
Effects of sputtering conditions on electrochemical behavior and
physical properties of Ni-Mo alloy electrode
HUANG Jin-zhao(黄金昭)1, XU Zheng(徐 征)1, LI Hai-ling(李海玲)2,
KANG Guo-hu(亢国虎)2, WANG Wen-jing(王文静)2
1. Key Laboratory of Luminescence and Optical Information, Ministry of Education,
Institute of Optoelectronic Technology, Beijing Jiaotong University, Beijing 100044, China;
2. Beijing Solar Energy Research Institute, Beijing 100083, China
Received 15 November 2005; accepted 2 March 2006
Abstract: Sputtering method was used to prepare Ni-Mo alloy electrodes for hydrogen production in alkaline solution. The influences of the working pressure during deposition and the substrate temperature on the electrochemical behavior of electrode were characterized by steady-state polarization plot and Tafel polarization curve measurements. And the physical properties of electrodes were characterized by XRD, SEM, AFM and EDS. It is found that the overpotential is significantly influenced by the working pressure which affects critically the electrode surface morphology, and two Tafel regions are observed for each sample. The overpotential value does not change very much with the substrate temperature. The XRD results indicates that the electrodes should be considered nanocrystalline. Thornton model for the microstructure of sputter-deposited electrodes is referred to explain the observed microstructure change.
Key words: hydrogen evolution reaction; overpotential; Ni-Mo alloy electrode
1 Introduction
Many transition metal alloys with binary and ternary compositions have been studied as hydrogen evolution reaction electrodes in water electrolysis[1-6]. The activity of such metal alloys for the hydrogen evolution reaction depends on at least two factors, i.e. the synergistic effect of alloy constituents and the surface area. As for the synergistic effect, JAKSIC[7-9] reported that, based on the Brewer-Engel valence bond theory, when metals of the left half of the transition series with empty or half-filled vacant d-orbital are alloyed with metals of the right half of the series with internally paired d-electron, there arises a well-pronounced synergism in electrocatalysis for the hydrogen evolution reaction. In some reports[5,10,11], the synergistic effects were actually verified to function, and the large surface area supported by such metal alloy electrodes is proved to be an important factor for the high catalytic activity. Among the materials, nickel and nickel based alloys [12,13], particularly Ni-Mo[14-18], have high catalytic activity.
Ni-Mo electrode is one of the best binary metal alloys for the hydrogen evolution reaction and can be fabricated by the methods such as electrolytic deposition [19-21], sputtering[15], and mechanical alloying. Sputtering is a relatively controllable method to prepare electrodes and the electrode characteristics are considered to depend on various sputtering parameters.
In this study, NiMo alloy thin films were deposited by RF magnetron sputtering under different conditions. The influence of the working pressure during deposition and the substrate temperature on the electrochemical behavior and microstructure of the electrode were studied. The properties were characterized by steady-state polarization plot, Tafel polarization curve, XRD, SEM, AFM and EDS.
2 Experimental
The Ni-Mo samples were prepared by RF magne- tron sputtering from a Ni-Mo alloy target which is 76.4 mm in diameter, 3 mm in thickness and 99.99% in purity. The molar fractions of Ni and Mo are all 50%. The sputtering parameters of working pressure (1-6 Pa) during deposition and substrate temperature (150-250 ℃) were changed for studying their effects on the sample preparation. The vacuum chamber was pumped to a pressure of less than 4×10-3 Pa with diffusion pump before sputtering. Then a pre-sputtering process (10 min) was conducted to clean the target surface and remove any possible contamination. The RF power was kept at 200 W. Nickel foil was used as a substrate for the electrochemical measurements, and Si wafer substrate for other characterizations.
The compositions of the fabricated samples were determined with EDS which was equipped with SEM (JEM-6301F). The phase compositions of the samples were measured by X-ray diffraction(XRD) using a D/max-RB X-ray diffractometer (RIGAKU, Japan) with filtered Cu Kα radiation and wavelength of 1.540 6 ?. The surface morphology was characterized by AFM (Quesant-ScopeTM 250) experiments. The electro- chemical measurements were carried out in a three-electrode cylindrical electrochemical cell with Luggin capillary in order to minimize the IR drop of the solution resistance, which consists of a standard calomel reference electrode(SCE) and a Pt counter electrode, as shown in Fig.1. All tests were conducted with a well defined electrode area of 1 cm2 in 1 mol/L potassium hydroxide (KOH) prepared using deionized water. The data were acquired at room temperature.
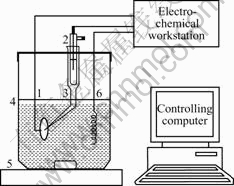
Fig.1 Schematic diagram of experimental set-up: 1 Working electrode; 2 Reference electrode; 3 Luggin capillary; 4 Electro- chemical cell; 5 Magnetic stirrer; 6 Counter electrode
3 Results and discussion
Fig.2 shows the relationship of the Mo content with working pressure and substrate temperature. With increasing working pressure, the Mo content increases. On the contrary, with increasing substrate temperature, the Mo content decreases.

Fig.2 Variations of Mo content with sputtering condition: (a) Working pressure; (b) Substrate temperature
Fig.3 shows the relationship of the hydrogen evolu- tion reaction overpotential value with the working pressure and substrate temperature. It is seen that increasing working pressure (i.e. the increase of Mo content) enhances the hydrogen evolution reaction activity. The overpotential increases with the increase of substrate temperature. While the influence of working pressure on the hydrogen evolution overpotential is much greater than that of substrate temperature.

Fig.3 Variations of overpotential with sputtering condition: (a) Working pressure; (b) Substrate temperature
In order to investigate the electrocatalytic activity of the prepared coatings, Tafel polarization curve measurements were made. Two Tafel regions are observed for every sample, as shown in Fig.4. It was discussed that the change of Tafel slope from the low overpotential range to high one could be due to the increase of hydrogen on surface[11]. By the polarization of the investigated bimetallic catalytic electrode to high overpotential, a large amount of atomic and molecular hydrogen are generated, which are then absorbed into the catalyst layer close to the electrolyte interface. Due to the combination of the right-hand side transition metal with almost-filled d-orbital (Ni) with the left-hand side metal with half-filled or almost-empty d-orbital (Mo), the binding energy for an M―H bond increases, and the strong bonding between metal and absorbed hydrogen atoms becomes a barrier to hydrogen evolution. So the modification of the electronic and structural property of the coating in the near-surface region causes the decrease in the Tafel slope and increase in hydrogen overpotential at high current densities. Therefore, the formation of hydrides at high overpotentials seems to be the major reason for the change of the Tafel slope.
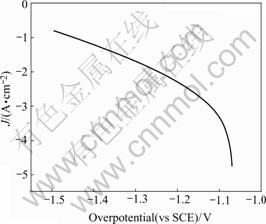
Fig.4 Tafel polarization curve of Ni-Mo alloy electrode
The XRD results are shown in Fig.5, and it can be seen that there is only one broadened diffraction peak at 2θ=43.29? and with the change of sputtering conditions (working pressure and substrate temperature) the peak pattern changes slightly. The broadened diffraction peak indicates that the crystal lattice distortion is possibly caused by codeposition of Ni with Mo. By Scherrer formula, the grain size can be calculated to be 2.3 nm. This shows that the grain size is very small. It is also found that the diffraction peak shape of the electrode is different from that of the amorphous state, so it could be considered nanocrystalline. The surface atoms on the nanocrystalline alloy possess higher reaction activity because of their higher surface energy, so the nano- crystalline NiMo electrode has specific electrochemical properties.

Fig.5 XRD patterns of Ni-Mo alloy electrode
The AFM images in Fig.6 show significant differences in morphology of the investigated electrodes prepared at different working pressures. The sample prepared at 1.0 Pa has a featureless flat surface while the sample prepared at 6.0 Pa has a granular structure. It is apparent that the latter sample has a larger surface area than the former. The variation in the overpotential value, which is observed by changing the working pressure, is considered to be mainly due to the change of surface morphology.
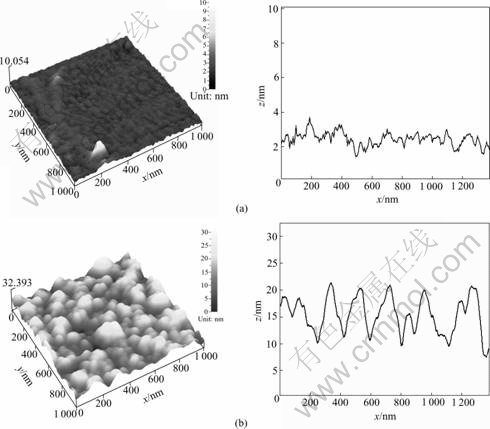
Fig.6 AFM 3D-images of surface morphologies for Ni-Mo alloy electrodes at substrate temperature of 150 ℃: (a) Working pressure of 1 Pa; (b) Working pressure of 6 Pa
The change in the surface morphology by the working pressure could be explained by the Thornton model for the microstructure of sputter-deposited films[22]. In this model, it is expected that the electrode which is composed of fine columns with many gaps is formed in the Zone 1 with high working pressure and low substrate temperature (Fig.7). On the other hand, the dense films made of packed fibrous columns are formed in the Zone 2 with low working pressure and low substrate temperature. It would be considered that the sputtering conditions applied to the samples on Figs.6(a) and (b) correspond to Zone 2 and Zone 1, respectively. The AFM observation for the samples prepared at 6.0 Pa and higher substrate temperature up to 250 ℃ does not show obvious changes in the surface structure (Fig.8 and Fig.6(b)). This is in accordance with the fact that the overpotential value does not change very much with the substrate temperature.

Fig.7 SEM images of cross-section of Ni-Mo alloy thin film (Scale is 6 ?m)
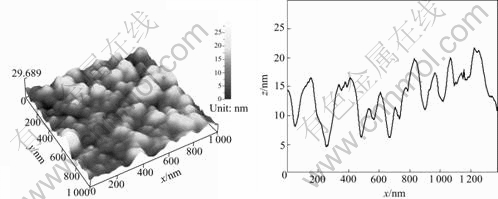
Fig.8 AFM 3D-images of surface morphology for Ni-Mo alloy electrode deposited at working pressure of 6 Pa and substrate temperature of 250 ℃
4 Conclusions
1) The NiMo alloy electrodes are deposited by RF magnetron sputtering with different parameters. The working pressure is the main factor affecting the physical and electrochemical properties of the films. The higher working pressure critically affects the film surface morphology. The electrochemical behavior is enhanced by increasing Mo content.
2) Two Tafel regions are observed. The over- potential value does not change very much with the substrate temperature.
3) The XRD pattern indicates that the electrodes should be considered nanocrystalline. The AFM, SEM experimental results are in accordance with Thornton model for the microstructure of sputter-deposited electrodes. NiMo nanocrystalline alloy is a promising hydrogen evolution reaction electrode.
References
[1] HU Lian-xi, WANG Er-de. Hydrogen generation via hydrolysis of nanocrystalline MgH2 and MgH2-based composites [J]. Trans Nonferrous Met Soc China, 2005, 15(5): 965-970.
[2] FAN C L, PIRON D L, MIAO H J, ROJAS M. Hydrogen evolution in alkaline water on cobalt electrodeposits prepared from baths containing different anions [J]. J Appl Electrochem, 1993, 23(10): 985-990.
[3] DE GIZ M J, MACHADO S A S, AVACA L A, GONZALES E R. High area Ni-Zn and Ni-Co-Zn codeposits as hydrogen electrodes in alkaline solutions [J]. J Appl Electrochem, 1992, 22(10): 973-977.
[4] ARUL R I. Nickel-based binary-composite electrocatalysts for the cathodes in the energy-efficient industrial production of hydrogen from alkaline-water electrolytic cells [J]. J Mater Sci, 1993, 28(16): 4375-4382.
[5] TOSHIO A, ASAHI K, EIJI A, HIROKI H, KATSUHIKO A, KOJI H. Hydrogen evolution characteristics of sputter-deposited Co-Mo, Co-Al and Co-Mo-Al alloy electrodes in NaOH solution [J]. Mater Trans JIM, 1998, 39(10): 1017-1023.
[6] CHANG R L, SUNG G K. Electrochemical stability of Co-Mo intermetallic compound electrodes for hydrogen oxidation reaction in hot KOH solution [J]. Journal Power Source, 2000, 87(1): 64-68.
[7] JAKSIC M M. Towards the reversible electrode for hydrogen evolution in industrially important electrochemical processes [J]. Advances in Hydrogen Energy, 1984, 2: 845-861.
[8] JAKSIC M M. Electrocatalysis of hydrogen evolution in the light of the Brewer―Engel theory for bonding in meals and intermetallic phases [J]. Electrochimica Acta, 1984, 29: 1539-1550.
[9] JAKSIC M M. Advance in electrocatalysis for hydrogen evolution in the light of the Brewer―Engel valence-bond theory [J]. Int J Hydrogen Energ, 1987, 12(11): 727-752.
[10] FAN C, PIRON D L, SLEB A, PARADIS P. Study of electrodeposited nickel-molybdenum, nickel-tungsten, cobalt- molybdenum, and cobalt-tungsten as hydrogen electrodes in alkaline water electrolysis [J]. J Electrochem Soc, 1994, 141(2): 382-387.
[11] FAN C, PIRON D L, PARADIS P. Hydrogen evolution on electrodeposited nickel-cobalt-molybdenum in alkaline water electrolysis [J]. Electrochimica Acta, 1994, 39(18): 2715-2722.
[12] MIOUSSE D, LASIA A, BORCK V. Hydrogen evolution reaction on Ni-Al-Mo and Ni-Al electrodes prepared by low pressure plasma spraying [J]. J Appl Electrochem, 1995, 25(6): 592-602.
[13] FAN C, PIRON D L, ROJS M. Polarization of cobalt-molybdenum and nickel-molybdenum hydrogen electrodes for alkaline fuel cells [J]. Int J Hydrogen Energ, 1994, 19(6): 529-533.
[14] ARUL RAJ I, VASU K I. Transition metal-based hydrogen electrodes in alkaline solution―electrocatalysis on nickel based binary alloy coatings [J]. J Appl Electrochem, 1990, 20(1): 32-38.
[15] ASAHI K, EIJI A, HIROKI H, KOJI H. Characterization of sputter-deposited Ni-Mo and Ni-W alloy electrocatalysts for hydrogen evolution in alkaline solution [J]. Mat Sci Eng A, 1997, 226-228: 905-909.
[16] BELTOWSKA-LEHMAN E. Kinetic correlations in codeposition of coatings of molybdenum-iron group metal alloys [J]. J Appl Electrochem, 1990, 20(1): 132-138.
[17] RODRIGUEZ-VALDEZ L M, ESTRADA-GUEL I F, ALMERAYA-CALDERON M A, NERI-FLORES M A, MARTINEZ-VILLAFANE A, MARTINEZ-SANCHEZ R. Electrochemical performance of hydrogen evolution reaction of Ni-Mo electrodes obtained by mechanical alloying [J]. Int J Hydrogen Energ, 2004, 29(11): 1141-1145.
[18] GENNERO DE CHIALVO M R, CHIALVO A C. Hydrogen evolution reaction on smooth Ni(1-x)+Mo(x) alloys(0≤x≤0.25) [J]. J Electroanal Chem, 1998, 448: 87-93.
[19] LUCIANA S S, SERGIO H D, CLAUDIA E B M, LUCIA H M. Characterisation of electrochemically deposited Ni-Mo alloy coatings [J]. Electrochemistry Communications, 2004, 6: 543-548.
[20] SIMPRAGA R, BAI L, CONWAY B E. Real area and electrocatalysis factors in hydrogen evolution kinetics at electrodeposited Ni-Mo and Ni-Mo-Cd composites: effect of Cd content and nature of substrate [J]. J Appl Electrochem, 1995, 25(7): 628-641.
[21] ELISA N, ZHIWEN C, SASHA O. Characterization of Ni, NiMo, NiW and NiFe electroactive coatings as electrocatalysts for hydrogen evolution in an acidic medium [J]. J Mol Catal A: Chem, 2005, 226(2): 179-197.
[22] THORNTON J A. Influence of apparatus geometry and deposition conditions on the structure and topography of thick sputtered coatings [J]. J Vac Sci Technol, 1974, 11(4): 666-670.
Foundation item: Project(2003CB214501) supported by the National Basic Research Program of China; Project(48010) supported by the Excellent Doctor’s Science and Technology Innovation Foundation of Beijing Jiaotong University, China
Corresponding author: XU Zheng; Tel: +86-10-51688605; Fax: +86-10-51683933; E-mail: zhengxu@center.njtu.edu.cn
(Edited by YANG Bing)