knowledge, is simply attributed to the very low lattice misfit between the primary Al3Sc particles and the Al matrix. In fact, it cannot be decided that either Sc or Ti has a better refining effect on aluminum just from the traditional theory: 1) Generally, peritectic reaction is preferred for a grain refiner, for it gives a clean interface of non-homogeneous nucleation sites. The reaction between the Al3Sc and α(Al) phases is an eutectic one, while that between the Al3Ti and α(Al) phases is a peritectic one[4, 9]. 2) Although the coherence between the L12 Al3Sc and α(Al) is much better than that between the tetragonal DO22 Al3Ti and α(Al)[11-12], which might lead to a better refining effect of Sc on aluminum than Ti, there is still another factor that definitely cannot be neglected, i.e., the number of nuclei in unit-volume melt. 3) By taking the solute concentration of 0.7% (mass fraction) as an example, the mass fraction of Al3Ti in the melt will be 2.65 times higher than that of Al3Sc[13]. Because the densities of Al3Sc and Al3Ti are close to each other (3.026 g/cm3 and 3.37 g/cm3), if the dimension of primary Al3Sc particle is close to that of Al3Ti, then the number of Al3Ti particles will be much larger than that of Al3Sc, which may offset the influence of the second factor.
It cannot be decided that either Ti or Sc is better for grain-refinement of aluminum with these three factors above. In the present work, experiments have been curried out to study the different grain-refining effects of Sc and Ti additions on aluminum, and the valence electron structures (VES) have been calculated, using the empirical electron theory (EET) of solids and molecules, which can help to achieve a deeper understanding of this interesting phenomenon.
2 Experimental
The different-composition alloys used in this work were prepared using pure aluminum (99.99%), Al-2.12Sc and Al-2.15Ti (%, mass fraction) master alloys. The cylindrical cast samples were produced by pouring the molten alloys with a 50 K superheat into a mould. A sample was taken from each casting and ICP-AES (inductively coupled plasma atomic emission spectroscopy) was employed to determine the final composition. These results are displayed in Table 1 and demonstrate that the actual compositions are very close to the nominal compositions.
Table 1 Nominal alloy compositions and analysis results (mass fraction, %)

To observe the grain-refining effect with naked eyes, the samples were etched using enriched Keller’s reagent after mechanical polishing. To reveal the grain structure for optical metallography, the samples were electropolished in the mixture of perchloric acid and acetic acid, and subsequently anodized in Baker’s reagent and examined under polarized light. For scanning electron microscope (SEM) examination, samples were lightly electropolished and examined to achieve backscatter electronic images in a FEI Quanta 200 FEG, operated at 15 kV. EDS was employed to determine the composition of different phases.
When a grain size of less than 500 μm was observed, the mean linear intercept method was used to measure the average grain size[14]. However, for the samples with very large grain (of the order of millimeters), the grain size was estimated directly from the samples.
3 Results and analysis
3.1 Experimental results
Fig.1 shows the naked-eye observation of the different refining effects of Sc and Ti additions on aluminum. The grain sizes of Al-Ti and Al-Sc against the Sc or Ti content are displayed in Fig.2. When the solute content is low (<0.6%, mass fraction), Ti has a better refining effect on aluminum than Sc, and an unrefined large grain size is measured in Al-Sc alloy. When the content of Sc is increased beyond a certain value (around 0.5%), a dramatic reduction in the grain size is observed in Al-Sc, thus Sc performs a much stronger refining effect on aluminum than Ti when the contents of Sc and Ti are larger than 0.7%.

Fig.1 Grain-refining effect comparison of Al-Ti and Al-Sc alloys with same solute content

Fig.2 Plot of grain size vs solute content of Al-Ti and Al-Sc alloys
Fig.3 presents metallographic images of the grain structures of Al-Ti and Al-Sc alloys. Equiaxed grains with an average size of 220 μm are achieved in Al-0.2Ti, while coarse columnar grains with the size order of millimetre still dominate in Al-0.2Sc. When the solute content is increased up to 1.2%, the grain size of Al-Ti alloy is around 90 μm without large difference compared with Al-0.2Ti. While for Al-1.2Sc, there is a total absence of dendritic sub-structure, the grain size is sharply decreased to around 25 μm.

Fig.3 Grain structures of Al-Ti and Al-Sc alloys: (a) Al-0.2Ti; (b) Al-0.2Sc; (c) Al-1.2Ti; (d) Al-1.2Sc
In the backscatter electronic images (Fig.4), it is obvious that Al3Ti in Al-1.2Ti is needle-like in 2D, with a dimension of 80-120 μm, while Al3Sc is cubic with a size of 5-10 μm.

Fig.4 Backscattered electron images of primary Al3Ti in Al-1.2Ti (a) and primary Al3Sc in Al-1.2Sc (b)
3.2 EET analysis
3.2.1 EET explanation of numbers of Al3Sc and Al3Ti
The empirical electron theory (EET) of solids and molecules, founded by YU[15], gives a description of valence electron structure (VES) of crystal on the basis of atom state using an ingenious method[16-17]. As one of the most promising electron theories[18], EET has been recently employed by many researchers, achieving inspiring results[19-23]. VES of Al3Sc and Al3Ti has been calculated by EET[13], and the results are shown in Table 2, in which Dnα stands for a covalent bond length between two atoms, nα for covalent electron pair number of the bond, and α for the sequence number of the bonds, marking as A, B, C, D, E, according to the length of bonds. It is necessary to indicate that, the hybrid level σ of Al in Al3Sc is chosen to be 6 in Ref.[13], which is considered to be not appropriate in this work. The hybrid level σAl is chosen to be 5 here because it is more reasonable when it is not under extreme condition[24-25].
Table 2 Valence electron structures of Al3Sc and Al3Ti[13]
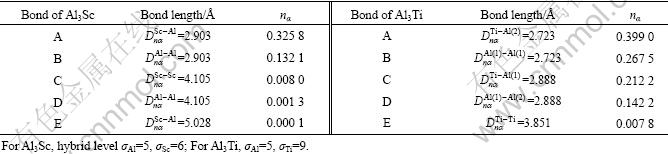
The nα values of the strongest bond A and the second stronger bond B of Al3Ti are higher than those of Al3Sc, respectively (Table 2). Even the fourth stronger bond D of Al3Ti has a higher nα value than the second stronger bond of Al3Sc. The strongest bond of α(Al) has a relative small nα value of 0.208 6[26]. These indicate that there is a strongest tendency to combine Al and Ti, and Al3Ti is easiest to precipitate during solidification, which is more difficult for Al3Sc unless the content of Sc in Al-Sc alloy is increased beyond a certain value. Therefore, if a small quantity (e.g. 0.2%) of Ti and Sc are respectively added to Al, Al3Ti tends to precipitate substantially, while Al3Sc does not. This also qualitatively explains why the critical content for Al3Sc to precipitate in Al-Sc is 0.55% (mass fraction) (0.33%, molar fraction), while for Al3Ti in Al-Ti alloy, it is only 0.15% (mass fraction) (0.08%, molar fraction). However, if the content of added element is increased beyond a certain value (around 0.7%, mass fraction), for Al3Ti there will be a more and more serious segregation. Although the quantity of Al3Ti is much larger than Al3Sc, excessive growth and serious reunion will lead to a smaller number of individual Al3Ti particles than individual Al3Sc particles, whose dimension is smaller because of the relatively low nα values.
3.2.2 EET explanation of crystallization efficiency of Al-Sc and Al-Ti alloys
In the DO22 Al3Ti, the nα values of bond A and bond B, which are located in the (001) crystal plane, are much higher than those of bond C and bond D, which are located in the (010) crystal plane (Fig. 5). Because of the stronger combining tendency of the atoms, the (001) crystal plane will extend more rapidly than the (010) crystal plane, which leads to anisotropy of the growth of Al3Ti. Therefore, Al3Ti grows to a needle-like shape in 2D (Fig.4), with a much larger dimension in the x and y directions than in the z direction (Fig.5), leaving only the (001) crystal plane facing the melt; and thus leads to a relatively low crystallization efficiency of Al matrix. Whereas, because of a high degree of symmetry of the atomic bonds in the L12 Al3Sc (Fig.5), it grows to a cubic structure with more crystal planes facing the melt (Fig.4), offering more nucleation sites for α(Al).

Fig.5 Diagrammatic sketches of crystal structures of Al3Ti and Al3Sc, and bonds with most valence electrons: (a) Al3Ti; (b) Al3Sc
Through calculation and analysis of electronic densities of two-phase interfaces in Al-Sc and Al-Ti alloys, the mechanism of grain refinement in the two alloys can be discussed further. There are following orientations on the two-phase interface in Al3Sc:
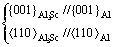 (1)
(1)
For the crystal plane (001) of Al3Sc, there are
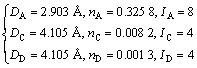 (2)
(2)
The total number of shared electrons on each covalent bond of this crystal plane is
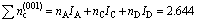 (3)
(3)
The area of the crystal plane is
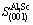 =a2=16.851 ?2 (4)
=a2=16.851 ?2 (4)
Covalent bond electron density of this crystal plane is
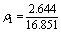 =0.157 ?-2 (5)
=0.157 ?-2 (5)
Similarly, the covalent bond electron density of the (001) crystal plane of α(Al) can be calculated:
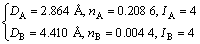 (6)
(6)
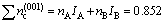 (7)
(7)
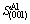 =a2=16.4 ?2 (8)
=a2=16.4 ?2 (8)
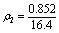 =0.052 ?-2 (9)
=0.052 ?-2 (9)
The difference of electron densities is
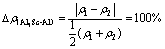 (10)
(10)
There are following orientations on the two-phase interface in Al3Ti:
 (11)
(11)
For the (001) crystal plane of Al3Ti, there are
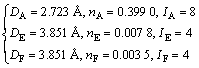 (12)
(12)
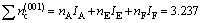 (13)
(13)
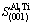 =14.822 5 ?2 (14)
=14.822 5 ?2 (14)
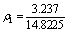 =0.218 ?-2 (15)
=0.218 ?-2 (15)
The difference of electron densities is
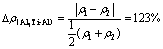 (16)
(16)
The interfacial electron density difference on the interface  , 100%, is smaller than that on
, 100%, is smaller than that on  , 123%, which means the electron density is more continuous on the interface
, 123%, which means the electron density is more continuous on the interface  than on
than on  , as shown in Fig.6. Thus, the interfacial stress and the interfacial energy between Al3Sc and α(Al) is smaller than that between Al3Ti and α(Al). Consequently, Al3Sc offers a better nucleation sites for α(Al) with a higher efficiency of crystallization than Al3Ti.
, as shown in Fig.6. Thus, the interfacial stress and the interfacial energy between Al3Sc and α(Al) is smaller than that between Al3Ti and α(Al). Consequently, Al3Sc offers a better nucleation sites for α(Al) with a higher efficiency of crystallization than Al3Ti.
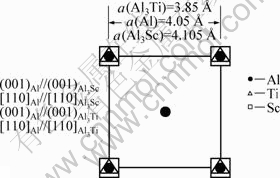
Fig.6 Schematic diagram of relation in interface 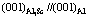 and
and 
4 Conclusions
1) The higher nα (covalent electron number of a bond) values of bonds of Al3Ti than Al3Sc lead to a stronger precipitation and reunion tendency of Al3Ti than Al3Sc, and thus a smaller number of individual Al3Ti particles than individual Al3Sc particles when the solute content is beyond a certain value.
2) The different nα values between bonds in different crystal planes of Al3Ti lead to anisotropy of the growth of Al3Ti, which grows to needle-like shape in two-dimension, leaving only the (001) crystal plane facing the melt. However, Al3Sc with a high degree of symmetry of atomic bonds, grows to a cubic structure with more crystal planes facing the melt, offering more nucleation sites for α(Al). Besides, the more continuous electron density at the interface in Al3Sc offers better nucleation sites for α(Al). Therefore, the crystallization efficiency of Al-Sc alloy is relatively high.
3) With a larger quantity of particles and higher crystallization efficiency, Sc is proofed a better grain-refiner of aluminum than Ti.
References
[1] FLEMINGS M C. Solidification processing [M]. New York: McGrawHill, 1974.
[2] MOHANTY P S, GRUZLESKI J E. Mechanism of grain refinement in aluminium [J]. Acta Metallurgica et Materialia, 1995, 43(5): 2001-2012.
[3] McCARTNEY D G. Grain refinement of aluminum and its alloy using inoculants [J]. International Materials Reviews, 1989, 34(5): 247-260.
[4] NORMAN A F, PRANGNELL P B, McEWEN R S. The solidification behavior of dilute aluminium-scandium alloys [J]. Acta Materialia, 1998, 46(16): 5715-5732.
[5] KOMURA S, BERBON P B, FURUKAWA M, HORITA Z, NEMOTO M, LANGDON T G. High strain rate superplasticity in an Al-Mg alloy containing scandium [J]. Scripta Materialia, 1998, 38(12): 1851-1856.
[6] FURUKAWA M, UTSUNOMIYA A, MATSUBARA K, HORITA Z, LANGDON T G. Influence of magnesium on grain refinement and ductility in a dilute Al-Sc alloy [J]. Acta Materialia, 2001, 49(18): 3829-3838.
[7] NIEH T G, KAIBYSHEV R, HSIUNG L M, NGUYEN N, WADSWORTH J. Subgrain formation and evolution during the deformation of an Al-Mg-Sc alloy at elevated temperatures [J]. Scripta Materialia, 1997, 36(9): 1011-1016.
[8] FULLER C B, KRAUSE A R, DUNAND D C, SEIDMAN D N. Microstructure and mechanical properties of a 5754 aluminum alloy modified by Sc and Zr additions [J]. Materials Science and Engineering A, 2002, 338(1/2): 8-16.
[9] MONDOLFO L F. Aluminum alloys: Structure and properties [M]. London: Butterworths, 1976.
[10] YIN Zhi-min, GAO Yong-zhen, PAN Qing-lin, ZHANG Yong-hong, YIN Song-bo. Effect of trace Sc and Zr on grain refinement of as cast Al-Mg alloys [J]. The Chinese Journal of Nonferrous Metals, 1997, 7(4): 75-78. (in Chinese)
[11] MURTY B S, KORI S A, CHAKRABORTY M. Grain refinement of aluminium and its alloys by heterogeneous nucleation and alloying [J]. International Materials Reviews, 2002, 47(1): 3-29.
[12] KHARAKTEROVA M L, ESKIN D G, TOROPOVA L S. Precipitation hardening in ternary alloys of the Al-Sc-Cu and Al-Sc-Si systems [J]. Acta Metallurgica et Materialia, 1994, 42(7): 2285-2290.
[13] LI Pei-jie, YE Yi-cong, HE Liang-ju. Valence electron structure analysis of refining mechanism of Sc and Ti additions on aluminum [J]. Chinese Science Bulletin, 2008, 53(11): 1345-1349. (in Chinese)
[14] PICKERING F B. Basis of quantitative metallography[M]. London: Institute of Metals, 1975.
[15] YU Rui-huang. The empirical electron theory of solids and molecules [J]. Chinese Science Bulletin, 1978, 23(4): 217-224. (in Chinese)
[16] LIU Zhi-lin, LI Zhi-lin, LIU Wei-dong. Electron structure of the interface and interfacial properties [M]. Beijing: Science Press, 2002. (in Chinese)
[17] ZHANG Rui-lin. Empirical electron theory in solids and molecules [M]. Changchun: Jilin Science and Technology Press, 1993: 225-427. (in Chinese)
[18] LI Wu-hui, REN Feng-zhang, MA Zhan-hong, JIA Shu-guo, TIAN Bao-hong, ZHENG Mao-sheng. Application of electron theories in materials science [J]. The Chinese Journal of Nonferrous Metals, 2008, 18(3): 494-504. (in Chinese)
[19] JIA Shu-guo, LIU Ping, ZHENG Mao-sheng, REN Feng-zhang, TIAN Bao-hong, ZHOU Gen-shu. Explanation based on electron theory for solid solution strengthening in copper alloy [J]. The Chinese Journal of Nonferrous Metals, 2008, 18(8): 1522-1525. (in Chinese)
[20] WU Wen-xia, GUO Yong-quan, LI An-hua, LI Wei. Analysis of valence electron structures and calculation of magnetic properties of Nd2Fe14B [J]. Acta Physica Sinica, 2008, 57(4): 2486-2491. (in Chinese)
[21] FANG Chang-zeng, SUN Li-ling, WEI Zun-jie, MA Ming-zhen, LIU Ri-ping, ZENG Song-yan, WANG Wen-kui. Valence electron structures of TaC and TaN [J]. Science in China: G, 2008, 38(12): 120-125. (in Chinese)
[22] PENG Ke, YI Mao-zhong, TAO Hui-jin, RAN Li-ping. Valence electronic structure analysis and cohesive energy calculation of MoSi2 [J]. The Chinese Journal of Nonferrous Metals, 2007, 17(2): 216-221. (in Chinese)
[23] LI Jin-ping, MENG Song-he, HAN Jie-cai, ZHANG Xing-hong, LUO Xiao-guang. Valence electron structure and properties of the ZrC1-xNx solid solution [J]. Rare Metal Materials and Engineering, 2008, 37(6): 980-983. (in Chinese)
[24] GAO Ying-jun, BAN Dong-mei, HAN Yong-jian, ZHONG Xia-ping, LIU Hui. Atomic bonding and mechanical properties of Al-Mg-Zr-Sc alloy [J]. Trans Nonferrous Met Soc China, 2004, 14(5): 922-926.
[25] YU Rui-huang. α and β phase of Al-Mg alloy and valence electron structure analysis of γ-Al12Mg19 [J]. Chinese Science Bulletin, 1979, 4(4): 54-75. (in Chinese)
[26] GAO Ying-jun, HUANG Chuang-gao, HOU Xian-hua. Atomic bonding and properties of Al-Mg-Sc alloy[J]. Materials Transactions, 2005, 46(6): 1148-1153.
Foundation item: Project(20050003042) supported by Research Fund for the Doctoral Program of Higher Education of China
Corresponding author: YE Yi-cong; Tel/Fax: +86-10-62788074; E-mail: yeyc@mails.thu.edu.cn
DOI: 10.1016/S1003-6326(09)60163-1
(Edited by YANG Bing)